Abstract
Background
Guidelines for treatment-resistant schizophrenia (TRS) advocate for a trial of clozapine monotherapy before the consideration of antipsychotic augmentation. Commonly cited justifications for augmentation include inadequate response to clozapine monotherapy and the potential to lower the necessary clozapine dose or serum concentration, thereby reducing dose-dependent adverse effects. Nonetheless, the degree to which these outcomes are realized in routine clinical practice, particularly among individuals with concurrent disorders, remains uncertain. This study aimed to explore the extent to which clozapine monotherapy is utilized before the initiation of antipsychotic augmentation strategies, and to assess the effects of antipsychotic augmentation on clozapine serum concentrations and the incidence of related adverse effects.
Methods
We retrospectively analyzed clinical and drug monitoring data from 80 adults with TRS and substance use disorder (SUD) comorbidity at a provincial inpatient centre for concurrent disorders. Antipsychotic augmentation was quantified using Defined Daily Dose (DDD). Generalized and linear mixed models compared the impact of monotherapy vs. augmentation on clozapine serum levels and adverse effects, adjusting for covariates.
Results
Most patients receiving antipsychotic augmentation (78%) did not have an adequate trial of clozapine monotherapy. Analysis revealed that clozapine with antipsychotic augmentation was modestly and negatively associated (B = –0.039; 95% CI = –0.078 - −0.001) with clozapine serum concentrations, particularly at higher DDD (≥2). Clozapine with antipsychotic augmentation was not associated with reduced incidence of dose-dependent adverse events (tachycardia, constipation, or overall anticholinergic medication use).
Conclusion
Findings from this study indicate that commonly cited rationales for combining clozapine with antipsychotic augmentation – namely, enhancing tolerability through clozapine dose reduction or mitigating inadequate response to monotherapy – are not consistently supported by real-world outcomes. These results underscore the necessity for clinical guidelines to incorporate context-sensitive recommendations that address the complexities inherent in managing individuals with TRS and comorbid SUDs, while integrating real-world considerations and the perspectives of those with lived experience.
Background
Approximately one-third of individuals diagnosed with schizophrenia have non-response to first-line antipsychotic medications. Treatment-resistant schizophrenia (TRS) is broadly defined as a lack of therapeutic response (≤20% reduction in positive and negative syndrome scale scores) after two “adequate” (dose: >600 mg chlorpromazine equivalent per day, and duration: 6 weeks per trial) trials of antipsychotic medications. 1 The diagnosis of TRS is associated with a substantially greater illness burden, including poorer functional outcomes, higher rates of comorbid substance use disorders (SUDs) and suicide, and greater use of healthcare services – factors that together drive significantly higher healthcare expenditures.2,3 Clozapine is currently the only antipsychotic medication recommended by clinical guidelines for TRS, with a significant body of evidence to support its efficacy and favourable risk-benefit ratio.1,4,5 Specifically, within the domain of highly prevalent SUD comorbidity, research has suggested that clozapine use is further associated with improvements in outcomes related to SUDs. 6
Individuals with SUD comorbidity represent a particularly challenging clinical subgroup, often characterized by high rates of treatment non-adherence and poor symptomatic remission. 7 Despite the considerable complexity of this population, real-world prescribing patterns of clozapine in TRS-SUD remain insufficiently examined. Current clinical guidelines for TRS advocate for clozapine monotherapy as the gold standard, reserving augmentation strategies for cases that meet criteria for clozapine-resistant schizophrenia (CRS) – typically defined by persistent psychotic symptoms despite at least 8 weeks of treatment at serum concentrations exceeding 350 ng/mL, or in the absence of therapeutic drug monitoring (TDM), a minimum daily dose of 500 mg.1,4
Although clinical guidelines recommend prioritizing clozapine monotherapy, adjunctive antipsychotics are often introduced early or prior antipsychotics are continued, resulting in increased pharmacological burden and a potential rise in adverse effects without clear evidence of improved symptom control or functional outcomes.8–13 Paradoxically, however, emerging evidence from large-scale observational studies suggest that clozapine-antipsychotic polytherapy may be associated with reduced all-cause mortality compared to clozapine monotherapy. 14 Furthermore, specific combinations – most notably with aripiprazole – have been linked to lower rates of psychiatric hospitalization due to a psychotic relapse, 15 thereby inviting reflection on the established views of the risks and benefits of clozapine-antipsychotic augmentation.
