Abstract
Objective
The effectiveness of current treatment options for depressive symptoms has been widely investigated with acknowledgment that some patients were either not adequately responding to treatment, finding the existing treatment intolerable, or otherwise prefer alternative options. There is increasing interest in microbiota modulation as an alternate form of depression treatment, with a growing number of trials and reviews on the subject published in the last five years. This systematic review aimed to analyze all completed randomized control trials (RCTs) that assessed depression symptoms in adults not using antidepressants, before and after oral methods of microbiota manipulation.
Method
All completed parallel-arm RCTs that assessed depression symptoms in adult participants before and after oral methods of microbiota manipulation were retrieved from four databases, MEDLINE, Embase, PsycINFO, and Cochrane Central Register of Controlled Trials. Data on study and intervention characteristics as well as RCT conclusions were collected independently and in duplicate, and each study's findings were summarized individually. Risk of bias was completed.
Results
We included 66 RCTs in our review, 34 of which concluded significant differences between the intervention and control group in depressive symptom using different interventions and measures. Of the 66 trials, 54 used probiotic interventions, seven used prebiotic, eight used synbiotic and two used oral fecal microbiota transplantation. Wide variation was observed in studies’ design, intervention composition and consumption methods across all 66 RCTs. No statistical synthesis or meta-analyses were possible due to the wide variety of interventions, measures and outcomes.
Conclusions
The heterogeneity of the existing RCTs did not allow for concrete conclusions on whether oral microbiota manipulation interventions are viable alternative treatment options for adults experiencing depression symptoms. We encourage the development of standardized guidelines for the design and reporting of microbiota studies in depression for the possibility of future intervention efficacy testing.
This is a visual representation of the abstract.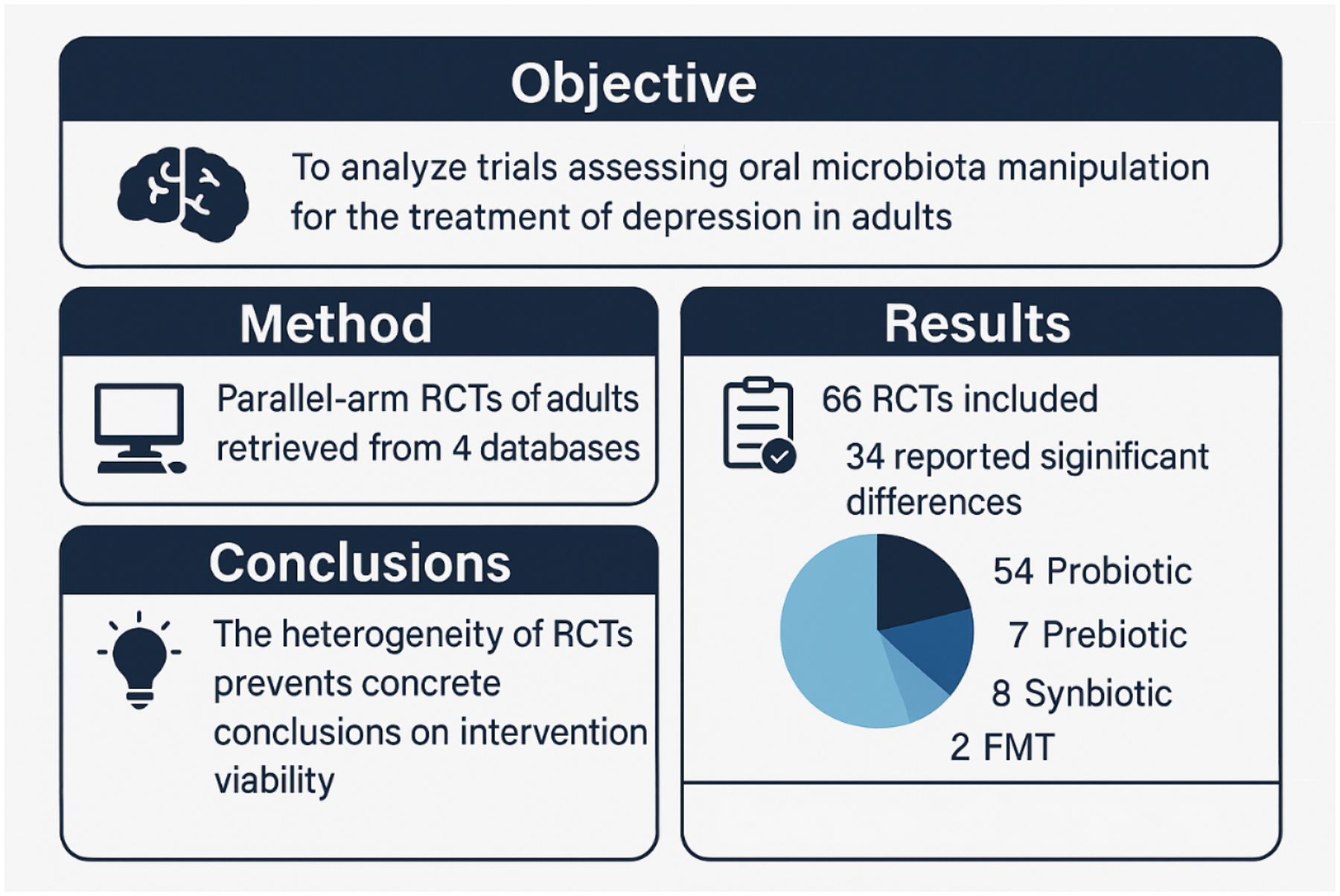
Introduction
Depressive symptoms are commonly treated with effective interventions, 1 however tolerability challenges and patients’ preferences for alternatives to pharmacotherapies have been seen. Such alternatives included microbiota manipulation 2 in depression.3,4
The gut microbiota, the microorganism population within the human gastrointestinal tract, is a dynamic, individually unique and complex system that has been found to affect nearly every aspect of the body and development. 2 Various factors affect gut microbiota makeup including infant delivery and feeding, age, diet, medications, stress, and geographical location. 5 An individual's microbiome is involved in overall health, metabolism, digestion, immune system function and protection, as well as central nervous system development. 6 The health and composition of the gut microbiome is crucial for the maintenance of the other body systems. Significant differences in microbial composition have been found when comparing patients with healthy controls in a variety of illnesses including inflammatory bowel disease, cancer, obesity, as well as depression4,7 Dysregulated gut microbiota has been linked to the development of gastrointestinal inflammation, cardiovascular and respiratory illnesses, and neuropsychiatric disorders, like depression and anxiety.8,9
