Abstract
Background
Sleep problems are common in older adults and associated with higher frailty risk.
Objective
To examine whether pain and social isolation explain the longitudinal association between sleep problems and frailty.
Methods
Data were from 925 adults aged 65+ in the 2013–2014 National Aging and Health Trends Study. Sleep problems were self-reported, and frailty was assessed using Fried’s phenotype. Mediators included pain, activity-limiting pain, number of pain locations, and social isolation. Multivariable logistic regression and Karlson–Holm–Breen method were used.
Results
Baseline pain and social isolation partially mediated the association between sleep problems and frailty at one-year follow-up, with pain accounting for 13.29%, activity-limiting pain 10.24%, pain locations 19.84%, and social isolation 12.77%, and pain and social isolation combined over 24% of the association.
Conclusions
Pain and social isolation explain part of the association between sleep problems and frailty, informing efforts to reduce sleep-related frailty risk over time.
• Sleep problems are associated with higher odds of pre-frailty and frailty over one year among older adults. • Physical pain (any pain, activity-limiting pain, multiple pain locations) and social isolation are linked with frailty risk. • Pain and social isolation partially mediate the sleep–frailty association, explaining 10–24% of the association.
• Interventions targeting sleep health, pain management, and social engagement may help delay frailty progression in older adults. • Findings support screening for sleep problems, pain, and social isolation in gerontological practice to identify high-risk individuals. • Results can inform policies and programs aimed at promoting healthy aging by addressing both physical and social determinants of frailty.What This Paper Adds
Applications of Study Findings
Background
Sleep is essential to health and aging. It affects many bodily functions, including hormone balance, immune strength, muscle and energy restoration, memory, and heart rate regulation (Itani et al., 2017; Van Cauter et al., 2008; Zielinski et al., 2016). Unfortunately, sleep problems are common and become more recurrent with advancing age (Foley et al., 2004; Kurina et al., 2013). About 50% of older adults report sleep hassles (Foley et al., 2004), and 12–20% are diagnosed with insomnia, meaning they regularly struggle with poor or insufficient sleep (Miner & Kryger, 2017; D. Patel et al., 2018). Importantly, sleep problems in later life are not limited to insomnia. Older adults also may experience short or long duration sleep, poor sleep quality, irregular sleep patterns, and poor daytime alertness, often occurring simultaneously (Stepnowsky & Ancoli-Israel, 2008).
At the same time, physical frailty is a growing concern in aging populations. Frailty is a medical condition where the body loses strength, energy, and its ability to recover from physiological stress. Frail individuals typically report muscle weakness, slow walking, low physical activity, unintended weight loss, and exhaustion (Fried et al., 2001). Frailty is associated with higher risk of falls, fractures, hospitalizations, disability, dementia, and death (C.-L. Chen et al., 2019; Fried et al., 2001; Kojima, 2015, 2017, 2018). That said, frailty can be prevented (Travers et al., 2019) and may also be reversible (Travers et al., 2019), especially if modifiable risk factors are identified early. Evidence based on cross-sectional and longitudinal studies suggests that sleep may be one such modifiable risk factor (Balomenos et al., 2021; Baniak et al., 2020; Muhammad et al., 2024; Sun et al., 2020). However, the processes underlying the sleep-frailty association remain largely unknown.
Potential Pathways Linking Sleep With Frailty
In this study, we examine two potential pathways. One is physical pain. Sleep problems and chronic pain often co-occur. Poor and insufficient sleep are risk factors for chronic pain (Afolalu et al., 2018), and experimental and longitudinal evidence suggests that sleep disturbances can increase pain sensitivity and symptoms over time (Sivertsen et al., 2015). Sleep disturbances are associated with lower pain thresholds, increasing susceptibility to pain stimulation and spontaneous pain symptoms (Finan et al., 2013). This may reflect disruptions in the body’s natural pain regulation system, including alterations in neurotransmitters like serotonin and dopamine, HPA axis dysfunction, and heightened inflammation (Haack et al., 2020; Koffel et al., 2019). For example, poor sleep is linked to higher levels of proinflammatory cytokines, which in turn adds to pain and tiredness—key frailty components. Because pain is also linked to reduced mobility, lower activity, and exhaustion, it may help account for the association between sleep problems and later frailty.
