Abstract
Vinyl-ester resin (VE) is a polymer matrix frequently used in several civil applications, but its long-term properties in humid and harsh environments can be a concern. Carbon nanotubes (CNTs) have remarkable mechanical, physical, and thermal properties. Adding them to the resin is a promising solution and makes it possible to manufacture cost-effective products with enhanced engineering properties. Our study investigated the chemical functionalization of multi-wall carbon nanotubes (MWCNTs) through acid oxidation to evaluate the durability-related properties of vinyl-ester-based nanocomposites. Samples containing 0.1 wt% of as-received CNTs and functionalized CNTs were characterized by scanning electron microscopy (SEM), elemental analysis, and X-ray photoelectron spectroscopy (XPS). A small amount of two dispersing agents (BYK 104S and BYK 9076) was added to the mixtures to produce high particle stability in the nanocomposites. Barrier properties and moisture diffusivity were determined by immersing samples in a water bath at 50°C. Aging was conducted by fully immersing samples in NaOH solution at 50°C. Matrix degradation was assessed with tensile tests, dynamic mechanical analysis (DMA), and differential scanning calorimetry (DSC) before and after conditioning. The results indicate that the addition of oxidized CNTs significantly improved the mechanical, physical, and durability properties of the composites. Additionally, unlike nanoclay-based nanocomposites, oxidized CNTs do not increase water absorption at saturation.
Introduction
Polymer nanocomposites (PNCs) are a new class of materials made up of multiple phases, one of which has dimensions below 100 nm, and the other acts as a polymer matrix. 1 Interest in PNCs has been a growing interest in PNCs in recent years due to their superior characteristics such as high thermal, 2 mechanical, 3 and barrier properties. 4 This novel group of materials is increasingly being used in a variety of industries, including sports and leisure, maritime, automotive, and transportation, as well as for civil infrastructure. 5 The matrix’s resistance to chemical degradation and the favorable properties of the nanofillers make PNCs novel, cost-effective, and high-performing materials. 6 Compared to conventional fillers, such as silica or nanoclay, carbon nanoparticles (CNPs) in the form of graphene, carbon nanotubes (CNTs), and carbon black (CB) are highly recommended for their exceptional properties. 7
CNTs have extremely small diameters and high aspect ratios, which make them ideal for improving the properties of the polymer matrix. CNTs in the shape of long cylinders of carbon molecules consisting of rolled-up sheets of single-layer carbon atoms have attracted great interest due to their unique structures. 8 There are four categories of CNTs: single-walled (SWCNTs), double-walled (DWCNTs), triple-walled, and multi-walled (MWCNTs). 9 MWCNTs are increasingly attractive and sought after for practical applications in polymer composites.10,11 Asymmetry and the number of walls play an important role in the electronic properties of CNTs. Due to their low density (1/3 to 2 g/cm3), excellent mechanical properties (strength and modulus), and thermal properties, CNTs are used in materials manufacturing.11,12 One downside is that, given their high surface area and van der Waals interactions, CNTs are insoluble and have poor dispersibility and interfacial bonding with usual matrices. This has restricted their use.9,13 Indeed, CNTs have their strong and attractive interactions, giving them a tendency to agglomerate in bundles, which produces a heavily entangled system. 14
Polymer modification of carbon nanotubes is an efficacious way for improving nanotube dispersion. The goal of such techniques is to increase chemical (covalent and non-covalent) interactions in order to improve the compatibility of nanoparticles and strengthen the bonding between the nanoparticles and matrix.12,15 Since pure CNTs are intrinsically hydrophobic owing to their nonpolar carbon–carbon bonds,
16
dispersing them in the more hydrophilic polymer matrices is challenging.
17
Employing dispersing agents (small molecules that interact with tube surfaces) promotes CNT wettability and produces electrostatic repulsion among individual tubes.13,18 Figure 1 illustrates the adsorption morphology of copolymers as dispersing agents on the surface of CNT particles. Dispersing agents are mostly hydrophobic block copolymers in which the hydrophobic tail is placed near the surface of CNTs, while their hydrophobic parts tend to interact with the resin matrix.
19
Yang et al.
14
investigated the effect of eight different dispersing agents on the properties of VE via a centrifugation method. They found that nanocomposites containing 0.1 wt% of CNTs with a suitable commercial agent (B60H) exhibited enhanced mechanical properties. The other consideration is if the tubes are changed by a chemical reaction that takes place on their surface or whether they are functionalized.
20
In this case, chemical functionalization such as oxidation, silanization, and amination generate functional groups on the surface of nanotubes that can react with the functional groups of matrices, forming long-lasting bonding.
21
The studies have shown that the oxidation of nanotubes led to a better interface between the resin and nanoparticles, resulting in a modest improvement in compression properties.
22
Schematic view of the morphologies of the agents as copolymer molecules in the vicinity of the surface of CNTs.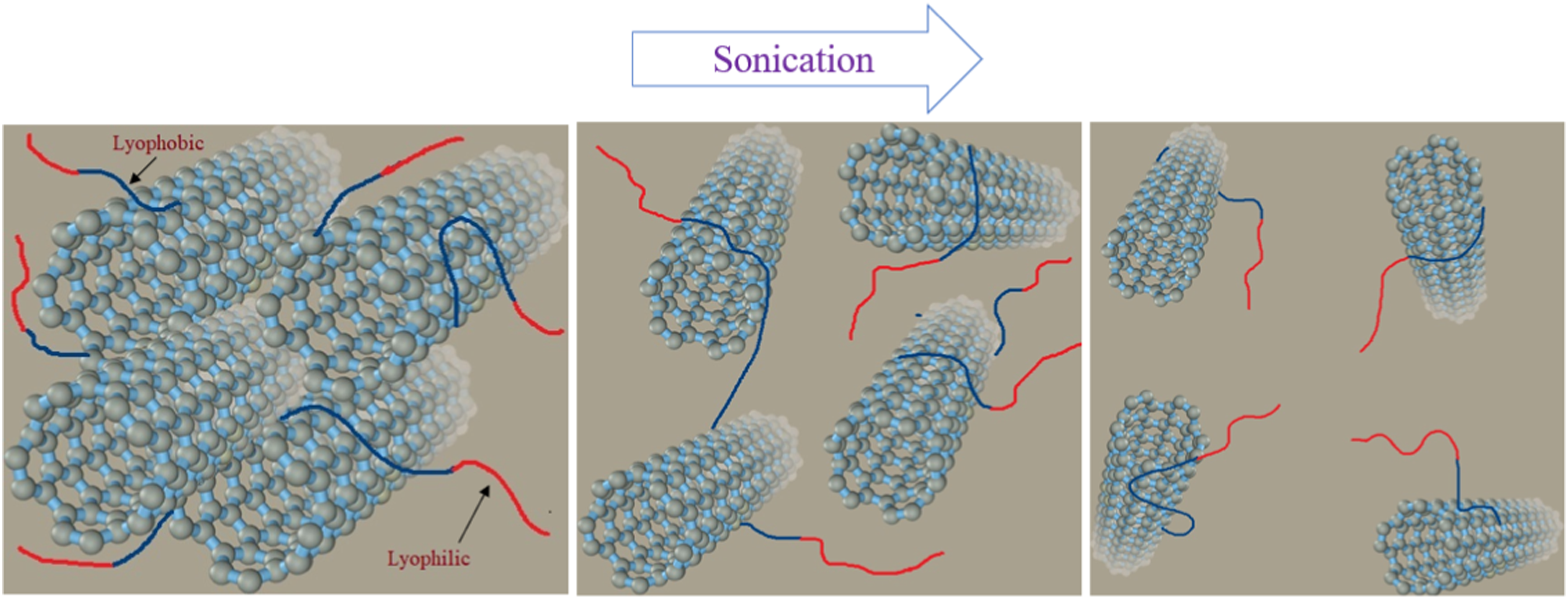
