Abstract
Purpose
To evaluate and estimate the association between Time and Go (TUG) test performance and mild cognitive impairment (MCI) or Alzheimer’s disease (AD) in older adults.
Methods
A systematic review and meta-analysis were conducted to synthesize data from studies examining the relationship between TUG test score and AD or MCI in adult subjects. The search strategy was applied to MEDLINE (OVID), Web of Science, Scopus, LILACS, and the Cochrane Central Register of Controlled Trials (CENTRAL). Studies were selected based on predefined inclusion criteria. For meta-analysis, effect sizes were pooled using a random-effects model to account for heterogeneity across studies. Heterogeneity was assessed using the I2 statistic, and publication bias was evaluated through funnel plot analysis. Sensitivity analyses were performed to test the robustness of the findings.
Results
A total of 25 studies were included in this review, of which 15 were incorporated into the meta-analysis. Participants with AD (4.22 [95% CI 2.76–5.69 s], p < 0.00001) and those with MCI (1.83 [95% CI 1.03–2.57 s], p < 0.00001) exhibited significantly longer times in the simple TUG test compared to healthy controls. The Dual-Task TUG test showed a stronger association with AD in the studies included in the analysis. AD participants demonstrated significantly longer test times (12.28 [95% CI 6.56–18.0 s], p < 0.0001) compared to cognitively normal controls.
Conclusions
This review highlights a potential association between TUG completion time and cognitive impairment in individuals with AD and MCI. The TUG test shows promise as a tool for the early identification and screening of cognitive decline and AD.
• Quantifies the association between TUG performance and cognitive impairment in older adults. • Identifies that dual-task TUG better distinguishes individuals with MCI and AD from healthy controls. • Supports the use of TUG as a simple and accessible tool for early detection of cognitive decline.
• The TUG test can be applied in both clinical and community settings as an early screening tool to identify older adults at risk for MCI or AD. • Beyond screening, it may help clinicians monitor changes in motor–cognitive function over time and can be incorporated into routine geriatric evaluations alongside cognitive tests to improve early detection and guide more effective treatment strategies.What this paper adds
Applications of study findings
Introduction
Alzheimer’s disease (AD) is one of the most prevalent neurodegenerative disorders among older adults worldwide. Currently, more than 55 million people live with some form of dementia, with approximately 60% of cases attributed to AD (Gustavsson et al., 2023). Aging is one of the primary risk factors for the development of this disease, and with increasing life expectancy, its prevalence is projected to rise significantly over the next 30 years, particularly in low- and middle-income countries (Monteiro et al., 2023). This growing burden positions AD as a significant global public health challenge.
Alzheimer’s disease is characterized by a progressive decline in functions related to memory processing, language, reasoning, executive functions, and motor task execution (Tiwari et al., 2019), primarily due to cellular degeneration caused by the abnormal accumulation of beta-amyloid (Aβ) and tau proteins in cortical areas responsible for these functions (Abubakar et al., 2022). In recent years, various studies have shown that although cognitive decline is one of the most prominent clinical signs of the disease, motor function impairment appears to be a significant risk factor for the development of AD (Buchman & Bennett, 2011) and may even precede the onset of cognitive symptoms (Montero-Odasso et al., 2020; Rudd et al., 2023). Motor impairment in AD can result from damage to the integrity of gray matter in motor-related brain regions, damage to white matter tracts connecting these gray matter areas, or a combination of both (Andrade-Guerrero et al., 2024).
Several studies have reported that motor function decline tends to be more pronounced in individuals with cognitive impairment compared to cognitively healthy adults of the same age. Lower levels of motor functions, such as muscle strength, gait, and dexterity, have been associated with an increased risk of AD and MCI (Koppelmans et al., 2020). In particular, gait pattern alterations have been observed in patients with AD and MCI, including reduced gait speed, shorter step length, and increased stance phase duration, as previously reported in various studies (Beauchet et al., 2008). The motor function thus appears to be an essential clinical symptom in the development and progression of AD.
The Timed Up and Go (TUG) test is a reliable and valid tool for assessing motor function in older adults in both clinical and home settings, as it evaluates lower limb function, balance, and gait speed (Christopher et al., 2021). Poor TUG performance has been linked to a higher risk of dementia and all-cause mortality in community-dwelling older adults (Chung & Byun, 2023). Additionally, studies have shown that total TUG time is linearly correlated with cognitive function scores, such as the Mini-Mental State Examination (MMSE). They may serve as a valuable predictor for the onset and progression of dementia (da Costa Teixeira et al., 2024; Mirelman et al., 2014). However, the TUG test is not specific to AD, and motor performance assessments exclusively designed for individuals with AD remain limited.
Most available tools are adapted from studies on healthy older adults or individuals with other forms of mild-to-moderate dementia (Åberg et al., 2023). Consequently, the use of motor assessment tests in AD patients represents a growing area of research with the potential to identify and evaluate motor deficits at different stages of the disease. Although the TUG test is not specific to AD, a focused review could be particularly valuable at this time because motor impairments might precede cognitive decline, and TUG may provide an accessible tool for the early detection of AD-related deficits. Given the recent increase in research linking mobility and cognition, synthesizing the evidence on TUG in AD could help clarify its potential role in clinical and research settings. This systematic review and meta-analysis aim to synthesize current evidence on the association between TUG performance and AD or MCI, estimate pooled effects across studies, and discuss its potential clinical applications as a screening tool for cognitive decline.
