Abstract
Background
The gut microbiome has been proposed as a potential modifiable target to treat mental illness. This double-blind randomized control trial investigated fecal microbiota transplant (FMT) in bipolar disorder (BD) to assess efficacy, safety, and feasibility. The primary outcome evaluated the effectiveness of standard approved therapy for BD depression + FMT in individuals not responding to standard treatment, measured by change in the Montgomery-Åsberg Depression Rating Scale (MADRS) score from baseline to week 24. Secondary outcomes included FMT's impact on anxiety, global function, side-effects, and safety. The feasibility of this novel intervention was also assessed. Microbial analysis utilized whole-genome shotgun metagenomic sequencing, comparing outcomes between allogenic (donor) and autologous (participants own) FMT.
Methods
A total of 35 participants (28 women and 7 men) with at least moderate depressive-phase BD (MADRS) were randomized to receive either allogenic FMT (n = 17) or autologous FMT (n = 18) via colonoscopy and were followed for 24 weeks.
Results
MADRS scores significantly improved from baseline to the last visit in both treatment arms. There was no significant difference between allogenic FMT (16.74-point improvement) and autologous FMT (15.4-point improvement) regarding clinical efficacy (t = −0.47, p-value = .64, 95% confidence interval [CI] = −7.3–4.6). Microbiota analysis showed that allogenic FMT let to a bacterial profile similar to the healthy donor and increased bacterial diversity at the 6-month mark, whereas those receiving autologous FMT did not. The intervention was well tolerated with no significant adverse events. Recruitment, randomization, and retention metrics support feasibility of a larger trial.
Conclusion
Feasibility and tolerability data indicate further investigation into microbial manipulation in BD is warranted. The absence of efficacy differences between the two types of FMT, despite microbial change, highlights the importance of a true placebo in future studies, as well as the importance of understanding exactly what bacteria are linked to improvements. ClinicalTrials.gov, NCT0327922
Plain Language Summary Title
Results of a Double-Blind Randomized Control Trial Investigating Fecal Microbiota Transplant (FMT) as an Add-on Treatment for Depression in Bipolar Disorder and Analyzing Microbial Diversity Changes Over 24 Weeks
Plain Language Summary
This study was a proof-of-concept, double-blind randomized control trial (RCT) that investigated Fecal Microbiota Transplant (FMT) as a modifiable target to treat mental illness, specifically focusing on depression in bipolar disorder (BD). The trial assessed the efficacy, safety, and tolerability of combining FMT with approved BD depression therapy in individuals not responding to standard treatment. Participants (35 total: 28 women and 7 men) were 18 years or older and presented in the depressed phase of BD of at least moderate severity, as measured by the Montgomery-Åsberg Depression Rating Scale (MADRS). They received either FMT from a healthy donor (allogenic, n = 17) or their own stool (autologous, n = 18) via colonoscopy and were followed for 24 weeks. Secondary measures included assessing anxiety via the Stait-Trait Anxiety Index and global function via the World Health Organization Brief Quality of Life Scale.
Introduction
Bipolar disorder (BD) is a serious mental health condition that causes dramatic shifts in mood with an estimated lifetime prevalence between 2 and 3 per cent. 1 While BD is diagnosed by the presences of episodes of both mania and depression, the depressive phase lasts significantly longer and can account for up to 72 per cent of the time during which a person is ill. 2 It is also associated with higher mortality. 3 Drug therapy remains the cornerstone treatment for BD and is mainly based on serendipitously discovered drugs that often display limited efficacy and poor tolerability, often repurposed from the treatment of other illnesses. 4 A recent review of treatments for the depressive phase of BD found no moderate or high evidence for any treatment, 5 thus alternative treatment options and investigations are warranted.
The microbiota-gut brain axis (MGB) axis is a bidirectional pathway between the brain and microbiota in the gastrointestinal system. 6 It is comprised of humoral, neural, and cellular pathways that ultimately connect the gut microbiota with the central nervous system (CNS). That the gut microbiota can impact the symptoms of mental illness through a variety of purported mechanisms has been extensively researched and work has been done to evaluate this connection as a potential therapeutic target via fecal microbial transplant (FMT) or probiotics.7-11 A growing body of literature has found that individuals with BD have alterations in the gut microbiome composition or function and that these alterations are associated with illness severity in BD.12-16 This has resulted in the intriguing possibility of MGB axis being a possible treatment target. The probiotic literature for mental illness is mixed, 17 however, in part because probiotics are not designed to change the gut microbiome. 18 FMT may offer a better biotherapeutic option as evidence suggest it may result in more long-term effects on the MGB axis.19,20
We therefore conducted a proof-of-concept randomized controlled trial (RCT) of the safety and efficacy of FMT in a population with BD depression (BDd), as well as to determine feasibility of doing a larger trail, a key consideration when evaluating novel interventions. The acceptability of FMT seems to vary across illness,21,22 but in the only published FMT RCT in depression, all feasibility targets were met or exceeded. Our own research regarding patient willingness to try microbiome-based therapies also highlighted interest in therapies targeting the gut in those with a mood disorder. 23 To our knowledge there are no published RCTs examining the use of FMT for BDd and this study is the first of its kind.
The primary objective was to examine the efficacy of adjunct FMT for depressive symptoms in individuals with BDd taking, but not responding to, standard treatments, comparing allogenic FMT (donor stool) to a control group receiving autologous FMT (an individual’s own stool). Secondary objectives were to examine the efficacy of FMT on anxiety and global function, as well as to look at the acceptability, safety, and tolerability of FMT in this population. Feasibility was also examined, as this study will inform the development of a larger multisite RCT and further our understanding of utilizing FMT for treating depression in patients with BD. The acceptability of FMT seems to vary across illness,21,22 but in the only published FMT RCT in depression, all feasibility targets were met or exceeded. Our own research regarding patient willingness to try microbiome-based therapies also highlighted interest in therapies targeting the gut in those with a mood disorder. 23 To our knowledge there are no published RCTs examining the use of FMT for BDd and this study is the first of its kind.
Methods
Study Design and Participants
This study was a phase 2/3, double-blind RCT. All participants with BD on, but failing to respond to, first-line treatment were given a medical grade bowel preparation and then randomized to receive either (1) allogenic FMT from healthy donors or (2) autologous FMT with their own feces by colonoscopy. FMT was manufactured into a liquid filtrate and administered via colonoscopy by a trained gastroenterologist. Participants were subsequently followed for 24 weeks for assessment of primary and secondary study outcomes. The study involved collaboration of three Canadian academic health centres affiliated with the University of Toronto in Toronto, Ontario, with recruitment and follow-up occurring at Women's College Hospital (Research Ethics Board [REB] # 2017-0099-B), FMT preparation occurring at Sinai Health (REB# 17-0289-E) and FMT administration occurring at University Health Network, Toronto Western Hospital (REB#17-5147). The study received a No Objection Letter from Health Canada (HC6-0240C201262). The trial is registered with ClinicalTrials.gov, number NCT03279224.
We recruited adults 18 to 65 years of age, who had a Mini-International Neuropsychiatric Interview 24 confirmed diagnosis of BD (type I or II) on a stable first-line treatment 25 for ≥8 weeks prior to study entry, who were currently experiencing a depressive episode. A participant met inclusion criteria if their current episode was characterized by a Montgomery–Asberg Depression Rating Scale (MADRS) 26 score at screening and baseline of ≥12. Exclusion criteria included a Young Mania Rating Scale (YMRS) 27 score of ≥12 at screening; Diagnostic and Statistical Manual of Mental Disorders IV (DSM-IV) criteria 28 for substance abuse within the past 6 months or lifetime dependency; active suicidal ideation, an active eating disorder; schizophrenia or schizoaffective disorder; chronic gastrointestinal diseases; conditions causing immunosuppression; a significant bleeding disorder; a history of incomplete colonoscopy; pregnancy, regular used of non-steroidal anti-inflammatory drugs, antibiotics, prebiotics or probiotics for medical purposes, or iron supplements within 3 months prior to study entry.