While several studies have been conducted on polymer composites containing CNTs, the barrier and durability properties of VE/CNTs nanocomposites have seldom been investigated. As for thermoset resins, vinyl-ester resins have been considered a promising alternative to the epoxy and polyester resins used for civil infrastructure. 23 VE resins have somewhat lower mechanical properties than epoxy but significantly greater chemical resistance. This is due to the presence of multiple large organic functions—for example, methyl or phenyl groups—that protect the entire matrix from hydrolysis or acid attack by protecting weaker groups.24,25 It is commonly accepted that water absorption and the diffusion of water molecules between polymer chains can produce a number of undesirable outcomes, such as swelling, that cause microcracks, plasticization, and in some cases, deterioration. 4
In this study, CNTs were chemically functionalized with the wet acid oxidation method. 26 The presence of functional groups was obtained by elemental analysis and XPS. The oxidized and non-oxidized CNTs were then used to prepare reinforced nanocomposites with the VE resin. The nanoparticles were directly dispersed within the resin via high-shear mixing and sonication methods. Two commercial dispersing agents (BYK104-S and BYK9076) as block copolymers, were incorporated during the process. Mechanical, thermal, durability, and barrier properties of samples were measured after conditioning in tap water and NaOH solution.
Experimental program
Materials
Materials employed in the research.

Nanotube oxidation
The first step of the oxidation process involved mixing 0.3 g of MWCNTs with 70 mL of 3.0 M nitric acid, stirring the mixture for 15 min, and sonicating in an ultrasonic bath for 2 h. The slurry was filtered and washed with distilled water. The second step involved repeating the first process with H2O2. The mixture was then filtered and dried in an oven at 60°C overnight.
Nanocomposite processing
Processing involved directly suspending 0.1 wt% of CNTs and 0.1 wt% oxidized CNTs in the liquid VE resin. Two types of dispersing agents (BYK 104-S and BYK 9076) were added to the mixtures. The combined quantity of the two dispersing agents (in a 1:1 ratio) equaled that of the MWCNTs and amounted to 0.1 wt%. The suspensions were homogenized in a high-shear mixer at room temperature based on a fixed protocol (2000 r/min for 30 min, 4000 r/min for 20 min, 6000 r/min for 10 min, and 8000 r/min for 5 min). Then, the mixtures were sonicated for 1 h with a 50–60 Hz ultrasonic bath before adding 0.1 wt% of curing agent (P16) to the mixtures and mixing mechanically until homogeneity was reached. Lastly, the nanocomposites were placed in a vacuum chamber to remove bubbles and cured in Teflon molds at 90°C for 2 h and post-cured at 120°C for 1 h.
Elemental analysis
The CHNS-O content was assessed with an elemental analyzer (Thermo Scientific Flash 2000 Organic Elemental Analyzer). Samples were analyzed in triplicates.
X-ray photoelectron spectroscopy
The XPS analysis was performed on a KRATOS Axis Ultra electron energy analyzer operating with Al Kα monochromatic source. The overviewing spectra were collected over a range of 0 to 1400 eV. High resolution peak was used for the C 1s peak. A specific binding energy of 284.2 eV for the C 1s peak was used to correct any energy shifts resulting from surface charging effects. Casa XPS was used as curve-fitting software to fit the peaks.
Differential scanning calorimetry
Differential scanning calorimetry (DSC) was used to determine the glass transition temperature (Tg) with a TA Instruments (TA Q10). Small specimens weighing 20 to 30 mg were placed in aluminum pans and the following procedure was applied: heating–cooling–heating cycles between 25°C and 200°C at a rate of 10°C/min. Two heating runs were performed on each specimen to assess how water affects the mobility of polymer chains, specifically looking at any reversible effects, such as plasticization.
Dynamic Mechanical Analysis
The storage modulus (E′) and tangent of the phase angle (Tan δ) of the nanocomposite samples (before and after conditioning) were recorded using dynamic mechanical analysis (DMA) with a PerkinElmer DMA D8000 equipped with a three-point bending device between 25°C and 180°C. Five samples of each batch were tested and the average values were reported.
Tensile test
Sample tensile properties were determined according to ASTM 638 with a ZwickRoell z050 tensile testing machine. The cross-head speed of the machine was set to 1 mm/min and the cross-head load was 5 kN. Five samples of each composite were tested, and the average values were reported.
Moisture absorption
Water absorption measurements were carried out based on ASTM D 570 by immersing samples in deionized water at 50°C. Five specimens with an average dimension of 20 mm × 20 mm weighing 1.4 to 1.7 g. The samples were periodically removed from water, dried, and immediately weighed until they achieved a constant mass, that is, full saturation. Equation (1) was used to calculate the moisture uptake, where %M represents the mass gain, M
cond
the mass after conditioning, and M
dry
the mass dried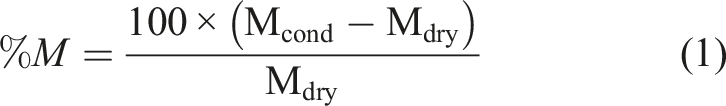
To calculate the coefficient of diffusion, the mass gain was plotted over time, according to Fick’s theory of diffusion. Equation (2) was used for short time periods, where D indicates the coefficient of diffusion, L is the thickness, and t is the time, Mt represents the percentage of water absorbed at time t; M∞ stands for the mass gain at equilibrium27,28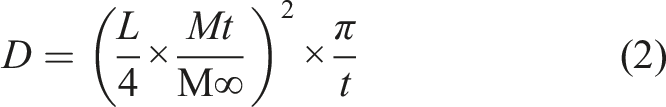
Scanning electron microscope
Cross sections were investigated using a Hitachi S-3000N (backscattering mode at 10 kV). All specimens were cut, polished, and sputtered with a gold-platinum alloy prior to analysis.