Methods
This review followed the recommendations of the Cochrane Collaboration and the guidelines outlined in the Cochrane Handbook for Systematic Reviews (Higgins et al., 2023) and the PRISMA protocol (Matthew et al., 2021). No protocol for this review was registered.
Eligibility Criteria
We included experimental, quasi-experimental, cohort, case–control, and cross-sectional studies involving adults diagnosed with AD or MCI that met the following criteria: studies reporting the measurement of the TUG test in at least one of these populations and their controls and studies examining the correlation between TUG performance and cognitive function decline in adults with AD and/or MCI. Studies including mixed dementia populations were considered only when AD- or MCI-specific data could be isolated and analyzed.
Exclusion criteria included studies involving populations with other neurodegenerative diseases, populations at risk of developing AD, and samples associated with chronic diseases or conditions unrelated to the diagnoses of interest. Also, studies including participants with mobility limitations due to musculoskeletal conditions, traumatic injuries, or other non-neurodegenerative causes were excluded.
Information Sources
The literature search was conducted following Cochrane’s recommendations. Medical Subject Headings (MeSH), Descriptors in Health Sciences (DeCS), and related text words were used. The search covered all published literature in PubMed, Scopus, Web of Science, and LILACS, including records from their inception until August 25, 2024.
To ensure literature saturation, references from relevant articles were identified through database searches, thesis repositories, and Google Scholar. No time limits, setting restrictions, or language exclusions were applied for article selection. The complete search strategy for each database is provided in Appendix 1.
Study Selection
A double-masked review was conducted, in which two researchers independently screened the titles and abstracts of articles retrieved from database searches to determine their potential relevance. Eligibility criteria were applied during the full-text review, and consensus resolved disagreements. A third reviewer decided if a consensus could not be reached. Inter-rater agreement for study screening and data extraction was assessed, and discrepancies were resolved through discussion.
Data Collection
Data were collected by two researchers using a standardized form. Independently, they extracted the following information from each article: study design, geographic location, author names, title, objectives, inclusion and exclusion criteria, age, sex distribution, main findings, number of patients included, TUG test scores, and cognitive function assessment scores for each group.
For statistical analysis, the following data were extracted: means, standard deviations, standard errors, interquartile ranges, minimum and maximum ranges, and confidence intervals for inflammatory marker levels and cognitive function tests. All data were standardized to means and standard deviations, except for minimum and maximum ranges, following Cochrane’s recommendations.
Methodological Quality and Risk of Bias
The Newcastle–Ottawa Scale (NOS) was used to assess the methodological quality of cross-sectional studies (adapted version), case–control studies, and cohort studies, as recommended by the Cochrane Collaboration. The NOS consists of eight items classified into three dimensions, with a total score ranging from 0 to 9 points. Studies were categorized as low quality (0–2 points), moderate quality (3–5 points), or high quality (6–9 points) (Ma et al., 2020). Clinical trials were assessed using the PEDro scale, which evaluates randomization, blinding, and outcome processing. A PEDro score above 6/10 was considered “good” (de Morton, 2009). Quasi-experimental studies were assessed using the Joanna Briggs Institute (JBI) critical appraisal checklist, which includes nine items assessing causal and effect variables, participants, control groups, pre- and post-intervention measurements, participant follow-up, outcome measures, and statistical analysis (Barker et al., 2024). Discrepancies between authors during the quality assessment were resolved through discussion. If consensus was not reached, a third author was consulted.
Data Analysis
Statistical analyses were performed using SPSS version 29.0.2.0 and RevMan 5.3. A random-effects model was used to calculate weighted mean differences for each outcome and 95% confidence intervals. Heterogeneity was assessed using the I2 test, with 25%, 50%, and 75% indicating low, moderate, and high heterogeneity, respectively. Publication bias was examined using a funnel plot to detect potential information or publication biases. A sensitivity analysis was conducted by removing weighted studies and recalculating effect estimates to identify differences.
