Abstract
In this work, the pyrolysis behavior of plastic waste—TV plastic shell—was investigated, based on thermogravimetric analysis and using a combination of model-fitting and model-free methods. The possible reaction mechanism and kinetic compensation effects were also examined. Thermogravimetric analysis indicated that the decomposition of plastic waste in a helium atmosphere can be divided into three stages: the minor loss stage (20–300°C), the major loss stage (300–500°C) and the stable loss stage (500–1000°C). The corresponding weight loss at three different heating rates of 15, 25 and 35 K/min were determined to be 2.80–3.02%, 94.45–95.11% and 0.04–0.16%, respectively. The activation energy (
Keywords
Introduction
Once touted as a ‘material of a thousand uses’, plastic meets our demand across various sectors and has become an essential part of daily life (Rahimi and García, 2017). Production and consumption of plastic has increased exponentially since the early 1950s. It is estimated that approximately 400 million tonnes (Mt) of plastic is produced globally each year and a whopping 8300 Mt of plastics was produced between 1950 and 2015 (Geyer et al., 2017). Concomitant with usage, the worldwide generation of plastic waste is rapidly increasing and the amount produced since 2015 is estimated to be 6300 Mt. However, only 9% of this waste has been recycled and 79% has ultimately accumulated in landfill sites or has been dumped in the natural environment (Geyer et al., 2017). It is well known that traditional plastics are difficult to decompose and thus the disposal of plastic waste poses a long-term threat to the natural environment. In order to tackle the plastic disposal problem, the development of alternative recycling or treatment technologies for them is mandatory. Traditional landfilling offers an inexpensive solution for the solid waste, but it will take up land resources and waste the energy intrinsic in plastics (Li et al., 2014). There are drawbacks to recent recycling methods, such as mechanical separation, pelleting and regeneration, attributed to their high labor cost and water contamination (Datta and Kopczyńska, 2016; Deng et al., 2017; Hamad et al., 2013).
Advanced thermal treatments are gaining interest, as these have the advantage of drastic volume reduction and energy recovery (Aguado et al., 2008; Cheng et al., 2019; Yu et al., 2019). Among these, pyrolysis is regarded as one potentially useful method, due to its lower emissions, reasonable cost and simple operation (Qi et al., 2019; Sharuddin et al., 2016). Understanding the process of pyrolysis of plastic waste and its kinetics is important for reactor design and the selection of optimization parameters in practice. Jung et al. (2013) studied the pyrolysis characteristics of waste high impact polystyrene (HIPS) and acrylonitrile-butadiene-styrene (ABS) using a fluidized bed reactor. Szabo et al. (2011) investigated the pyrolysis behavior of a waste polymethylmethacrylate-ABS blend. Liu et al. (2017) investigated the thermal degradation of polymer blends (ABS/polyvinyl chloride (PVC), ABS/nylon 6 (PA6) and ABS/polycarbonate (PC) and analyzed pyrolysis kinetics using the Coats–Redfern (CR) and the Starink methods. Balart et al.(2006) determined the thermal properties of PC-ABS mixtures and studied the kinetics using an autocatalytic model. It is worth noting that reports on the pyrolysis of plastic waste in the form of TV plastic shell are sparse. In addition, it is difficult to precisely describe the pyrolysis kinetics using several methods. Therefore, in this work, a series of thermogravimetric (TG) experiments on plastic waste was conducted using a simultaneous thermal analyzer with different heating rates of 15, 25 and 35 K/min. Based on the TG analysis, the activation energies (
Experimental
Materials
The waste TV plastic shell was collected from a large-scale electrical and electronic equipment recycling plant located in Shandong province, China. It was mainly composed of ABS. It is difficult to crush this plastic using traditional mechanical methods. Therefore, it was ground using a LD-450 cryogenic grinder (Jiangyin Yifang Machinery Co. Ltd., China) with a grinding temperature of −33.1°C.
Experimental procedure
TG analysis is the most widely used technique for studying the thermal decomposition of solids (Ashraf et al., 2019; Bach and Chen, 2017). During the test, approximately 2 mg of the plastic sample was placed on the ceramic crucibles and heated from 20°C to 1000°C under a helium gas flow of 50 mL/min. The tests were repeated at different heating rates of 15, 25 and 35 K/min. The sample weight and temperature changes during the thermal process were recorded to obtain the TG and derivative thermogravimetric (DTG) profiles. The schematic diagram of this study is illustrated in Figure 1.
The schematic diagram of this work.