Microbiota manipulation is a process that aims to alter one's microbiota population, composition, and metabolic output, 10 the most common methods included dietary changes (e.g., anti-inflammatory diet, ingestion of fermented foods), fecal microbiota transplantations (FMTs), and probiotic, prebiotic or synbiotic consumption.8,11 The terminology for the varying methods of oral microbiota manipulation is often inconsistently described and not clearly defined, though there are a few notable differences between them. FMT consists of the replacement or integration of one individual microbiota into another individual, transferring fecal microbiomes of healthy individuals into ill individuals in hopes of repairing the assumed imbalanced native microbial composition. 12 There are several methods to introduce FMT in humans including oral capsules to be ingested by the individual, gastric tube, colonoscopy insertion or endoscopy through oesophagogastroduodenoscopy. 13 The focus of this review is on the oral capsules.
Other oral microbiota manipulation methods include probiotics, prebiotics and synbiotics. Probiotics can fall under the classification of a dietary supplement and are defined as microecologics, strictly selected live strains of microorganisms that are used to help with whole-body immunity, intestinal flora structure and inhibit harmful microorganism overgrowth. 14 While similar to FMT capsules, probiotics are made up of a small and limited number of bacterial cultures, stemming from a variety of locations throughout the human intestinal tract, as well as animal- and plant-based sources as opposed to direct sampling from a donor stool. 15 Prebiotics consist of fibre sourced from non-digestible carbohydrates and oligosaccharides and have been reported to selectively stimulate and modulate the growth and activity of colon microbial species and reduce the severity of gastrointestinal symptoms such as diarrhea and inflammation. 16 Synbiotics14,16 are a combination of prebiotics and probiotics, mixed in a synergistic manner with the aim of improving the beneficial qualities and survival of both substances. The prebiotic compounds selectively favour the growth and increase the metabolism of probiotic organisms, assisting in their passage through the upper intestinal tract and implantation within the colon.
The relationship between the gut microbiome and the central nervous system, the gut–brain axis, has been studied in great depth 17 suggesting bidirectional communication. This bidirectional communication network extends from the gastrointestinal tract to the central, autonomic and enteric nervous systems as well as the hypothalamic pituitary adrenal axis, linking gut function to the cognitive and emotional centres of the brain. 18 Impacting one another, gastrointestinal symptoms such as indigestion, bloating, constipation and heartburn have been seen to arise with emotional or psychosocial events and disturbances in gut microbiome composition. Decreased microbiota diversity has been linked to the development and worsening of several psychiatric conditions including depression and anxiety.19,20 With evidence of such a dynamic relationship, there has been an uptake of research examining microbiome manipulation on psychological pathologies and behaviour.21,22
There has been an increasing interest in microbiota modulation as an alternate form of depression symptom treatment, with a growing number of trials and reviews on the subject published in the last few years.23–25 These studies included various forms of microbiota including pre, pro and synbiotics, food supplements, and a limited number of trials. The most recent review published in 2023 included only seven trials 26 and the review that included FMT was conducted in 2021 23 and identified only one trial thus providing limited data to understand the role of FMT in depressive symptoms. The objective of this systematic review is to identify completed randomized control trials (RCTs) that assessed oral microbiota manipulation intervention in adults with depression symptoms not using antidepressants.