Results
A total of 362 studies were identified through database searches. After removing duplicates, 85 studies were screened by title and abstract. Of these, 26 were excluded for being systematic reviews, letters to the editor, intervention protocols, or irrelevant to the topic of interest. Subsequently, 59 full-text studies were assessed, with 34 excluded for not meeting the inclusion criteria. Ultimately, 25 studies (Ansai et al., 2017, 2018; Borges Sde et al., 2015; Bramell-Risberg et al., 2005; Caliskan et al., 2023; Cedervall et al., 2012; Fujisawa et al., 2017; Gonçalves et al., 2018; Gras et al., 2015; Harper et al., 2020; Liu et al., 2023; McGough et al., 2011; Muir-Hunter et al., 2015; Mumic de Melo et al., 2019; Nadkarni et al., 2009; Ng et al., 2022; Nielsen et al., 2018; Nilsson et al., 2021; Parvin et al., 2020; Ries et al., 2009; Seifallahi et al., 2022; Sobol et al., 2016; Souza et al., 2022; Yao et al., 2012; Yoon et al., 2020; Åhman et al., 2020) were included in this review (Figure 1). Flowchart of Study Selection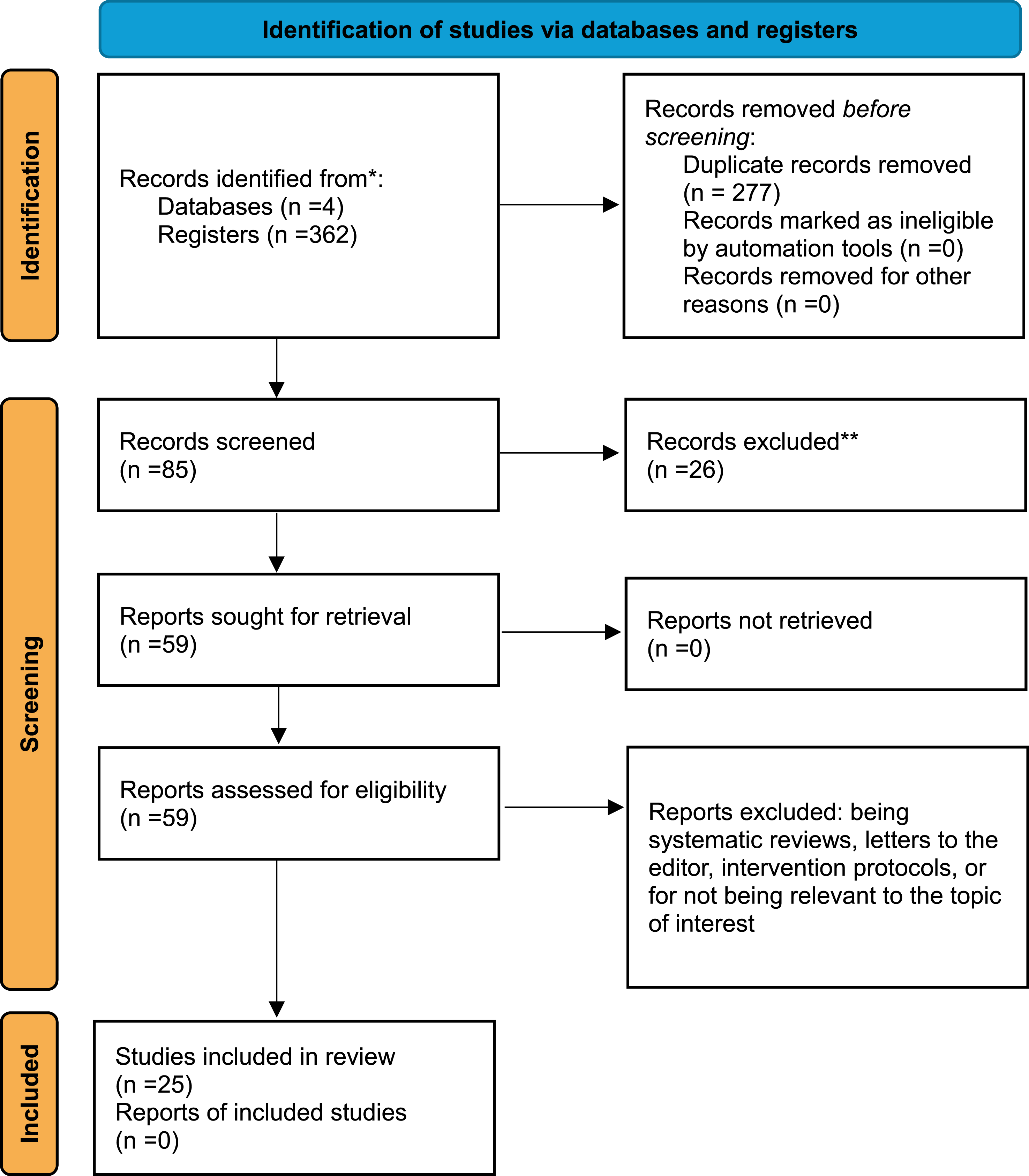
Characteristics of Excluded Studies
The excluded articles did not focus on the intervention or outcome of interest. Additionally, letters to the editor, systematic/narrative reviews, animal studies, studies involving pediatric populations, individuals with comorbidities, other types of dementia, Down syndrome, and other neurodegenerative disorders were excluded.
Characteristics of Included Studies
The 25 studies included in this review were published between 2005 and 2024. Among them, 17 were cross-sectional studies (Ansai et al., 2017; Borges Sde et al., 2015; Bramell-Risberg et al., 2005; Cedervall et al., 2012; Fujisawa et al., 2017; Gras et al., 2015; Liu et al., 2023; McGough et al., 2011; Muir-Hunter et al., 2015; Mumic de Melo et al., 2019; Nadkarni et al., 2009; Nilsson et al., 2021; Ries et al., 2009; Seifallahi et al., 2022; Sobol et al., 2016; Yoon et al., 2020; Åhman et al., 2019), 5 were prospective cohort studies (Ansai et al., 2018; Gonçalves et al., 2018; Harper et al., 2020; Nielsen et al., 2018; Souza et al., 2022), 1 was a case–control study (Caliskan et al., 2023), 1 had a quasi-experimental design (Yao et al., 2012), and 1 was a randomized controlled trial (Parvin et al., 2020). Geographically, the studies were conducted in Brazil (6 studies), North America (7 studies), Asia (2 studies), the Middle East (3 studies), Europe (6 studies), and Oceania (1 study) (Table S1, S2).