We also examine “social” pain, measured as social isolation, as a factor associated with the sleep-frailty relationship. Sleep-deprived individuals often feel more tired, angry, irritable, emotionally reactive, and are cognitively compromised (Audigier et al., 2023; Díaz-Leines et al., 2017)—factors that may de-motivate social interactions, social activities, and social engagement. Persons with poor sleep also report lower perceived relationships quality than peers who sleep well (Audigier et al., 2023). Research finds that sleep-deprived persons tend to retreat socially and perceive others as less friendly or welcoming (Ben Simon & Walker, 2018). Moreover, even those who sleep well report feeling lonelier when interacting with sleep-deprived individuals, which may be indicative of a “spreadable” effect of social isolation (Ben Simon & Walker, 2018). In fact, in their laboratory study, Ben Simon and Walker (2018) found that just one night of poor sleep can cause others to avoid a person, which may lead to further feelings of loneliness. Over time, social withdrawal is associated with adverse physical health, including higher inflammation, reduced physical activity, and poorer health management (Hawkley & Cacioppo, 2010; Van Bogart et al., 2022)—all risk factors for frailty.
The Present Study
While prior studies have shown that poor sleep is linked to frailty, very few have explored factors relevant to the sleep-frailty association. Accordingly, we examine whether pain and social isolation help account for the longitudinal association between sleep problems and frailty using statistical mediation models. Per our knowledge, ours is the first to longitudinally examine physical and social pain as potential mediators of the sleep-frailty association. We rely on multiple indicators of sleep, including dissatisfaction with sleep, poor daytime alertness, suboptimal sleep timing, inefficiency and inadequate sleep duration. Most prior studies use one or two measures of sleep, mostly sleep duration, sleep quality or insomnia symptoms (Pourmotabbed et al., 2020; Souza et al., 2025). While useful, single measures often fail to encompass the full extent of sleep hassles that older persons encounter. Though self-reported, the inclusion of multiple sleep indicators is a strength of this study, and it allows us to capture a broader range of sleep-related difficulties that may be relevant for frailty risk. Moreover, by examining factors linked to the sleep-frailty association, our findings may help inform policy and interventions aimed at reducing the risk of frailty over time. In Figure 1, we present a conceptual diagram suggesting potential mediation of pain measures and social isolation in the association between sleep problems and frailty. Conceptual diagram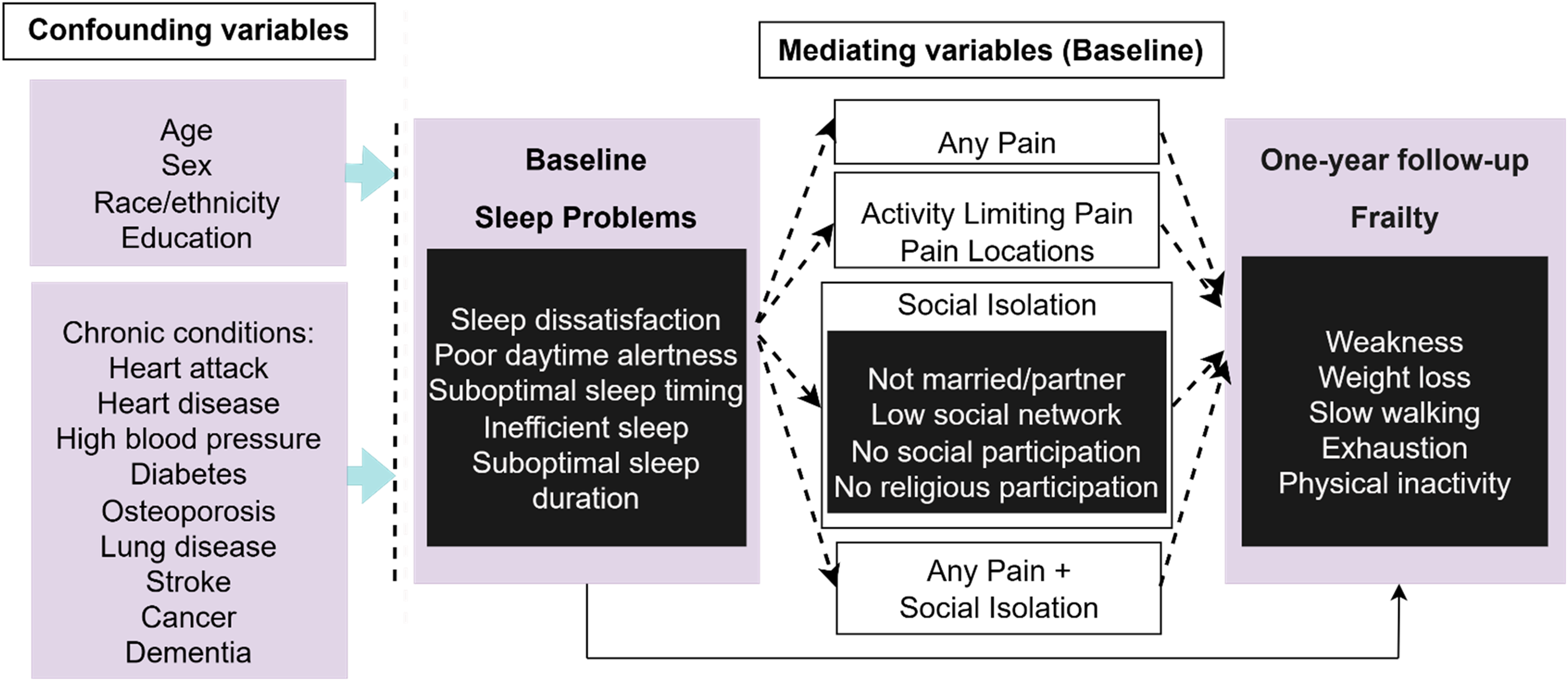
Methods
Data
This study is a secondary data analysis of the NHATS, a probability-based sample of U.S. Medicare beneficiaries aged 65+, representing approximately 96% of older adults in the United States and followed annually from 2011 to 2021 (Freedman & Kasper, 2019). We used data from waves 3 and 4 (2013–2014) of NHATS, during which approximately one-third of participants were randomly selected (n = 1844) to complete the sleep module and provided information on sleep measures. We excluded older adults residing in nursing homes or formal care facilities, as restricted access to social and physical activities may influence sleep, pain, and frailty differently than among community-dwelling older adults. We analyzed data from 925 older adults 65+ who self-completed the survey and had complete data on sleep problems, baseline and follow-up frailty, pain measures, social isolation, and chronic conditions.