To help achieve adequate participant enrollment, flyers were placed in the community at public venues and at the participating academic hospitals. Family physicians and psychiatrists providing clinical care at those sites were informed of the study to assist with identification of potentially eligible participants and ads were also placed on social media. The healthy donors who provided the allogenic FMT were recruited as part of the Microbial Therapeutics Outcome Program (MTOP) at the University of Toronto using procedures described previously. 29
Randomization and Masking
Participants were randomly allocated (1:1) to groups receiving either allogenic or autologous FMT. Randomization occurred according to a blocked randomization list (4 patients per block), generated in Statistical Analysis System (SAS) Procedure PLAN (proc PLAN). Blinding was maintained for all participants and study personnel, apart from the designated lab technologist who handled FMT preparation.
Study Procedures
FMT administration was delivered via colonoscopy after a standard bowel cleanse consisting of 4 L of an over-the-counter polyethylene glycol laxative solution (Table 1). FMT were manufactured and frozen following MTOP procedures, as previously described. 30
Participants Schedule of Events.
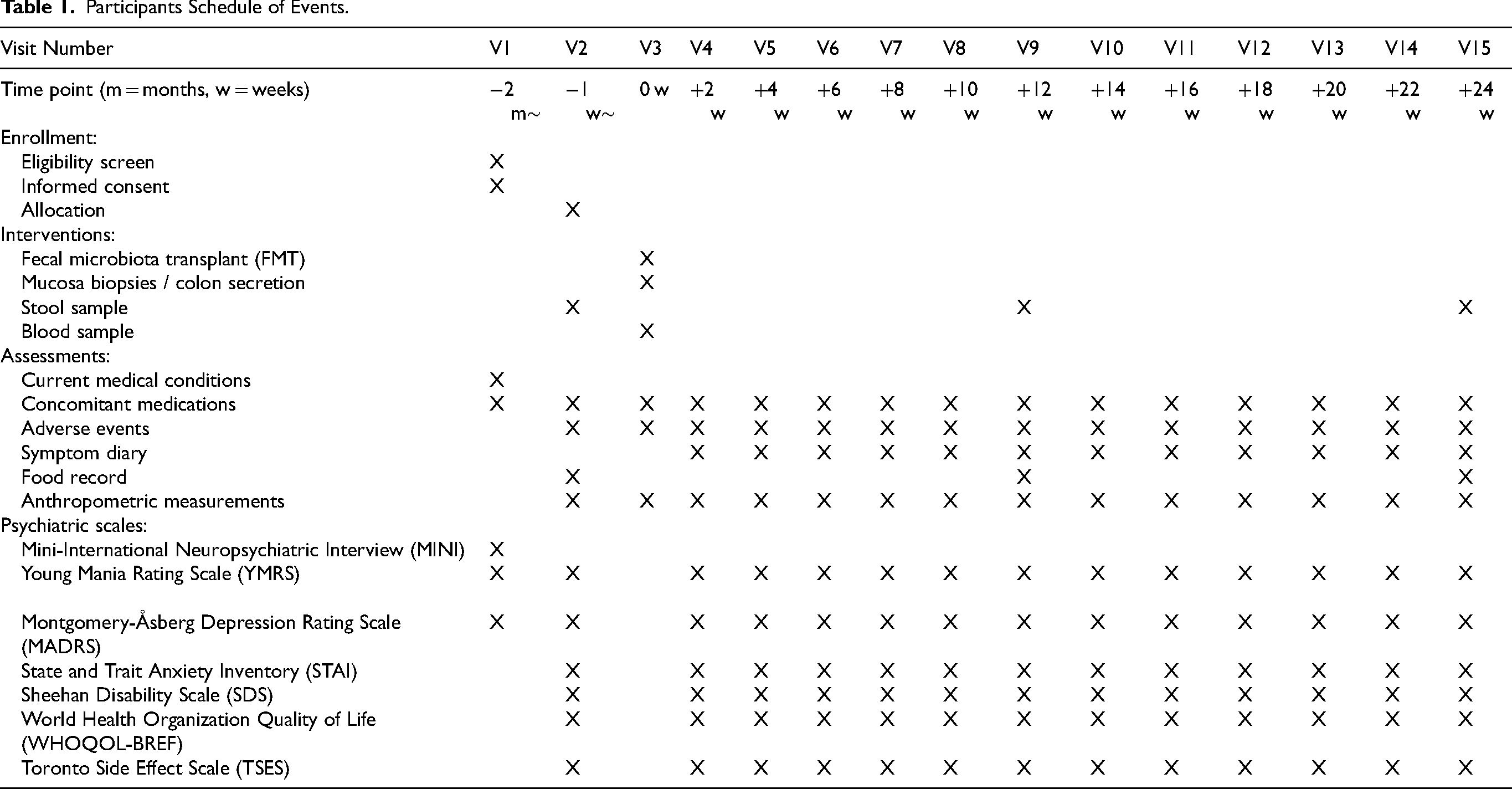
Briefly, 50 g of screened donor or participant feces was homogenized for 30 min using a Stomacher® Paddle Blender with 30 mL sterile 0.9% NaCl + 10% glycerol in a sterile, 330-μn microfilter-separated, double-compartment polyethylene bag. Fecal filtrate volume corresponding to 50 g of original feces was aliquoted, labeled with a recipient ID number to maintain blinding, and stored at −80 °C. Prior to colonoscopy, three falcon tubes of frozen, concentrated fecal filtrate, corresponding to a total of 150 g of feces was thawed, diluted to a total volume of 300 mL with 0.9 N NaCl and packaged into 6 × 50 mL syringes for colonoscopy. Colonoscopy was then performed by an experienced gastroenterologist with fecal filtrate in one syringe deposited in the terminal ileum and five in the cecum.
A MADRS score, a 10-item questionnaire use to measure the severity of depressive episodes in patients with mood disorders, was determined at baseline and every 2 weeks after dosing for a total of 24 weeks of follow-up. Secondary outcome measures were also measured at the same time points, as was concomitant medications use and anthropometric measurements (waist-to-hip ratio, and body mass index [BMI]). The secondary outcome measures included the YMRS, 27 an 11-item clinician-administered interview scale used to assess manic symptoms, the State Trait Anxiety Index (STAI), 31 a commonly used measure of anxiety and the Sheehan Disability Scale (SDS), 32 a brief self-report tool developed to assess functional impairment in work/school, social life, and home life or family responsibilities domains. The World Health Organization Brief Quality of Life Scale (WHOQOL-BREF), 33 a quality-of-life assessment designed to be applicable cross-culturally, was also administered.
To control for diet change, food intake was recorded for 3 days (2 weekdays and 1 weekend day), and the completed forms collected at baseline (week 1), week 12, and week 24. To minimize confounding effects, participants were asked to maintain their usual diet and exercise level. Participants also itemize their food intake using a validated form developed by the Fred Hutchinson Cancer Research Center. 34 A symptom diary to record adverse events was also provided and reviewed at every subsequent study visit. In addition, the Toronto Side Effects Scale (TSES), 35 a 28-item instrument designed to establish incidence, frequency, and severity of CNS, gastrointestinal, and sexual side effects was administered and weight changes were evaluated.
Outcomes
The primary outcome of this study is to determine the effectiveness, safety, and tolerability of FMT as an adjunctive treatment in adults with BD depression, as well as the viability of a larger trial. Feasibility was measured by participant recruitment, retention and completion rates, adherence to the intended study protocol, participant acceptability, and robustness of study methodology. Effectiveness, the primary clinical outcome, was measured by change in MADRS score from baseline to the end of follow-up. If participants received the intervention and follow-up data was available, they were assessed for the primary endpoint. We also assessed response, defined as ≥50 per cent reduction in MADRS scores from baseline and remission, defined as a MADRS score of ≤7. Changes in YMRS, STAI, WHOQOL, and the SDS between baseline and the primary endpoint were also assessed. Dietary data was analyzed using NUTRITIONIST PRO diet analysis software (Axxya Systems) to assess intake of macro- and micro-nutrients.
Safety was measured by the assessment of severe adverse events while Tolerability was monitored by the TSES, with each symptom rated for both frequency and severity on a five-point scale (1 = Never/No trouble; 5 = Everyday/Extreme trouble), as well as weight changes. Adverse events were also assessed via a symptom diary reviewed at each visit.
Stool Sample Collection
Participants receive instructions and two kits for collecting two stool samples at home. Each kit contains a plastic collection container for fresh stool and an OMNIgene•GUT tube (DNAgenotic™). Samples were collected at baseline prior to FMT and 3 and 6 months post-FMT. Samples were collected at home, and immediately stored in a home freezer and returned to the research facility within 8 h where they were stored at −80 °C.
