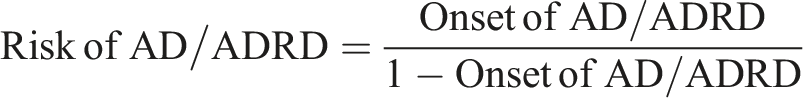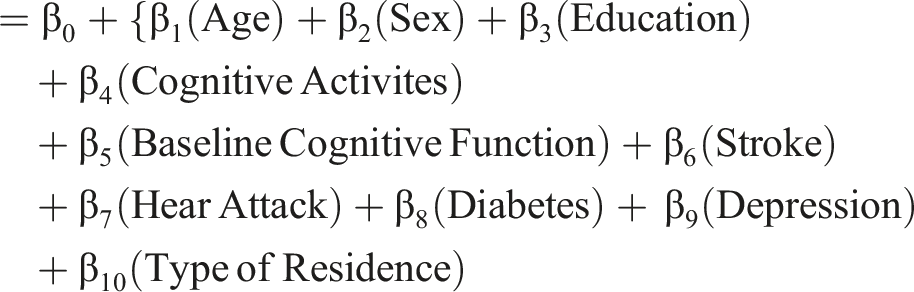Abstract
This study examined the relationship between participation in cognitive activities (CA), the risk of Alzheimer’s disease and related dementias (AD/ADRD), and the residential setting using Health and Retirement Study data from 2012 to 2020 (n = 18,111). A Cox proportional hazards regression model assessed risk factors. Findings revealed that residential setting significantly predicted dementia risk. Older adults living in nursing homes were 3.57 times more likely to develop AD/ADRD than those residing in the community (95% CI [2.23, 5.07]). Although both groups showed reduced risk with increased CA participation, community dwellers experienced a 12% risk reduction compared to only 2% among nursing home residents, even when both participated in cognitive activities three to four times per week. These results underscore the dual importance of engaging in cognitive activities and considering environmental context in mitigating AD/ADRD risk among older adults.
• Regular engagement in cognitive activities (e.g., reading, writing, games) is associated with decreasing the risk of Alzheimer’s diseases and related dementia. • Community-dwelling older adults gain greater cognitive health benefits from these activities than nursing home residents, even when participation levels are equivalent, underscoring the influence of residential context. • High baseline cognitive function is associated with a lower dementia risk, whereas the presence of chronic health conditions is related to an increased dementia risk.
• Utilize digital health devices (e.g., mobile apps) to deliver sustainable and personalized cognitive programs to all older adults, maximizing dementia risk reduction. • Provide nursing home residents with social interaction opportunities and relationship-building activities similar to those in community settings to enhance cognitive training benefits.What this paper adds
Applications of study findings
The high prevalence of dementia worldwide presents a growing public health challenge due to a rapidly aging global population. The Centers for Disease Control and Prevention (CDC) estimates that the number of people diagnosed with dementia will double approximately every 20 years and potentially surpass 131 million by 2050 (CDC, 2021). As individuals age, they tend to experience cognitive decline, which is considered a natural aging process that is characterized by reduced attention span, memory loss, and slower processing speed (Dexter & Ossmy, 2023; Hedden & Gabrieli, 2004). Although advancing age remains the primary non-modifiable risk factor, other adverse contributors such as physical inactivity and limited social engagement further exacerbate the risk of dementia by accelerating cognitive decline (Hwang et al., 2023; Rolandi et al., 2020).
Given the increasing numbers and increasing rates of dementia cases, researchers have stressed the importance of designing, developing, and implementing dementia prevention strategies (Kivipelto et al., 2018; Livingston et al., 2024). Research has provided evidence that activity-based prevention approaches that include cognitively stimulating tasks, physical exercise, and community-based social interaction are effective in delaying the progression of cognitive decline and moderating dementia risk factors (Scarmeas et al., 2004; Stern, 2002). Initiating preventive efforts during the early stages of cognitive impairment may help reduce not only the individual burden of dementia but also its broader societal and economic impacts.
Recently, substantial evidence has been presented that participation in cognitive activities can mitigate cognitive decline and lead to noticeable improvements in cognitive health (Mellow et al., 2025; Silva et al., 2025). These researchers have characterized cognitive activities as activities that stimulate and support brain activities such as puzzles, crafting, playing musical instruments, listening to music, and reading. These studies have provided evidence that participation in such activities is associated with improved cognitive outcomes and a reduced risk of dementia. Kim and Lee (2002) also investigated the contribution of engaging in intellectually enriching tasks that included language learning, creative writing, and attending educational lectures and found that they were effective in delaying cognitive decline in older adults.