A common rationale for clozapine-antipsychotic augmentation is the potential to use lower clozapine doses or serum levels, thereby minimizing dose-related side effects.16–22 Nevertheless, it remains unclear whether this benefit is realized in real-world clinical practice, particularly in individuals with TRS-SUD, or if such prescribing patterns primarily reflect an intention to limit high clozapine dose exposure. Although previous studies have investigated how patient-specific factors affect clozapine exposure – typically assessed by serum concentrations relative to dose – due to the high interindividual variability in clozapine metabolism,23–26 the effects of co-prescribed antipsychotics on clozapine serum concentrations and associated adverse events have received limited attention.
To address these knowledge gaps, this study assessed: (1) clinician utilization of clozapine monotherapy prior to antipsychotic augmentation, (2) the direction and magnitude of the effect of antipsychotic augmentation on clozapine serum concentrations, and (3) the occurrence of dose-related adverse events among those treated with clozapine monotherapy versus antipsychotic augmentation. We hypothesized that antipsychotic augmentation would be associated with lower clozapine serum concentrations, and based on the previous evidence, a higher likelihood of anticholinergic agent prescribing for antipsychotic-related adverse effects.27,28
Methods
This study is reported according to the STROBE statement 29 of cohort studies (Supplementary Table S5). Ethics approval, including waiver of consent, was obtained from the University of British Columbia Clinical Research Ethics Board as well as BC Mental Health and Substance Use Services ethics committee, and employed principles highlighted in the Declaration of Helsinki.
Study Setting and Population
This was a retrospective cohort study using routine care electronic medical record data of patients admitted to the Red Fish Healing Centre for Mental Health and Addiction (RFHC) in British Columbia, Canada.
RFHC is a 105-bed provincial tertiary treatment facility that provides multidisciplinary integrated treatment for adults (19 years of age or older) with co-occurring SUDs and severe and persistent mental illnesses. According to the access protocol, the patients must have not found success in other psychiatric and SUD treatment programs on a regional level.
Eligibility Criteria
Electronic medical records of adult patients with diagnosis of TRS (clozapine prescription used as a proxy) were reviewed for inclusion from the time of RFHC's opening (Oct 8, 2021) to July 31, 2024. Patients were included if they (1) had stayed at the centre for a minimum of 90 days during the study period (the 90 days was considered the minimum required stay to finish treatment at the facility) and (2) were prescribed clozapine during their stay at the facility. Patients on clozapine for whom TDM was not completed were excluded. Patients with abnormal renal or liver function (eGFR<60 mL/min or liver enzymes (AST/ALT/GGT) ≥ 3× ULN) were also excluded due to potential for artificially increased clozapine serum levels.
Clozapine serum concentrations were included if the blood samples were drawn 12 +/- 2 h post-dose. 30 Clozapine serum levels obtained during the initial titration phase were excluded, as these were associated with standing dose escalation orders and had not yet reached the intended target dose. Clozapine serum levels collected during periods of active inflammation or infection were excluded, given the potential for inflammatory states to elevate clozapine concentrations. 23 “Inflammatory state” was defined by any of the following criteria: current use of antibiotics, antivirals, an elevated C-reactive protein level (CRP > 10), elevated white blood cells (>11.0 × 10^9 cells per liter), or documentation in progress notes indicating an active infection or inflammatory process at the time of blood sampling.
Measurement of Clozapine Serum Concentrations
Clozapine serum concentrations were measured using liquid chromatography-tandem spectrometry at the LifeLabs® accredited laboratory. Given the inpatient setting, many patients had repeated clozapine TDM. Clozapine levels were analyzed independently of the corresponding norclozapine level, noting that predicted clozapine level thresholds have been established for both efficacy and dose-dependent adverse drug effects. Clozapine levels were collected as nmol/L and converted to ng/mL with a conversion ratio of 3.06 nmol/L = 1 ng/mL.