Measures
Sleep Problems
Sleep problems at baseline were assessed using self-reported measures covering five key dimensions based on the SATED model proposed by Buysse: sleep satisfaction, daytime alertness, sleep timing, sleep onset latency, and sleep duration (Buysse, 2014). In each dimension, respondents were classified as having either “good” or “poor” sleep based on criteria established in previous studies (Furihata et al., 2017; S. Lee et al., 2022). Sleep dissatisfaction was defined as reporting fair, poor, or very poor overall sleep quality, and poor daytime alertness as having trouble staying awake during the day on some days, most days, or every day. Suboptimal sleep timing was defined as having a typical mid-sleep time earlier than 2:00 AM or at or after 4:00 AM. Sleep onset latency was defined as taking 30 min or longer to fall asleep, and suboptimal sleep duration as sleeping fewer than 6 hr or 9 or more hours per night. A composite sleep problem score (0–5) was created by summing poor sleep dimensions, with higher scores indicating greater sleep problems.
Physical Frailty
Frailty at baseline and one-year follow-up was assessed using the physical frailty phenotype on five components adapted from Fried’s criteria: exhaustion, low physical activity, weakness, slowness, and weight loss (Fried et al., 2001). Participants were classified as exhausted if they reported low energy or fatigue limiting activities, and as having low physical activity if they never walked for exercise or engaged in vigorous activities. Shrinking was defined as a BMI less than 18.5 kg/m2 or unintentional weight loss of 10 or more pounds in the past year. Slowness was determined by a walking speed test, with thresholds based on sex and height, while weakness was assessed using dominant hand grip strength, with thresholds varying by sex and BMI. Missing data for any of the components (due to safety concerns or inability to complete tests) were imputed with 0, allowing for up to two missing items. Participants were classified as non-frail (0), pre-frail (1–2), or frail (3–5).
Pain Measures at Baseline
Physical pain was assessed using a question asking whether participants had been bothered by pain in the previous month. In addition to its face validity, other studies have utilized this NHATS pain measure to estimate the effect of pain among U.S. older adults (K. V. Patel et al., 2013). Participants reporting pain were asked whether it limited their activities (yes/no) and to identify specific pain locations experienced during that time. Pain sites included back, hips, knees, legs, feet, hands, wrists, arms, shoulders, stomach, head, and neck, with each site counted as one point toward a total pain site score, mirroring prior studies (Rundell et al., 2025). Participants could also specify additional pain sites not included in the predefined list; these were counted as one additional site, yielding a total possible pain site score ranging from 0 to 13.