Considering the importance of engagement in cognitive activities, residential setting has been found to be a significant determinant of dementia risk, especially when comparing nursing home with community-dwelling older adults (Appelhof et al., 2017; Jao et al., 2018). These two distinct residential settings are associated with unique environmental, psychosocial, and activity-related environments for older adults that affected the quality of engagement in health behaviors and the effectiveness of cognitive health outcomes (Olsen et al., 2016; Scocco & Nasuato, 2017). Prior studies have further shown that homelike residential care models can positively influence emotional well-being, behavior, and social engagement (Ausserhofer et al., 2016), that variations in quality-of-life indicators among nursing home residents are partly dependent on cognitive status (Missotten et al., 2009), and that community-dwelling older adults in diverse cultural contexts such as Indonesia and China tend to exhibit better cognitive function and lower prevalence of cognitive decline compared to their nursing home counterparts (Setiyani & Iskandar, 2022; Yang et al., 2019). Typically, nursing home residents often experience reduced autonomy, limited cognitive and social engagement, and higher levels of isolation, all of which are known contributors to declines in cognitive health (Lyu & Fan, 2024; Rafnsson et al., 2017). On the other hand, community-dwelling older adults are known to have more opportunities to participate in cognitively and socially stimulating activities that support and preserve cognitive function and delay the occurrence of dementia (Kim & Lee, 2002). These disparities underscore the importance of accounting for environmental factors when investigating the association between cognitive activity participation and dementia risk.
While research has provided empirical evidence of the importance of cognitive activity participation as a strategy that delays the risk of dementia and improves cognitive function, there is a critical research gap in the impact of the type of residential setting on the risk of dementia by comparing older adults that reside in nursing homes to those residing in the community. To confirm the impact of cognitive engagement in preventing the risk of dementia, our study investigated the longitudinal relationships between cognitive activities, the risk of AD/ADRD, and covariates in older adults to determine whether cognitive activity participation is associated with the risk of dementia and other covariates such as demographic factors and comorbidities.
In addition, while the benefits of cognitive activities have been well validated, further examination is needed to estimate the required level of cognitive activity participation necessary to achieve a significant reduction in the risk of AD/ADRD based on a visualized dose-response relationship. A dose–response relationship in behavioral health describes how changes in the amount of a behavioral factor (the dose) are statistically related to changes in a health outcome (the response). This relationship helps establish potential causality and inform recommendations for optimal behavioral interventions, serving as a useful strategy to confirm the threshold levels of cognitive activity for practical implications for the design of dementia healthcare services for older adults who are experiencing rapid cognitive decline (Guo et al., 2022). Further, little research has been conducted that examines the differences in the risk of AD/ADRD associated with different types of living environments, specifically between nursing home and community dwelling older adults. Comparison of the changes in risk associated with the type of residence will shed light on the different levels of efficacy, even when participants in different living environments engage in the same level of cognitive activity. Our findings will provide valuable insight into whether nursing home residents require more activity participation or require other types of healthcare resources.
Methods
Data and Study Participants
We used core data from the 2012 to 2020 Health and Retirement Study (HRS) that is published biannually and has tracked study participants aged 50 and above since 1992 in the United States. The HRS collects a wide range of health-related data including pensions, occupational status, leisure behaviors, and socioeconomic information to understand older life from a longitudinal perspective. HRS data collection has been implemented either through telephone or face-to-face interviews based on participant characteristics such as cognitive function. To maintain reliability in data collection, the HRS employed two types of data collection strategies. Patient-Reported Outcome (PRO), the default setting, has been used with participants who can provide reliable answers without support from caregivers or family members. Proxy-Measured Outcome (PMO) is an optional method that is used to collect data from participants who give unreliable answers to questions. The interview administrator decides which data collection strategy to use after evaluating the responsiveness of participants in the initial interview stage. We used the data collected only from participants included in the PRO section who were expected to have reliable and independent responses even if they were living with cognitive challenges or had been diagnosed with AD/ADRD.
The HRS uses a Household (HH)-based data collection protocol to construct the longitudinal dataset in which HRS assigns a unique HH number, and assigns each member within the HH, such as family members, a Person Identification Number (PN). The PN within the HH represents the number of family members and may change in subsequent data collections due to deaths, births, or divorce. The HRS obtained ethical approval for data collection from the Institutional Review Board of the academic institution, and HRS administrators received consent forms from the participants. Most HRS data are publicly accessible on their website, except for sensitive data that require approval by the administrator. Data users are not required to obtain further ethical approval to make use of these data.