Methods
This review was not pre-registered. We conducted a systematic review following the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA). 27 This review included all completed RCTs that investigated methods of oral microbiota manipulation on the depressive symptoms measured using validated depression symptoms scale in adult participants. No Language restrictions were placed on the search strategy (see Supplemental e-Tables 1 and 2 for the inclusion criteria and search strategy). We excluded studies that were not an RCT design; followed a cross-over design; did not exclude participants who were receiving antidepressant treatment; used interventions combined with vitamins or supplements; or used diet manipulation as the primary intervention. We searched MEDLINE (1946 to 08 March 2023), Embase (1974 to 08 March 2023), PsycINFO (1987 to 2023 February) and Cochrane Central Register of Controlled Trials (2023 February) from inception to 9 March 2023. We re-run the search on 22 February 2024, to identify any additional trials. Title and abstract, full-text screening, and data extraction were completed independently and in duplicate by eight reviewers (AO, AM, JS, OS, GH, MB, JA, MH) in Covidence 28 following the Cochrane guidelines for systematic reviews. 29 Data extraction included study inclusion/exclusion criteria, participants’ baseline characteristics, the measure of depressive symptoms, the type of intervention, the duration, statistical analyses used, limitations and conclusions (see Supplemental e-Table 6: Systematic Review Data Extraction Variables). Each study's results were entered separately in Excel with the aim of synthesizing the data for meta analyses however no meta-analyses were possible due to the diversity of the studies identified. Risk of bias (RoB) was assessed independently and in duplicate by five reviewers (AO, OS, GH, MB, and JA) using the Cochrane RoB Tool. Screening, data extraction and assessment disagreements were resolved through discussion. The reporting of this review follows the PRISMA recommendations. We reported probiotic strain by genus, species, subspecies and alphanumeric designation as directed by the World Gastroenterology Organisation 30 and reported study limitations using standardized methods for RCTs. 31
Results
Study Selection
The search strategy identified 4,979 studies, of which 2,876 were duplicates and were removed by Covidence 28 and 31 duplicates were identified manually. The remaining 2,072 articles were screened through title and abstracts, 1,781 irrelevant articles were excluded, leaving 291 articles eligible for full-text screening. Of the 291 articles eligible for full-test screening, 225 studies were excluded, 132 of which were published protocols or ongoing trials with no published results and only one study was published in Farsi that met the inclusion criteria 32 focused on patients with hypothyroidism and fatigue and was excluded as it was unclear how depression symptoms were measured including the validation of the scale used. The second most common reason for excluding articles was the presence of a co-existing treatment within the trial or study population (k = 35). We included 66 studies in our systematic review. The study selection flow diagram is presented in Figure 1.