Social Isolation at Baseline
Social isolation was measured using a modified version of the Social Network Index, assessing three primary domains (Pohl et al., 2017): marital/partner status, family and friends, and social participation. The adapted criteria capture both structural and functional aspects of social connectedness. Structural aspects included having no spouse or partner, speaking with fewer than three people about important matters in the past year, and having no in-person visits with friends or family outside the household in the past month. Functional aspects included engagement in religious services, clubs or classes, recreational outings, and volunteer work within the past month. Although structural aspects are more stable than functional ones, both may serve as mediators. For example, poor sleep can gradually erode close relationships and social contacts (Gordon et al., 2021), which, together with diminished participation, may heighten frailty risk. Each indicator was coded 1 to indicate social isolation or activity and 0 otherwise, and the scores were summed ranging 0–7, with higher scores indicating greater social isolation. Details of the indicators are presented in Table S1.
Combined Pain and Social Isolation
To assess the joint contribution of physical pain and social isolation, a composite measure was created by standardizing the social isolation score (range: 0–7) and summing it with the binary indicator of physical pain (0–1). Higher values of this composite reflect greater co-occurrence of pain and social isolation.
Covariates
We included conceptually relevant covariates known to influence sleep and frailty, including age (in years), sex (male, female), education (less than high school, high school, more than high school), and race/Hispanic ethnicity (non- Hispanic White vs Hispanic and non-White). Although the sample comprises older adults, age was retained to capture residual variability within this group. Sensitivity analyses showed that including age did not meaningfully change the findings, suggesting minimal risk of overadjustment. Also included were chronic conditions based on self-reports of physician-diagnosed high blood pressure, heart attack, heart disease, arthritis, osteoporosis, diabetes, lung disease, stroke, cancer, dementia, or Alzheimer disease. Arthritis was excluded due to potential collinearity with physical pain. The chronic conditions count ranged from 0–9.
Statistical Analysis
Data were analyzed using Stata version 18 (StataCorp, 2023, p. 16). Descriptive analyses for demographic and health characteristics with weighted percentages at baseline were performed. Additionally, descriptive statistics were used to summarize the prevalence of pre-frailty and frailty at baseline. Multinomial logistic regression was used to model the association between sleep problems and pre-frail and frail categories, adjusting for baseline frailty to account for initial differences. We further conducted a longitudinal mediation analysis to examine whether baseline pain, activity-limiting pain, number of pain locations, and social isolation mediate the association between baseline sleep problems and frailty at one-year follow-up. Models were weighted using the NHATS wave 3 weights. Although both the exposure and mediators were measured at baseline, we conceptualized the mediators as potential pathways linking the exposure to the outcome (see the directed acyclic graph (DAG) in Figure S1). Conceptually, the simultaneous measurement of exposure and mediators is appropriate for this study, as pain or social isolation in response to poor sleep may occur immediately or proximally (Finan et al., 2013). However, given the concurrent measurement of exposure and mediators, causal interpretations should be made cautiously.
For the mediation analysis, the Karlson–Holm–Breen (KHB) method (Kohler et al., 2011) was employed. KHB is a method that enables decomposition of effects in nonlinear models while incorporating survey weights from nationally representative data (Kohler et al., 2011). Specifically, the KHB method compares a full model that includes the mediator(s) with a reduced model in which the mediator(s) are replaced by their residuals from a regression of the mediator(s) on the key independent variable, thereby accounting for the rescaling inherent in nonlinear probability models. This approach extends the decomposition framework of linear mediation models to settings with binary outcomes. It decomposes the total effect (the effect of independent variable on dependent variable without mediating variable) into direct (the effect of independent variable on dependent variable after controlling for mediating variable) and indirect effects (the effect of independent variable on dependent variable through the proposed mediating variables). The percentage of the total effect explained by the mediator was calculated on the log-odds scale as the ratio of the indirect effect to the total effect [ln(indirect effect OR)/ln(total effect OR) × 100], providing an estimate of the extent of mediation.
We estimated both unadjusted and fully adjusted models; the latter controlled for baseline frailty and covariates, including age, sex, education, race/ethnicity, and number of chronic conditions. We report mediation estimates from separate models for any pain, activity limiting pain, pain locations, and social isolation, including total, direct, and indirect effects along with the percentage mediated. An additional model examined the mediation effect of the combined measure of any pain and social isolation. Mediation analyses were conducted following AGReMA guidelines (H. Lee et al., 2021). The analyses assume no unmeasured confounding of the exposure–mediator, exposure–outcome, and mediator–outcome relationships, and correct model specification. Odds ratios (ORs) and 95% confidence intervals (CIs) are reported for total, direct, and indirect effects.