Kinetic modeling
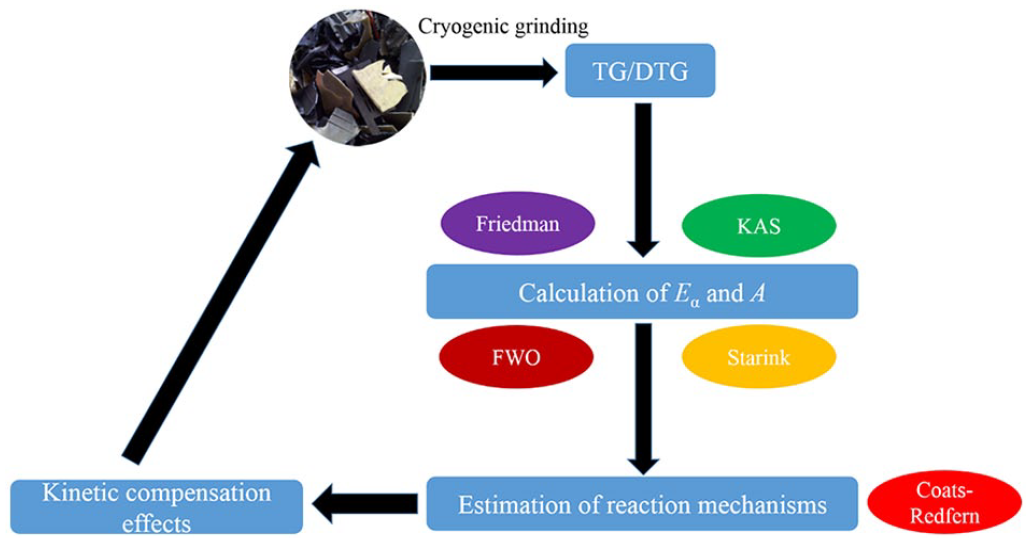
The basic theory of kinetic modeling for solid fuels is presented here; more details can be found elsewhere (Papari and Hawboldt, 2015, Tanaka, 2005, Yao et al., 2019a, Yao et al., 2019b). There are two major methods, the model-fitting and the model-free method, which are employed for the study of the decomposition of solids (Khawam and Flanagan, 2005; Vyazovkin et al., 2011). Model-fitting methods are commonly applied because of their ability to determine the kinetic parameters directly and to offer information about possible reaction mechanisms. Different reaction models have been proposed (Aboulkas and El Bouadili, 2010; Chong et al., 2017; Khawam and Flanagan, 2006; Ma et al., 2018) and kinetic parameters can be determined based on these models. The expressions of
Expressions of
The TG results can be expressed as a function of degree of conversion (

where
The solid conversion rate (d

where
The

where
Under constant temperature ramp conditions, combining equation (2) and equation (3) yields

CR method
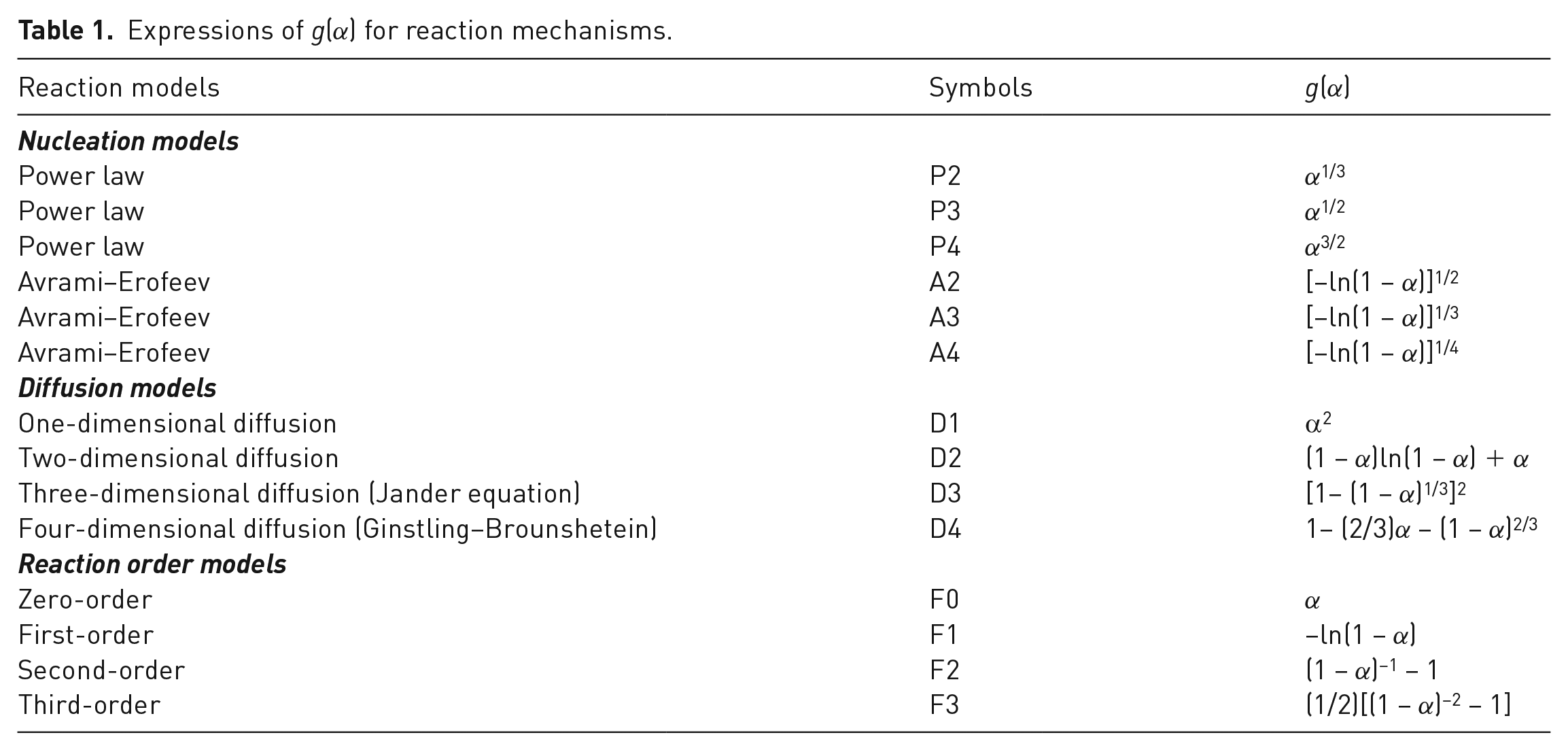
The CR method is an integral model-fitting method developed by Coats and Redfern (1964), which uses an asymptotic series expansion for estimation of the temperature integral. The integral form of the reaction model can be obtained by integrating equation (4) as (Çepelioğullar et al., 2016; Özveren and Özdoğan, 2013)
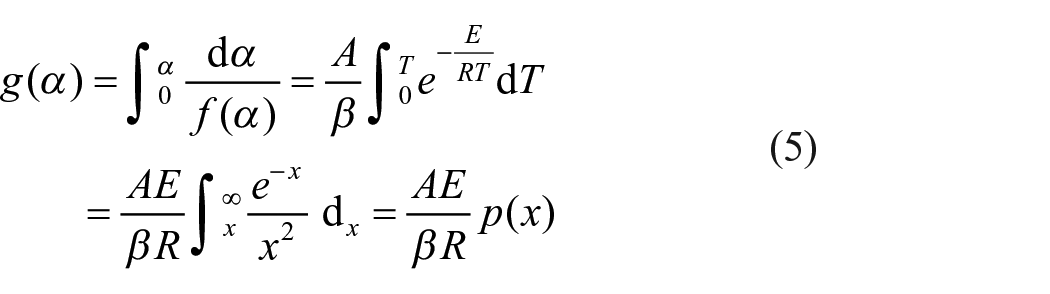
where
By introducing an approximation
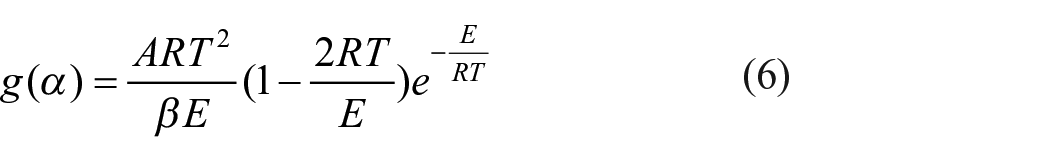
Taking the natural logarithm of both sides of equation (6) yields
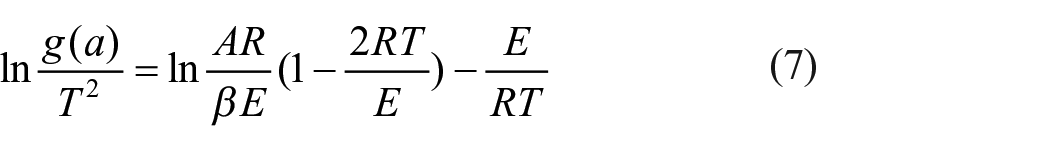
Since 2

For a fixed
Friedman method
Both differential and integral isoconversional methods were used for determination of the activation energy. The Friedman method (Friedman, 1964) is the most common differential isoconversional method for evaluating the activation energy as a function of
