Abstract
Plain Language Summary Title
Citalopram vs. Escitalopram for major depression: No Real Difference in Efficacy or Safety, Just Higher Cost.
Plain Language Summary
Background:
Citalopram is an antidepressant. Escitalopram is a closely related drug—basically a slightly modified version of citalopram—that drug companies promote as being safer, more effective, and faster acting. In Canada, escitalopram costs about twice as much as citalopram. We wanted to know how these two drugs are actually used in British Columbia and whether escitalopram really works better or is any safer.
What We Did:
We looked at prescription records for the nine most common antidepressants in BC between 2005 and 2024. We also reviewed all the studies that directly compared citalopram with escitalopram. We focused on whether any differences were large enough to matter to patients, whether study results were reliable, and whether studies had problems such as bias or conflicts of interest.
What We Found:
By 2013, escitalopram had become the most prescribed antidepressant in BC, while citalopram use dropped. We found 16 studies that compared the two drugs. Many had problems such as missing data, selective reporting, or funding from the drug manufacturer. None of the studies showed meaningful differences in effectiveness. Claims that escitalopram works faster were weak and inconsistent. Side effects were essentially the same, including risks for heart rhythm changes (QT prolongation).
Introduction
Citalopram is a racemic selective serotonin reuptake inhibitor (SSRI) composed of the R(−) enantiomer and S(+) escitalopram, the pharmacologically active S-enantiomer. Escitalopram has been promoted as safer, more efficacious, and faster acting than citalopram for major depressive disorder (MDD). Escitalopram is twice as expensive in Canada, raising the question of whether preferential prescribing is justified. Our objectives were to examine patterns of escitalopram and citalopram use in British Columbia (BC), and to determine whether claims favouring escitalopram are based on science.
Methods
We analyzed the annual prevalence of antidepressant use per 1,000 persons in BC from 2005 through 2024 using PharmaNet, 1 which captures all community pharmacy dispensing except for people covered by Indigenous health services or by the Government of Canada. We included adults ≥19 years diagnosed with depression, based on hospitalizations, emergency visits, or physician billing records.
To evaluate comparative efficacy, we reviewed head-to-head randomized controlled trials (RCTs) identified from reviews, an updated Medline search, product monographs, regulatory submissions (US FDA, Health Canada), and international health advisories. We translated RCTs published only in Chinese. We assessed whether reported differences met minimal clinically important thresholds, examined robustness of dichotomous outcomes against early withdrawals using the Fragility Index, and applied the Cochrane Risk of Bias tool (ROB2). 2–4
Results
Escitalopram use increased after 2009, surpassing citalopram by 2013. It remained BC's most prescribed antidepressant through 2022 (32 per 1,000 persons/year). Over the same period, citalopram use declined to 12 per 1,000 persons/year (See Figure 1).
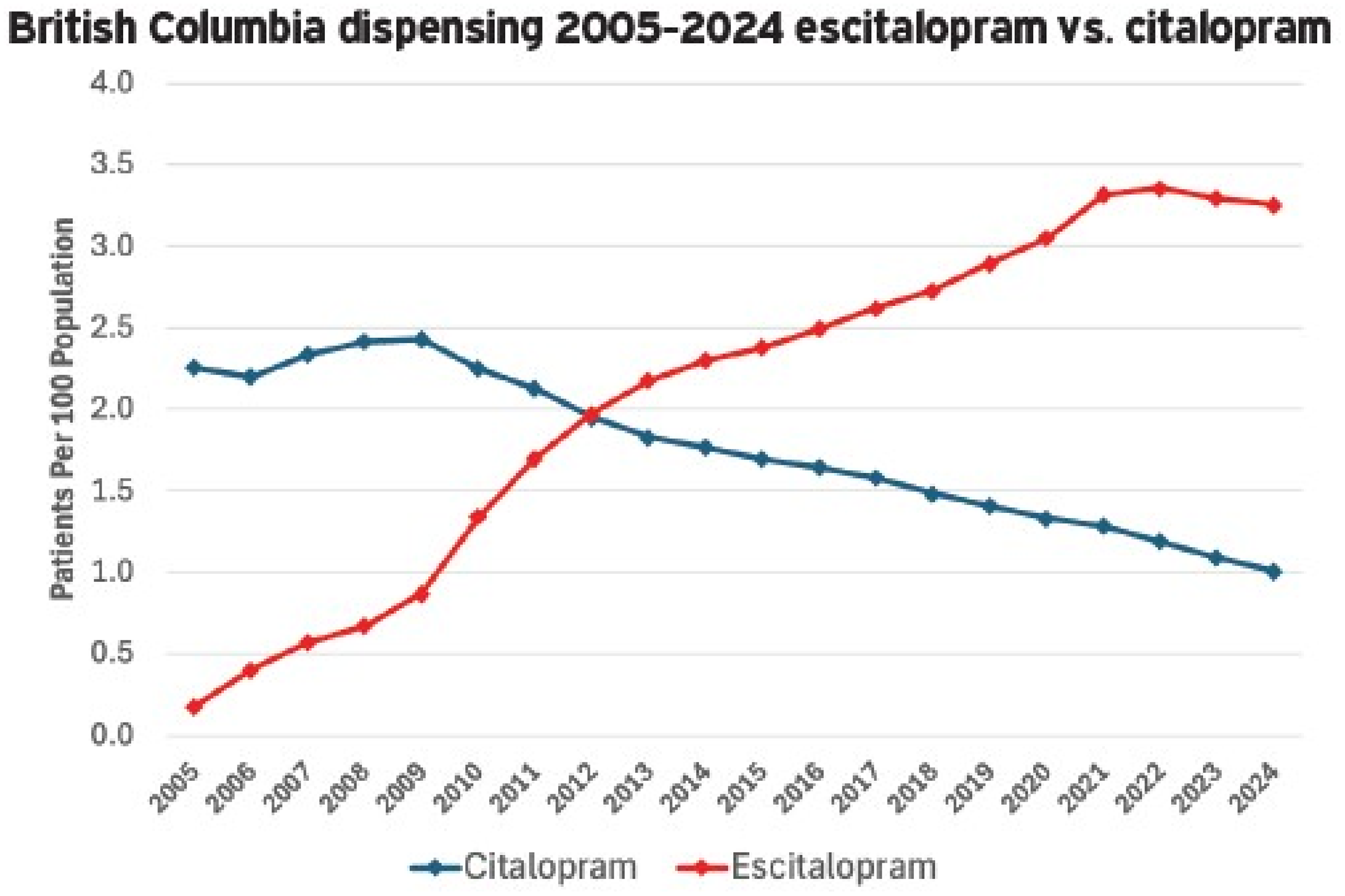
Antidepressant use in British Columbia from 2005 to 2024.