Sample conditioning
Samples were immersed in a NaOH solution with a concentration of 3.2 g/L (pH = 12.9) at 50°C. The conditions were applied to simulate practical use conditions based on a chemical resistance guide published by Owens Corning. After a period of 100 days, which corresponds to the maximum point of moisture absorption, the samples were taken out, dried, and tested.
Results and discussion
Oxidation of carbon nanotubes
Elemental analysis was performed on both AR and OX to verify the oxidation process and assess the oxygen content. It revealed that the pristine CNTs displayed an oxygen content of 0.1 wt%. According to the data, the oxidation with nitric acid and hydrogen peroxide resulted in further groups that contain oxygen, which were introduced on the surface of the carbon nanotubes. The weight percentage of oxygen contents in the oxidized CNTs was determined to be 0.83% through the CHNS-O elemental analysis, which was approximately eight times greater than the oxygen content found in the as-received one.
Furthermore, the XPS spectra of the first layer of the materials showed the presence of carboxylate groups on the oxidized CNTs. Figure 2 presents the spectra of AR (a) and OX (b). In the case of OX, a high resolution scan of the C1s main peak was performed: four distinct components at energy levels of 284.2, 285.2, 286.6, and 289.1 were observed. The main peak at 284.2 eV was related to sp2 hybridized carbon atoms, and the peak at 285.2 was attributed to sp3 hybridized carbon atoms. The two peaks at 286.8 and 289.1 were attributed to the presence of C-O and COO- groups, respectively. Table 2 provides the relative percentage of carbon atoms in the CNTs and oxidized CNTs samples. The results show a 6% content of carboxylate groups. The collected data is consistent with Liu et al.’s (2014) research on using oxidized CNTs for drug delivery purposes .
29
XPS spectra of (a) pristine CNTs and (b) oxidized CNTs. Relative percentages of carbon atoms in the CNTs and oxidized CNTs.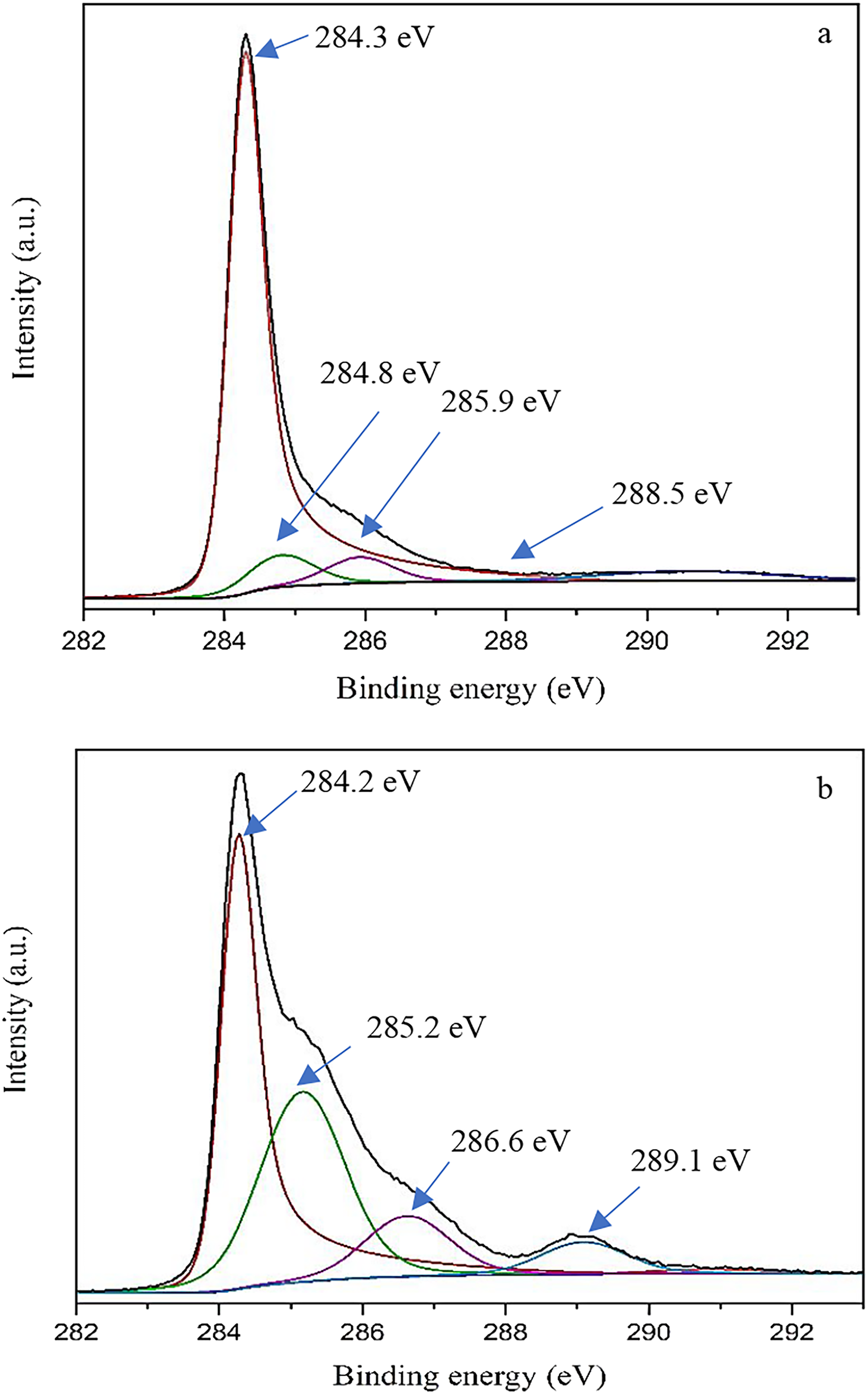

SEM micrographs
Based on past studies,
14
the addition of optimized amounts of dispersing agents can help to obtain a favorable dispersion and the long-term stability of CNTs within vinyl-ester resin. In fact, the dispersion process is challenging due to the presence of Van der Waals forces between CNT bundles,
30
a mismatch in chemical properties, and poor interfacial adhesion between CNT particles and VE resin. The fracture surface of the samples was examined to gain further insight into the morphology of vinyl-ester reinforced CNT composites. Figure 3 shows the SEM micrographs of VE/AR (a, b), VE/AR/AG (c, d), and VE/OX/AG (e, f). According to the micrographs, nanotubes appear as bright spaghettis in a dark polymer matrix. The dispersion of CNTs was poor in the VE/AR sample, since some agglomerates are visible. While additional mixing could improve the dispersion, the high energy of the process might also damage or shorten the CNTs. Voids were detected in the VE/AR composite. These voids—probably generated by a post-curing effect—might reduce the composite’s ductility. Studies indicate that a thermal residual stress might be generated during the post-curing process at high temperatures. This can create voids and microcracks in the nanotube agglomerates, diminishing the material’s mechanical properties.