TUG and Cognitive Assessment of Studies
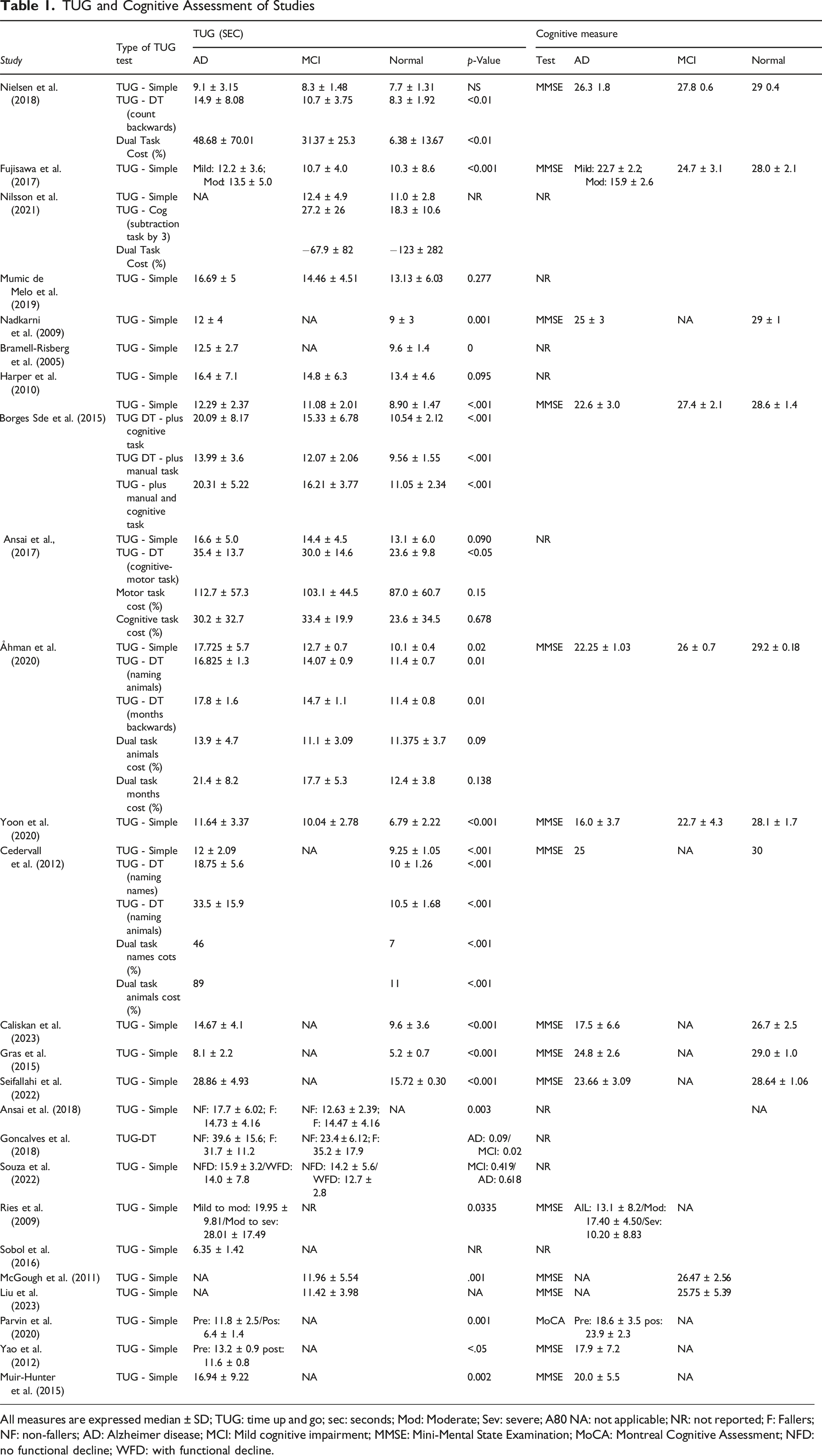
All measures are expressed median ± SD; TUG: time up and go; sec: seconds; Mod: Moderate; Sev: severe; A80 NA: not applicable; NR: not reported; F: Fallers; NF: non-fallers; AD: Alzheimer disease; MCI: Mild cognitive impairment; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; NFD: no functional decline; WFD: with functional decline.
Methodological Quality
The 17 cross-sectional studies included in this review had an average quality score of 6, with 76% classified as having good or high methodological quality. The 5 cohort studies had an average score of 6, with 80% demonstrating good or high quality. The case–control study (6/9) and the randomized controlled trial (8/11) were also considered to have good methodological quality. The quasi-experimental study was evaluated using the JBI tool, scoring 5/9, which the research team deemed as a moderate risk of bias. The complete methodological quality assessment for all studies is presented in Tables S3, S4, S5, and S6.
Evaluation of the Timed Up and Go (TUG) Test
Two types of TUG tests were assessed in the studies: Simple TUG—performed without additional tasks—was reported in 24 out of the 25 included studies, and Dual-Task TUG (TUG-DT)—combining the standard test with a cognitive, motor, or cognitive-motor task—was reported in 7 studies (Ansai et al., 2017; Borges Sde et al., 2015; Cedervall et al., 2012; Gonçalves et al., 2018; Nielsen et al., 2018; Nilsson et al., 2021; Åhman et al., 2019). Among these, 5 (Cedervall et al., 2012; Gonçalves et al., 2018; Nielsen et al., 2018; Nilsson et al., 2021; Åhman et al., 2019) used cognitive tasks, 1 (Borges Sde et al., 2015) assessed cognitive and motor tasks separately, and 1 (Ansai et al., 2017) incorporated a combined cognitive-motor task. Most studies evaluating TUG-DT used cognitive tasks such as counting numbers or naming animals.