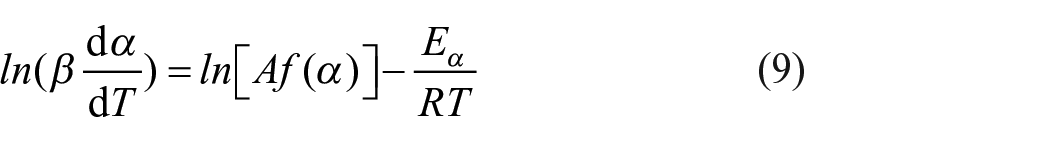
A series of TG experiments at different heating rates enables the extraction of data for the same
KAS method
The integral term

For a constant value of
FWO method
The FWO method is a model-free method developed by Flynn and Wall (1966) as well as Ozawa (1965). It uses Doyle’s equation for the approximation of the temperature integral (Doyle, 1965). Taking into account the approximation
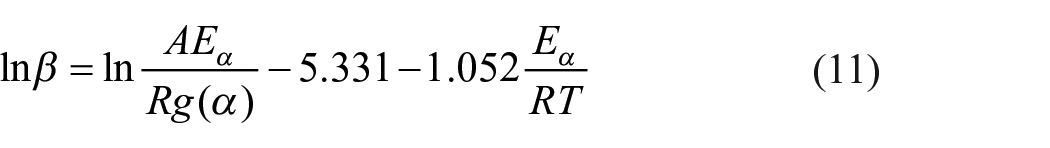
For a fixed value of
Starink method
By integrating the approximation

For a series of
Results and discussion
TG analysis
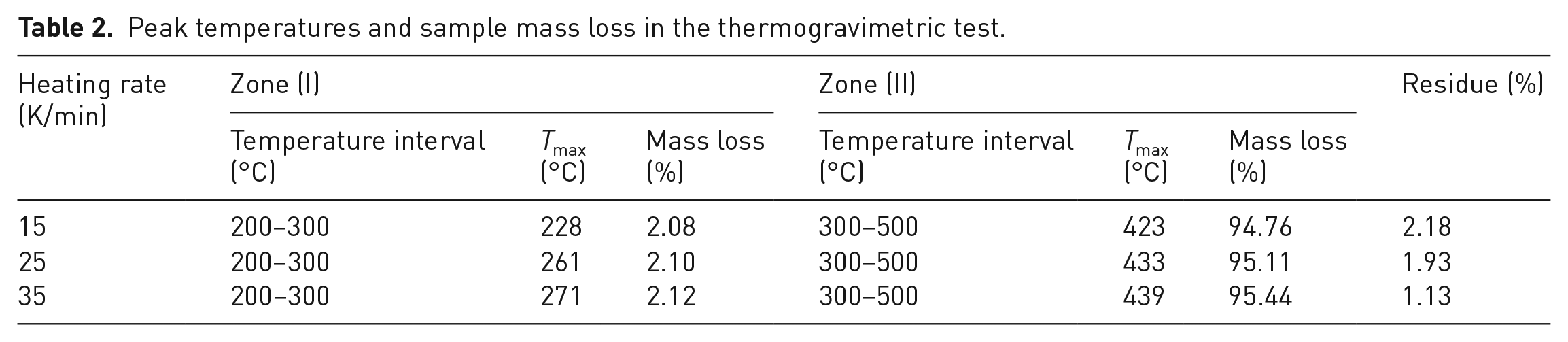
The conversion rate and DTG curves of plastic waste at different heating rates are displayed in Figure 2. From Figure 2(a), it can be seen that the conversion displayed a similar trend for samples at different heating rates. The whole thermal process can be divided into three stages: the minor loss stage (20–300°C), the major loss stage (300–500°C) and the stable loss stage (500–1000°C). The weight loss at three heating rate were determined to be 2.80–3.02%, 94.45–95.11% and 0.04–0.16% for these stages, respectively, which was consistent with the decomposition of ABS polymer (Ma and Pang, 2015; Polli et al., 2009). As for the thermal degradation of pure ABS, some researchers observed one or two steps. The discrepancy mainly derives from the sample molecular weight, sample mass, heating rate, etc. From the DTG profiles in Figure 2(b), it was found that the DTG curve can be divided into three zones. Among them, zone I is the initiation of pyrolysis, during which the backbone of the polymer ruptured into monomers, such as styrene, acrylonitrile and polybutadiene (Yang et al., 