Results
Characteristic Distribution of the Sample by Baseline Frailty Status
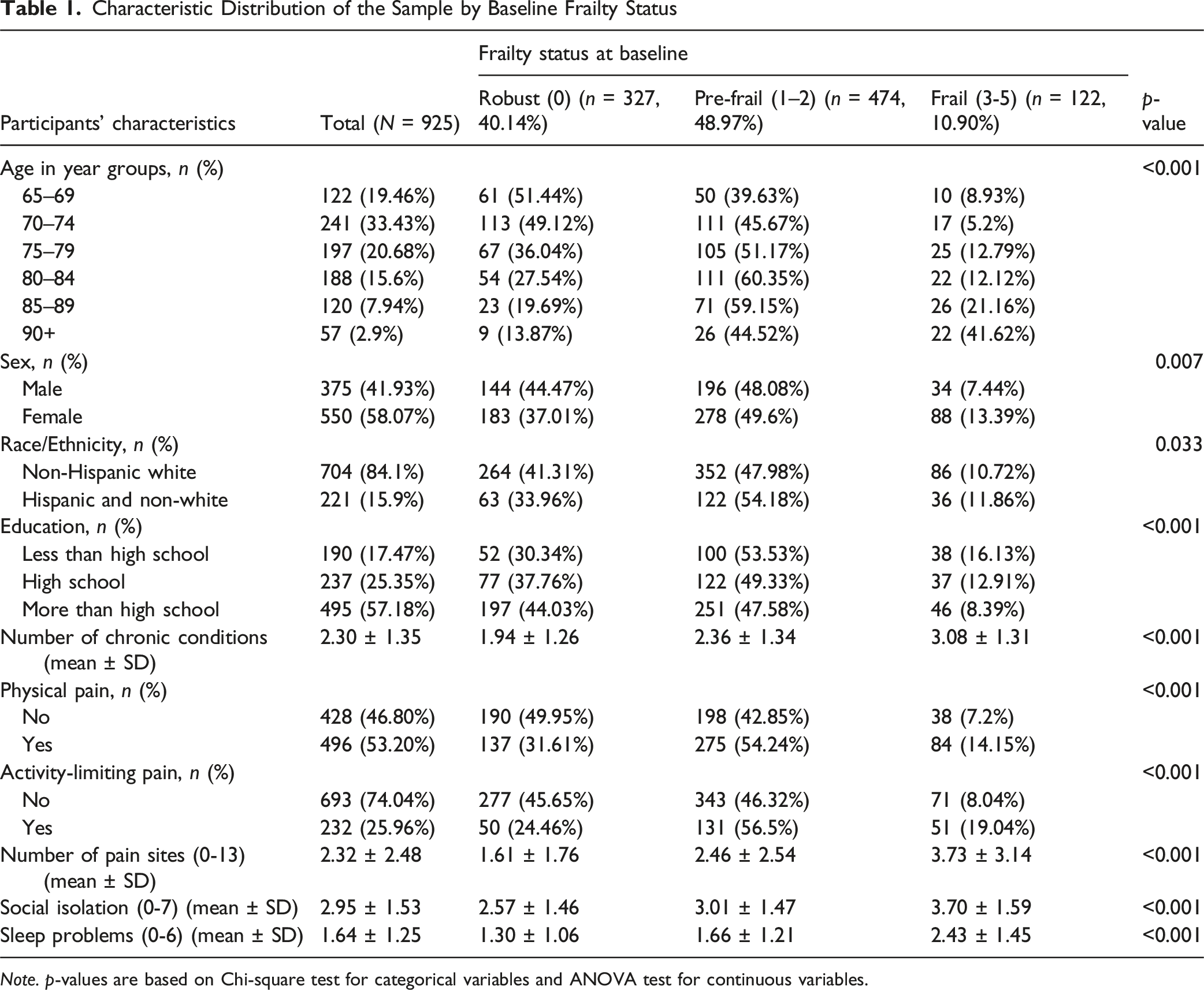
Note. p-values are based on Chi-square test for categorical variables and ANOVA test for continuous variables.
The prevalence of frailty among adults aged 65 years and older was 10.90%, with 48.97% prefrail, and 40.14% robust. Expectedly, higher frailty prevalence was observed among older persons, women and racial/ethnic minorities, persons with less than high school education, those with pain experiences, and social isolation. The extent of variation was striking, with frailty prevalence around twofold higher among those with pain experiences than those without (14.15% vs 7.2%), and more than fourfold higher among the oldest age group relative to the youngest (41.62% vs 8.93%).