All studies with control groups, except one (Nielsen et al., 2018), reported significantly longer TUG times in participants with AD and MCI compared to cognitively normal controls. Studies that assessed both Simple TUG and TUG-DT showed a significant increase in completion time when an additional cognitive or motor task was incorporated. However, De Melo Borges et al. found that a cognitive dual-task condition resulted in longer completion times across all groups (AD, MCI, and controls) than a manual dual-task condition. Additionally, Ansai et al. was the only study to include a combined cognitive-motor dual task, reporting a significant increase in completion time across all three groups compared to Simple TUG.
Among studies without control groups, three studies (Ansai et al., 2018; Gonçalves et al., 2018; Liu et al., 2023) reported longer TUG times in AD participants compared to MCI participants, while one study (Souza et al., 2022) found no significant differences between the groups. This review’s only randomized controlled trial showed an improvement in Simple TUG times following a physical/cognitive exercise intervention in AD patients (Table 1).
Cognitive Function Assessment
Cognitive function was reported in 16 (Borges Sde et al., 2015; Caliskan et al., 2023; Cedervall et al., 2012; Fujisawa et al., 2017; Gras et al., 2015; Liu et al., 2023; McGough et al., 2011; Muir-Hunter et al., 2015; Nadkarni et al., 2009; Nielsen et al., 2018; Parvin et al., 2020; Ries et al., 2009; Seifallahi et al., 2022; Yao et al., 2012; Yoon et al., 2020; Åhman et al., 2019) out of the 25 studies included in this review. All studies used the Mini-Mental State Examination (MMSE) as the primary measure of cognitive function, except for one study, which reported the Montreal Cognitive Assessment (MoCA). Additionally, six studies included supplementary cognitive assessments, such as the Clock Drawing Test, Verbal Fluency Test, Trail Making Test A (TMT-A), and Addenbrooke’s Cognitive Examination (ACE) score.
Association Between TUG and Alzheimer’s Disease
Of the 25 studies, 15 (Ansai et al., 2017; Borges Sde et al., 2015; Bramell-Risberg et al., 2005; Caliskan et al., 2023; Cedervall et al., 2012; Fujisawa et al., 2017; Gras et al., 2015; Harper et al., 2020; Mumic de Melo et al., 2019; Nadkarni et al., 2009; Nielsen et al., 2018; Nilsson et al., 2021; Seifallahi et al., 2022; Yoon et al., 2020; Åhman et al., 2019) were included in the meta-analysis. Means and standard deviations of TUG completion time (in seconds) were extracted for statistical analysis using a random-effects model. The Simple TUG completion time for patients with AD was reported in 14 studies and compared with healthy controls. The analysis revealed that AD patients had significantly longer TUG times (p < 0.00001), with a mean difference of 4.22 seconds (95% CI: 2.76–5.69 s) compared to healthy controls (Figure 2). Analysis of TUG Score in Subjects With Alzheimer’s. Meta-Analysis Plot Summarizing the Effect Sizes (With 95% Confidence Intervals) of Levels of TUG Score in Alzheimer’s Subjects. Each Horizontal Line Represents an Individual Study, With the Square Indicating the Effect Size and the Line Representing the Confidence Interval. The Square Size Reflects the Study’s Weight in the Meta-Analysis. The Diamond at the Bottom Represents the Pooled Effect Size and Confidence Interval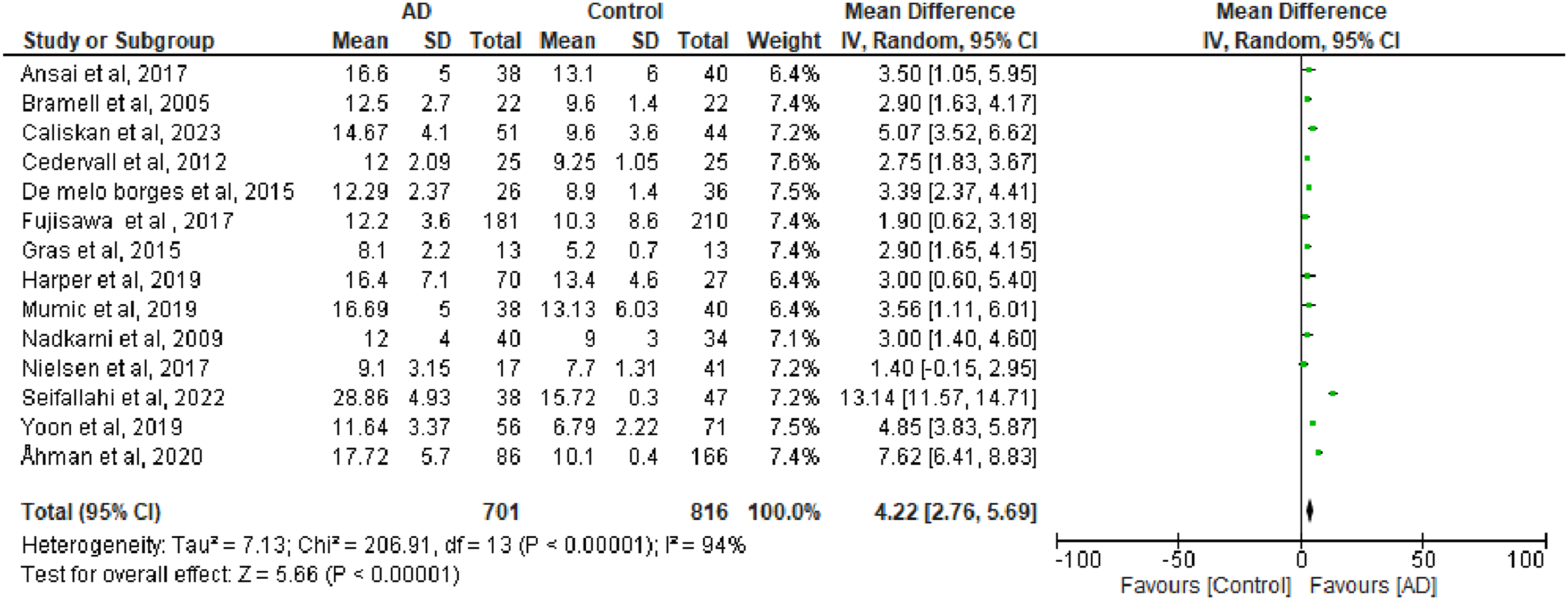