31
Furthermore, the poor dispersion of MWCNTs was obvious when no appropriate dispersant was used. The VE/AR/AG composite provided ideal dispersion of nanoparticles in the matrix. The incorporation of BYK 104 S and BYK 9076 in the mixture not only increased the compatibility but also stabilized the dispersed particles and improved the interfacial adhesion of the nanoparticles and matrix.
32
In the case of VE/OX/AG, the hydroxyl group of the vinyl ester interacted with carboxylic acid groups on the surface of the oxidized CNTs and created chemical bonding. This interaction has a positive effect on the stress transfer mechanisms within the material, leading to a significant increase in its tensile strength.
33
SEM micrographs of VE/AR (a, b), VE/AR/AG (c, d), and VE/OX/AG (e, f).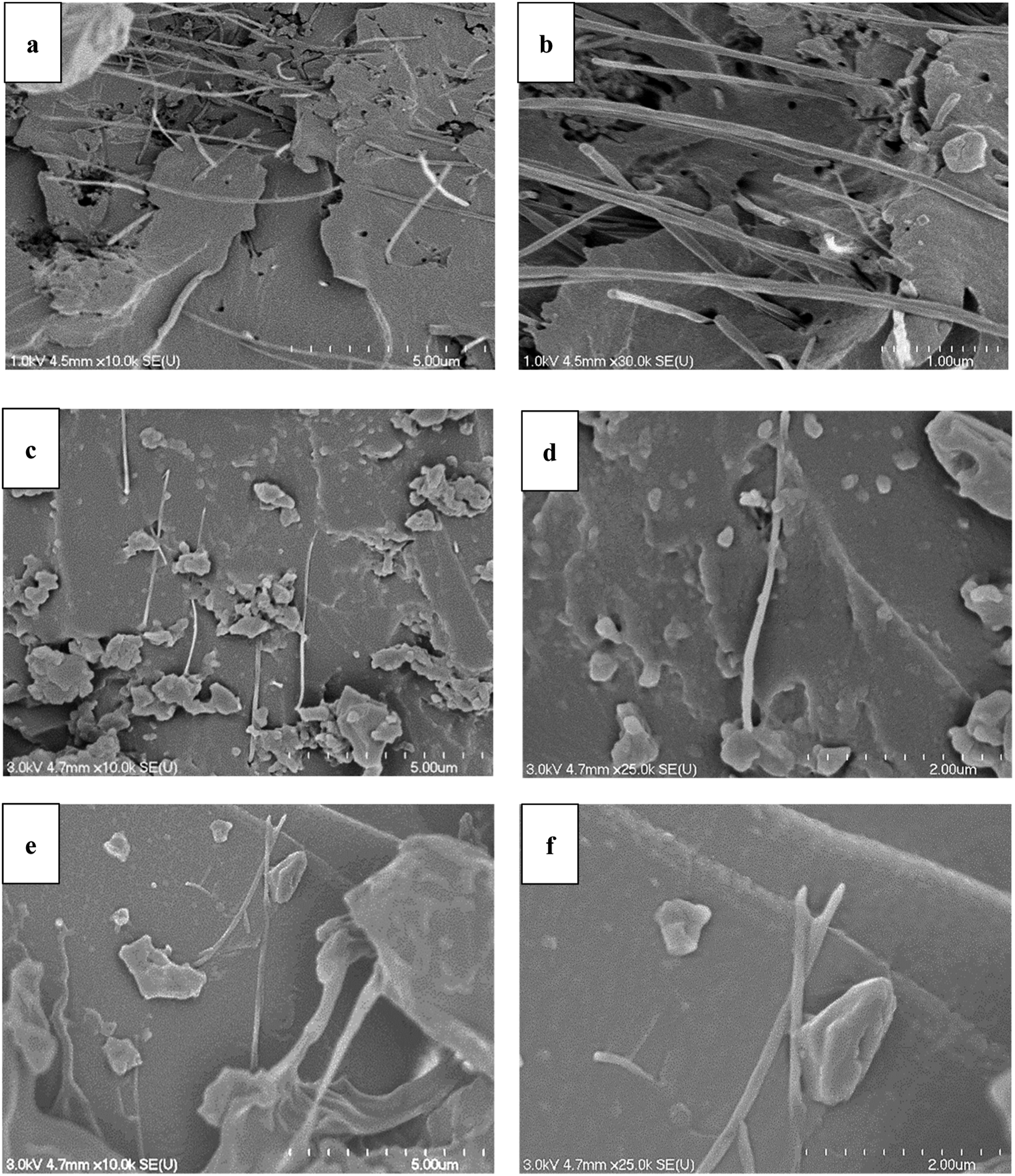
Tensile testing measurements
Mechanical properties of samples with and without dispersing agents.

In the next step, the tensile strength (б) and Young’s modulus (E) of the nanocomposites were measured to investigate the effect of adding CNTs and oxidized CNTs before and after conditioning in alkaline solution (Figures 4 and 5). Before conditioning was applied, the incorporation of 0.1 wt% of oxidized nanotubes increased the tensile strength and Young’s Modulus by 43% and 1.4 times, respectively, due to the improvement of the interface. The functionalization of nanotubes leads to the formation of covalent bonds between the nanoparticles and matrix, which restricts chain mobility and enhances the stiffness and tensile strength.