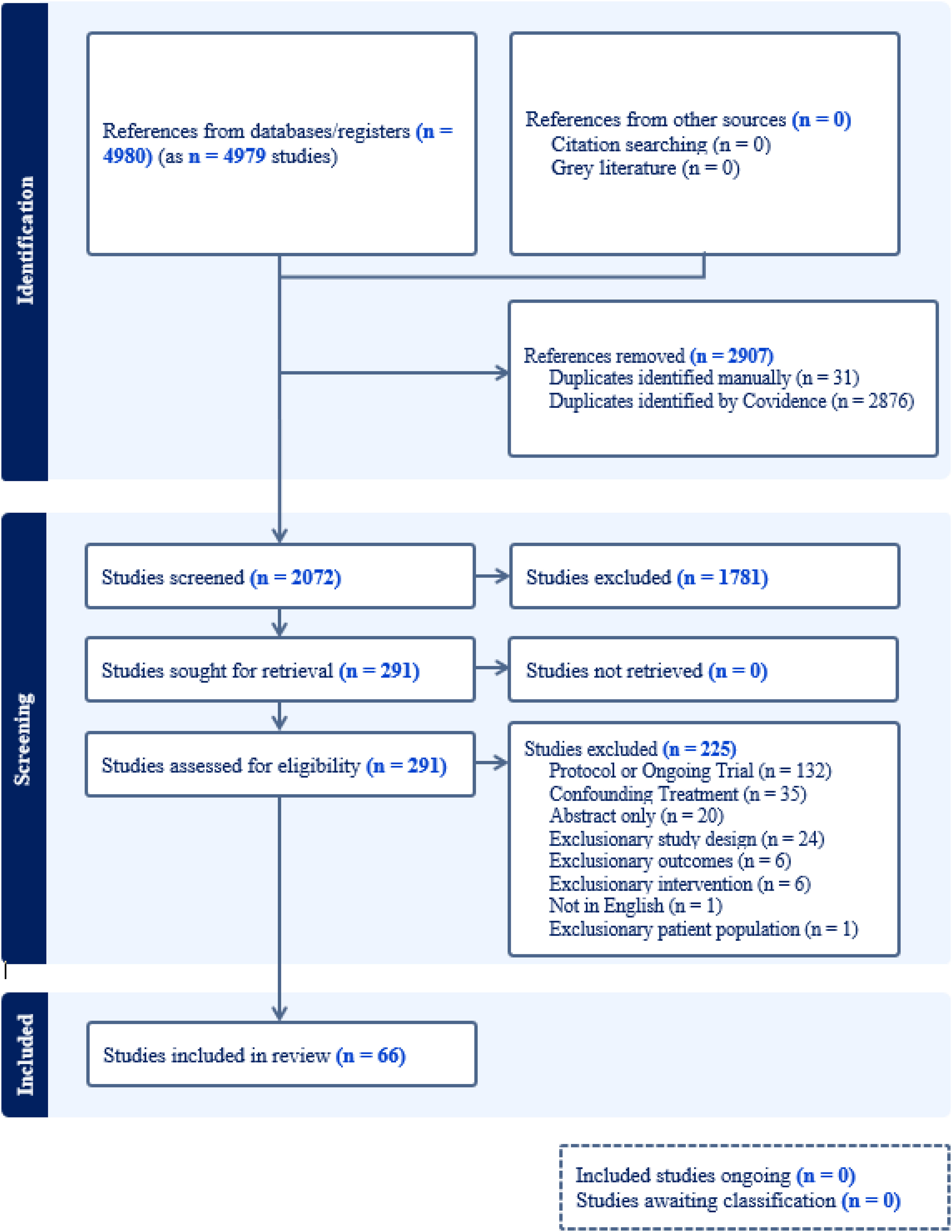
PRISMA flow diagram.
RoB Assessment
The RoB for studies included in our study is summarized in Supplemental eTable 12 (RoB and eFigure 2: RoB – Robvis Traffic-light Plot, eFigure 3: RoB Robvis Summary Plot). Of the 66 included studies in our review, 16 (24%) were assessed to be at high overall RoB and 23 (35%) were assessed to have some concerns. Randomization was generally deemed to be low for RoB, with only two (3%) studies reported with a high RoB. Assignment to intervention RoB showed 20 (30%) studies with some concerns and three (5%) with high RoB, and adherence to intervention was generally deemed low for RoB (k = 58 low). Missing outcome data had two (3%) studies deemed high for RoB and 19 (29%) with some concerns, and selection of reported result was generally deemed low for RoB (k = 56 low).
Study Characteristics
Each of the 66 studies was found to have unique characteristics, outlined in Supplemental eTable 7. We found that the included studies originated from various countries, the most common countries of studies were Iran (n = 12) and China (n = 6). The sample size of the studies also varied greatly from as low as n = 18 to as high as n = 400 (median n = 59.5). The reporting of participants’ demographics varied across the studies. The lowest mean age reported was 19.4 years, while the highest mean age reported was 77.2 years. Most studies (k = 46) reported a mean or median age less than 50 years old. The percentage of female participants also varied greatly. We found three studies that did not report sex proportion, one study included exclusively male participants, and nine studies included exclusively female participants. Most of the included studies reported a majority (more than 50%) female population (k = 50). The most common population type included in the studies was healthy adults (k = 13), followed by patients with gastrointestinal disorders (k = 9), healthy adults with subclinical depression, anxiety or stress symptoms (k = 8) and psychiatric patients diagnosed with major depressive disorder (MDD) or bipolar disorder (k = 6).
Intervention Characteristics
Of the 66 trials included, 54 used probiotic interventions, seven used prebiotics, eight used synbiotics, and two studies used oral FMTs. All data pertaining to study intervention characteristics are summarized in Supplemental eTables 8 (eTable 8. Probiotic Study Intervention Characteristics), 9 (eTable 9. Prebiotic Study Intervention Characteristics, 10 (eTable 10. Synbiotic Study Intervention Characteristics), and 11 (eTable 11. Oral FMT Study Intervention Characteristics).