In this study, we used participant data from 2012 to 2020 that totaled 22,554 participants (PNs) as described in Figure 1. A total of 18,111 participants were screened based on the inclusion criterion of being 60 years of age or older. In the final sample, 804 participants had been newly diagnosed with AD/ADRD between 2012 and 2020. Follow-Up Participant From 2012 to 2020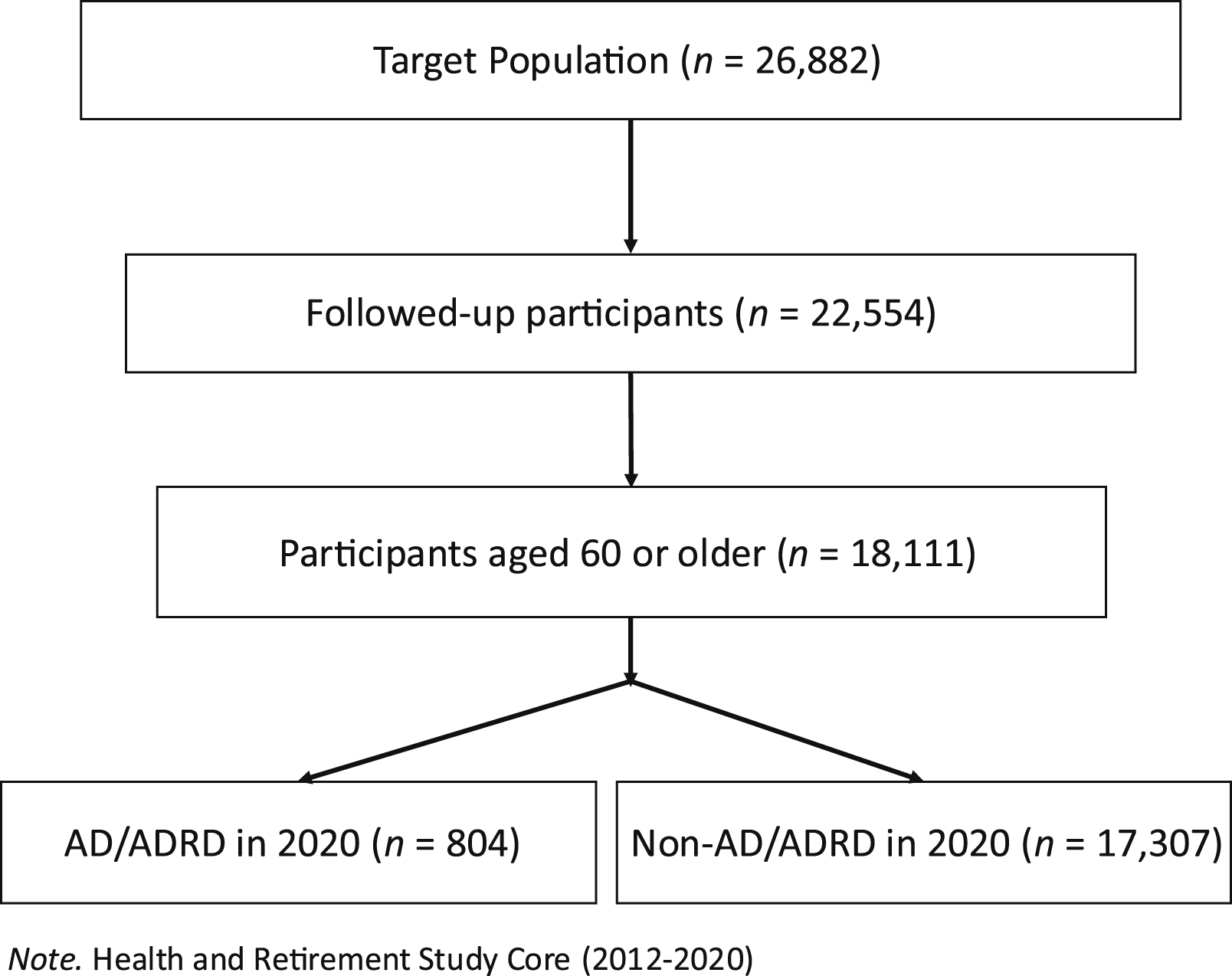
Instruments
Input Variables
The Psychosocial module (LB) in the HRS core encompasses data describing 21 leisure-related health behaviors, a subset of which are considered cognitive activities (CAs). Using established criteria from prior HRS research (Lee et al., 2022), we extracted six CA specific items: reading (books, magazines, newspapers), engaging with word games (e.g., crosswords, Scrabble), playing cards or board games (e.g., chess), writing, participating in educational training courses, and crafting (e.g., knitting, making clothes). The frequency of participation in these cognitively and intellectually stimulating activities was measured on a seven-point Likert scale (1 = “Daily” to 7 = “Never”). The scale was reverse-coded (i.e., 1 = “Never relevant,” 7 = “Daily”), ensuring higher values corresponded to more frequent participation. A total CA score was calculated by summing six specific CA items from 2012 to 2020, and the average level of CA participation was entered as an input variable, consistent with the protocol used in HRS studies (Lee et al., 2022; Liu et al., 2020; Rimmele et al., 2009). The CA scores ranged from 6 to 42.
Output Variables
Alzheimer’s Disease and Related Dementias
Self-reported physician diagnoses of Alzheimer’s disease and Alzheimer’s disease–related dementias were used to identify cases. Individuals who reported a new diagnosis of AD or ADRD between 2012 and 2020 were included, whereas those reporting a diagnosis before 2012 were excluded. To maintain data integrity, only individuals with consistent diagnostic histories were included. Specifically, participants who reported a dementia diagnosis in multiple survey waves were required to affirm that diagnosis in both earlier and subsequent assessments.
Two questions were used to identify participants diagnosed with AD/ADRD. The first question asked was, “Since we last asked you, has a doctor told you that you have Alzheimer’s disease?” The second question inquired, “Since we last asked you, has a doctor told you that you have dementia or any other serious memory impairment?” Responses to both questions were coded as 1 (“Yes, diagnosed”) or 0 (“No, non-diagnosed”). Following the approach of a previous study using HRS data to assess cognition (Crimmins et al., 2016), participants were categorized as having AD/ADRD if they answered “Yes” (1) to either question.