2004, Yu et al., 2019). Suzuki and Wilkie (1995) investigated the degradation of pure ABS and found that the evolution of butadiene commenced at 340°C and styrene at 350°C, while the evolution of monomeric acrylonitrile initiated at 400°C. Zone II is the major conversion stage, where a significant mass loss of approximately 95% was observed. The decomposition rates elevated from 0.12 to 0.28 %•s−1 with the heating rate increasing from 15 to 35 K/min. In this stage, monomers resulting from zone I decomposed to form small molecules, such as HBr, CO2, CO and CH4 (Yu et al., 2019). In zone III, the degradation rate significantly decreased to 2.13E−5–55.05E−5 %•s−1 and a minor weight loss of 0.04–0.40% was observed. Larger brominated derivatives decomposed and smaller molecules dominated the major gaseous products (Yu et al., 2019). This indicated that the decomposition of TV plastic shell was mainly occurred at less than 500°C, namely, in zone I and zone II. From Figure 2 and Table 2, the maximum peak temperatures were observed at 228, 261 and 271°C for three heating rates in zone I and 423, 433 and 439 °C in zone II, respectively. The obvious shift toward higher peak decomposition temperatures with increasing heating rate has been reported in the literature for coal (Song et al., 2016) and biomass (Cortés and Bridgwater, 2015). This may be attributed to the increasing effect of heat transfer limitations, which cause temperature gradients within the sample and inside each particle. At a lower heating rate, the heating of plastic particles occurs gradually leading to an improved heat transfer to the inner portions and among the particles. However, an increase of
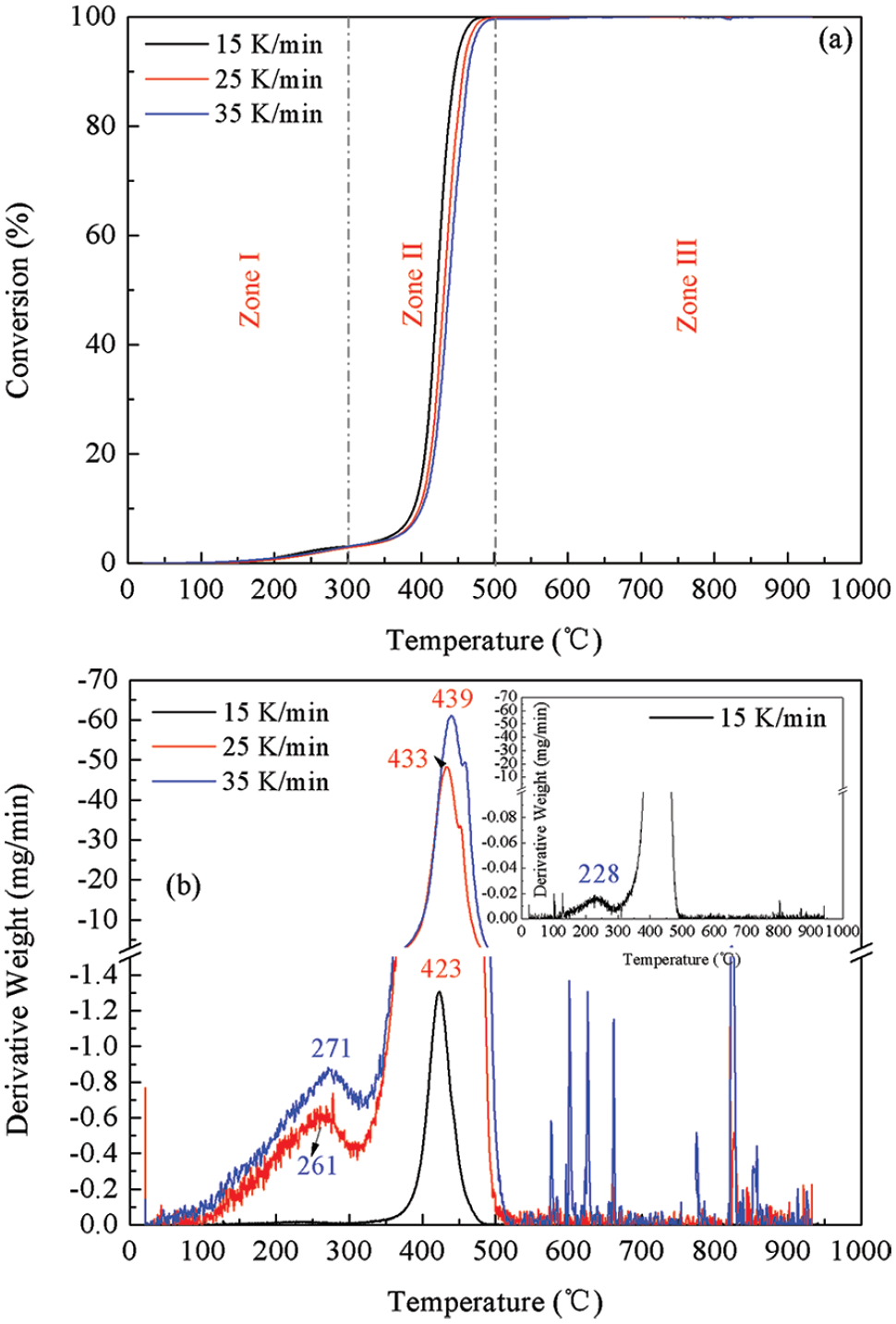
(a) Conversion rate and (b) derivative thermogravimetric (DTG) curves of plastic waste at different heating rates.