Nine studies reported the Simple TUG completion time for MCI participants. The analysis showed that individuals with MCI had significantly longer TUG times (p < 0.00001), with a mean difference of 1.83 seconds (95% CI: 1.03–2.57 s) compared to healthy controls (Figure 3). Analysis of TUG Score in Subjects With Mild Cognitive Impairment (MCI). Meta-Analysis Plot Summarizing the Effect Sizes (With 95% Confidence Intervals) of Levels of TUG Score in MCI’s Subjects. Each Horizontal Line Represents an Individual Study, With the Square Indicating the Effect Size and the Line Representing the Confidence Interval. The Square Size Reflects the Study’s Weight in the Meta-Analysis. The Diamond at the Bottom Represents the Pooled Effect Size and Confidence Interval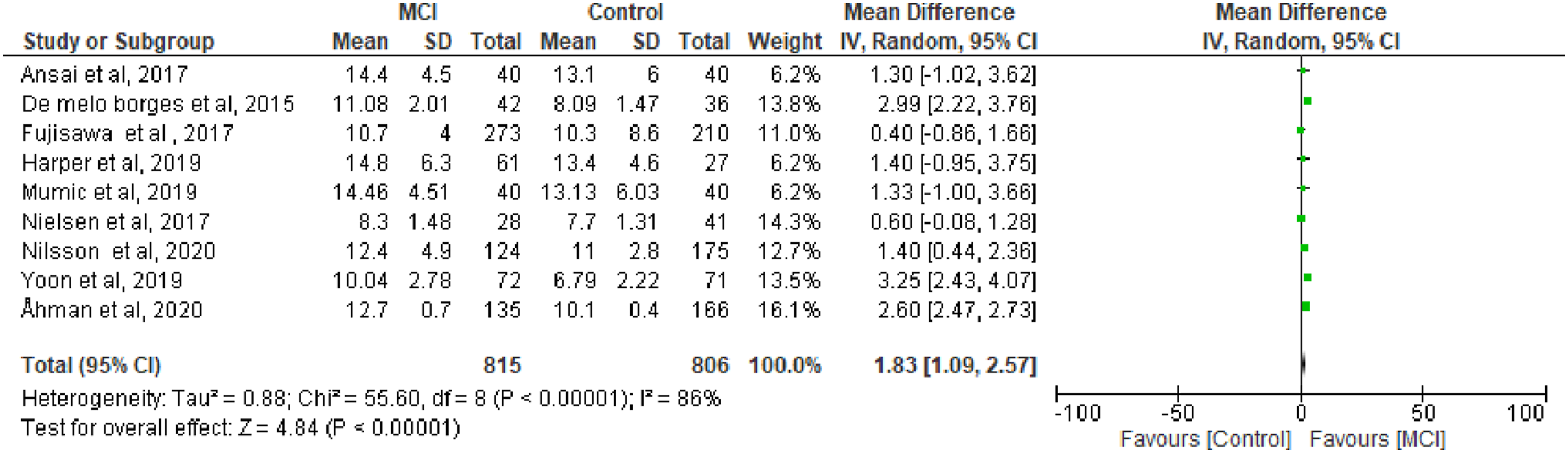
For the Dual-Task TUG, only four studies were included in the meta-analysis. Results indicated that participants with AD had significantly longer completion times (p < 0.0001), with a mean difference of 12.28 seconds (95% CI: 6.56–18.0 s) compared to cognitively healthy controls (Figure 4). Analysis of TUG-DT (Dual Task) Score in Subjects With Alzheimer’s. Meta-Analysis Plot Summarizing the Effect Sizes (With 95% Confidence Intervals) of Levels of TUG Score in Subjects With Alzheimer’s. Each Horizontal Line Represents an Individual Study, With the Square Indicating the Effect Size and the Line Representing the Confidence Interval. The Square Size Reflects the Study’s Weight in the Meta-Analysis. The Diamond at the Bottom Represents the Pooled Effect Size and Confidence Interval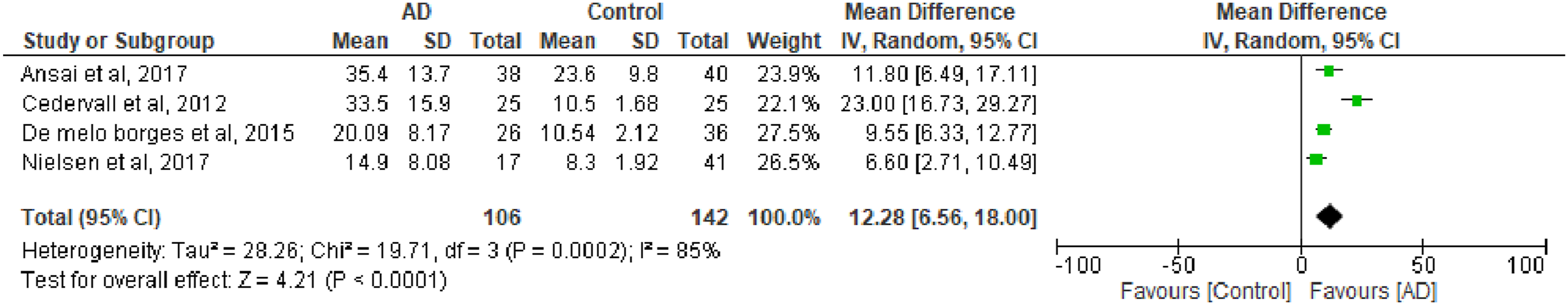
Sensitivity analyses indicated that any individual study did not significantly influence the overall test results. Despite standardizing all test outcomes to the same unit of measurement (seconds), significant heterogeneity was observed in the comparisons between groups.
No significant risk of publication bias was detected based on the funnel plot analysis. The studies were relatively evenly distributed, and most reported the number of test repetitions while documenting at least one negative result or lack of correlation between TUG scores and cognitive function assessment in the target population, suggesting a low risk of selection bias.
Discussion
This systematic review and meta-analysis aimed to evaluate the association between TUG performance and cognitive impairment in older adults with AD and MCI, highlighting its potential as a clinical screening tool. The findings suggest that individuals with AD and MCI take significantly longer to complete the TUG compared to cognitively normal controls, and that completion time nearly triples in AD patients when a secondary task is incorporated, underscoring the value of dual-task TUG in detecting early deficits.
Our results indicate that individuals with MCI and AD consistently require more time to complete the TUG test across all its variations. Several studies have shown that slower TUG performance is associated with poorer global cognition, processing speed, attention, and executive function and has been linked to an increased risk of dementia, including AD (Kuate-Tegueu et al., 2017). The connection between motor function and cognitive decline has been widely studied, particularly concerning gait disturbances in the preclinical stages of the disease (Dumurgier et al., 2017; Liu-Ambrose et al., 2008; Sabia et al., 2017).