Covariates
We included covariates to account for the confounding factors that were related to study variables, and to provide a more reliable understanding of the relationship between the input and output variables (Rothman et al., 2008). First, we incorporated demographics such as age, sex, and educational attainment. Previous research has provided evidence that demographic factors such as age, sex, and educational level can introduce bias when examining the health status of older adults with preexisting conditions.
Second, baseline cognitive function assessed in 2012 was included as a covariate as baseline cognitive function is linked to the risk of AD/ADRD and confounds the association between health behaviors and the long-term risk of AD/ADRD (Crimmins et al., 2011). The HRS assesses cognitive function using the Telephone Interview for Cognitive Status-27 (TICS-27) in which three cognitive tests evaluate memory, working memory, and attention and processing speed. Memory was measured through immediate and delayed recall of 10 random words (e.g., orange, water, cloud). Participants recalled as many words as possible immediately after hearing them, and then again after a five-minute interval. Each correct response was worth one point, for a summed possible total of 20 points. Working memory was measured using a subtraction test that required participants to sequentially subtract seven from 100 over five trials (e.g., 100–7 = 93, 93–7 = 86). Each accurate answer was awarded one point for a maximum of five points. Attention and processing speed were assessed using a reverse counting test from 20 to 10 over two trials, with each correct count receiving one point for a total of two possible points. Overall scores on the TICS-27 range from 0 to 27.
Third, the impact of comorbidities was assessed in the analysis model. The history of diagnosis with chronic diseases is closely related to the risk of AD/ADRD and hinders the estimation of the benefits of CAs on that risk (Livingston et al., 2020; Norton et al., 2014). Various chronic health conditions such as diabetes, heart attack, stroke, and depression were included to clarify the dose-response relationship. For example, “Since we last asked you, has a doctor told you that you have diabetes?” Responses to both questions were coded as 1 (“Yes, diagnosed”) or 0 (“No, non-diagnosed”).
Fourth, we integrated the type of residence into our analysis, which is a crucial determinant of the risk of AD/ADRD in older adults (Luppa et al., 2010). The HRS collected data from the study participants that describe their residence type as, “Are you living in a nursing home or other healthcare facility?” The HRS defined nursing home or other health care facility as, “The facility that provides all of the following services for some or all of its residents, such as 24-hour nursing assistance and supervision, dispensing of medication, personal assistance, and room and meals.” The residence type was binary coded as 1 (nursing home residence) and 0 (community residence).
Analysis
Before the main analysis, a descriptive analysis was conducted to explore the demographic characteristics of study participants. A Cox proportional hazards regression model was used to estimate changes in the risk of AD/ADRD associated with increasing levels of CA participation while accounting for demographics, baseline cognitive function, comorbidities, and type of residence. The dose-response relationship was visualized, and the risk of AD/ADRD was compared according to residence type. All statistical analyses were conducted using SPSS version 29.0.
The regression equation we used is to test research hypotheses presented below
The type of residence is associated with differential risk levels for AD/ADRD
Individuals residing in nursing homes exhibit a higher risk of developing AD/ADRD compared with those living in community settings
Results
Demographic Characteristics
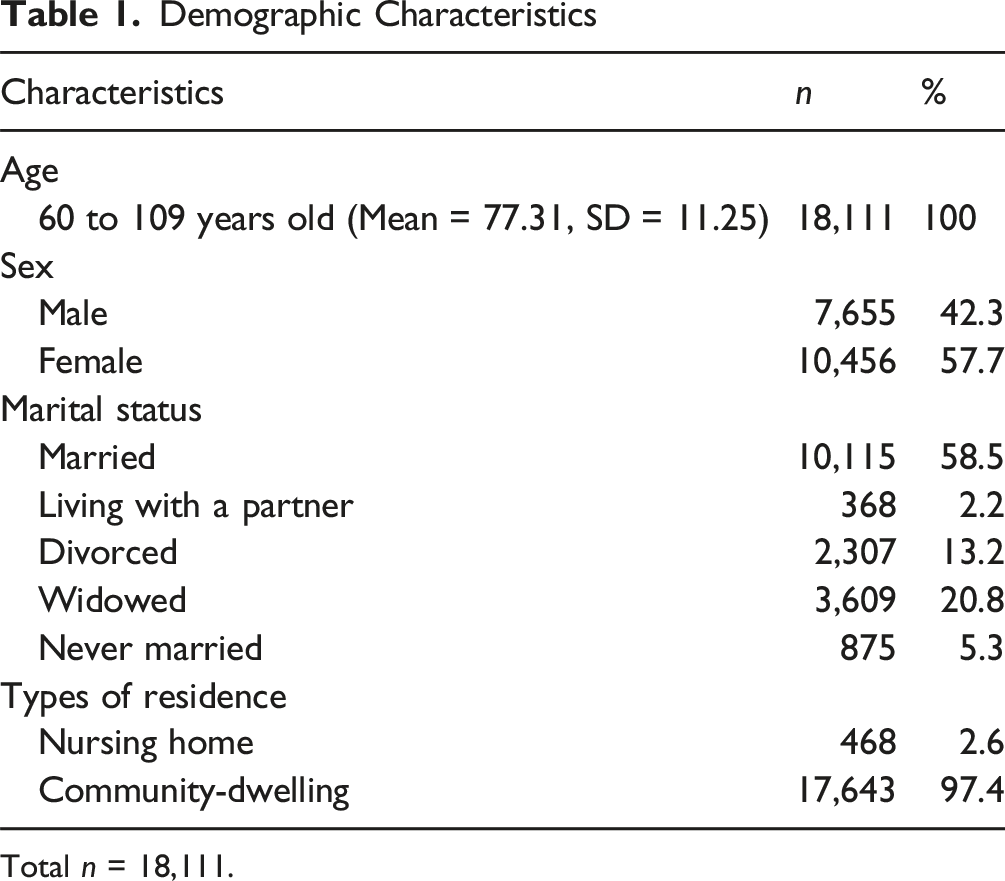
Total n = 18,111.