For the 54 studies that used a probiotic intervention, the most common method of consumption was a capsule (k = 34), followed by a powder (k = 19). One study used a beverage probiotic intervention. The probiotic studies varied in length; the shortest duration study reported a seven-day time frame, and the longest duration study a six-month time frame. The most common intervention length found amongst the probiotic trials was eight weeks (k = 14), followed by 12 weeks (k = 13). The most common intervention consumption dosing schedule was one dose daily (k = 35), with instructions varying with regards to the time of day to consume the intervention, with or without food, and the amount and/or type of liquid to consume the intervention with. Intervention compositions were also of wide variety with 36 different strains identified, distinctions made by genus, species and subspecies. Many of the trials (k = 32) used multi-strain probiotics, the number of bacterial strains used in an intervention varied from 1 to 14, most trials using a single strain intervention (k = 22) or a combination of two to four strains in their intervention (k = 23). We found that the most common bacterial species used was Lactobacillus acidophilus (k = 23), followed by Bifidobacterium bifidum (k = 18) and Bifidobacterium longum (k = 19). Dosing with regards to colony-forming units was also varied.
For the seven studies that used a prebiotic intervention, the most common method of consumption was a powder (k = 4), followed by delivery through a capsule (k = 1), a gummy (k = 1) and a jelly (k = 1). The prebiotic studies varied in duration; the shortest study reported a three-week duration (k = 1), the majority reported a four-week duration (k = 4), and the longest studies reported an eight-week time frame (k = 2). The intervention composition and consumption schedules also varied greatly across included trials, with each intervention using a unique recipe of dietary fibres and unique dosing instructions.
For the eight studies that used a synbiotic intervention, the most common methods of consumption were a capsule (k = 3) and a powder (k = 5), one study intervention with an unclear delivery format. The synbiotic studies also varied in length; the shortest study reported a six-week time frame, and the longest study a 24-week time frame. The most common intervention length found amongst the synbiotic trials was eight-weeks (k = 3). Intervention compositions varied, each presenting with a unique combination of bacterial strains and dietary fibres. The intervention consumption schedules for the synbiotic trials were for the majority one dose daily (k = 5), instructions varying with regards to time of day, with or without food, and amount or type of liquid to consume the intervention with.
For the two studies that used an oral FMT intervention, both used capsules as their delivery method. The oral FMT studies also varied in length, with one study reporting a three-week time frame and the other a 12-week time frame. The intervention compositions were unique to each study, and the intervention consumption schedules differed in number of capsules per dose and timing of dosage.
Depression Symptoms Outcomes
Table 1 provides the depression outcomes from each study. Thirty four out of 66 trials (total sample size for the 34 trials, n = 2,957) concluded the intervention showed a significant improvement in depression symptom. The baseline depression severity across the studies varied from “healthy” to severe symptoms based on the various measurement scales. Measurement of depression symptoms also varied including up to four scales used in the same study. Most studies used one scale to measure depressive symptoms (k = 37). A total of 34 different scales were used across the 66 studies, the most common depression assessment tool was Beck Depression Inventory II (BDI-II) (k = 16), followed by Hospital Anxiety and Depression Scale (HADS) (k = 11), and BDI (k = 8). The reporting of results was also inconsistent across the studies, with results presented as mean scores at baseline and checkpoints, mean score changes, proportion of participants improved, as well as only visually. The most common study limitations reported were related to outcome measures (k = 54), underpowered studies (k = 30) and study duration limitations (k = 18).
Depression Outcome.