Longitudinal studies have demonstrated that slower gait speed is associated with an increased risk of dementia in community-dwelling older adults (Kuate-Tegueu et al., 2017). Additionally, among individuals with cognitive impairment, gait speed appears to decline progressively based on the severity of cognitive decline: 0.11 m/s in individuals with MCI, 0.20 m/s in those with mild dementia, and 0.41 m/s in cases with moderate dementia, compared to healthy controls (Montero-Odasso et al., 2019).
A longer TUG completion time has also been observed in populations with physical frailty, which is associated with a higher risk of cognitive frailty progressing to dementia (Wada et al., 2023). Additionally, TUG time has been significantly correlated with changes in frailty, showing that frail individuals take longer to complete the test than pre-frail and non-frail individuals (Ansai et al., 2019; Montero-Odasso et al., 2012). Our review reports that in two of the included studies, individuals with AD with a history of falls exhibited longer TUG completion times than those without previous falls. These findings suggest that frailty and fall history might amplify the impact of cognitive impairment on mobility, reinforcing the role of TUG as a multidimensional marker that captures not only motor decline but also the combined burden of physical vulnerability and cognitive dysfunction in AD and MCI populations.
Although most studies in this review reported significant differences in TUG performance between groups, two studies did not find differences in simple TUG completion time, and this could be related to the variability of symptoms during disease progression or a higher baseline level of physical capacity in individuals with AD at the onset of motor decline (Allan et al., 2005; Kato-Narita et al., 2011; Werner et al., 2017). However, in these studies, where no significant differences were found in TUG completion time, introducing a secondary cognitive task resulted in prolonged test duration, producing a significant difference, particularly in individuals with AD compared to healthy controls. These findings might also be explained by demographic characteristics, such as younger age or higher baseline functional capacity of participants, as well as by the inclusion of individuals at earlier stages of AD or MCI, in whom subtle motor deficits could remain undetected in simple TUG but become apparent when cognitive load is increased. Our results are consistent with longitudinal evidence linking gait speed and TUG performance to dementia risk, but they diverge from studies reporting no differences in simple TUG among early-stage patients. These differences underscore the importance of disease stage and baseline physical function when interpreting TUG results. From a clinical perspective, the greater sensitivity of dual-task TUG could support its use as a feasible, low-cost tool for early detection of cognitive impairment. Integrating dual-task TUG into routine assessments in memory clinics, geriatric evaluations, or community screenings might allow earlier identification of at-risk individuals and help guide timely interventions.