Data Collection
Data collection occurred between August 2024 and February 2025. Included patients were anonymized by medical record number with a study ID and a chart review was conducted via electronic medical records. Documents reviewed included referral packages, clinical notes, patient records, lab results, consultations, admission and discharge documentations, pharmacists’ review of previous medication trials (An evaluation conducted by clinical pharmacists based on electronic records of prior healthcare use and community-sourced documentation), and medication administration records. Data were extracted by one author (L.F.) utilizing a custom data extraction template. The template included patient demographics, medical and psychiatric diagnoses, the Health of the Nation Outcome Scales (HONOS) scores (a clinician rated instrument comprising scales measuring domains such as cognition, hallucination and delusions, mania, depression, severity of SUDs and anxiety rated on a five-point item of severity of 0–4), prescribed medications at the time of each clozapine serum level measurement, monthly weight measurements, clozapine serum concentrations, heart rate (taken as close as possible up to 7 days from the clozapine serum levels), and positive urine drug screen (UDS)/breathalyzer results. Procedures for gathering and assessing UDS data have been previously described. 31
For patients who discontinued their clozapine prior to discharge from the facility, reasons for discontinuation were collected. Clinicians’ notes and pharmacists’ review of previous medication trials were examined to determine whether clozapine monotherapy had ever been attempted or if a diagnosis of CRS had been established prior to the initiation of antipsychotic augmentation.
Antipsychotic Augmentation
Antipsychotic augmentation was quantified using the Daily Defined Dose (DDD), a standardized unit established by the World Health Organization to represent the assumed average maintenance dose per day for a drug used for its primary indication in adults. 32 This continuous dosing metric was selected to capture potential dose–response relationships, rather than dichotomizing augmentation as a binary variable. Within this framework, augmentation was defined as a DDD greater than zero, whereas monotherapy corresponded to a DDD of zero.
Adverse Events
Markers and proxy markers were utilized to assess clozapine's dose-dependent adverse events as dichotomous dependent variables. Sinus tachycardia was defined as either HR > 100 or concomitant usage of a beta-blocker confirmed via clinical notes to be prescribed for clozapine-induced tachycardia. Constipation was defined as usage of laxative medications including PEG, sennosides, lactulose (prescribed regularly or ≥4 days of a PRN medication within 7 days of clozapine level). All topical and systemic anticholinergic use was identified through medication administration records. This included agents such as atropine and tropicamide eye drops (used sublingually), ipratropium nasal spray (used sublingually), as well as systemic medications including benztropine, procyclidine, diphenhydramine, and oxybutynin prescribed regularly or PRN use ≥ 4 days within 7 days of clozapine serum level.
Covariates
Covariates were selected based on their previously reported impact on clozapine serum concentrations,23–26 including: sex, age, smoking status (dichotomous variable, either positive or negative, as data on smoking intensity was not available), weight, frequency of relapse to substance use (measured as the number of positive UDSs per duration of stay), time from admission to the date of blood sample collection, and primary psychotic disorder diagnosis (schizophrenia versus schizoaffective disorder).
Statistical Analysis
Jamovi version 2.5 33 was used for all analyses. Baseline demographics were summarized using descriptive statistics. Continuous variables are presented as mean ± standard deviation (SD). Categorical variables are reported as frequencies and percentages.
To account for the variable number of measurements per patient in our study population, the effect of antipsychotic augmentation and other covariates on clozapine serum concentrations was estimated by multivariate linear mixed model analysis. Study ID was used as a cluster variable. Random coefficients were “Intercept|Study ID” and “Time from Admission to Level|Study ID”. Clozapine serum concentrations were log10 transformed to correct the positive skewness of the residuals. Residuals were then visually inspected via histogram to ensure normal distribution. Multicollinearity was evaluated for the independent variables; and dependent variables underwent outlier analysis. An alpha threshold of 0.05 was used for all tests. To assess the impact of antipsychotic augmentation on dose-related adverse events and anticholinergic use, logistic generalized mixed models were utilized to account for the variable number of measurements across patients.