Peak temperatures and sample mass loss in the thermogravimetric test.
Calculation of activation energy
In this section, the activation energy (
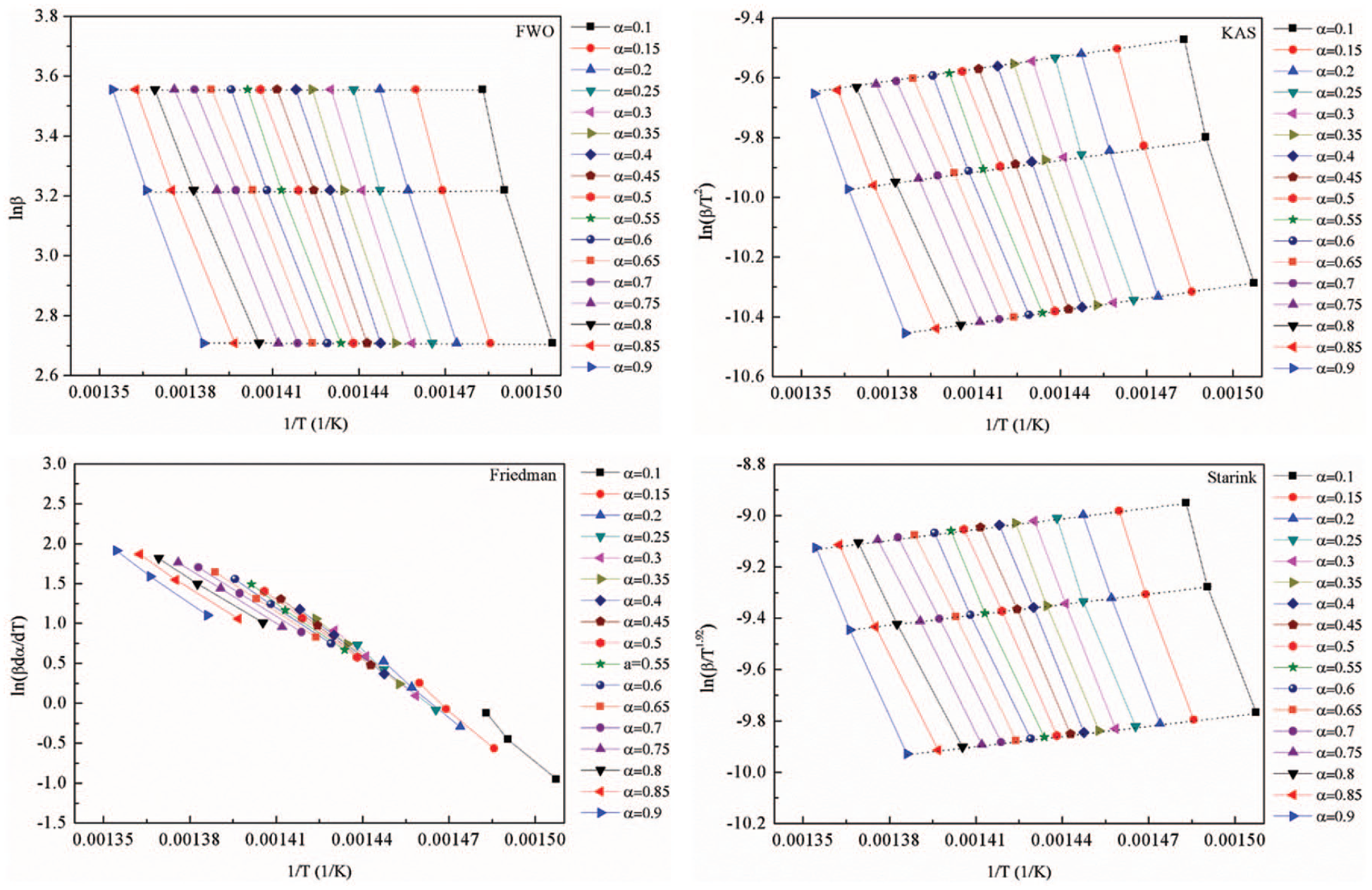
The linear fitting curves under different conversions for model-free methods.
The kinetic parameters determined using the model-free methods.