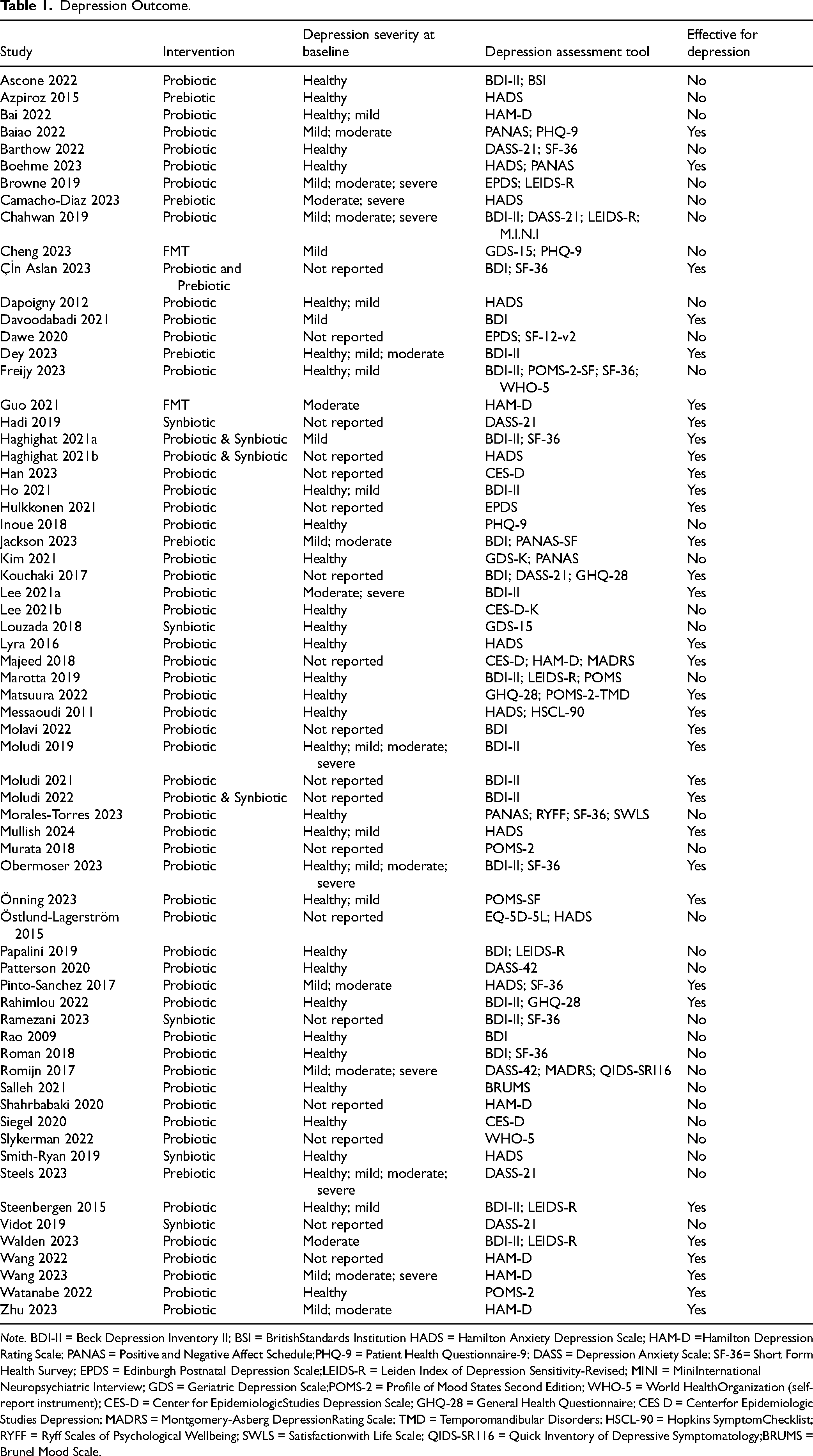
Note. BDI-II = Beck Depression Inventory II; BSI = BritishStandards Institution HADS = Hamilton Anxiety Depression Scale; HAM-D =Hamilton Depression Rating Scale; PANAS = Positive and Negative Affect Schedule;PHQ-9 = Patient Health Questionnaire-9; DASS = Depression Anxiety Scale; SF-36= Short Form Health Survey; EPDS = Edinburgh Postnatal Depression Scale;LEIDS-R = Leiden Index of Depression Sensitivity-Revised; MINI = MiniInternational Neuropsychiatric Interview; GDS = Geriatric Depression Scale;POMS-2 = Profile of Mood States Second Edition; WHO-5 = World HealthOrganization (self- report instrument); CES-D = Center for EpidemiologicStudies Depression Scale; GHQ-28 = General Health Questionnaire; CES D = Centerfor Epidemiologic Studies Depression; MADRS = Montgomery-Asberg DepressionRating Scale; TMD = Temporomandibular Disorders; HSCL-90 = Hopkins SymptomChecklist; RYFF = Ryff Scales of Psychological Wellbeing; SWLS = Satisfactionwith Life Scale; QIDS-SR116 = Quick Inventory of Depressive Symptomatology;BRUMS = Brunel Mood Scale.