36
Ultimate tensile strength of samples before and after conditioning. Young’s modulus of samples before and after conditioning.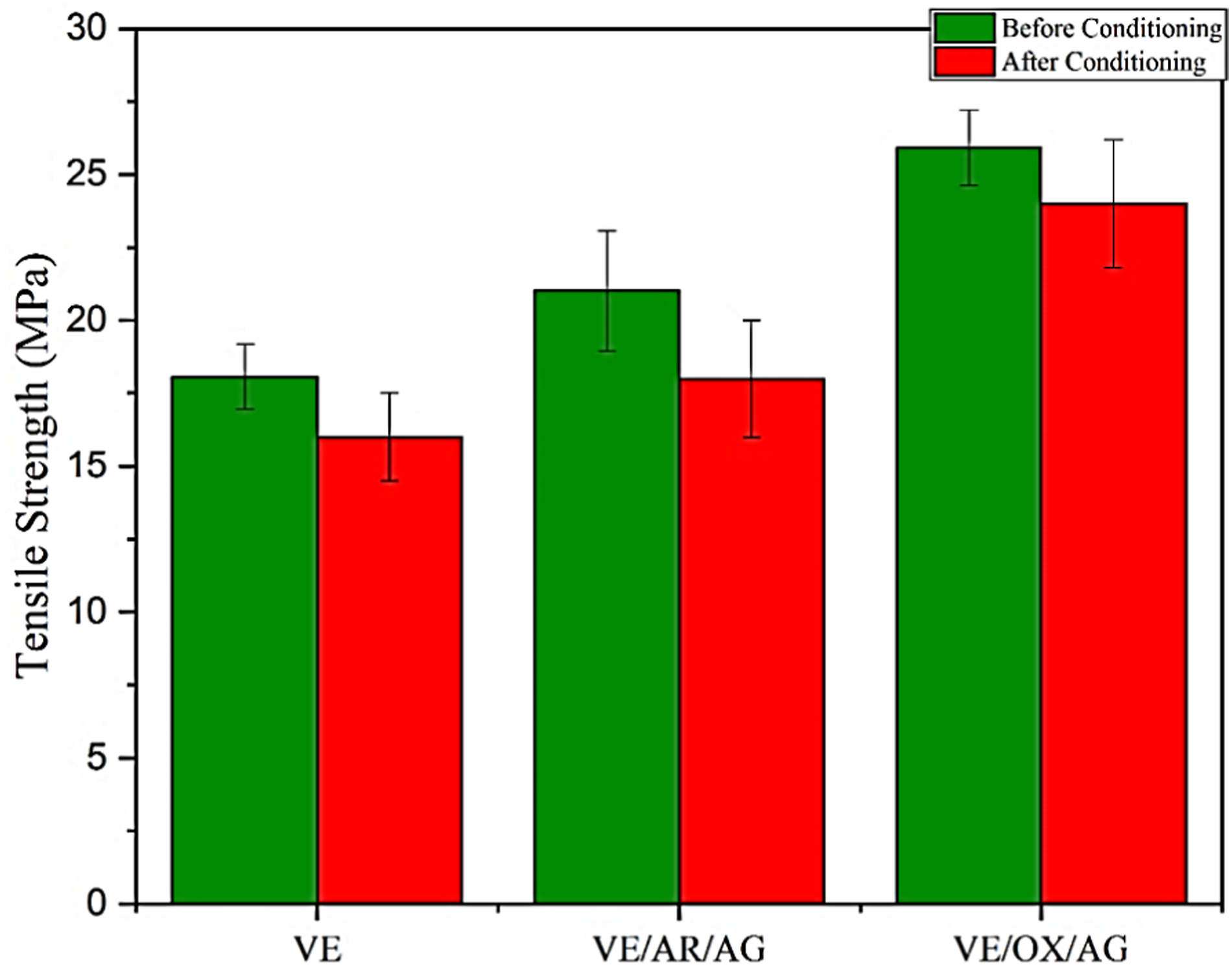
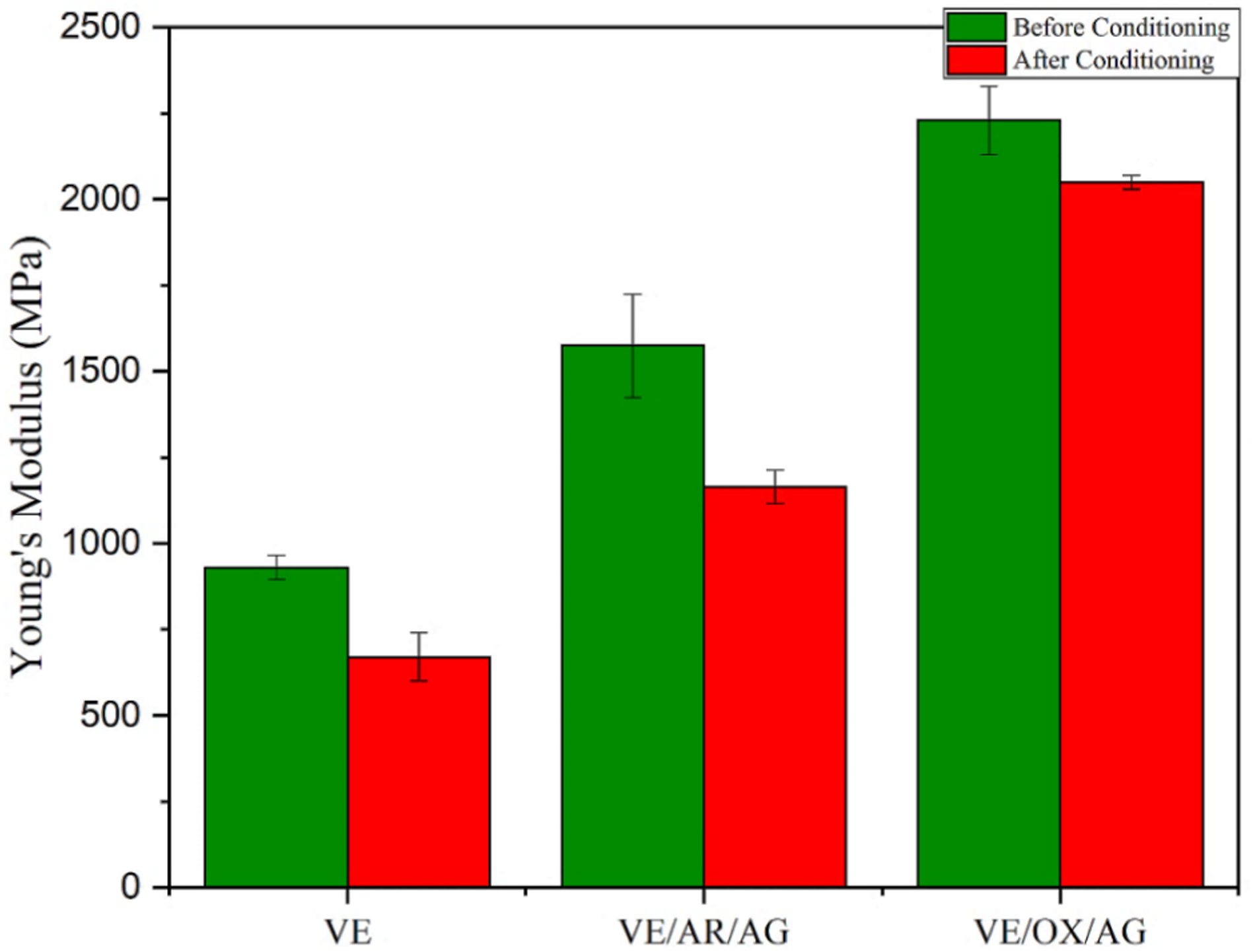
After the samples were conditioned, the tensile strength and Young’s modulus were significantly affected by the alkaline environment. The plasticization of VE—with or without carbon nanoparticles—caused matrix deterioration, resulting in the increase of porosity and stress concentration. The results show that the tensile strength of samples was reduced by 4% to 14%. The most reduction in ultimate tensile strength (UTS) was observed with the VE and VE/AR/AG composites, while the least was observed with the VE/OX/AG composite. Moreover, the stiffness of the VE and VE/AR/AG composites, related to Young’s modulus, decreased by 28% and 26%, respectively. This reduction can be related to the absorption of water and NaOH molecules by the material, which might have weakened the interfacial adhesion. Additionally, the occurrence of hydrolysis, swelling, or plasticization phenomena in the VE resin further contributed to the reduced stiffness.37,38 No notable decline in stiffness was observed in the case of the VE/OX/AG composite.