Four prior reviews cite 12 RCTs comparing escitalopram with citalopram. We identified an additional 4 RCTs for a total of 16. One large multinational RCT was published twice, and 1 of 2 US RCTs (SCT-MD-02) was not published. In 2001, Forest Laboratories submitted to Health Canada and the US FDA the unpublished results of SCT-MD-02 showing no difference between escitalopram and citalopram. A 2014 publication presented a pooled analysis of 3 Chinese RCTs, all negative, but counted them as 1 RCT in a prior review. After translating 5 Chinese RCTs, we included data not previously available in English. These steps corrected mistaken accounting in prior reviews and expanded the evidence base.
The high or unclear risk of bias for all RCTs rendered meta-analysis inappropriate. 5 Frequent concerns included industry sponsorship of at least 11 of 16 RCTs, selective outcome reporting, incomplete data, and small-study effects. Industry funding is consistently linked to overestimation of benefits, further limiting confidence in reported differences.
No RCT reported an efficacy difference exceeding a clinically important threshold. Of the 16 RCTs, 3 reported statistically significant differences in clinical response or remission favouring escitalopram. However, in 1 RCT, both response and remission were fragile; in another, response was reported but fragile; and in a third, response was fragile but remission was not. Three RCTs suggested earlier symptom improvement with escitalopram at Week 1, but no early difference persisted, and Week 1 differences were trivial or indeterminable. Adverse drug reactions, including QT prolongation, were comparable, with no evidence that the R-enantiomer of citalopram worsens safety outcomes.
Discussion
Evidence from RCTs does not demonstrate clinical superiority of escitalopram compared with racemic citalopram. Our findings are consistent with the Long et al. review of single-enantiomer vs racemic drugs that included 8 RCTs comparing escitalopram with citalopram, of which 7 showed no difference in efficacy. Through literature search, translation of RCTs published in Chinese, and inclusion of unpublished RCT data submitted to Health Canada, we incorporated previously inaccessible data.
It is notable that the unpublished RCT conducted from 1999 to 2000 by Forest Laboratories was negative. In SCT-MD-02, both citalopram and escitalopram failed to show superiority over placebo for the treatment of MDD. In addition, an RCT published in 2003 reported data from the full 8-week study that had been selectively published in 2001. Our review is the most complete accounting to date of RCTs directly comparing escitalopram with racemic citalopram.
Claims of escitalopram's superiority arose from preclinical findings. In vitro and animal studies suggested faster onset or enhanced serotonergic activity; however, these differences were not borne out clinically. Both drugs prolong the QT interval, and at recommended doses, no RCT has shown QT-related adverse events unique to either drug. More extensive regulatory warnings for citalopram likely reflect its longer market history.
Limitations include incomplete information for 3 RCTs subsumed in a 2014 publication and missing information about funding and methodology for 6 RCTs. Our translations of RCTs published in Chinese are available from the corresponding author.
Conclusion
Preferential prescribing of escitalopram and claims of superiority are not supported by science. Comparative efficacy and safety data indicate that escitalopram and citalopram are therapeutically equivalent, although escitalopram is twice as potent as the racemate.
Footnotes
Acknowledgments
Arrietty Song for translating non-English RCTs
Data Source Declaration
Access to data provided by the BC Ministry of Health is subject to approval but can be requested for research projects through the Data Stewards or their designated service providers. The following data sets were used in this study: PharmaNet, Medical Services Plan, Discharge Abstract Database, National Ambulatory Care Reporting System, and Client Roster. All inferences, opinions, and conclusions drawn in this publication are those of the authors, and do not reflect the opinions or policies of the Data Stewards. The data were provisioned under ISA 16-036. Details of our analysis can be requested from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