Results
Patient Characteristics
Among the 97 patients prescribed clozapine during the study period, 80 completed the program while remaining on clozapine, were discharged with a clozapine prescription, and were included in analysis. From these individuals a total of 306 clozapine plasma concentrations were collected (Figure 1). Table 1 shows clinical and demographic characteristics of the study population. Most patients were male of European ancestry with a diagnosis of schizophrenia. All patients had a non-nicotine SUD, with stimulant use and opioid use disorders being most common. Mean HONOS scores (which include relevant ratings of behaviour, drug use, cognitive problems, and hallucinations/delusions) were similar between the clozapine monotherapy and polytherapy groups (Table 1). Out of 306 clozapine serum levels, 40 were obtained from individuals concurrently prescribed low-dose fluvoxamine to enhance clozapine concentrations (mean fluvoxamine dose: 29.17 mg, SD: 17.9). Among all samples, 82 levels were in the low range (<250 ng/mL), 169 were within the recommended range (≥250 ng/mL to ≤550 ng/mL), and 55 were in the higher range (>550 ng/mL). 34
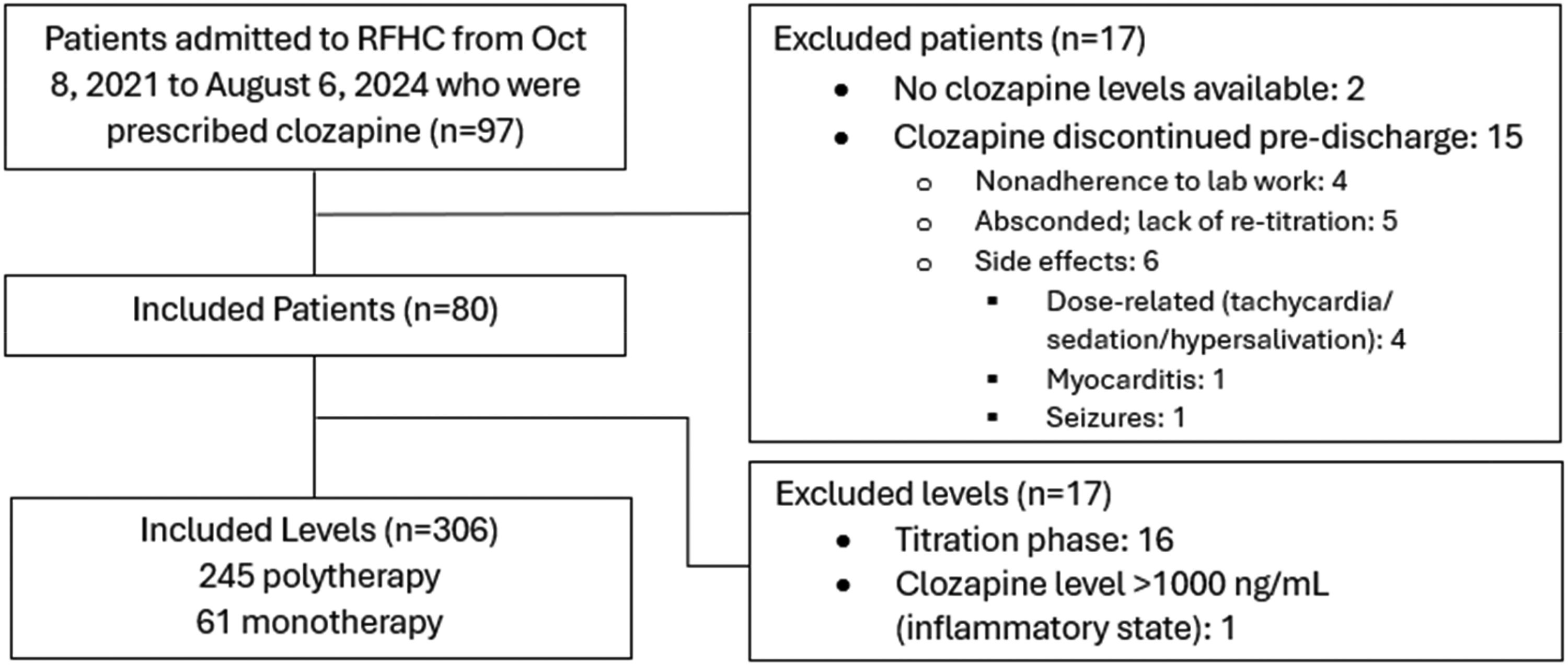
Flowchart of patient selection.