Descriptive Statistics

Note. Cognitive activities represent the mean score of six types of activity participation between 2012 and 2020, assessed using a seven-point Likert scale, with scores ranging from six to 42. Baseline cognitive function was assessed using the Telephone Interview for Cognitive Test-27 in 2012. Only cases in which the response regarding the presence or absence of disease following its onset remained consistent from 2012 to 2020 were included. ADRD = Alzheimer’s Diseases and Related Dementias.
First, we investigated how the risk of AD/ADRD changes in relation to the average level of CA participation from 2012 to 2020. As shown in Figure 2, the risk of AD/ADRD decreases with increasing levels of CA participation, providing evidence of a dose-response relationship. Although CA participation was not included as a separate predictor in the regression model, the pattern observed in Figure 2 is consistent with the presence of a protective effect related to participation in cognitive activities over time on the onset of AD/ADRD. Dose-Response Curve Between Cognitive Activities and AD/ADRD Risk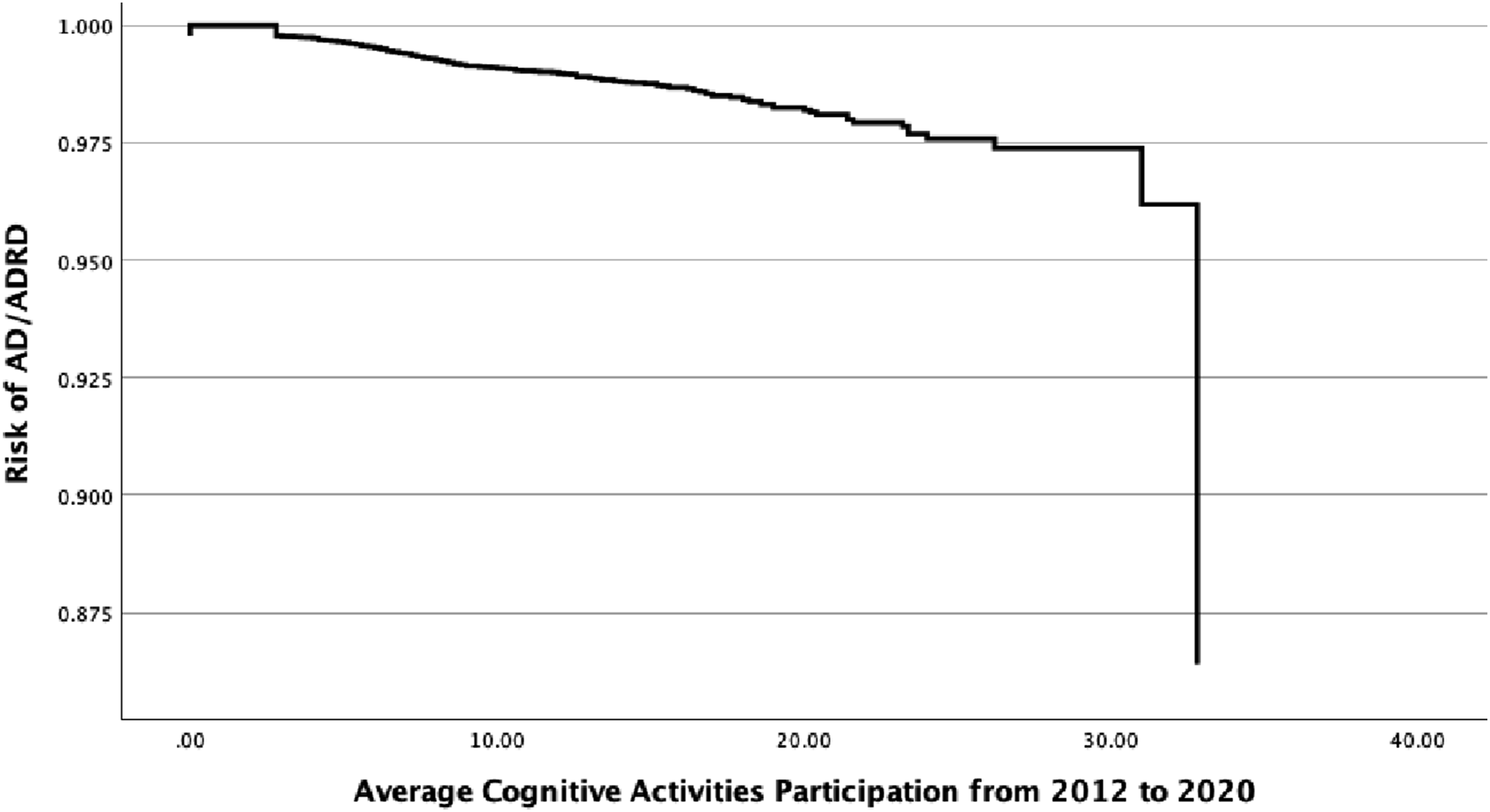
Cox Regression Results for the Risk of AD/ADRD
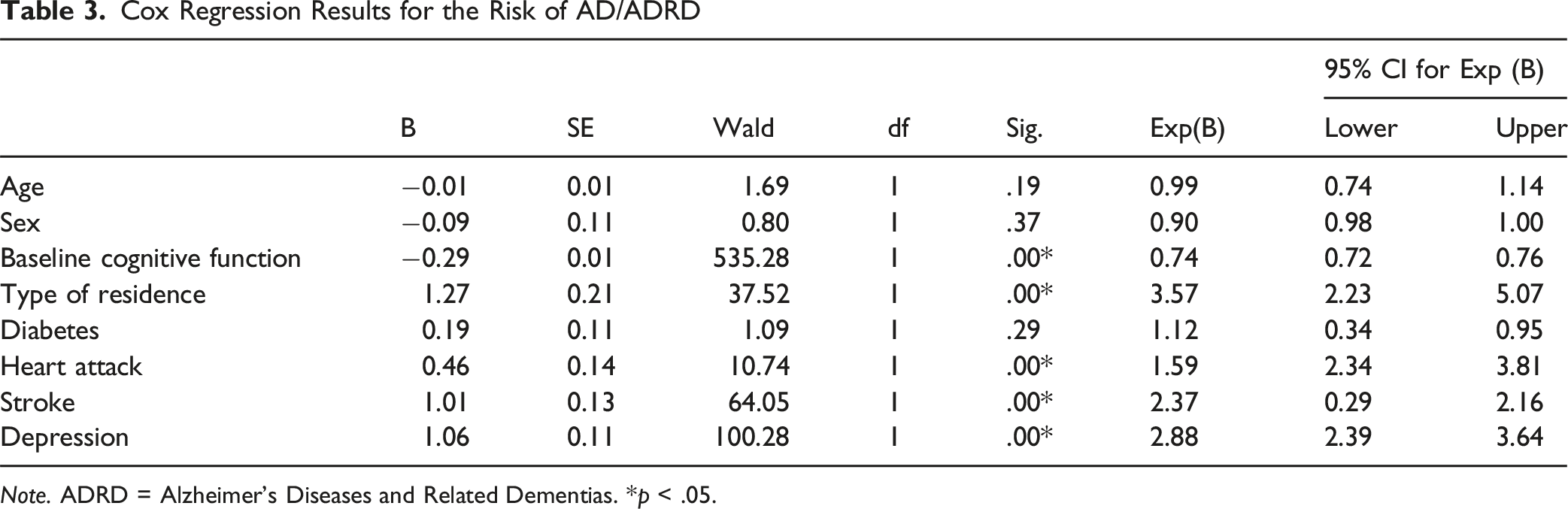
Note. ADRD = Alzheimer’s Diseases and Related Dementias. *p < .05.