Previous studies have suggested that motor tasks involving cognitive components, such as dual-task TUG, are more sensitive for early detection of cognitive impairment (Cohen et al., 2016). The dual-task paradigm posits that performing two tasks simultaneously can lead to interference if they utilize, share, or compete for the same cognitive resources or functional systems, particularly those related to executive function and attention (Beauchet et al., 2012).
The neural networks and brain regions involved in gait control, including the prefrontal cortex and hippocampus, are crucial for cognitive function and are highly susceptible to pathological changes caused by neurodegenerative diseases. Slower baseline TUG times have been associated with faster cognitive decline in individuals with subjective cognitive impairment and even in healthy controls. Furthermore, longer TUG completion times have been linked to reduced cortical thickness in motor, executive, associative, and somatosensory cortical areas (Mielke et al., 2013; Snijders et al., 2007; Studenski et al., 2011). The association between longer TUG times and structural brain changes suggests that mobility assessments could indirectly reflect underlying neurodegeneration. Clinically, this might encourage the use of TUG as a simple bedside tool to identify patients who could benefit from neuroimaging or targeted interventions, such as gait training, balance rehabilitation, or combined motor-cognitive therapies.
The dual-task TUG demonstrated a positive association between neurodegeneration biomarkers in cerebrospinal fluid and white matter lesion volume in AD patients in one of the studies included in this review. Previous research has reported that neurodegeneration biomarkers, such as total tau, phosphorylated tau, and beta-amyloid proteins detected in CSF, are closely linked to the volume of white matter lesions in AD patients (Tangen et al., 2023). These lesions, visible on magnetic resonance imaging, reflect damage to neuronal connection fibers and are associated with motor function impairment. The accumulation of these biomarkers in CSF indicates the progression of neurodegenerative processes, affecting cognitive and motor functions due to the disintegration of neural circuits essential for movement control and coordination. This motor decline, resulting from white matter alterations, leads to reduced precision in movement, impaired walking, and balance deficits, increasing the risk of falls and functional dependency in daily activities (Cullen et al., 2018; Tangen et al., 2014). As cognitive abilities deteriorate, particularly in areas such as memory, attention, and planning, there is a significant impact on motor function, affecting coordination, balance, and the ability to perform daily activities.
The TUG test is a simple, quick, easy-to-administer assessment that evaluates functional mobility, gait, and dynamic balance. It includes subtasks such as walking in a straight line, turning, and performing transitions, all of which require cognitive integration, visual processing, and additional afferent information compared to simply walking straight. When performed under a dual-task condition, an extra cognitive load is introduced, allowing for the evaluation of an individual’s ability to manage simultaneous physical and cognitive demands.
This review highlights a potential association between TUG completion time and cognitive impairment in individuals with AD and MCI. It underscores the importance of evaluating functional mobility and incorporating cognitive tasks, as these can reveal postural, gait, and mobility alterations, as well as an increased risk of falls and disability in individuals with cognitive decline. The combination of walking and performing a dual-task emerges as an effective diagnostic tool for early AD detection, offering greater sensitivity than single-task assessments, thus making it valuable for implementing therapeutic interventions.
Additionally, the evidence suggests that TUG is a reliable test for predicting future cognitive impairments, as motor and mobility impairments precede cognitive decline. In this regard, previous studies have demonstrated that gait and motor function deficits may precede cognitive impairment and the development of AD.
Limitations
Among the main limitations of this review are the heterogeneity of the study population and the age difference between AD participants and controls, as the latter were generally younger. Moreover, the number of studies evaluating dual-task TUG was limited, with methodological variability making it difficult to generalize the findings. Differences also influenced variability in TUG administration, as some studies used a single measurement, while others averaged multiple trials. Additionally, small sample sizes and the lack of a standardized cognitive assessment across studies posed challenges. While many studies used the MMSE, its limited sensitivity in early AD stages may have impacted the findings.
Conclusions
Despite these limitations, this review highlights the TUG test as a promising tool for the early identification and screening of cognitive decline and AD, given its ability to integrate motor and cognitive assessments. However, while TUG demonstrates high specificity and predictive accuracy, its sensitivity in early disease stages remains challenging.
For this reason, TUG is recommended not to be used in isolation but rather in combination with other cognitive assessments, ensuring a more comprehensive evaluation of an older adult’s cognitive status and contributing to a more integrated diagnostic approach.
Supplemental Material
Supplemental Material - Association of the Timed Up and Go Test With Alzheimer’s Disease: Systematic Review and Meta-Analysis
Supplemental Material for Association of the Timed Up and Go Test With Alzheimer’s Disease: Systematic Review and Meta-Analysis by Maria Fernanda Serna Orozco, Hilary Reinosa Rivera, Jennifer Jaramillo-Losada, Harold Andres Payan-Salcedo, and Mildrey Mosquera Escudero in Journal of Applied Gerontology.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by the Direccion General de Investigaciones de Universidad Santiago de Cali under call No. DGI-01-2025.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