Studies With a Diagnosis of MDD
Five trials with sample size varied from n = 40 to n = 79 (total n = 305) investigated microbiota in individuals with MDD.33–37 Three of these studies33,35,36 (n = 155) used probiotic intervention and concluded reduction in depressive symptoms while two studies (n = 150) also used probiotic intervention34,37 did not find a significant difference in depressive symptoms between intervention and control groups. Two studies that showed an improvement in depressive symptoms included individuals with comorbid irritable bowel syndrome.35,36
Discussion
We identified 66 completed parallel-arm RCTs that assessed depressive symptoms in adults before and after oral methods of microbiota manipulation. Among the 66 studies, we found 54 probiotic intervention trials, seven prebiotic, eight synbiotic and two oral FMT intervention trials. The trial durations varied from two weeks to six months, and the total participant sample size varied from 18 to 400 participants. Although 34 of the studies reported finding significant differences in depression symptom outcomes between the intervention and the control groups, the wide variety in interventions, populations, duration, measurements and outcomes preclude drawing a firm conclusion regarding the efficacy of microbiota for relieving depressive symptoms.
When comparing the studies that concluded significant reduction in depressive symptoms in the intervention group, no obvious differences were seen in demographic details, duration of intervention (except for the two studies of FMT) or total sample size.
However, the symptoms severity at baseline as measured by depressive symptoms scales varied among the included trials. For example, 29 studies reported “healthy” when describing baseline depression symptoms scores, (nine of which reported mixed baseline scores of healthy, mild, moderate and severe), 19 out of 29 of those studies reported no significant effects of the microbiota manipulation on depressive symptoms. Studies that included “mild” and “moderate” depressive symptoms at baseline (k = 14) reported no significant differences between intervention and control in 10 out of 14 studies. Many trials (k = 18) did not report the baseline depressive symptoms severity, 11 of which concluded a significant difference between intervention and control groups.
These findings highlight the need to assess whether initial depressive symptom severity impacts the effectiveness of microbiota manipulation interventions on depressive symptoms.
When looking at the trials’ reporting of significant findings by intervention type, we identified 29/54 (54%) of the probiotic intervention studies, 2/7 (29%) of the prebiotic intervention studies, 4/8 (50%) synbiotic intervention studies, and 1/2 (50%) of the oral FMT intervention studies concluded significant differences in depressive symptoms between the intervention and control groups. The two trials that investigated oral FMT,38,39 one trial reported a significant effect and the other did not. The main differences between these two studies are the duration of the intervention was longer in the positive trial (12 weeks compared to three weeks) and none were conducted in a primary population of MDD.
It is important to note the common limitations amongst the included trials. The most common limitation across the included studies was the lack of participants’ microbiome analysis before and after intervention. The inclusion of these data in microbiota manipulation trials has many benefits, including more accurate adherence, confirmation of intervention saturation, and the possibility for identification of the most beneficial bacterial strains in composite interventions. While most trials used probiotics, the significance of multi-strain probiotic interventions components remains largely unknown despite the availability of multiple approaches and best practice guidance to analyze the microbiome. 40
Although the ability of probiotics to modify the gut microbiota and relieve various disease symptoms has been reported, there is evidence of ideal probiotic intervention compositions for condition-specific symptom relief. 41 Therefore, isolated microorganism trials that investigate changes in depressive symptoms must identify specific strains for the effective treatment of depressive symptoms, as well as specific dosing standards. We should highlight that our review did not include RCTs that solely focused on depression patients, and while 47/66 studies reported analyzing the effects of the intervention on depression as a primary outcome, only five trials included MDD participants. This subgroup of trials (k = 5) had modest sample size (minimum n = 40 to maximum n = 79) used PHQ-9, HADS, BDI, MADRS, and HAM-D and two trials included comorbid IBS. Despite having participants with MDD, all used probiotic as the intervention for various durations, these studies provided inconsistent results precluding any recommendations to be made on the use of probiotics in MDD.
The use of prebiotic, synbiotic and oral FMT as microbiota manipulation interventions for depressive symptoms is less common than probiotic interventions. 24 while trials have been conducted, there is a need for standardization and more precise analyses of these types of interventions using best practice approach informed by multi-omics for example. 40 In this review, we identified inconsistency in the composition of the intervention, dosing and consumption schedules, making it difficult to compare the studies and identify whether microbiota manipulation has a promise as a potential intervention for depression symptoms.
The heterogeneity in study methods, including varied intervention composition and dosing, depression symptom measurement tools, and reporting methods, did not allow a quantitative synthesis or meta-analysis to be conducted. The variety in study design also indicates a need for standardized study protocols. Amongst the 66 trials we included in our review, we found the use of 34 different depression symptom rating scales and different methods used to report study findings. While significant differences in sensitivity or specificity have not been established between the various depression rating scales, 42 the inconsistency in assessment of outcomes and the reporting of the trials makes synthesis of results unfeasible. To allow for meaningful quantitative synthesis and meta-analysis, future studies should follow consensus guidelines when planning, conducting and reporting the results of studies, as well as standardizing intervention duration and increasing trial sample sizes.