Microbiome Analysis
Genomic DNA extraction and Illumina library preparation were performed as previously described in Shekarriz et al. (2025). 36
Metagenomics Processing Pipeline
Raw reads were filtered to remove low-quality reads and human-derived DNA using KneadData. The resulting high-quality, non-host reads were processed using the MetaPhlAn4 37 and HUMAnN3 38 pipelines for taxonomic and functional annotations, respectively. Taxonomic profiling was performed with MetaPhlAn4 (v4.0.4) using the -t rel_ab option to compute relative species abundances. Microbial pathway abundances in Counts Per Million (CPM) were obtained using HUMAnN3 (v3.5). Taxonomic profiles at the species level were merged using the “merge_metaphlan_tables.py” script, and diversity indices—including Shannon, Aitchison, and Bray-Curtis—were calculated with the “calculate_diversity.R” script from MetaPhlAn4.
Final taxonomic and functional profiles were analyzed and visualized in R (v4.1) using the Tidyverse library and custom scripts. The top 20 microbial families by relative abundance were visualized for all participant and donor samples using ggplot2. Taxa not among the top 20 families were aggregated and labeled as “Other” in the legend. Shannon alpha diversity and pairwise Bray-Curtis beta diversity metrics were compared between patients receiving autologous and allogenic FMT across different time points. Statistical comparisons were conducted using the Wilcoxon rank-sum test, with significance annotated via the “geom_signif()” function from the ggpubr package.
Sample Size
The purpose of the study was to confirm feasibility of a BDb FMT trials and to gain preliminary information about the magnitude of effect and distribution of outcome measures. Therefore, no formal sample size calculation was conducted and we initially aimed for 60 participants.39,40 A review by Hertzog suggests a range of 20 to 40 participants to allow for sufficient variability in acceptability assessment of an intervention and given challenges posed for recruitment by COVID, we therefore aimed to enroll within that range. 41
Statistical Analysis
We summarized demographic and baseline clinical characteristics overall and by randomization group with descriptive statistics. Feasibility was also reported descriptively. Change in MADRS score between baseline and last visit was assessed overall and in each group using paired t-tests. The primary efficacy analysis compared the mean change in MADRS score from baseline to last visit between allogenic and autologous FMT using an independent t-test. Response and remission rates were also examined with and without using the Last Observation Carried Forward (LOCF) method for missing data and compared between groups using chi-square tests. To further understand changes in depression over time and between groups, we fitted linear mixed models to account for the hierarchical structure of the MADRS scores. Fixed effects included group, time, a time-squared term to allow for non-linear change. Interaction terms of group by time and group by time-squared were also assessed. Random effects consisted of a random intercept and random slopes for time and time-squared, allowing patient-specific quadratic trajectories. A likelihood ratio (LR) test with maximum likelihood was used to confirm the necessity to include the time-squared fixed effect. The participant-specific quadratic random effect of time was added based on a significant LR test result with restricted maximum likelihood. 42 The secondary outcomes included the YMRS, STAI, WHOQOL, and SDS. All were assessed for changes from baseline to last visit and with a comparison between groups. For tolerability comparison, the prevalence and proportion of participants reporting side-effects and adverse events were calculated and compared with Wilcoxon rank-sum test, Pearson's chi-square test, and Fisher's exact test. Adverse effects were assessed using the TSES with each symptom rated for both frequency and severity severity and compared within groups. Mean change of frequency and severity ratings from baseline to final visit were compared between groups with t-tests. All analyses were conducted using R version 4.3.1.
Metagenomics Analyses
To identify differentially abundant microbial taxa and metabolic pathways at mid-point (week 12) and endpoint (week 24), taxonomic and functional profiles generated by MetaPhlAn4 and HUMAnN3, respectively, were analyzed. MetaPhlAn4 outputs relative abundance values, while HUMAnN3 reports pathway abundances in Reads Per Kilobase of transcript per Million mapped reads (RPKM) units; these were used as input data for linear models that were implemented in R (v 4.4.1). For univariate analyses of temporal changes, we fit separate linear models for each species and pathway using the formula Abundance ∼ Time point within either allogenic or autologous FMT group, with time point treated as a categorical variable and baseline (week 1), prior to FMT, as the reference. Coefficients, P-values, and false discovery rate (FDR)-adjusted P-values (<0.05) were extracted to evaluate differences at 12 and 24 weeks relative to baseline.
Role of the Funding Source
The funder of the study (Stanley Medical Research Institute) had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Results
From January 2018 to September 2021, 75 participants with BD were screened for enrollment, of whom 31 (41.3%) were ineligible or declined to participate, resulting in 44 participants being enrolled. An additional 9 participants withdrew or were deemed ineligible prior to randomization. Thirty-five participants were randomly assigned to receive one of the two study interventions: allogenic fecal microbiota transplantation (n = 17, comprising 13 individuals with bipolar disorder I [BD I] and 4 with bipolar disorder II [BD II]) or autologous FMT (n = 18, comprising 15 individuals with BD I and 3 with BD II). In the allogenic group, one participant withdrew prior to treatment allocation, and one withdrew after receiving FMT but prior to providing a follow-up visit. Of those receiving the intervention and a minimum one follow-up visit (n = 15), one participant was excluded at week 8 (secondary to antibiotic use), one was lost to follow-up after week 2 and another after week 16 while another was unblinded due to medical reasons unrelated to the study at week 22. In the autologous group, one participant withdrew prior to treatment allocation. Of those receiving the intervention and a minimum one follow-up visit (n = 17), another withdrew after week 4 secondary to personal reasons. Twenty-seven participants completed the entire follow-up period. All participants who received the intervention and had at minimum one data point (n = 32) were included in the final analysis (Figure 1).
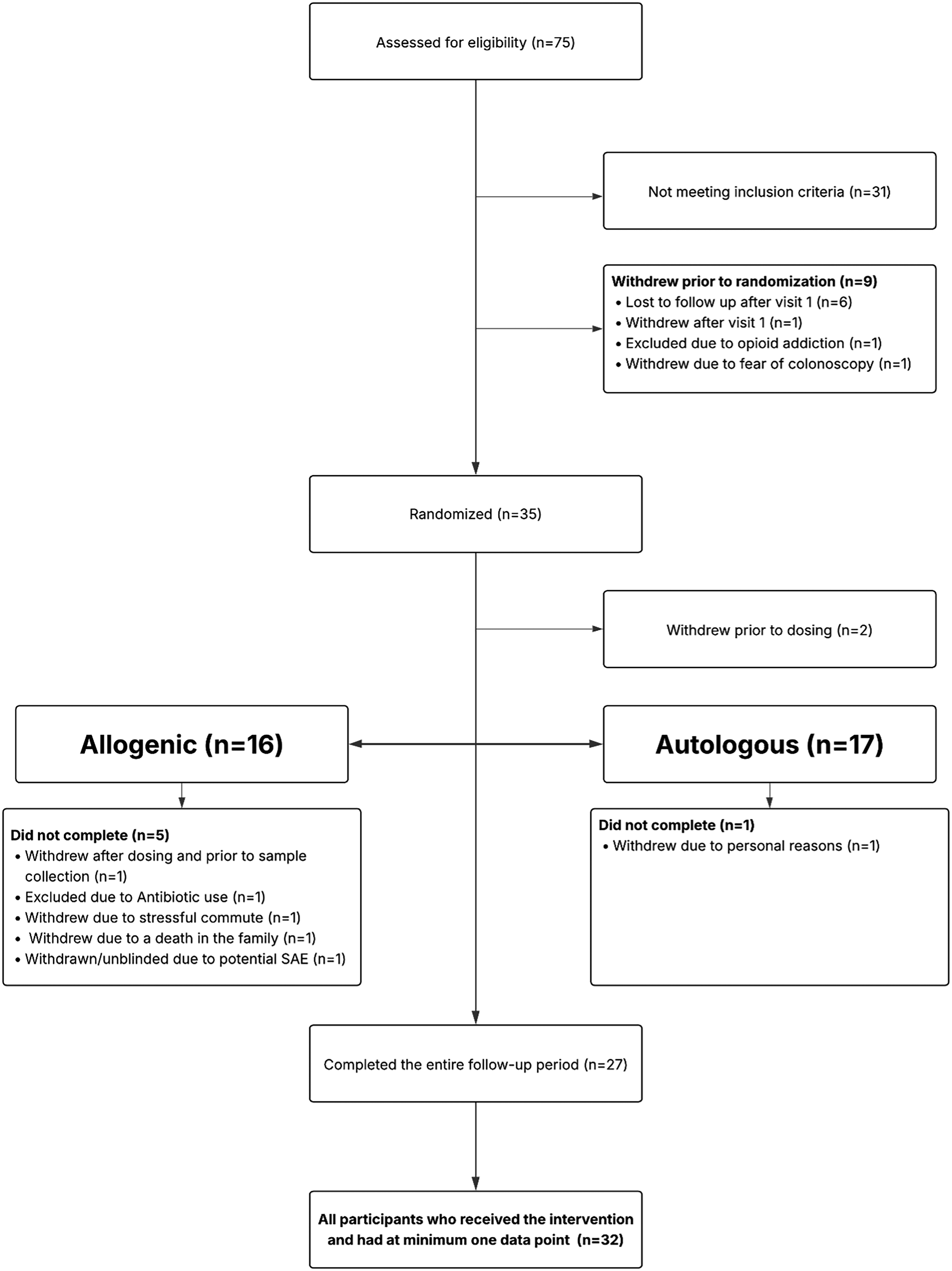
Participant flow diagram.