Dynamic mechanical properties
DMA results before and after conditioning.
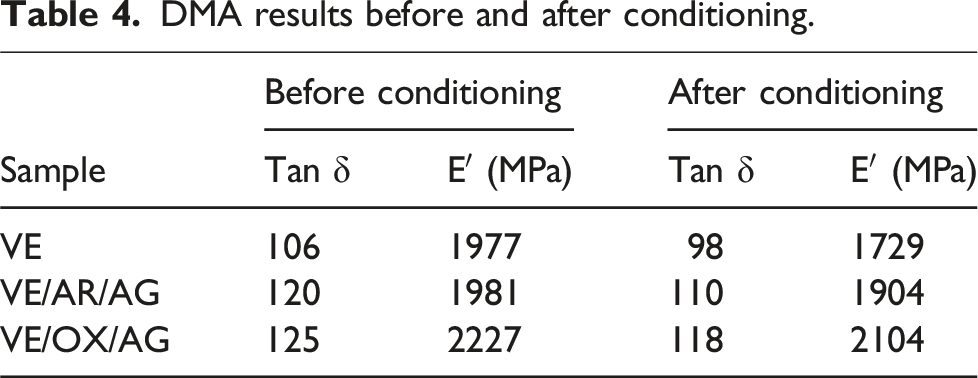
Figure 6 gives the storage modulus versus temperature before conditioning. As shown, the resin’s thermal stability increased with the addition of CNT and CNTO particles. This also improves the interfacial adhesion between the resin and particles. Since the nanoparticles have a high aspect ratio of the nanoparticles, there is more contact with the matrix, which strengthens the interface. Storage modulus versus temperature diagram.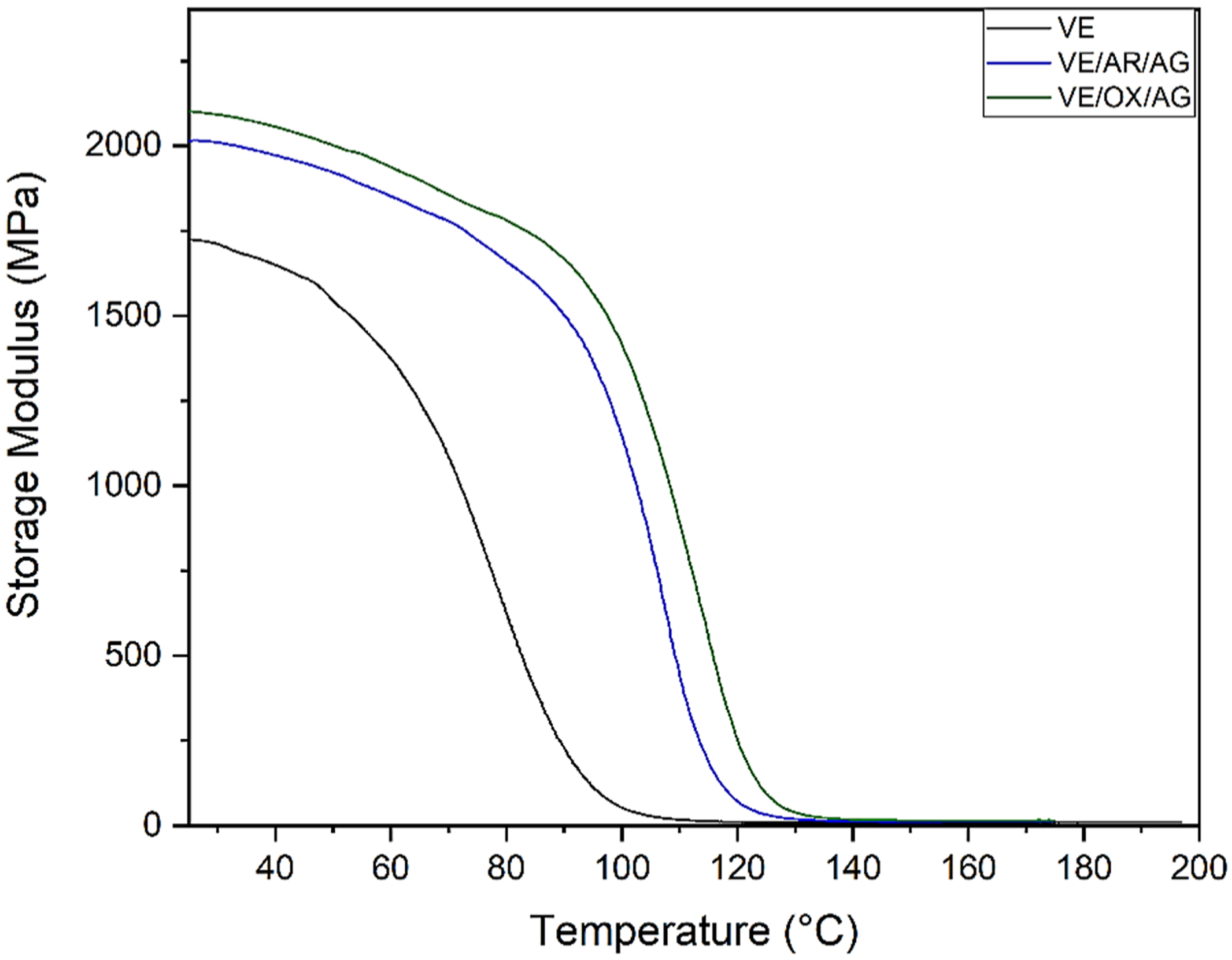
After conditioning, the Tan δ and E′ of VE, VE/AR/AG, and VE/OX/AG composites exhibited decreases ranging from 5% to 8% and 4% to 13%, respectively. The lower reduction for the VE/OX/AG sample can be attributed to the barrier effect of functionalized CNTs, which mitigates moisture ingress and improves the compatibility and interfacial adhesion between the particles and resin. These factors contribute to a lower drop in the mechanical properties of the samples containing oxidized CNTs, which is consistent with the outcomes obtained from the tensile testing analysis.