Clinical and Demographic Characteristics of the Study Population.

DDD=Defined Daily Dose; CRS=clozapine-resistant schizophrenia; SD=standard deviation; LAI=long-acting injectable.
Nearly half (n = 38) of patients had clozapine initiated during admission, while the remainder had clozapine initiated prior to admission. Nearly three-quarters (n = 57) of the patients were receiving antipsychotic augmentation at discharge. The choice of augmentative antipsychotic therapies varied, with aripiprazole, quetiapine, and paliperidone being the most utilized (Supplementary Table S1).
Clozapine Monotherapy Utilization
Out of 57 patients on antipsychotic augmentation, only 13 (23%) of these patients had a clozapine monotherapy trial in the past. The remaining patients had clozapine added onto their previous antipsychotic regimen. Out of those 13 patients, only 2 (15%) patients had documented diagnosis of CRS.
Impact of Antipsychotic Augmentation on Clozapine Serum Concentration
Antipsychotic augmentation had a negative correlation with clozapine serum concentrations, (B = –0.039; 95% CI = –0.078 - −0.001) (Table 2, Supplementary Figure S1). Greater body weight adjusted by height, and longer duration from admission to blood sampling were positively associated with clozapine serum concentrations, whereas male sex was negatively associated (Table 2).
Multivariate Mixed Model for Impact of Independent Variables on Variability in Clozapine Serum Concentrations.a
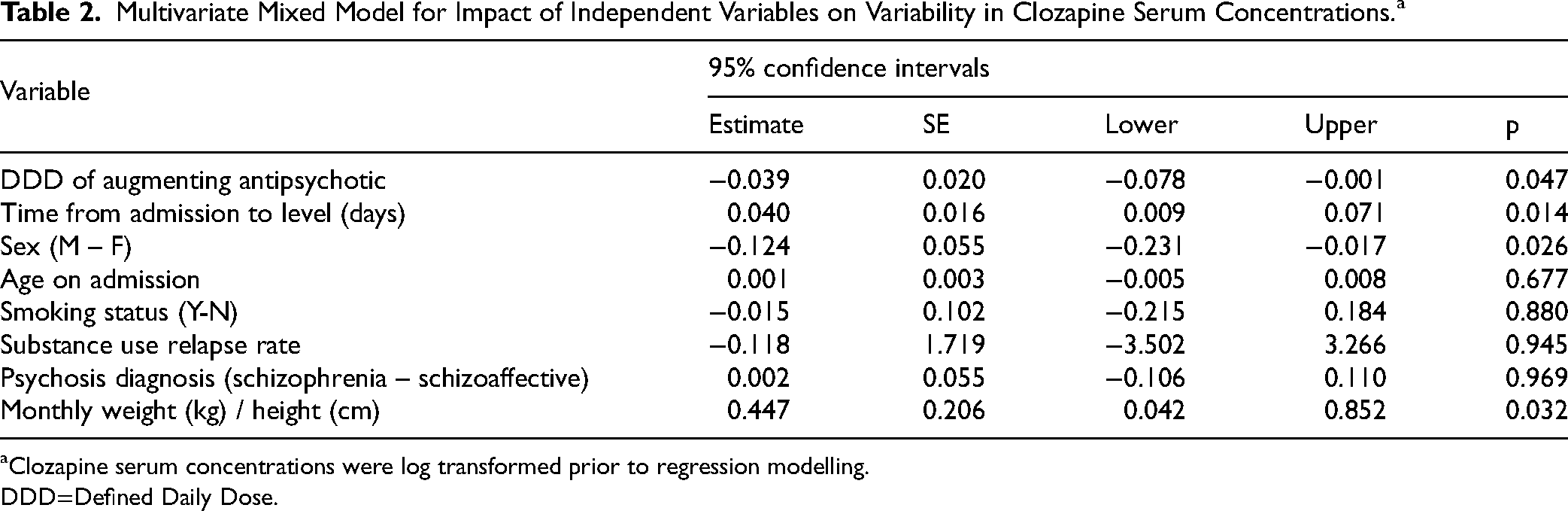
aClozapine serum concentrations were log transformed prior to regression modelling.