The current literature shows a growing interest in testing microbiota manipulations in depression symptoms.26,43,44 However, like our findings, the published reviews commented on the heterogeneity of the findings and the inability to make conclusive recommendations on the effectiveness of the interventions, urging for the standardization of microbiota manipulation interventions specifically for depression and mood disorders. Our review differs from a recent review 24 in several aspects including the focus on depression and depressive symptoms as the outcome, while the previous review included all psychiatric disorders with a confirmed diagnosis. The included studies were not all RCTs, and the included depression studies also included co-interventions such as vitamin B7 and antidepressants, e.g., escitalopram and fluoxetine. Our review excluded co-interventions to distinguish the effects of the microbiota as a stand-alone intervention. While there have been reviews concluding significant effects of probiotic interventions on depressive symptoms,25,45 the published reviews exclusion criteria did not outline concurrent anti-depressant use, which may have impacted the study findings. Our exclusion of trials that allowed for concurrent antidepressant use and/or vitamin supplementation during the intervention timeline aimed to ensure the effects of microbiota manipulation are independently investigated.
The current study has limitations including potential known or unknown confounding factors such as receiving concurrent psychotherapy or other interventions that were not reported. The lack of trials’ data synthesis and meta-analysis precludes the current study from making firm conclusions on the use of microbiota manipulation in depression. Another limitation of the current review is the lack of pre-registration of the systematic review protocol.
Conclusion
While the topic of this review is accelerating in interest, it is evident that there is a need for further investigations on the effectiveness of oral microbiota manipulation as potential alternatives or additional options for depression symptom treatment. We identified 66 trials, 34 of which reported a significant effect of the intervention on depression symptoms. While these reports seem promising, there are many inconsistencies in studies’ methods, interventions used, reporting and analysis leading to reduced confidence in the overall effects of oral microbiota manipulation on depression symptoms. To enhance the knowledge generated by future studies, standardized protocols regarding depression symptom assessment and reporting, harmonization of criteria and guidelines within the field of oral microbiota manipulation interventions, investigating the individual probiotic strains to isolate the most effective intervention compositions that is disorder specific and identifying equivalent dosing of the interventions would be beneficial and necessary if these interventions translate into clinically impactful treatment options for patients with depression.
Supplemental Material
sj-docx-2-cpa-10.1177_07067437251394369 - Supplemental material for Oral Methods of Microbiota Manipulation for Depression Symptoms: A Systematic Review
Supplemental material, sj-docx-2-cpa-10.1177_07067437251394369 for Oral Methods of Microbiota Manipulation for Depression Symptoms: A Systematic Review by Anna Oprea, Joe Steinman, Grace Huang, Omolara Soyinka, Megan Brookbank, James Abesteh, Maya Hartman, Alannah McEvoy, Joanna C. Dionne, Roumen Milev and Zainab Samaan in The Canadian Journal of Psychiatry
Supplemental Material
sj-xlsx-3-cpa-10.1177_07067437251394369 - Supplemental material for Oral Methods of Microbiota Manipulation for Depression Symptoms: A Systematic Review
Supplemental material, sj-xlsx-3-cpa-10.1177_07067437251394369 for Oral Methods of Microbiota Manipulation for Depression Symptoms: A Systematic Review by Anna Oprea, Joe Steinman, Grace Huang, Omolara Soyinka, Megan Brookbank, James Abesteh, Maya Hartman, Alannah McEvoy, Joanna C. Dionne, Roumen Milev and Zainab Samaan in The Canadian Journal of Psychiatry
Footnotes
Registration and Protocol
The review was not registered and was part of a senior student (AO) final thesis. No protocol was published for this review.
Data Access
Supplementary data are provided in appendices, additional data available by reasonable request.
Author Contributions
All authors contributed to the study question, design, data collection, analysis and writing of the manuscript. AO led the search strategy, data collection, and writing first draft. AO, AM, JS, OS, GH, MB, JA, MH completed screening, data extraction and quality checks. AO, OS, GH, MB, JA completed risk of bias assessment. AM contributed to the study design, data collection form design, and analyses. JD provided expertise on microbiota and interpterion of trials methods and results. RM contributed to the study design, data interpretation and manuscript review. ZS led the study design, implementation, analyses, interpretation, manuscripts revisions and final draft.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Zainab Samaan holds SEAMO Chair in Research and Innovation from The Southeastern Ontario Academic Medical Organization (SEAMO) and Queen's University. The funder has no role in the study design, interpretation or reporting.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