Moisture absorption
Vinyl ester can absorb up to 1.6% water, which causes plasticization and decreases Tg. Figure 7 and Table 5 show the water uptake versus the square root of time, which corresponds to typical Fickian behavior. Three critical parameters—water absorption rate (v), moisture absorption at saturation (M∞), and diffusion coefficient (D)—were calculated. Moisture absorption of VE, VE/AR/AG, and VE/OX/AG. Diffusion coefficients, absorption rates, and moisture contents at equilibrium.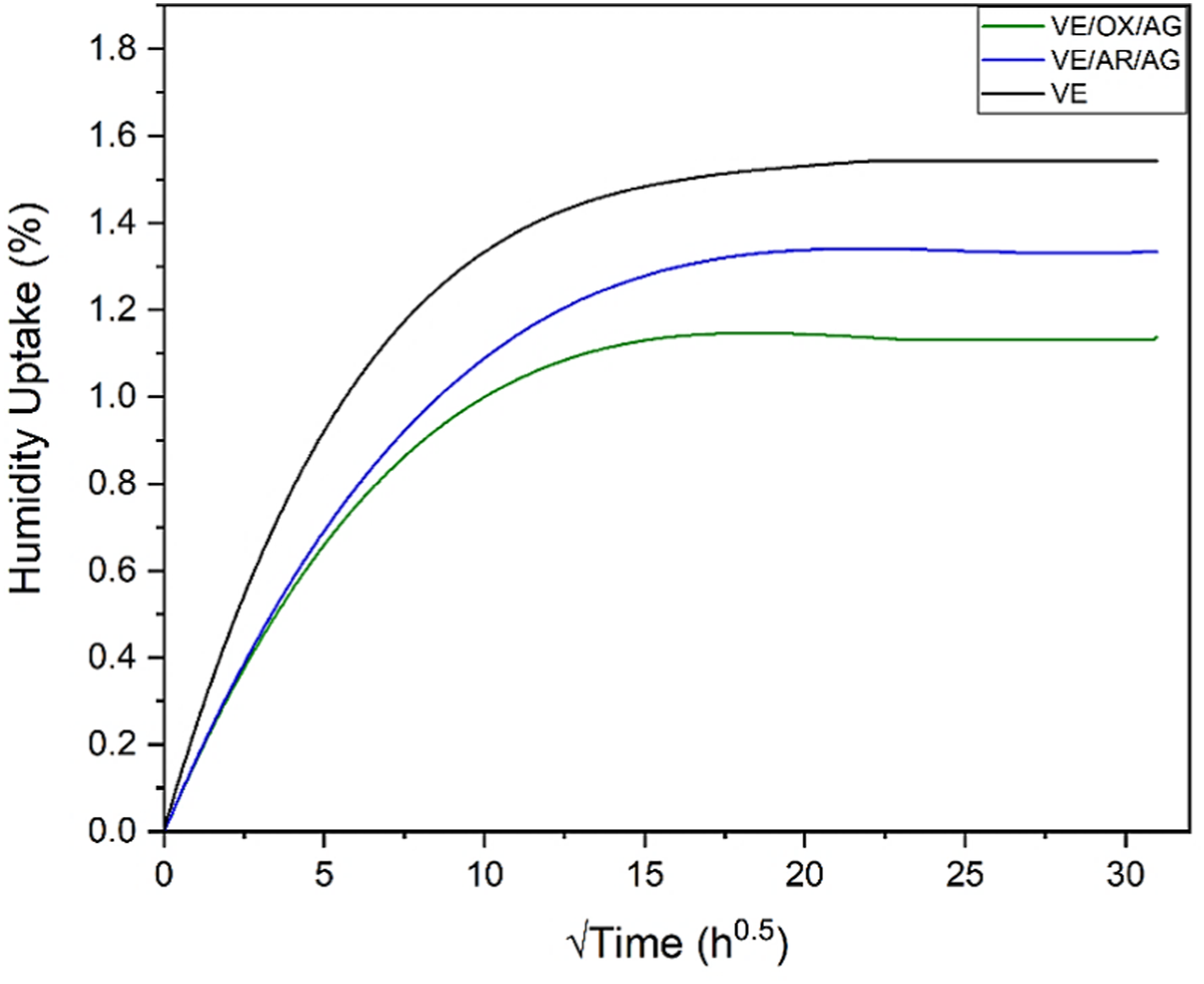

The rate of water absorption can be shown as the slope of the curves during the preliminary stage (Figure 8). Indeed, it represents the initial rate of diffusion of water molecules in the material. The addition of low amounts of CNTs and CNTOs decreased the water absorption rate by 21% and 26%, respectively. In both composites, the nanoparticles acted as water barriers by creating a tortuous pathway within the resin, which decreased the speed of water molecules during penetration, ultimately improving the water resistance parameter. To be more specific, nanoparticles are extremely small in size with limited spaces between them. This forces water molecules to move through microscopic channels. The speed at which water penetrates the material is significantly impeded by this inherently restricted movement and consequently, the overall penetration of water molecules is delayed.
36
Evolution of the diffusion coefficient over time.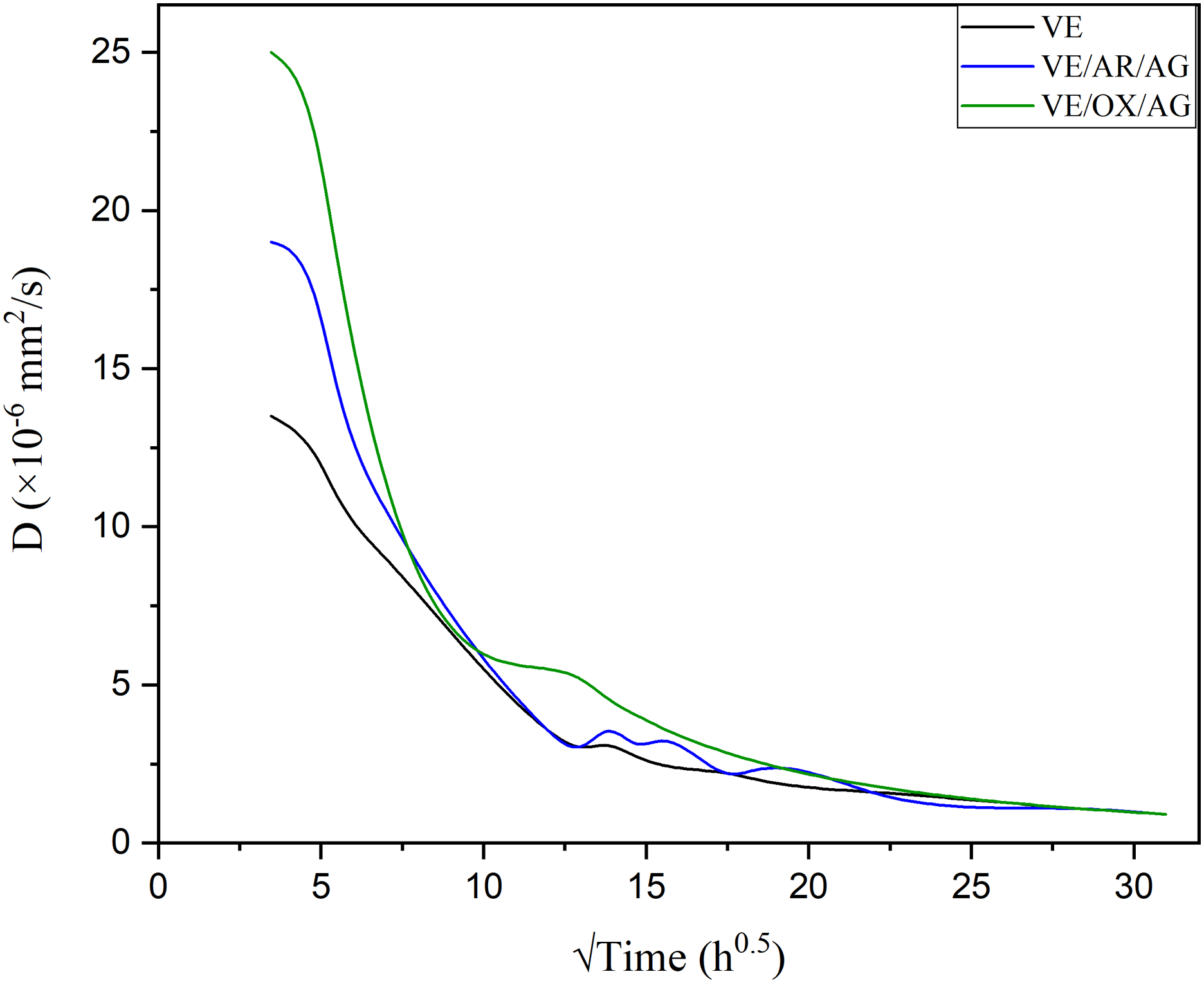
The moisture absorption at saturation dropped by 14% and 26% due to the presence of nanotubes and oxidized nanotubes, which are intrinsically hydrophobic and repel water. The presence of nanoparticles effectively boosted the resin’s stiffness while simultaneously enhancing chain mobility within the polymer. This dual effect limits the space for water molecules to enter the composite, thereby reducing the material’s capacity for molecular rearrangement. On the other hand, the improved resin/particle interface formed a physical and chemical barrier to prevent water molecules from entering the composite. Another noteworthy consideration is the role of dispersing agents. It can be hypothesized that dispersing agents can contribute to reducing the affinity for moisture absorption. Both BYK 9076 and BYK 104S have hydrophobic properties. The hydrophobic tail of these agents can interact with the polar functional groups on the surface of the oxidized CNTs, while the hydrophobic parts of the agents tend to interact with the VE matrix, leading to the formation of a more continuous network and reduction of pathways for moisture ingress. 34
Table 5 gives the coefficient of diffusion. In general, there was an inverse relationship between the moisture absorption at saturation and the diffusion coefficient. 