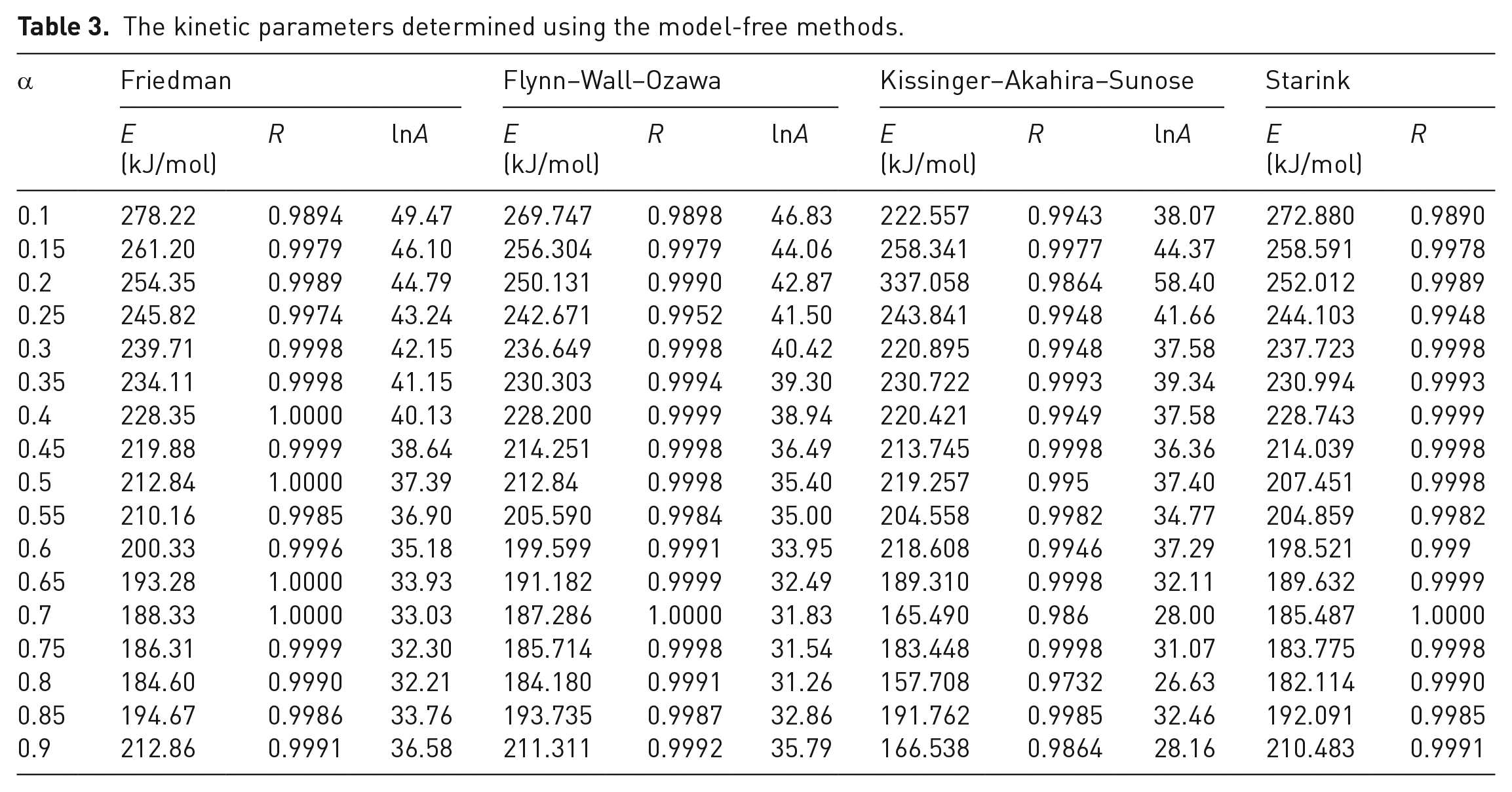
The kinetic parameters calculated using the KAS method displayed a similar trend to those from the Friedman method, although they were smaller than the latter (Figure 4). This was consistent with the literature (Yuan et al., 2017), although an increasing tendency with an increase of
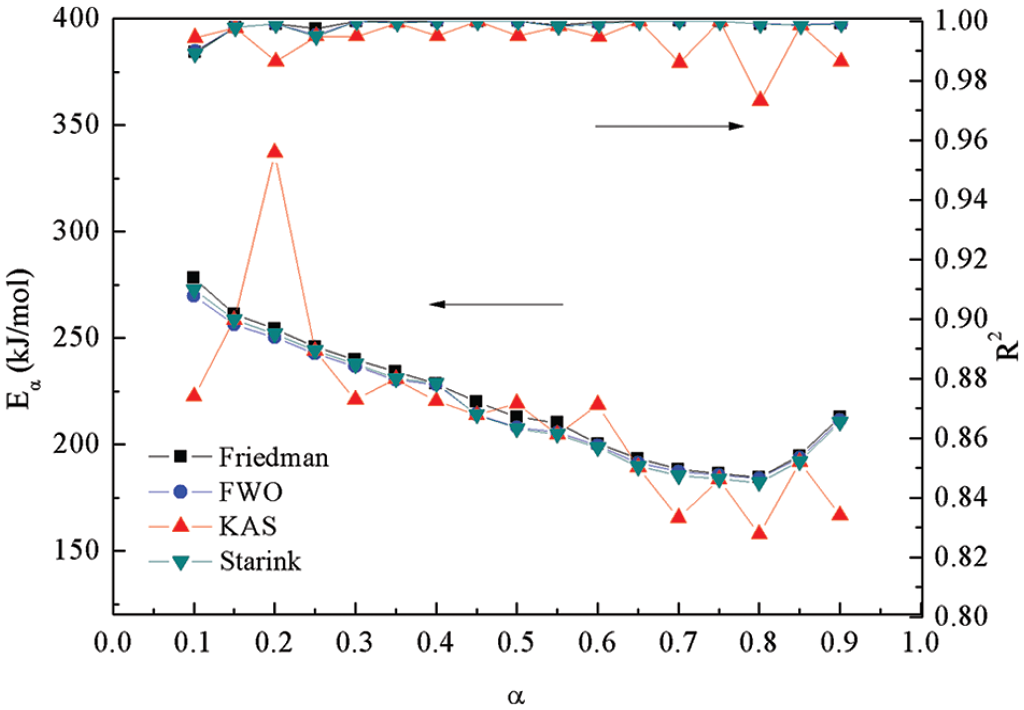
Comparison of kinetic parameters derived from model-free methods.
The FWO method is an integral method, which is also independent of the degradation mechanism. According to equation (11), numerous pairs of ln
The kinetic parameters calculated using the Starink method displayed the same trend as those derived from the FWO method. This was consistent with the literature (Wu et al., 2018), although the activation energy displayed three stages for the entire
When the four model-fitting methods are compared, similar activation energies and correlation coefficients were observed for the Friedman and KAS methods. The profiles are displayed for these and for the other two methods in Figure 4. However, the
Estimation of reaction mechanisms
Employment of the CR method for TG data can determine the most probable mechanism function and enable the pre-exponential factor (ln
The kinetic parameters calculated using the model-fitting methods.