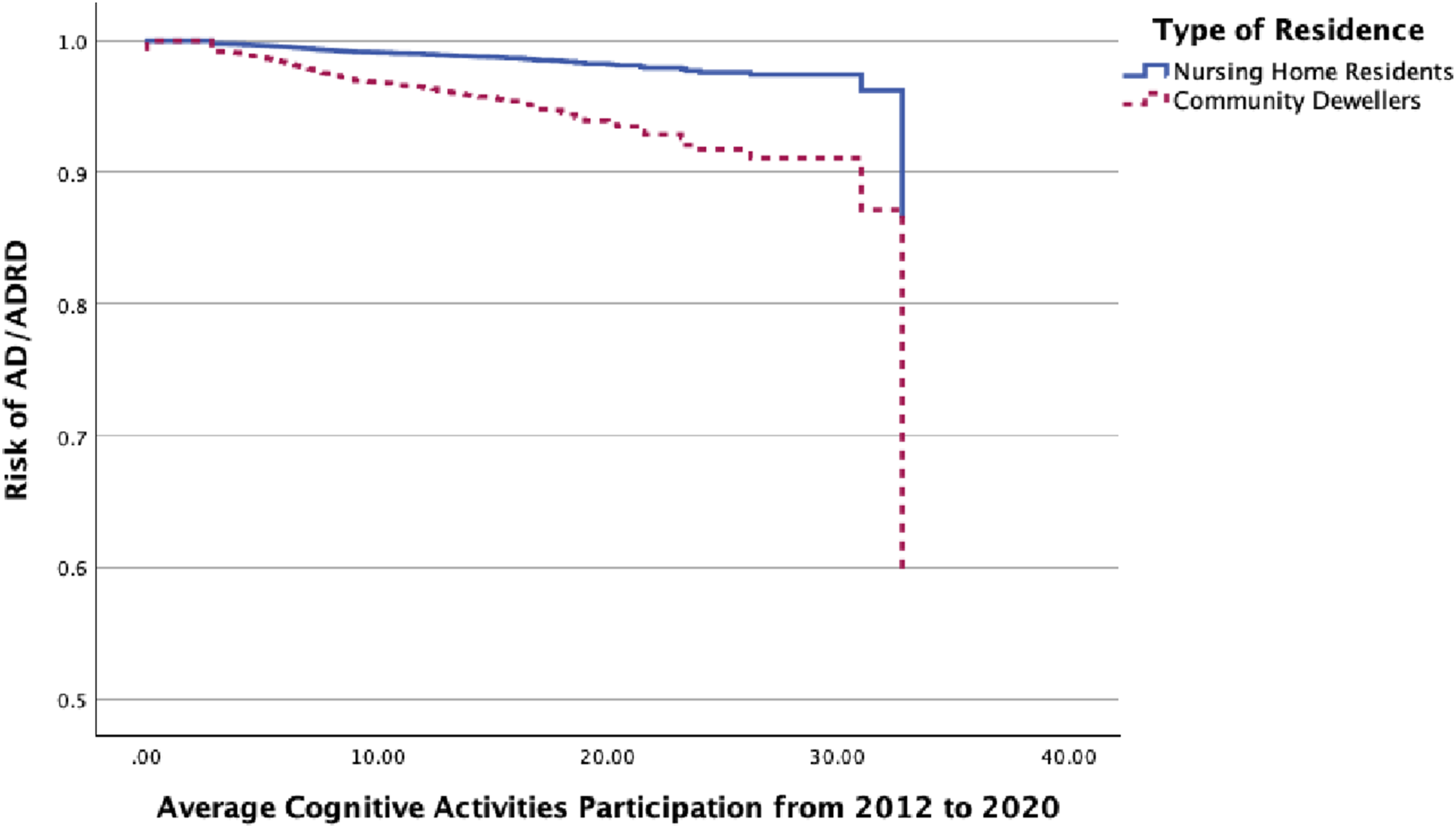
Risk Comparison Between Community Dwellers and Nursing Home Residents
In addition to these primary relationships, the baseline cognitive function (B = - 0.29, Wald = 535.28, Exp(B) = 0.74, 95% CI [0.72, 0.76]), indicates that each one-point increase in the baseline cognitive score was significantly associated with a 26% reduction in the risk of AD/ADRD (Table 3). Other covariates, including comorbidities, were significantly related to the risk of AD/ADRD. Heart attack (B = 0.46, Wald = 10.7, Exp(B) = 1.59, 95% CI [2.34, 3.81]) was associated with a 59% higher risk of AD/ADRD compared to those without a heart attack, while stroke (B = 1.01, Wald = 64.05, Exp(B) = 2.37, 95% CI [0.29, 2.16]) increased the risk by 137%. We found that depression (B = 1.06, Wald = 100.28, Exp(B) = 2.88, 95% CI 2.39, 3.64]) was associated with the largest increase among these conditions, at 188%. Conversely, age (B = −0.01, Wald = 1.69, p = .19) and sex (B = −0.09, Wald = 0.80, p = .37) did not reach statistical significance, indicating no substantial differences in AD/ADRD risk associated with these factors. Although diabetes (B = 0.19, Wald = 1.09, p = .29, Exp(B) = 1.12) was associated with a 12% higher risk, this effect was not statistically significant.
Discussion
In this longitudinal study, we examined the relationship between cognitive activity participation and the risk of AD/ADRD across different residential settings. The findings of our study demonstrate the positive impact of cognitive activity engagement on reducing the risk of AD/ADRD among both nursing home and community-dwelling older adults. Indeed, community-dwelling older adults who participated in cognitive activities exhibited a greater reduction in dementia risk compared to their counterparts who resided in nursing homes. This finding highlights not only the protective role of cognitive activity engagement but also the impact of residential context on cognitive health among older adults.
Prior studies have demonstrated that participation in cognitive activities reduced the risk of dementia by improving cognitive functioning (Mellow et al., 2025; Silva et al., 2025). These studies stressed the importance of designing and implementing structured cognitive interventions that promote cognitive health among older adults. The findings of our study support the idea that nursing home residents and community-dwelling older adults both benefited from cognitive activity participation to reduce the risk of AD/ADRD. Our results also suggest the need to promote cognitive activity participation be older adults as a preventive strategy to support long-term cognitive health and reduce dementia risk.