Baseline demographic and clinical variables were well balanced between treatment groups, suggesting successful randomization. There were no notable differences in baseline MADRS, YMRS, STAI, and SDS scores. Age and sex were similarly distributed across groups (Table 2). Discontinuation/drop-out rates were also comparable between groups.
Baseline Statistics.
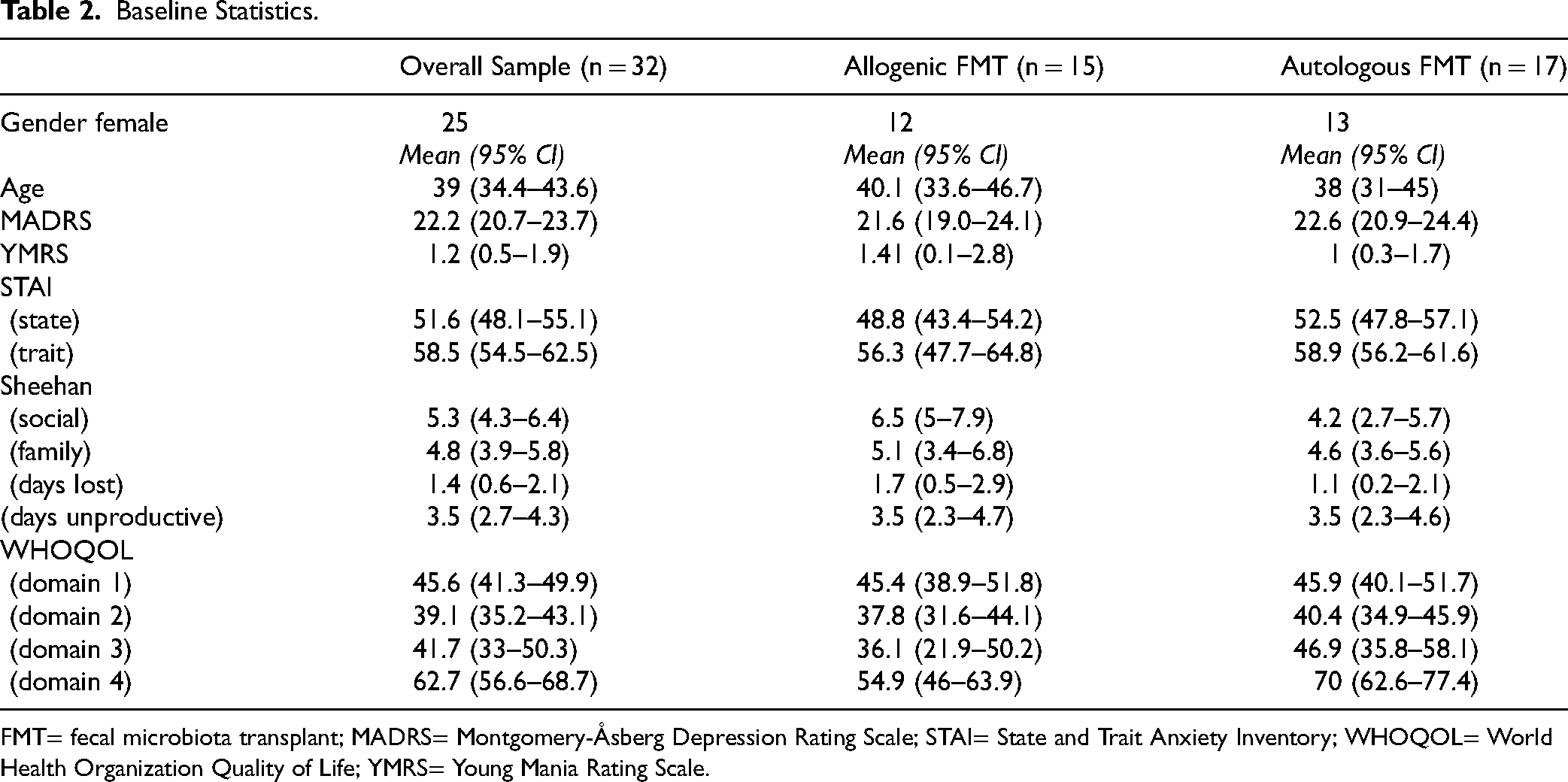
FMT= fecal microbiota transplant; MADRS= Montgomery-Åsberg Depression Rating Scale; STAI= State and Trait Anxiety Inventory; WHOQOL= World Health Organization Quality of Life; YMRS= Young Mania Rating Scale.
Primary Outcome
The primary goals of this proof of concept study were to determine the effectiveness, safety, and tolerability of FMT in adults with BD depression. With respect to efficiency, both allogenic FMT (16.7-point improvement, 95% confidence interval [CI] 11.9–21.5) and autologous FMT (15.4-point improvement, 95% CI = 11.2–19.5) showed significant within-group improvements. However, the independent t-test showed no significant difference in MADRS change between the two FMT groups (t = −0.47, P-value = 0.64, 95% CI = −7.3–4.6). Overall, MADRS scores significantly improved from baseline to the last visit with a paired t-test (15.9-point improvement, t = −11.2, P < 0.001) (Figure 2(a)).
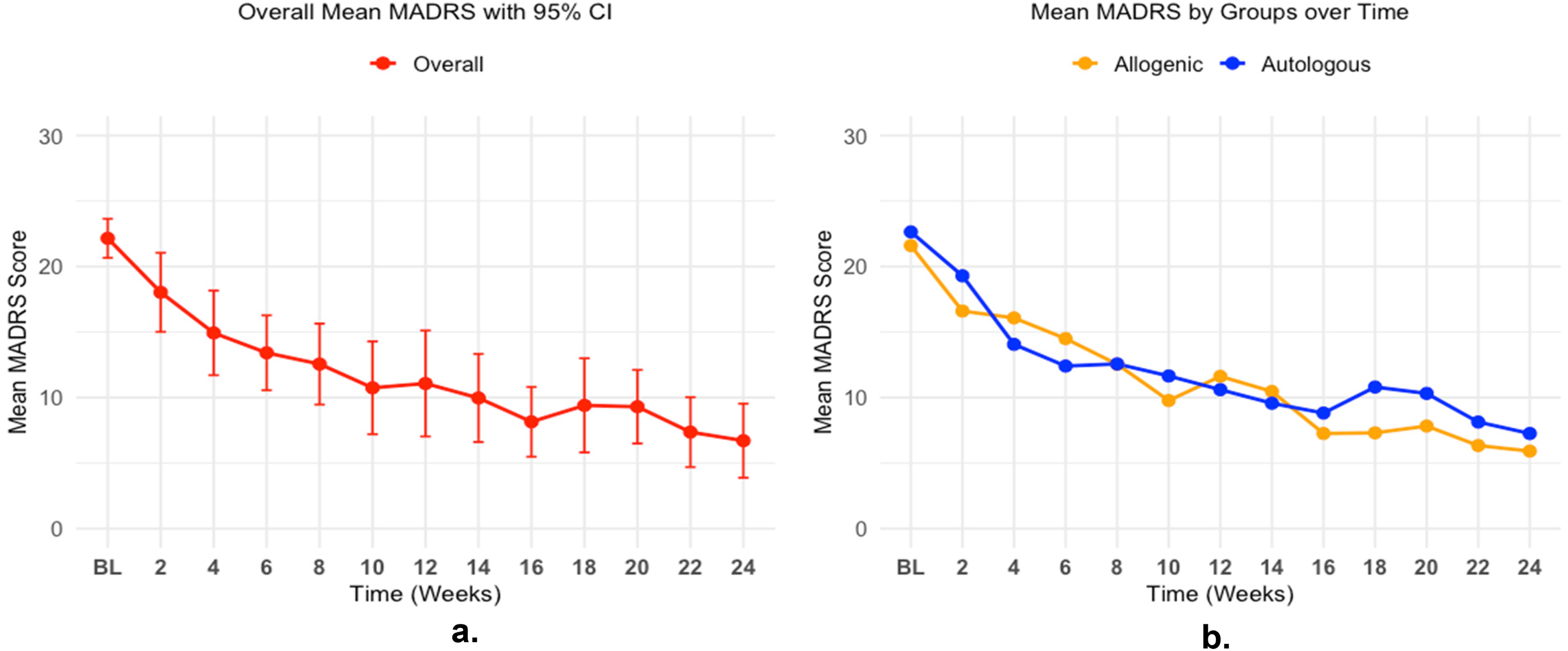
Changes in MADRS scores over time for all FMT participants (2a) and by FMT type (2b). FMT=fecal microbiota transplant; MADRS=Montgomery-Åsberg Depression Rating Scale.