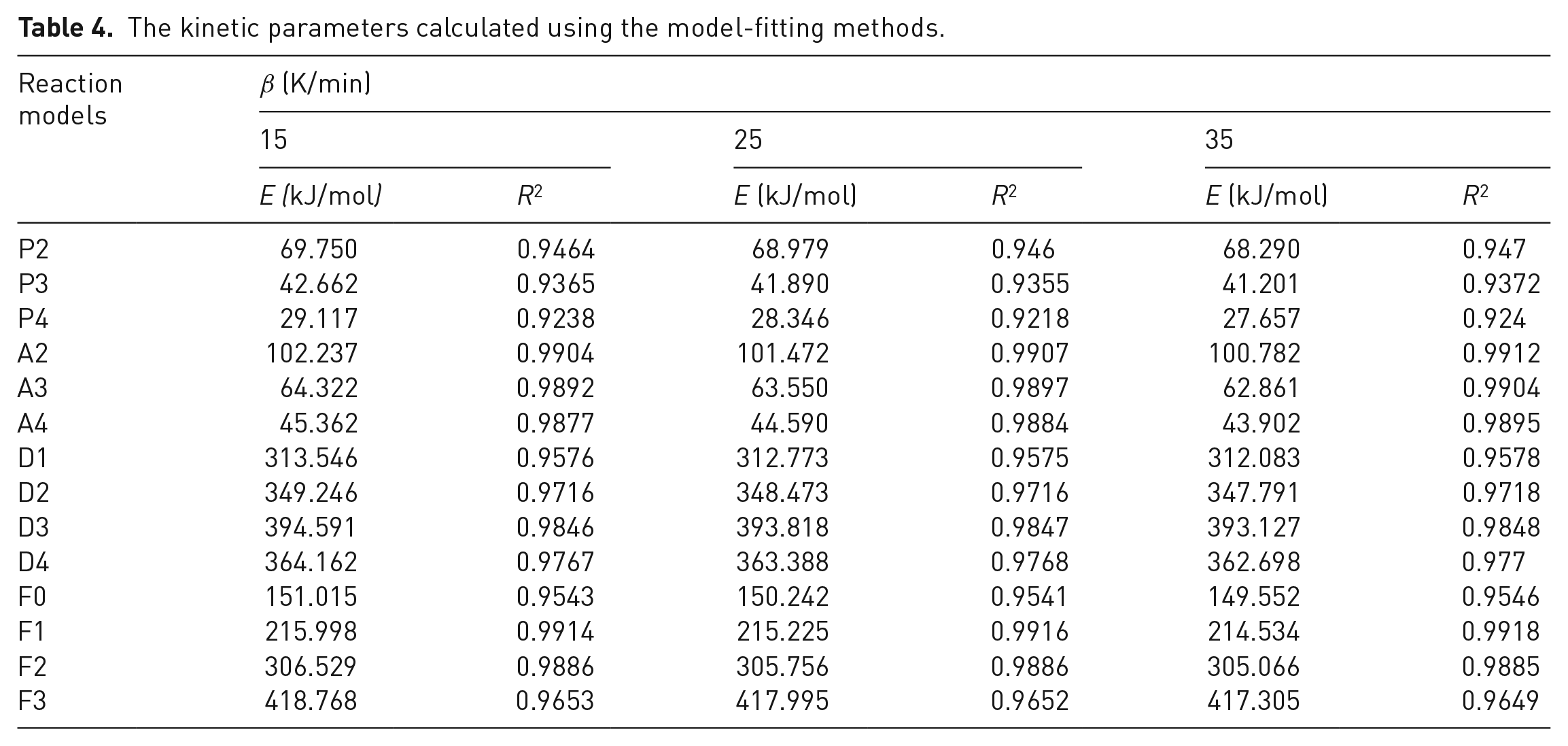
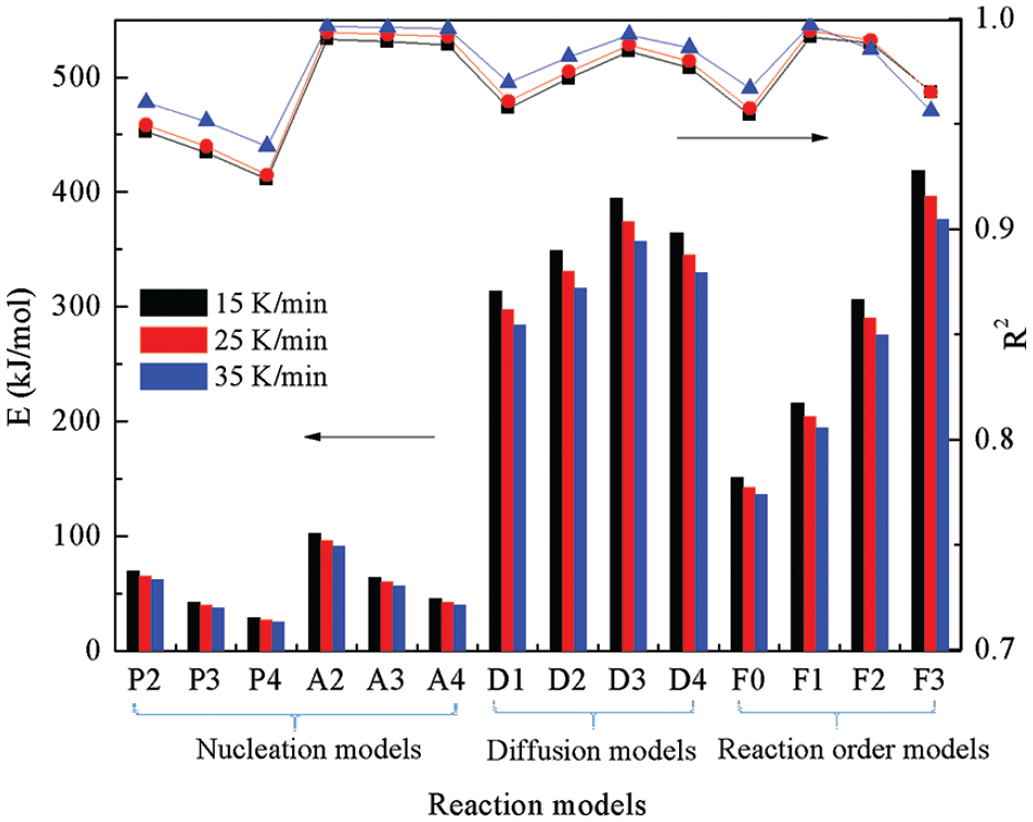
Comparison of kinetic parameters calculated from model-fitting methods.
