Abstract
Objective
In this study, we evaluated the concordance between urine drug screening (UDS) and self-reported use in a pragmatic randomized clinical trial.
Methods
Our data was drawn from OPTIMA, a 24-week pragmatic multicentric open-label randomized-controlled trial comparing flexible take-home dosing of buprenorphine/naloxone to the methadone standard model of care for treating prescription-type opioid use disorder. A total of 272 participants were randomized (1:1 ratio) to methadone or buprenorphine/naloxone. Following treatment initiation, participants were followed-up every 2 weeks for 24 weeks. During each visit, participants provided urine samples for UDS and self-reported their substance use over the past 2 weeks. Self-reported use was dichotomized to align with UDS detection windows. Tetrachoric correlations and 2 × 2 contingency tables were used to estimate the sensitivity, specificity, positive predictive value and negative predictive value of self-reported use. A generalized linear mixed model assessed how substance type, time in the study, treatment assignment, study site, unstable housing, and sex impacted self-report accuracy.
Results
Significant differences were found between substance types (p < 0.001) and study sites (p < 0.001). Fentanyl, cannabis, and amphetamines consistently showed the greatest concordance between measurement methods. Hydromorphone, oxycodone, heroin, and benzodiazepines had low sensitivity and low positive predictive value. Participants from Québec showed higher concordance between UDS and self-reported use compared to those from British Columbia, Alberta, and Ontario. There was no moderating effect of treatment assignment (p = 0.174), time in the study (p = 0.221), unstable housing (p = 0.733), or sex (p = 0.321) on the concordance between UDS and self-reported use.
Conclusions
Our results indicate that concordance between UDS and self-reported use is impacted by several factors. Combining UDS and self-reported use could help provide a more accurate assessment of substance use.
Clinical trial registration
This study was registered in ClinicalTrials.gov (NCT03033732).
Introduction
Thought to be more objective than self-report measures, urine drug screening (UDS) is sometimes referred to as the ‘gold standard’ for evaluating changes in substance use. This is partly due to decreased recall bias, where individuals are unable to properly recall their use, or social desirability bias, where individuals may underreport substances to conform to social expectations.1–3 UDS may also be advantageous when individuals are not aware of substance contamination – for instance, an individual might not know that the opioids they have purchased also contain benzodiazepines.
Despite the apparent objectivity of UDS, it also has some disadvantages. Practically speaking, UDS is more costly than self-report measures. 4 Secondly, UDS will test positive for various drugs within different time windows, potentially leading to an overestimation of use for substances with longer detection windows and an underestimation of use for substances with shorter ones. These detection windows can also be affected by individual variations in drug metabolism, amount of drug used, and route of administration. 5 Furthermore, UDS used in clinical research are often simple dichotomous measures (yes or no), which limits their ability to detect changes in substance use patterns when a person is not abstinent (i.e. change in daily frequency or numbers of days of use). 6 This is especially important given that reductions in use are a commonly desired and potentially achievable goal of treatment.7,8 False positives may occur when lipophilic substances (e.g. fentanyl, cannabis) are consumed chronically and remain detectable in urine for weeks after the last use. 9 False positives may also occur due to drug interactions, including with certain frequently prescribed drugs. 10
When UDS is only tangentially related to study goals, participants may be more likely to find the process demeaning. 4 Some may consider it invasive and find that it demonstrates a lack of trust in the individual to accurately self-report their use. 11 Some studies suggest that inaccurate reporting of drug use is more likely when access to treatment relies on a specific result, or when individuals are part of a stigmatized community.3,12 However, in clinical settings, individuals are most likely to accurately self-report use of the drug being targeted in treatment. 3
In non-abstinence-based clinical trials, self-reported use may better capture changes in substance use, including frequency of use and routes of administration, than UDS. One self-report tool, the Timeline Followback (TLFB), measures substance use for up to 30 days prior, which can be useful when participants miss some visits, potentially reducing missing data. Continuous self-report measures also allow for greater statistical flexibility, power and precision when evaluating the effectiveness of addiction interventions. 6 However, previous studies have shown that the accuracy of self-report may be influenced by many factors, including drug type, social and environmental contexts, and individual characteristics, including age, sex/gender, race, psychiatric comorbidities and housing instability.3,13–15
Assessing the concordance between self-reported use and UDS is particularly relevant in the context of opioid use disorder (OUD) treatment. First, opioids often have similar metabolites, which could reduce the specificity of UDS when testing is being done for a specific opioid. 11 In addition, UDS alone does not make it possible to differentiate between what people think they were using and what they were actually exposed to. This is especially relevant considering the increased adulteration of the illegal drug supply.16,17
Our study evaluated the concordance between self-reported drug use data (TLFB) and point-of-care UDS in a pragmatic clinical trial setting, where participants with OUD were randomized to either methadone or buprenorphine/naloxone models of care. We investigated the following substances: cannabis, cocaine, amphetamines, benzodiazepines, oxycodone, heroin, fentanyl, and hydromorphone – comparing groups over time and across study sites.