Odds Ratios from Multinomial Logistic Regression Models for Pre-Frailty and Frailty by Exposure/Mediators
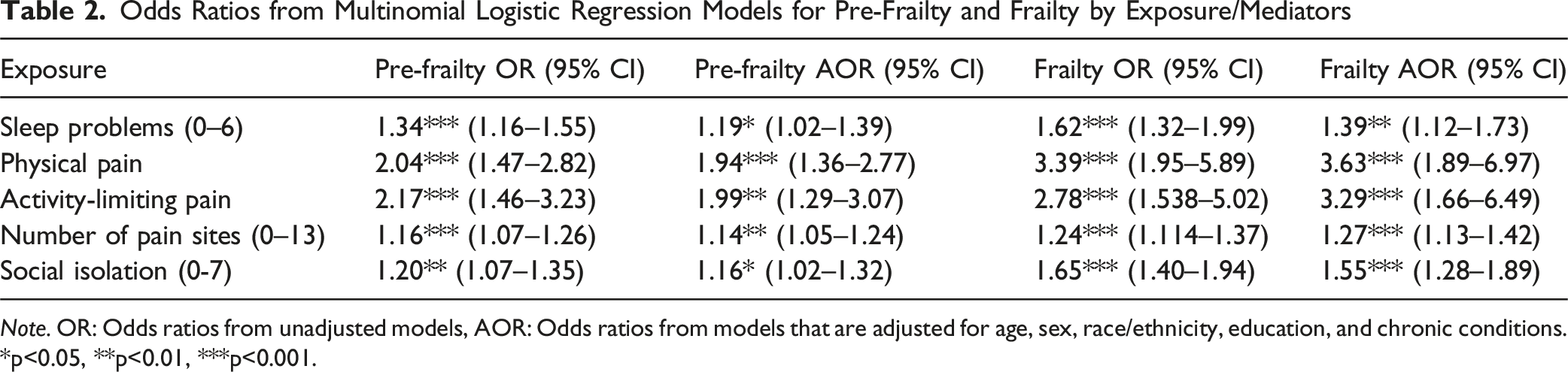
Note. OR: Odds ratios from unadjusted models, AOR: Odds ratios from models that are adjusted for age, sex, race/ethnicity, education, and chronic conditions. *p<0.05, **p<0.01, ***p<0.001.
Odds Ratios from Unadjusted and Adjusted Path Decomposition Models (KHB) for Frailty by Mediators
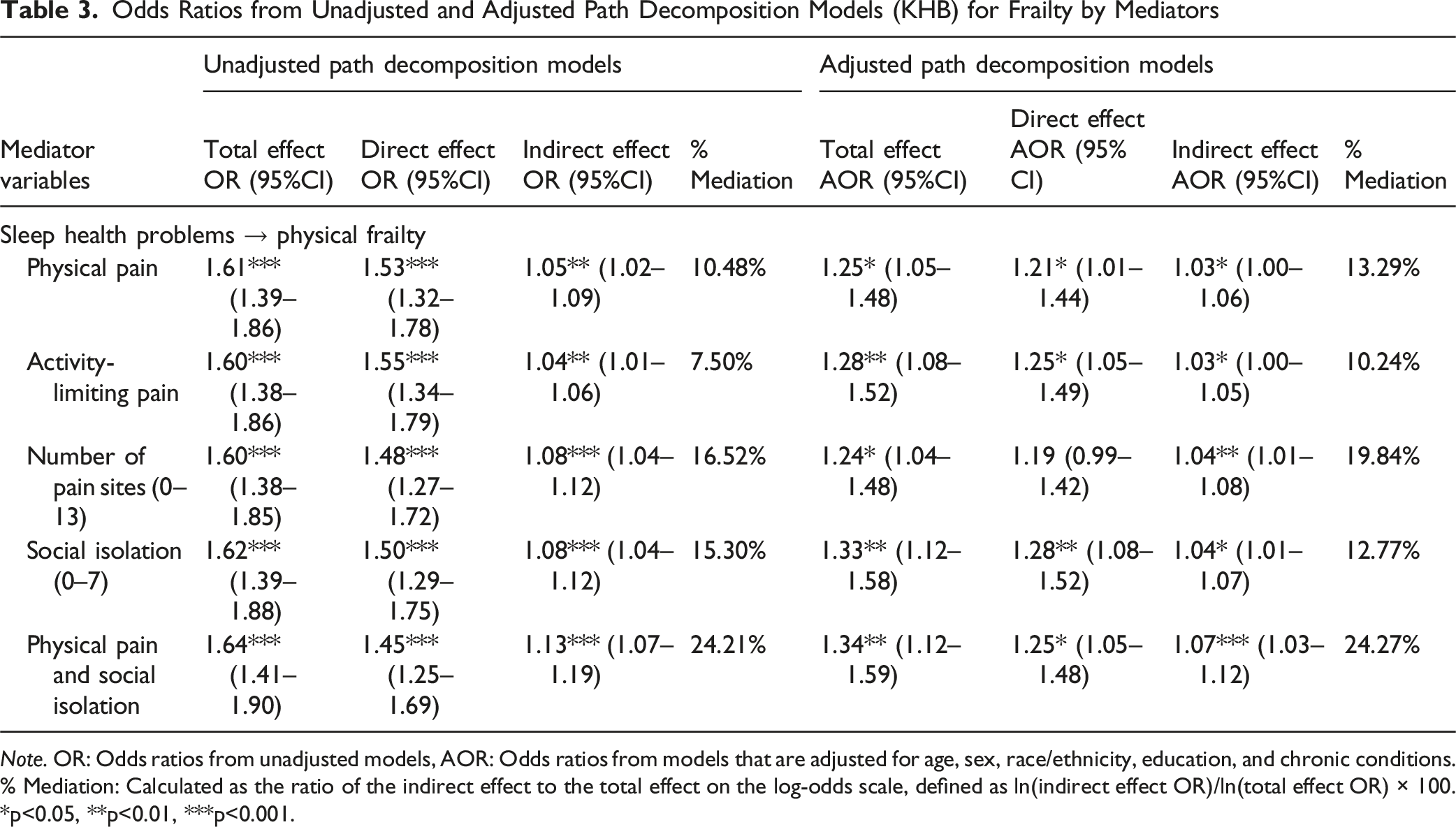
Note. OR: Odds ratios from unadjusted models, AOR: Odds ratios from models that are adjusted for age, sex, race/ethnicity, education, and chronic conditions. % Mediation: Calculated as the ratio of the indirect effect to the total effect on the log-odds scale, defined as ln(indirect effect OR)/ln(total effect OR) × 100. *p<0.05, **p<0.01, ***p<0.001.
Discussion
This study offers worthful insights into the association between sleep problems and physical frailty among older Americans, with physical pain, activity-limiting pain, number of pain locations, and social isolation acting as potential mediators. Upon considering several sociodemographic characteristics, chronic conditions, and baseline frailty status, older adults reporting more sleep problems had a higher likelihood of becoming pre-frail and frail over a one-year period. These patterns add to a growing body of work suggesting that sleep may be more than just a correlate; it may represent an important marker of risk for physiological debility with advancing age (Balomenos et al., 2021; Baniak et al., 2020; T.-Y. Chen et al., 2022; Muhammad et al., 2024; Sun et al., 2020). Cross-sectional and longitudinal studies have consistently indicated that sleep issues, including sleep disruptions, insufficient or protracted sleep, poor sleep quality, and insomnia are associated with frailty in later life.