Response and remission rates of MADRS scores at the final visit are summarized in Table 3 for both FMT groups. Among completers at this time point, the overall remission rate was 74.1 per cent (20 out of 27); allogenic FMT had a remission rate of 81.8 per cent (9 out of 11), with 2 non-responders, while the autologous FMT had a remission rate of 68.8 per cent (11 out of 16). When including all patients with the LOCF, the overall remission rate is 71.9 per cent (23 out of 32); allogenic FMT had a remission rate of 80 per cent (12 out of 15) while the autologous FMT group has a response and remission rate of 64.7 per cent (11 out of 17). Fisher's exact tests comparing remission and response rates between the two groups were non-significant (P-value = 0.66; P-value = 0.44 with LOCF), indicating no significant difference in the proportion of responders or remitters between the two FMT groups.
Number of Participants Achieving Response and Remission at Last Visit.
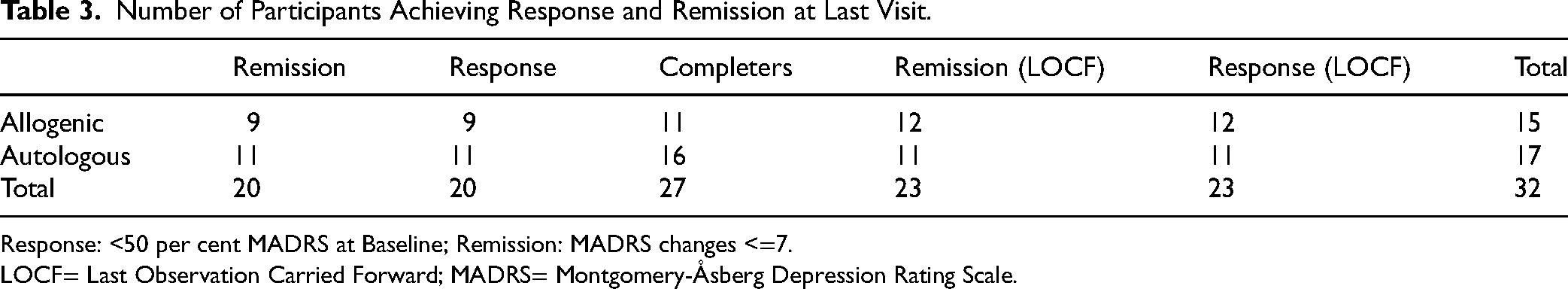
Response: <50 per cent MADRS at Baseline; Remission: MADRS changes <=7.
LOCF= Last Observation Carried Forward; MADRS= Montgomery-Åsberg Depression Rating Scale.
The final linear mixed model results are shown in Table 4, including the fixed-effect estimates for group, time, and time-squared. The MADRS scores decreased significantly over time (β = −2.3, t = −5.38, P < 0.001), with a significant positive quadratic time (β = 0.09, t = 3.39, P = 0.001) indicating a diminishing rate of decrease in MADRS scores over time. There was no significant group effect nor significant interaction terms between group and times, indicating similar MADRS trajectories overall between FMT types (Figure 2(b)).
Linear Mixed Model Results With Fixed-Effect Parameter.
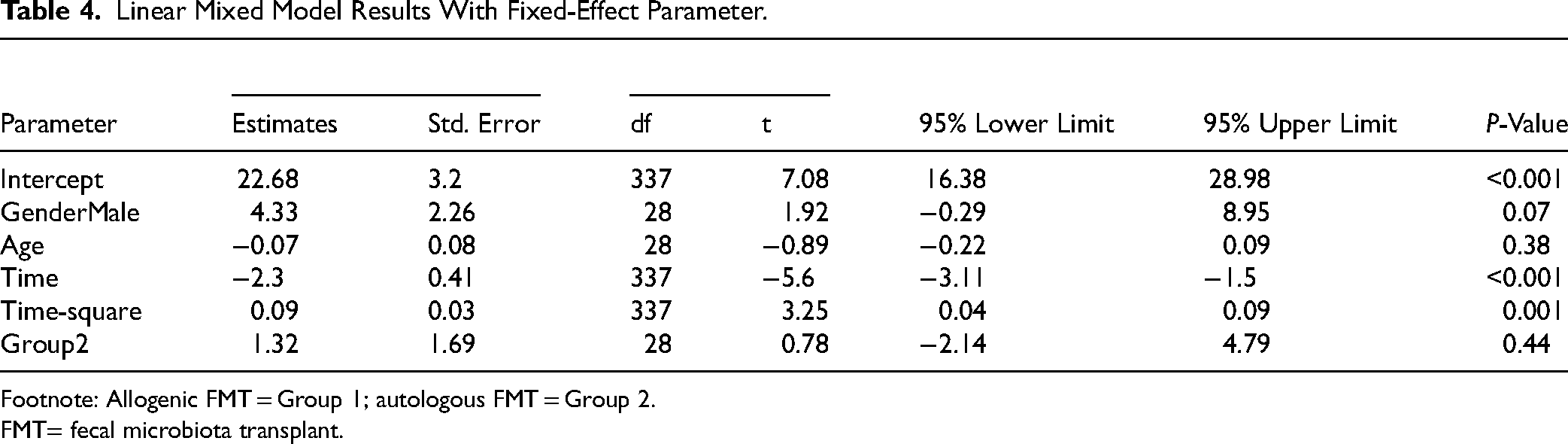
Footnote: Allogenic FMT = Group 1; autologous FMT = Group 2.
FMT= fecal microbiota transplant.
Secondary Outcomes
The changes in secondary outcome domains from baseline to final visit were summarized in Table 5. Statistically significant improvements were observed in measures of anxiety, manic symptoms, functional impairment, and quality of life overall. The findings showed a consistent pattern of symptom reduction and improvement post bowel cleanse and FMT. There were no significant between-group differences in change scores for any secondary outcome subdomain. There was no impact of dietary composition as analyzed by Nutritionist Pro dietary software or on medication exposure.
Secondary Outcomes.