Methods
Overview of Study Design
Our study is a secondary analysis of OPTIMA,18,19 a 24-week pan-Canadian, two-arm, open-label, multicentric, pragmatic randomized-controlled trial designed to compare flexible take-home dosing of buprenorphine/naloxone to the methadone standard model of care in treating prescription-type OUD. Prescription-type opioids were defined as any legal or illegal opioids other than heroin, including fentanyl. The trial protocol was registered at ClinicalTrials.gov (NCT03033732). Main outcomes are described in the literature. 18 This study was conducted in accordance with the Good Clinical Practice guidelines from the International Conference on Harmonization (Declaration of Helsinki), Canadian (Tri-Council Policy Statement and Health Canada Division 5), provincial, and institutional ethical guidelines. The local institutional ethics review board of each study site approved the protocol. The statistical analysis plan was not pre-registered.
Participants
Individuals with OUD were recruited between October 2, 2017 and March 23, 2020 in one of seven hospital and community-based centres: The Rapid Access Addiction Clinic (British Columbia); the Portland Hotel Society Medical Clinic (British Columbia); the Opioid Dependency Program Clinic (Alberta); the Centre for Addictions and Mental Health (Ontario); the Ontario Addiction Treatment Centre (Ontario); the Centre hospitalier de l'Université de Montréal (Québec); and the Centre de recherche et d'aide pour narcomane (Québec). Inclusion criteria were being aged between 18 and 64 years and having a diagnosis of moderate to severe OUD according to DSM-5 criteria requiring an opioid agonist treatment. Exclusion criteria were having an unstable psychiatric or medical condition precluding safe participation, having a chronic pain condition requiring opioids, using heroin as the most frequent opioid in the past 30 days, taking medications that could interact with study treatments, a history of an adverse reaction to either study medications, having pending legal action preventing study completion, and being pregnant, breastfeeding, or of childbearing potential and planning to conceive.
Study Procedures
A total of 272 eligible participants were randomized at a 1:1 ratio to either the standard methadone model of care (n = 134) or buprenorphine/naloxone flexible take-home dosing (n = 138). Participants having at least one observation for self-reported use (TLFB) coinciding with a UDS at any timepoint (n = 271) were included. Clinicians initiated treatment following Canadian clinical guidelines for the management of OUD.20–26 However, clinicians were allowed to adapt treatment initiation based on their clinical judgment. Participants had in-person follow-up visits every 2 weeks for 24 weeks, during which questionnaires were administered, and urine samples were collected.
Measure of Interest
Urine samples were collected at each study visit (every 2 weeks for all participants) by the study nurse. Heath Canada-approved Rapid Response™ Multi-Drug one-step Test Panel temperature-controlled urine drug test cups were used to analyse urine specimens through immunoassay. 27 As it is often the case in clinical care, immunoassay UDS results were not confirmed with gas chromatography or mass spectrometry. Urine samples were screened for morphine, oxycodone, fentanyl, methadone and its metabolite (EDDP), tramadol, buprenorphine, as well as for other commonly used drugs i.e. benzodiazepines, amphetamines and methamphetamine, delta-9-tetrahydrocannabinol, and cocaine. Hydromorphone and 6-monoacetylmorphine (heroin metabolite) were assessed using Heath Canada-approved Rapid Response™ one-step single test strips. 28 Urine samples collected as part of regular clinical care were not available for research purposes.
Past 2-week substance use was self-reported with the TLFB at each study visit. The TLFB is a validated, commonly used self-report instrument assessing quantity and frequency of substance use in the past 30 days, as opposed to a dichotomous yes/no measure of substance use. 29 The TLFB was administered by the clinical research staff. Participants were asked to retrospectively self-report their substance use since their last visit (up to a maximum of 30 days if a previous visit was missed). The following substances were systematically investigated: cannabis, cocaine, amphetamines, oxycodone, heroin, hallucinogens, sedatives, benzodiazepines, inhalants, methadone, buprenorphine, fentanyl, hydromorphone and nicotine. Participants were allowed to report use of other substances.