Missing largely from this literature is the examination of factors that may help account for the sleep-frailty association. Our study, as such, is among the first to quantify the proportion of this association that may operate via presence of any pain, activity limiting pain, number of pain locations, and the experience of social isolation. Any pain experience, activity-limiting pain, and pain across multiple body locations, emerged as factors contributing to the sleep-frailty association. This fits with earlier research showing that poor sleep is linked to pain. (Aili et al., 2018), for example, noted that study participants with sleep issues were more likely to develop chronic widespread pain over time. In a study by Uhlig et al. (2018), insomnia was found to raise the risk of chronic musculoskeletal complaints, and chronic widespread musculoskeletal complaints in three or more parts of the body. Together, these findings are consistent with our observations over the one-year period examined in this study: Older adults who do not sleep well report for more widespread and disabling pain, which may increase frailty vulnerability by restricting activity (Reid et al., 2015), reducing strength (Leveille et al., 2001), and increasing depression (Tomás et al., 2023) and exhaustion (Norton et al., 2024).
We also found that the number of pain sites accounted for around 20% of the sleep-frailty association, more than any other individual mediator. Widespread pain is associated with higher risk of disability and mortality in later life (Butera et al., 2019; Leveille et al., 2001; Wade et al., 2016). This may be because widespread pain makes movement difficult, not only due to physical discomfort but also because of the emotional and social distress it causes (Butera et al., 2019). Over time, less movement and multi-site pain are associated with muscle weakness and balance problems (Evans, 2010), fatigue, and functional decline—all linked to frailty.