DDD=Defined Daily Dose.
Adverse Events with Antipsychotic Augmentation
No associations between antipsychotic augmentation and incidence of dose-dependent clozapine adverse events (tachycardia, constipation, or anticholinergic agent use) were detected (See Supplementary Tables S2, S3, S4).
Discussion
In this study, we examined clozapine prescribing patterns in a provincial residential treatment centre specializing in severe concurrent mental health and SUDs. We found that clozapine augmented with other antipsychotics was nearly 2.5 times more prevalent than clozapine monotherapy, despite 78% of these patients never having received a guideline-recommended “adequate” trial of clozapine monotherapy. Our analysis of the time variable indicated that the longer individuals stayed at RFHC, the more practitioners tended to increase their clozapine levels, potentially reflecting adjustments prompted by substance use relapse or ongoing residual symptoms.
Antipsychotic augmentation had a modest inverse association with serum clozapine levels, reflected in a shallow decline in the slope of dose–response relationship (Supplementary Figure S1), without any detectable reduction in adverse events in comparison with monotherapy. The modest association between polytherapy and lower clozapine serum concentrations may reflect clinicians’ inclination to reduce clozapine doses when additional antipsychotics are prescribed. However, the effect on the dose–response relationship (Supplementary Figure S1) is most pronounced at higher DDDs of co-prescribed antipsychotics, raising the possibility that these adjustments occur unconsciously, driven by caution regarding cumulative antipsychotic exposure, rather than as a fully deliberate clinical strategy. Our findings call into question the commonly cited clinical justifications for antipsychotic augmentation – namely, to address persistent symptoms non-responsive to clozapine monotherapy or consciously targeting a lower clozapine exposure for better tolerability.
The decision to use clozapine monotherapy or antipsychotic augmentation in TRS introduces a set of nuanced clinical challenges. Moreover, little is known about the relationship between clinicians’ initial prescribing rationale and the long-term outcomes of antipsychotic augmentation. While continuation of prior antipsychotics during clozapine cross-titration is common, many patients remain on these agents even after achieving target clozapine doses – possibly due to concerns about relapse or non-adherence, especially when long-acting injectables (LAIs) are involved.35,36 However, continuing antipsychotics for which a patient has already shown non-response may provide little benefit, potentially increasing medication burden without improving outcomes. In patients with co-occurring SUDs, this burden may also exacerbate adverse events and influence substance use trajectories through dopaminergic suppression, potentially increasing sedation, parkinsonian symptoms, cravings, and substance use relapse risk.37,38
Our study was not designed to assess the possible superiority or inferiority of antipsychotic augmentation compared with clozapine monotherapy in this difficult-to-treat TRS-SUD population. While preliminary observational evidence has suggested that clozapine-LAI combinations are associated with decreased psychiatric hospitalization rates in outpatient settings,34,35 our findings in inpatient settings showed comparable reductions in underlying symptoms and SUD severity (based on HONOS scores) across both clozapine monotherapy and antipsychotic augmentation groups. However, due to potential selection bias, we cannot infer equivalence – only that our findings align with existing literature showing limited benefit from antipsychotic augmentation in TRS.9,39 The observed departure from the prioritization of guideline-recommended clozapine monotherapy in this population highlights the pressing need for updated, pragmatic clinical guidance that accounts for real-world practices and the complexities inherent in TRS with comorbid SUD. Current monotherapy-focused guidelines do not sufficiently address the multifaceted challenges faced in managing this high-risk subgroup. Residual pharmacodynamic effects from previously prescribed antipsychotics may obscure the assessment of early clozapine treatment response, while the ongoing paucity of robust long-term data limits confidence in the use of clozapine-antipsychotic polypharmacy. Future clinical guidance should provide practical recommendations regarding the use of antipsychotic augmentation therapies, including considerations for clinical rationale, documentation standards, monitoring strategies, cost analysis, and outcome evaluation. Such guidance is essential to support evidence synthesis, and inform safer, more effective prescribing practices in this complex population.