40 More specifically, the water molecules moved slowly through the material when it had a lower coefficient of diffusion. In this case, the material took longer time to reach equilibrium. The material might absorb more moisture through this prolonged period, increasing the moisture absorption at saturation. The initial high D values for the VE/AR/AG and VE/OX/AR composites are 12.9 and 16.2 10−6 mm2/s, respectively, attributed to the saturation of carbon-based particles being more hydrophilic than pure resin (9.85 × 10−6 mm2/s). As the coefficient of diffusion is a measure of the rate at which a substance diffuses from a high-concentration to a low-concentration area, it decreased with time until the saturation point was reached. In addition, this parameter was divided into two steps: (1) a sharp decline during the initial period (t < 100 h) and (2) a homogenous diffusion after (t > 200). The trend of D after 200 h immersion was consistent and close to that of pure resin. 27
To sum up, moisture absorbed by the resin can cause swelling, hydrolysis, and plasticization, ultimately negatively affecting durability properties. Adding non-functionalized and functionalized carbon nanotubes to the resin, however, can prevent these effects without impacting the initial diffusion rate or the value of D over extended periods of time.
Thermal properties before and after conditioning (DSC)
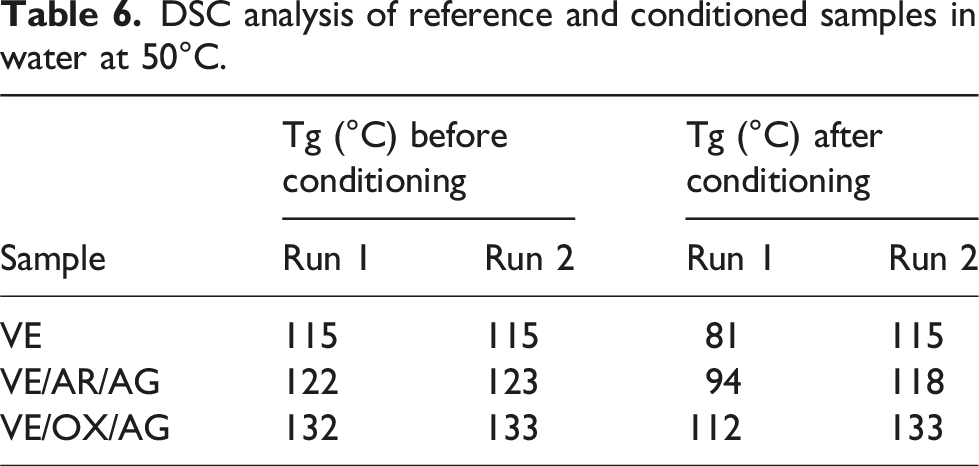
DSC analysis of reference and conditioned samples in water at 50°C.
Conclusion
In this study, CNT particles were functionalized through wet acid oxidation. Nanocomposites were prepared by adding CNTs and oxidized CNTs at 0.1 wt% to vinyl-ester resin. Two dispersing agents (BYK 9076 and BYK 104 S) were added to improve the dispersion and compatibility. The moisture diffusion and chemical degradation of the samples were studied by conditioning in water or alkaline solution at 50°C.
SEM micrographs revealed significant improvements in the dispersion and interfacial adhesion of samples containing CNTs and oxidized CNTs in combination with dispersing agents. Tensile and DMA tests were conducted to further characterize the effect of these additives on the vinyl ester. The results show that the tensile strength and Young’s modulus increased by 43% and 1.4 times, respectively, for composites containing oxidized CNTs. Upon conditioning, the stiffness values of VE and VE/AR/AG composites decreased by approximately 26% to 28%, which can be related to their higher hydrophilicity than the VE/OX/AG. Remarkably, the latter showed good protection and stable mechanical properties. DMA test results support these findings.
Moisture diffusion and DSC analyses were performed to investigate the barrier properties and reversibility of Tg changes. Oxidized CNT-based composites exhibited the lowest water uptake at equilibrium and the lowest decrease in Tg compared to the other samples immersed in water. This suggests that the presence of oxidized CNTs limited matric plasticization.
In conclusion, the incorporation of oxidized CNTs as reinforcement in PNC applications holds great promise. Despite their low content of only 0.1 wt%, which is notably less than conventional additives (10 times or more), oxidized CNTs lead to significant enhancements in mechanical, thermomechanical, durability, and barrier properties.
Footnotes
Acknowledgments
The authors express their gratitude to the National Science and Engineering Research Council (NSERC) of Canada for financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