Kinetic compensation effects
The kinetic compensation effect is usually used to characterize the dependence of
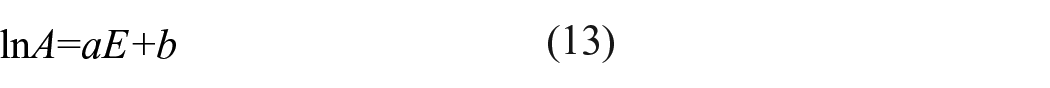
where
The F1 mechanism was selected for determining the pre-exponential factors as a function of conversion to validate the kinetic parameter. The activation energy calculated using the FWO method was more reliable and was used in this section. Substituting
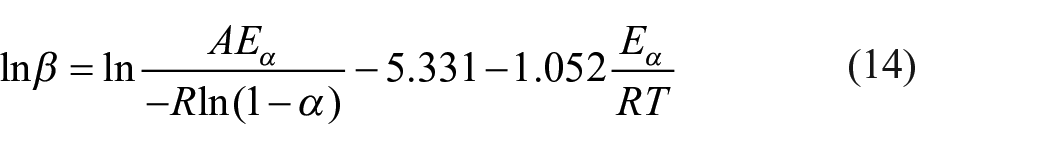
From the
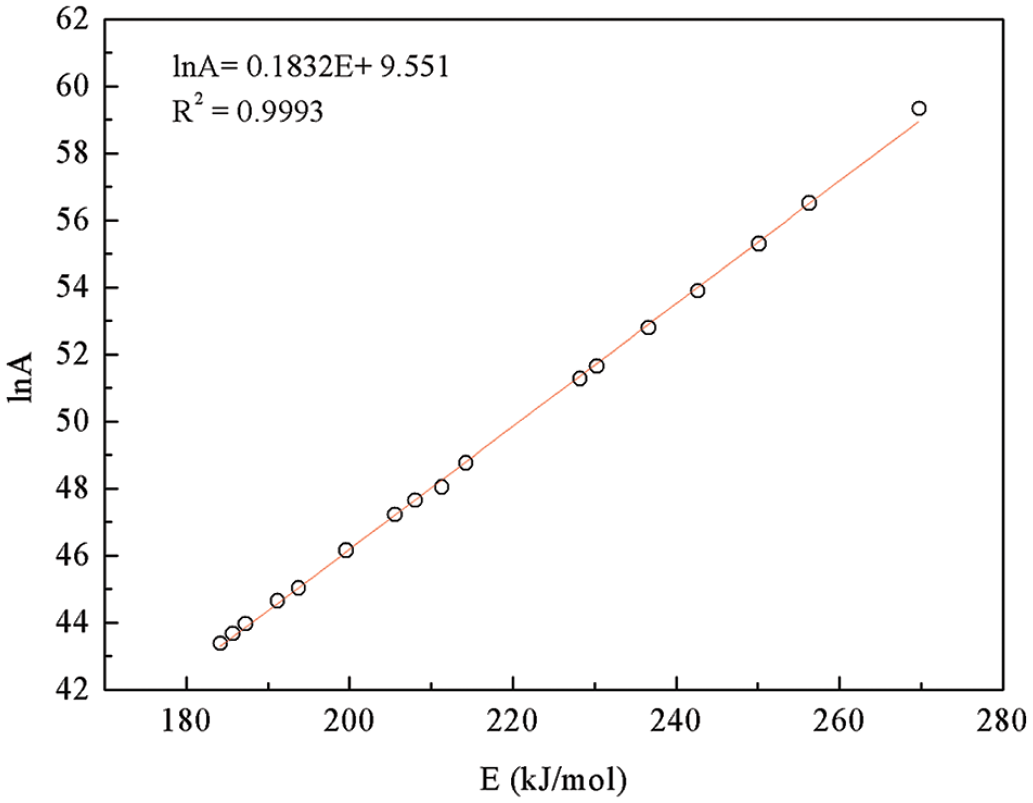
Compensation plot of ln
Conclusions
In this work, a series of TG experiments was conducted to investigate the pyrolysis kinetics of plastic waste. The TG analysis revealed that the decomposition process can be divided into three stages with corresponding weight losses of 2.80–3.02%, 94.45–95.11% and 0.04–0.16%, respectively. The maximum peak temperatures were observed at 423, 433 and 439°C for three heating rates in the second stage. Based on the TG analysis, the activation energy and linear correlation coefficient were determined at different conversion rates using four model-free methods. The kinetic parameters calculated using the Friedman method displayed a similar trend to those determined by the KAS method. The same profiles were also observed for the FWO and Starink methods. However, the
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Zhejiang Provincial Natural Science Foundation of China (Grant no. LY19B070008) and the National Natural Science Foundation of China (Grant no. 51911530460 and 51606055).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