Several limitations should be noted. First, we used electronic health records to collect information, and this approach can result in some data being entered incorrectly or omitted. Second, for individuals admitted already on clozapine, we depended on medication histories compiled by pharmacists to evaluate prior treatment trials, which may be subject to inconsistencies or missing information from community-based records. Third, proxies were used to assess adverse events incidence (i.e., beta-blocker, laxative, and anticholinergic medication use) which may result in some adverse events where these proxies were not used going undetected. Although we confirmed via clinical notes that these medications were prescribed for the purpose of treating these adverse events, this approach is likely to underestimate the true incidence rate of these events and therefore caution should be exercised in interpreting these results. Fourth, we did not explore the impact of augmentation with individual antipsychotics, and therefore further study is required to determine whether certain augmenting agents may be more effective than others which has been suggested by previous publications. 12 Finally, our study examined patients in a controlled inpatient setting with overall excellent medication adherence, and results may not be generalizable to outpatient settings with high levels of non-adherence.
In conclusion, clozapine monotherapy – despite being the recommended standard in TRS guidelines – was markedly underutilized. Over three-quarters of patients receiving clozapine therapy with antipsychotic augmentation had not undergone a guideline-concordant, adequate trial of clozapine monotherapy. Furthermore, antipsychotic augmentation had a modest effect on lowering clozapine concentration targets and did not reduce antipsychotic-related adverse events. Collectively, these findings challenge the commonly cited clinical justifications for clozapine augmentation – namely, the management of persistent symptoms unresponsive to monotherapy or the deliberate use of lower clozapine doses to enhance tolerability. Future clinical guidelines should incorporate context-sensitive recommendations that address the complexities of treating individuals with SUD comorbidity. Specifically, guidance should be provided on clinical decision-making frameworks, standardized documentation practices, appropriate monitoring protocols (including safety and efficacy parameters), cost-effectiveness considerations, and meaningful outcome metrics that align with both psychiatric and addiction-related treatment goals.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251408172 - Supplemental material for Clozapine Therapy With or Without Antipsychotic Augmentation: A Retrospective Evaluation of Prescribing Practices in a Canadian Provincial Residential Treatment Centre for Concurrent Disorders: Traitement par la clozapine avec ou sans intensification à l’aide d’antipsychotiques : évaluation rétrospective des pratiques de prescription dans un centre de traitement résidentiel provincial canadien pour les troubles concomitants
Supplemental material, sj-docx-1-cpa-10.1177_07067437251408172 for Clozapine Therapy With or Without Antipsychotic Augmentation: A Retrospective Evaluation of Prescribing Practices in a Canadian Provincial Residential Treatment Centre for Concurrent Disorders: Traitement par la clozapine avec ou sans intensification à l’aide d’antipsychotiques : évaluation rétrospective des pratiques de prescription dans un centre de traitement résidentiel provincial canadien pour les troubles concomitants by Laura Frankow, Chad A Bousman, Nickie Mathew and Reza Rafizadeh in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
We would like to recognize that our work takes place on historical and contemporary Indigenous lands. We also acknowledge the many Indigenous communities forged in Canada's urban centres.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
RR, LF and NM have none to declare. CAB is founder and CEO of Sequence2Script Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Reza Rafizadeh is supported by 2025 Michael Smith Health Research BC / BC Schizophrenia Society Foundation Health Professional-Investigator award.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