Additionally, although both general and activity-limiting pain mediated the sleep-frailty association, the strength of the mediation by activity-limiting pain was relatively weaker than that of general pain. While this may seem counterintuitive, general pain was more prevalent and may capture a fuller range of physical discomfort, even if not severe enough to constrain activity, potentially making it a more sensitive mediator in this sample. Also, general pain may reflect underlying chronic conditions or inflammation associated with frailty. Together, these patterns highlight the need to differentiate between types of pain when assessing frailty risk among older adults with sleep problems.
In addition to physical pain, we found that “social” pain—that is, social isolation—also partly accounted for the sleep-frailty link. Recent work has found social isolation linked with an elevated frailty risk in older adults (Zhang et al., 2025). Studies also have noted that social relationships can reduce mental distress and improve cognitive health among older adults with frailty (Mehrabi & Béland, 2024). Our findings extend this evidence by suggesting that sleep problems are associated with social withdrawal and factors linked to frailty risk (Krause et al., 2017). Even in experimental research, sleep-deprived individuals are perceived to be less socially appealing, leading others to avoid interacting with them (Ben Simon & Walker, 2018). Our findings resonate with previous research patterns, showing that social isolation accounted for nearly 13% of the sleep-frailty association. Patterns we observe are important given evidence that mortality risk is highest among frail older adults who also are socially isolated (Hoogendijk et al., 2020).
Limitations and Future Directions
Despite its meaningful contributions, this study has important limitations. First, primary variables, including sleep problems and pain, were self-reported. This may lead to issues of accurate recall or reporting bias. While self-report measures are typical in large surveys, future studies should consider objective assessments of sleep (e.g., actigraphy) and pain (e.g., clinical examination). Second, we cannot ascertain causal mechanisms using observational data. It is plausible that broader underlying health decline, such as increasing disease chronicity, inflammation, declining functional and mental acuity, may simultaneously compromise sleep and contribute to greater pain, social isolation, and the onset and progression of frailty. Third, reverse causality cannot be ruled out, as frailty may be associated with sleep troubles, greater pain, and increased social isolation via fatigue, reduced mobility, inflammation, and loss of autonomy (Soysal et al., 2016).
Fourth, differing time frames used to measure components of social isolation (e.g., past year vs. past month) may introduce temporal inconsistency, which could bring into question the precision of the mediation estimates. Fifth, although the one-year follow-up allows detection of early frailty changes while minimizing attrition, particularly among high-risk older adults as shown in previous studies (Rasmussen et al., 2024; Tay et al., 2023), it may limit the assessment of longer-term frailty progression. Sixth, while we considered several important mediators in this study, future research should explore biological mediators, including inflammatory markers such as C-reactive protein and interleukin-6, which are found linked to sleep and frailty (Pourmotabbed et al., 2020). Lastly, studies that more clearly separate the timing of sleep, mediators, and frailty (e.g., sleep at baseline, mediators at follow-up, and frailty at subsequent follow-up) and that utilize non-observational data designs may help clarify causal and temporal inferences related to the pain-frailty relationship.
Conclusions
In this inquiry, we observe that sleep problems are significantly associated with frailty among older Americans, with pain—especially activity-limiting and multi-site pain—and social isolation accounting for some of this association. Though with caution, these findings pinpoint the relevance of integrated interventions that address not only sleep, but also its physical and social repercussions. As populations continue to age, recognizing and disrupting these pathways may help reduce disability and dependence in later life.
Supplemental Material
Supplemental material - Pain and Social Isolation as Mediators of the Longitudinal Association Between Sleep Problems and Frailty in U.S. Older Adults
Supplemental material for Pain and Social Isolation as Mediators of the Longitudinal Association Between Sleep Problems and Frailty in U.S. Older Adults by T. Muhammad, Manacy Pai, David M. Almeida and Soomi Lee in Journal of Applied Gerontology
Footnotes
Ethical Considerations
This study used secondary data from the NHATS study that was approved by the Johns Hopkins Bloomberg School of Public Health Institutional Review Board.
Consent to Participate
Informed consent was obtained from all individual participants in NHATS.
Funding
The authors disclosed no financial support for the research, authorship, and/or publication of this article. Dr Soomi Lee reports she received funding support from National Institutes of Health (R01HL163226, PI: Lee), outside this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