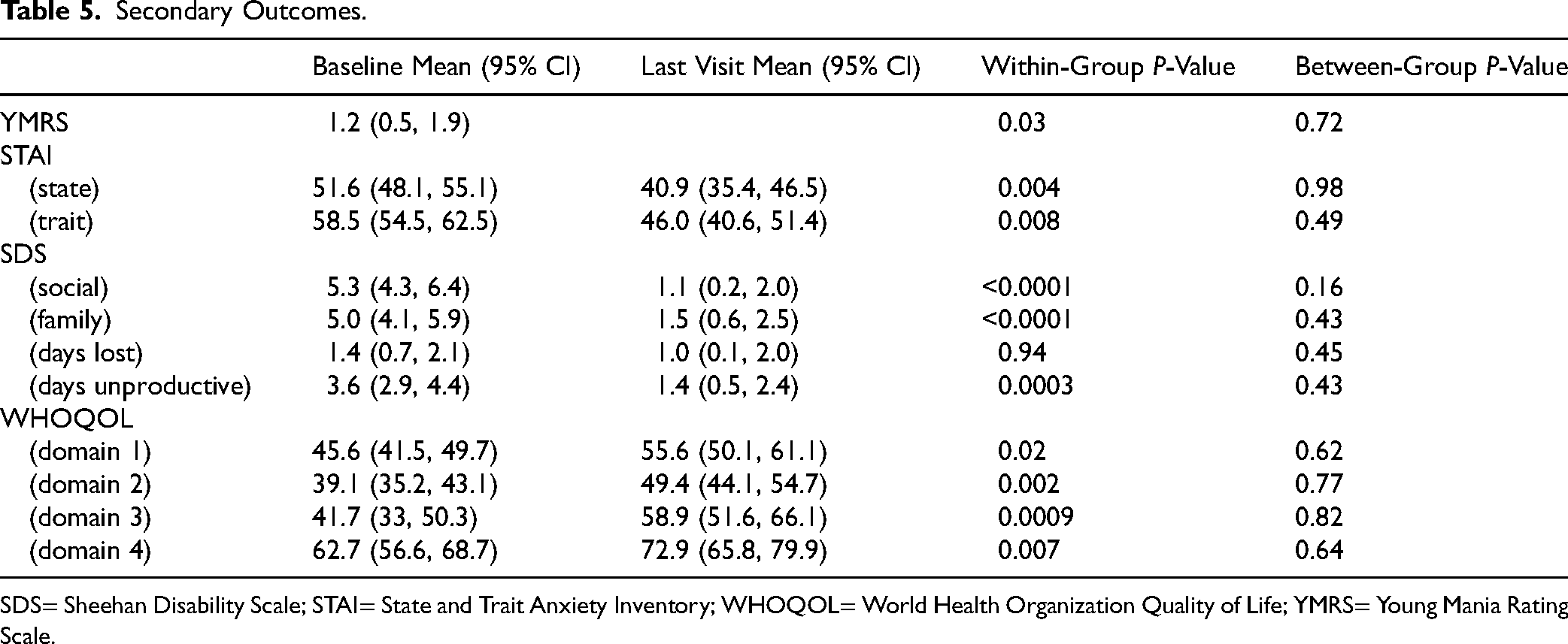
SDS= Sheehan Disability Scale; STAI= State and Trait Anxiety Inventory; WHOQOL= World Health Organization Quality of Life; YMRS= Young Mania Rating Scale.
Adverse Effects, Safety, and Tolerability
With respect to safety, no serious adverse events associated with treatment were observed during the study across both groups, and no participants discontinued treatment secondary to any study-related side-effect and no clinically significant weight gain was reported.
Most TSES symptoms were rated at the low end of the scale on average at baseline and remained low at the last visit. Several symptoms that were rated at moderate levels at baseline (≥2) showed statistically significant reductions in both frequency and severity, including nervousness, agitation, drowsiness, and fatigue (all P < 0.01). Between-group comparisons in changes from baseline to last visit showed that the majority of TSES symptoms did not differ significantly. However, the symptom of dyspepsia (upset stomach) demonstrated a statistically significant between-group difference. Participants in allogenic group experienced a greater reduction from baseline to the final visit than those in autologous group for both frequency (P = 0.025) and severity (P = 0.042) (Supplemental Table 1).
Feasibility
Feasibility of a decisive trial was confirmed in that only 10 eligible subjects were not randomized, indicating successful recruitment. Under a modified intention to treat analysis only three participants were excluded from the analysis. Twenty-seven members of the analyzed sample (84%) were successfully followed to the end of the protocol, indicating acceptability.
Microbiome Results
Of the 27 participants who completed the study, 25 were included in the microbiome analysis. Two participants from the allogenic FMT group were excluded due to missing baseline stool samples. The final dataset comprised nine participants who received allogenic FMT (7 responders and 2 non-responders) and 16 participants who received autologous FMT (11 responders and 5 non-responders). For each participant, three fecal samples collected at baseline (week 1), mid-point (week 12), and endpoint (week 24) were subjected to shotgun metagenomic sequencing to assess longitudinal changes in microbial composition and function. Supplemental Figure 1 shows the sample distribution and relative abundance of the top 20 microbial families across time points.
To investigate whether FMT induced distinct microbial signatures, both taxonomic (species-level) and functional (pathway-level) profiles were analyzed. Linear models were applied to assess microbial changes within participants following either allogenic or autologous FMT at week 9 and week 15. At the taxonomic level, a significant increase in the relative abundance of Phascolarctobacterium succinatutens was observed at week 15 within the allogenic group. In the autologous group, increased abundance of Oscillibacter sp., Ruminococcaceae sp., Lachnospira eligens, and Alistipes communis was detected at week 9 (linear model, FDR-adjusted P < 0.1; Figure 3). At the functional level, numerous microbial pathways were significantly enriched at week 15 in the allogenic group (linear model, FDR-adjusted P < 0.05; Figure 4). These pathways were primarily associated with Odoribacter splanchnicus, P. succinatutens, Bacteroides ovatus, Bacteroides clarus, and Bacteroides vulgatus, and involved in essential amino acid biosynthesis (e.g., L-histidine, L-lysine, L-valine, and L-rhamnose). In contrast, the autologous group showed significant enrichment of microbial pathways belonging to Lachnospira eligens at week 9, but not at week 15 (linear model, FDR-adjusted P < 0.05; Figure 4).
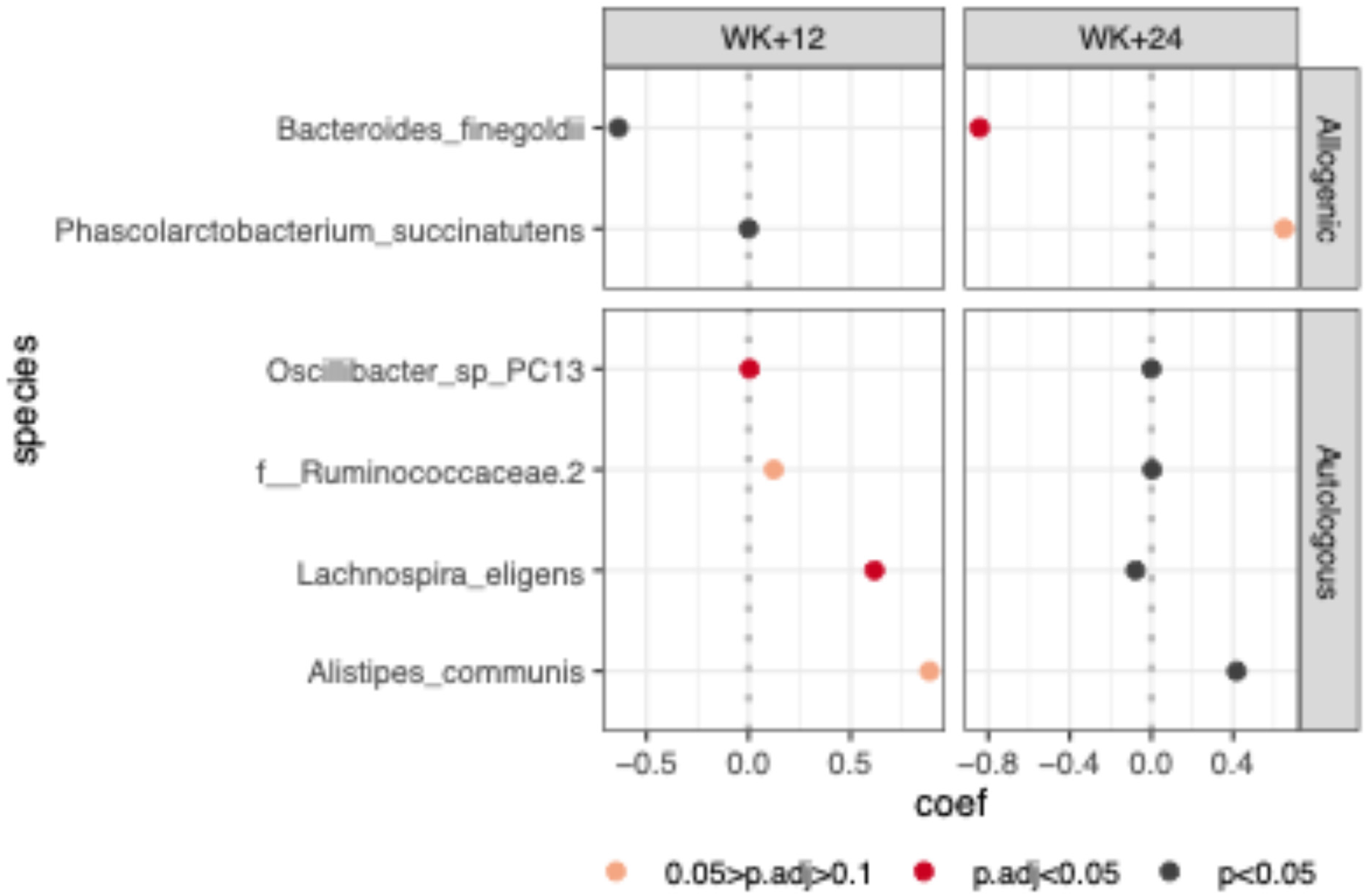
Differentially abundant microbial species at week+12 and week+24 compared to baseline (week 1). Species on the y-axis shows significant changes in abundance at one or more time points within either the allogenic or autologous FMT group, based on linear modeling with FDR-adjusted P-values < 0.1. The x-axis displays the estimated model coefficients for each species, indicating the direction and magnitude of change relative to baseline. Species with statistically significant changes (FDR-adjusted P < 0.05) are highlighted in red. FDR= false discovery rate; FMT=fecal microbiota transplant.