Prior research has provided evidence that residents in long-term care facilities often report lower levels of quality of life and higher rates of the behavioral and psychological symptoms of dementia (Lyu et al., 2024; Olsen et al., 2016; Rafnsson et al., 2017; Scocco & Nasuato, 2017). The findings of these studies have also provided evidence that such residents typically engaged in lower levels of activity participation and were more likely to be prescribed psychotropic medications that resulted in diminished quality of life. Consistent with these previous findings, the findings of our study provide empirical evidence that while nursing home residents benefited from participation in cognitive activities, they were less likely to experience a significant reduction in AD/ADRD risk compared to community-dwelling older adults. This finding indicates the critical influence of environmental factors such as residential setting on dementia-related behaviors and outcomes. Possible explanations include the greater prevalence of multimorbidity and frailty among nursing home residents, qualitative differences in the nature and level of stimulation provided by cognitive activities, and the impact of limited physical activity and social interactions in institutional settings, all of which may attenuate the protective effects of cognitive engagement (Robertson et al., 2016). In particular, the level and nature of social participation may be an important differentiator between the two settings. Nursing home residents may engage in cognitive activities primarily on an individual basis, whereas community-dwelling older adults are more likely to participate in such activities jointly with peers they choose voluntarily. In our study, only two activities, playing cards and participating in courses, necessarily involved other people, suggesting that many cognitive activities in nursing homes may lack a socially engaging component. Prior work has shown that social participation can enhance the cognitive benefits of activities, even when the frequency of cognitive activity itself is low (Robertson et al., 2016). For instance, the cognitive reserve hypothesis suggests that enriched and varied stimulation, often more accessible in community environments, can buffer neurodegeneration (Stern, 2012), and clinical studies have shown that reduced mobility and sensory stimulation in institutional settings are associated with accelerated cognitive decline despite a similar frequency of activity participation (Ballesteros et al., 2015). Moreover, perceptions of aging may also interact with these environmental and activity-related factors. Negative perceptions of aging have been shown to contribute to declines in psychological, physical, and cognitive functioning, both in the short and long term (Robertson et al., 2016). Longitudinal evidence indicates that such perceptions are independently associated with declines in verbal fluency and self-rated memory over time, suggesting that they may be a modifiable factor to target in interventions aimed at reducing cognitive decline among older adults.
As regards the impact of comorbid health conditions on the risk of dementia, our findings show that heart attack, stroke, and depression were associated with an increased risks of AD/ADRD among older adults. Substantial evidence exists in the research of strong links between these conditions and elevated dementia risk in older adults (Livingston et al., 2020; Norton et al., 2014). The findings of our study are consistent with these previous findings that these comorbid health conditions are associated with an increased risk of AD/ADRD, which provides evidence of the complex interplay between these health conditions and their cumulative effect on the cognitive decline of older adults.
Several limitations of the study must be addressed. While our study focused on the longitudinal relationships between cognitive activity engagement, the risk of AD/ADRD, residential contexts, and a limited number of comorbid health conditions, there could be other potential confounders (e.g., sleep, nutrition, activity preferences, and other comorbidities) that may affect cognitive activity engagement and the risk of dementia. Due to limitations in HRS Core data availability, future research should employ machine learning models to investigate the underlying mechanisms that comprise the dynamic interactions among confounding variables using accessible datasets. Such approaches may provide deeper insights into the differences in AD/ADRD risk based on residential type and contribute to the development of comprehensive strategies to support cognitive health for older adults.
In addition, we dichotomized residential types into nursing home residents and community-dwelling individuals. Although various types of nursing homes and associated care services exist, the limited nature of the available information prevented a more extensive categorization of residence types such as assisted living facilities and day care service units. For instance, some day care service users intermittently receive care from nursing homes while living independently in the community. These cases necessarily complicate the comparison of AD/ADRD risk by residential status. Furthermore, the proportion of nursing home residents in our sample was relatively small (2.6%), which may have reduced the statistical power for between-group comparisons and potentially limited the generalizability of the findings to the broader nursing home population. Future studies should adopt qualitative approaches to enhance understanding of the factors contributing to these differences in AD/ADRD risk individually.
Conclusions
Despite these limitations, the findings of our study provide evidence that higher levels of participation in cognitive activities were associated with a lower risk of developing AD/ADRD that suggests a protective effect over time. While this benefit was observed in both community dwellers and nursing home residents, older adults residing in nursing homes consistently exhibited a higher risk of AD/ADRD. Further, baseline cognitive function and chronic health conditions such as heart attack, stroke, and depression were found to be significant predictors of increased AD/ADRD risk. The findings of our study provide critically needed empirical evidence of the preventive role of cognitive activity engagement by older adults, with particular emphasis on nursing home residents. Given the elevated risk of AD/ADRD observed in nursing home residents, even when they engage in a similar level of activity as community residents, customized interventions that promote consistent engagement in cognitive activities is essential. Thus, our study suggests the need for the development and implementation of tailored cognitive training interventions such as mobile-based cognitive programs aimed at reducing the risk of dementia.
Footnotes
Author Notes
This manuscript has not been published or will not be submitted elsewhere for publication while being considered by Journal of Applied Gerontology.
Ethical Considerations
This study used publicly available anonymized databases, and as such, was exempt from ethical compliance review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.