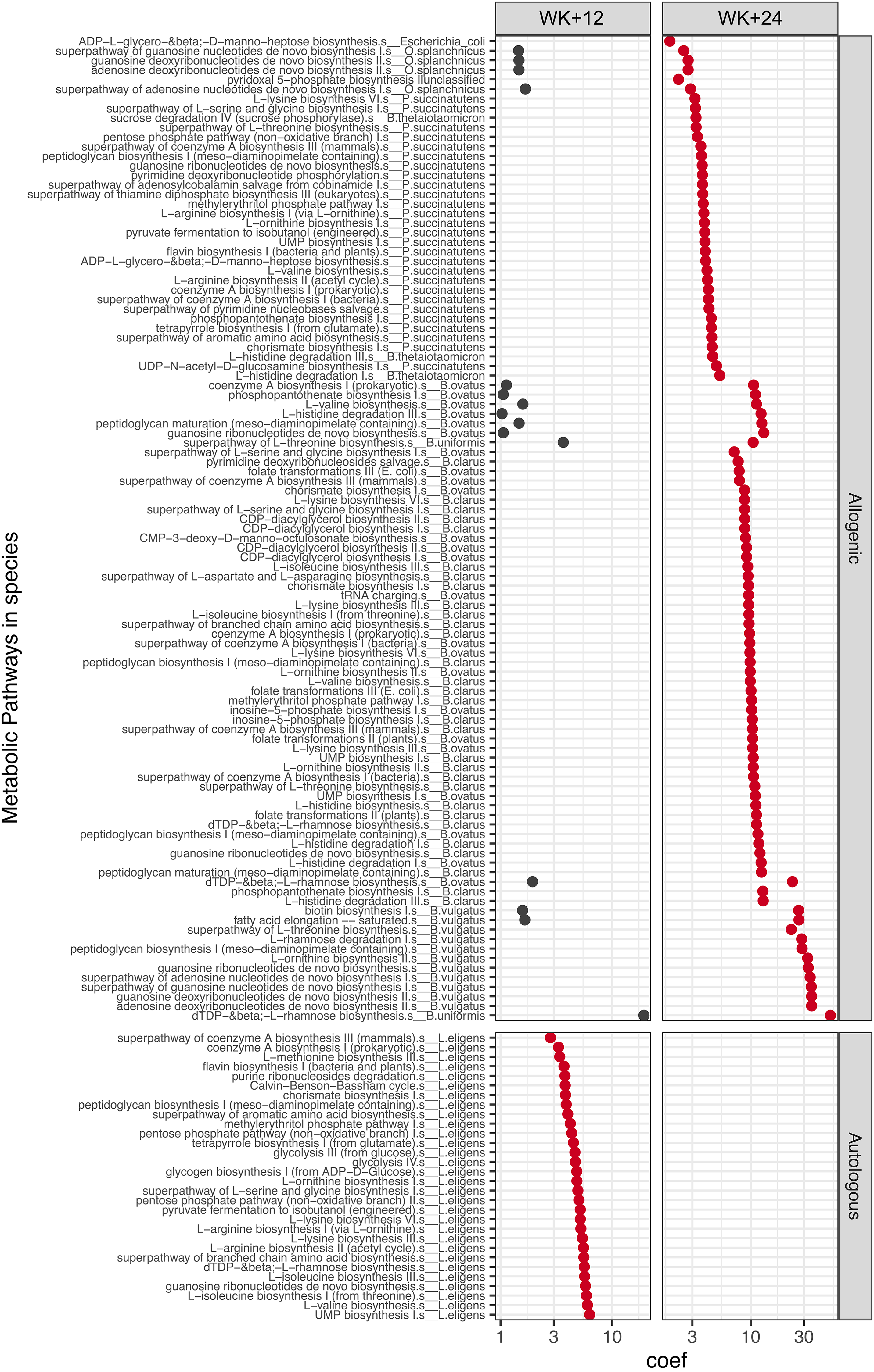
Enriched microbial pathways at week+12 and week+24 compared to baseline (week 1). Metabolic pathways on the y-axis represent those with significant increases in abundance at one or more time points within either the allogenic or autologous FMT group, based on linear modeling with FDR-adjusted P-values < 0.1, and effect size (coefficient) > 1. The x-axis shows the estimated model coefficients, indicating the direction and magnitude of change relative to baseline. Pathways with statistically significant changes (FDR-adjusted P < 0.05) are highlighted in red and FDR-adjusted P < 0.1 > 0.05 in black. Each pathway is annotated by the contributing species from which it was identified. Note that Lachnospira eligens pathways were identified as Eubacterium eligens by HUMAnN3 (v3.5) and corrected here to L. eligens consistent with the updated taxonomy in MetaPhlAn4 and with Figure 3. FDR= false discovery rate; FMT=fecal microbiota transplant.
To evaluate whether FMT was associated with changes in microbial diversity, Shannon diversity was assessed based on metagenomic reads. In the allogenic group, a trend toward increased Shannon diversity was observed at week 9 compared to baseline, accompanied by reduced inter-individual variability, as reflected by narrower boxplot boundaries (Supplemental eFigure 2). Although this increase did not reach statistical significance, the findings suggest an overall gain in microbial richness and convergence among recipients of allogenic FMT. No such trend was observed in the autologous FMT group, where Shannon diversity remained largely unchanged across time points (Supplemental eFigure 2).
The extent to which recipient microbiota shifted toward the donor microbiota was assessed by using pairwise Bray-Curtis dissimilarity based on metagenomic taxonomic profiles. In the allogenic FMT group, recipient microbiomes became significantly more similar to the donor microbiome at week 9 compared to baseline (P < 0.05, Wilcoxon rank-sum test), with a continued, albeit non-significant, trend toward increased similarity at week 15 (Supplemental eFigure 3). In contrast, no such pattern was observed in the autologous FMT group; instead, patient microbiomes became significantly more dissimilar from their own baseline profiles at both week 12 and week 24.
Discussion
This study represents the first RCT examining FMT as a treatment for BDd. The results suggest that manipulation of the gut microbiota could be a promising strategy for improved symptoms of BD. An overall remission rate of 74 per cent, with the allogenic group showing 84 per cent remission, is encouraging and clinically meaningful, given that these participants had not responded to at least one first-line treatment. For comparison, a recent study comparing the efficacy outcomes of electroconvulsive therapy (ECT) and algorithm-based pharmacological treatment in treatment-resistant BDd found remission rates of 34.8 versus 30.0 per cent respectively. 43 FMT was well tolerated and safe, consistent with its established use as a treatment for C. difficile infection 44 and its recent investigational use for the neurological conditions such as Parkinson's disease. 45 We observed no severe adverse events (SAEs) associated with FMT, consistent with a systematic review that found SAEs in only in 1.4 per cent of FMT procedures done in the past 20 years, and then only in those with mucosal barrier injury. 46
While all participants who received FMT showed significant improvement (P < 0.0001), there was no significant between group differences in those receiving allogenic or autologous FMT. We had originally conceptualized autologous FMT as a control condition. However, our finding is consistent with other recently FMT trials that suggest that autologous FMT may actually be an active treatment intervention. For example, a recent study of FMT in Parkinson's disease showed both allogenic and autologous FMT were significantly effective until the 6-month mark. 45 Another study in Irritable Bowel Syndrome also showed a significant effect of FMT as an intervention, with no significant difference between FMT types. 47 The study where the autologous and allogenic groups separated at the 24-week mark. This may be because autologous FMT is not a true placebo in a pharmacological sense, 47 with significant alterations in the gut microbiome noted in other studies using autologous FMT for medical indications. 47 Simply the process of collecting, processing (e.g., freezing, cryoprotecting, and thawing), and reinfusing a patient's own stool, especially after a bowel preparation protocol, can lead to changes in the gut microbiome. Autologous FMT also subsequently reintroduces an inoculum of the patient's own microbes into this less competitive environment and the subsequent microbial community may re-establish in different proportions or with altered spatial organization compared to the pre-preparation state. This could lead to transient shifts in dominant species, metabolite production, and interaction with the host, potentially influencing symptoms.45, 47
While our clinical findings revealed no significant difference in symptom improvement between the allogenic and autologous groups, microbiome profiling demonstrated clear compositional and functional distinctions. Of note, several of the enriched taxa identified in the allogenic group have previously been implicated in succinate metabolism 48 and targeting succinate pathways within the gut microbiota has been proposed as a promising therapeutic approach in inflammatory bowel disease. 49 The allogenic group also demonstrated increased microbial diversity, something that has previously been associated with lithium treatment, 50 while lack of diversity was associated with illness. 16
The study's strengths include the length of follow-up of 24 weeks and the correlation to microbiome changes. Unfortunately, given there is mounting evidence to show separation between allogenic and autologous FMT emerges beyond the 6-month mark, extending the follow-up to see if the trends we observed would have continued would have been helpful. We also recruited a treatment-resistant population that had failed at minimum one treatment, a factor previously identified in depression trials as being linked to a smaller placebo effect 51 but a placebo effect cannot be excluded. This is a limitation, and future studies should therefore be designed to include a true placebo, given the mounting evidence that autologous FMT is not likely to be an inert control, as originally conceptualized.
There are limitations associated with FMT methodology, related to the many unknowns still existing in this field, including the use of a single dose FMT, as there is still debate regarding the best frequency of FMT to produce appropriate results. 52 Coloscopy was chosen as the method of intervention as it reasonable to believe that better FMT distribution increases the chance of donor microbiota engraftment, which has been shown to be associated with treatment outcome, 53 and current evidence indicates this is better achieved with colonoscopy. 54 A recent study of FMT by enema for depression, however, indicates other methods of administration may be adequate as well however. 55 No antibiotic was used as evidence indicated that it could impact engraftment,56,57 but again, the data is equivocal and thus further work needs to be done. 58 The use of bowel preparation may also have an impact on outcomes, 59 but evidence from studies showing efficacy after the 6 months indicates that the effect may be transient. 45 We also notice trends toward increased diversity in the allogenic group, indicating something other than the bowel preparation was having an effect.
This study was designed as a feasibility study to inform outcomes of future research, as such studies are essential to inform key next steps. It adds to the positive findings of two case reports reporting the efficacy of FMT in refractory BDd 60 and a probiotic RCT showing improvements in mania, 61 building on the evidence base supporting microbial manipulation being a viable therapeutic target. A crucial next step 61 is to expand on this work with a larger trial, designed to include an inert placebo and to explore exactly what bacterial changes seem associated with symptom changes observed. It is essential to ensure that clinical translation in the mental health space is informed by well-designed pilot studies with a focus on clear outcomes measured via validated tools, in well-characterized populations, focused on symptom burden and supported by sophisticated microbiome approaches. 62 A lack of adequate treatments for mental illness can generate premature enthusiasm for alternative therapies but it also represents a compelling opportunity. 63 The Gut-Brain Axis (GBA) is an area where, if preclinical scientists partner with clinical and trial experts, we can augment our understanding of the etiology and treatment of mental illness.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437261420877 - Supplemental material for The Safety, Efficacy, and Feasibility of Fecal Microbiota Transplantation in a Population With Bipolar Disorder During Depressive Episodes: A Pilot Parallel Arm Randomized Controlled Trial: Sécurité, efficacité et faisabilité de la transplantation de microbiote fécal chez une population atteinte de troubles bipolaires, au cours d’épisodes dépressifs : essai pilote contrôlé à répartition aléatoire et à groupes parallèles
Supplemental material, sj-docx-1-cpa-10.1177_07067437261420877 for The Safety, Efficacy, and Feasibility of Fecal Microbiota Transplantation in a Population With Bipolar Disorder During Depressive Episodes: A Pilot Parallel Arm Randomized Controlled Trial: Sécurité, efficacité et faisabilité de la transplantation de microbiote fécal chez une population atteinte de troubles bipolaires, au cours d’épisodes dépressifs : essai pilote contrôlé à répartition aléatoire et à groupes parallèles by Shahrokh Shekarriz, Simone N. Vigod, Teresa Bianco, Asem Bala, Chenhui Hao, Johane P. Allard, Susy Hota, Susan Poutanen, Michael G. Surette and Valerie H. Taylor in The Canadian Journal of Psychiatry
Supplemental Material
sj-docx-2-cpa-10.1177_07067437261420877 - Supplemental material for The Safety, Efficacy, and Feasibility of Fecal Microbiota Transplantation in a Population With Bipolar Disorder During Depressive Episodes: A Pilot Parallel Arm Randomized Controlled Trial: Sécurité, efficacité et faisabilité de la transplantation de microbiote fécal chez une population atteinte de troubles bipolaires, au cours d’épisodes dépressifs : essai pilote contrôlé à répartition aléatoire et à groupes parallèles
Supplemental material, sj-docx-2-cpa-10.1177_07067437261420877 for The Safety, Efficacy, and Feasibility of Fecal Microbiota Transplantation in a Population With Bipolar Disorder During Depressive Episodes: A Pilot Parallel Arm Randomized Controlled Trial: Sécurité, efficacité et faisabilité de la transplantation de microbiote fécal chez une population atteinte de troubles bipolaires, au cours d’épisodes dépressifs : essai pilote contrôlé à répartition aléatoire et à groupes parallèles by Shahrokh Shekarriz, Simone N. Vigod, Teresa Bianco, Asem Bala, Chenhui Hao, Johane P. Allard, Susy Hota, Susan Poutanen, Michael G. Surette and Valerie H. Taylor in The Canadian Journal of Psychiatry
Footnotes
Data Access
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Shekarriz reports receiving a Mitacs postdoctoral fellowship through McMaster University supported by Taylored Biotherapeutics and being an inventor on patents planned, issued, or pending related to Taylored Biotherapeutics (WO 2025/129338, US 63/613,222). He has also received consulting fees from eDNAtech, a company developing environmental DNA-based monitoring technologies in a field unrelated to the subject of this manuscript. Dr. Vigod receives royalties from UpToDate Inc. related to authorship of materials on depression and pregnancy and holds unpaid leadership roles in MONA, CANMAT, and CAPE. Asem Bala has received consulting fees from Taylored Biotherapeutics for work on development of the company's clinical and regulatory platform and holds stock or stock options in Taylored Biotherapeutics, a company developing live bacterial therapies for mental health disorders. Dr. Hota has received consulting fees, honoraria, and advisory board payments from Ferring Canada and reports travel reimbursement and a voluntary, unpaid leadership role as President-Elect of the Association for Medical Microbiology and Infectious Disease Canada (AMMI Canada). Dr. Poutanen has received honoraria for advisory board participation and presentations from Ferring. Dr. Surette has received consulting fees from Taylored Biotherapeutics for Scientific Advisory Board activities and is listed as an inventor on Taylored Biotherapeutics patents WO 2025/129338 and US 63/613,222. Dr. Taylor is the founder of Taylored Biotherapeutics, holds equity in the company, and is the inventor of a U.S. patent application (No. 63/613,222, “Bacterial Compositions for Treatment of Bipolar Disorder or Symptoms Thereof”) derived from secondary analysis of microbiome data that are not part of the work reported in this manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This clinical trial was supported by a The Stanley Medical Research Institute (SMRI) grant (15T-009). The funder played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