Since the substances measured by TLFB and UDS differed, we selected the following substances to investigate in our study: cannabis, cocaine, amphetamines, benzodiazepines, heroin, oxycodone, hydromorphone, and fentanyl.
Statistical Analyses
All available TLFB and UDS data were included in our analyses. No imputation method was used to reflect the actual accuracy of these measurements in the context of a pragmatic clinical trial. Self-reported measures were matched to the UDS detection windows for each substance. For example, cocaine is detectable in urine for up to 4 days. Therefore, self-reported cocaine use was dichotomized based on whether a participant reported using cocaine in the past 4 days. Detection windows for all substances were determined based on previous reports.5,11,30 We used the average detection windows reported in previous reports, although it should be noted that these detection windows can vary significantly due to individuals’ metabolism and drug–drug interactions. Calibrator analytes and detection windows are presented in Table 1. Detailed analytes and cut-offs for each substance are available in Supplemental Table S1.
Analytes and Detection Window Used for Each Substance.
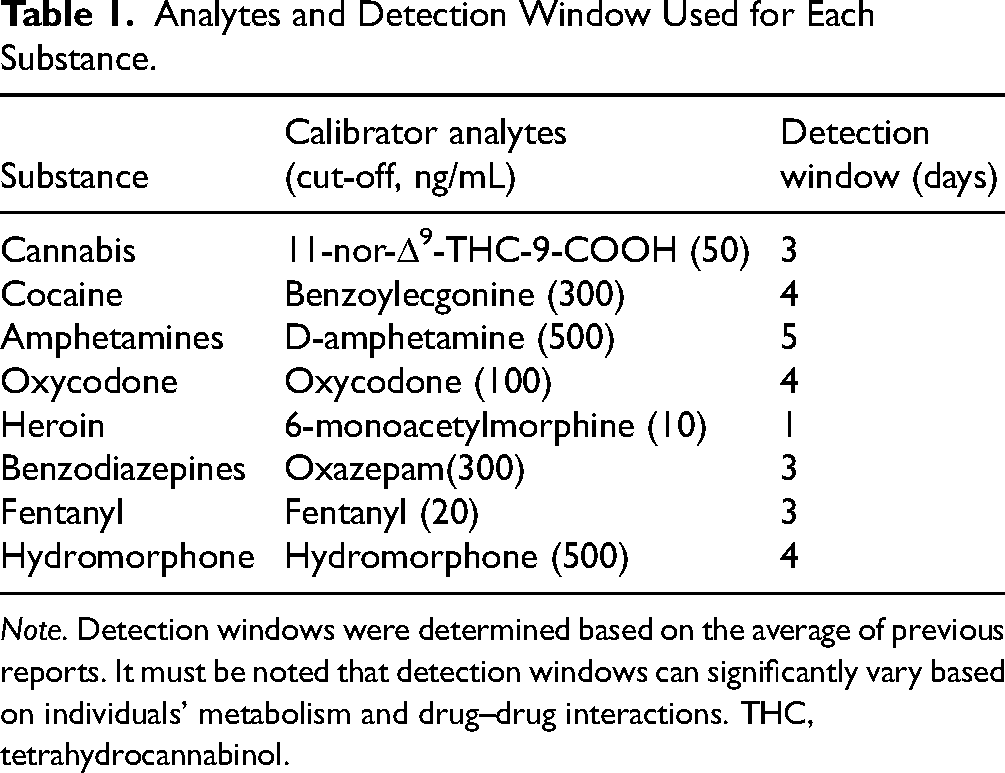
Note. Detection windows were determined based on the average of previous reports. It must be noted that detection windows can significantly vary based on individuals’ metabolism and drug–drug interactions. THC, tetrahydrocannabinol.
To measure the concordance between self-reported use and UDS, we conducted tetrachoric correlations and estimated the sensitivity, specificity, positive predictive value, and negative predictive value of each substance using 2 × 2 contingency tables. For all analyses, UDS was used as the reference (‘true’) value.
We then estimated a generalized linear mixed model (GLMM) with UDS as a predictor of self-reported use to assess the likelihood of self-reporting substance use based on whether the urine sample was positive/negative for a specific substance. We additionally included moderating effects for substance type, time in the study, assigned treatment, study sites (combined by province), unstable housing and sex. Post-hoc pairwise comparisons were conducted to assess statistical significance. All analyses were conducted using RStudio (v2024.04.1 + 748, Comprehensive R Archive Network) assuming a two-tailed 5% significance level. Tetrachoric correlations and 2 × 2 contingency tables were done using the caret package. 31 GLMM and pairwise comparisons were estimated using the lme4 and emmeans packages, respectively.32,33
As a sensitivity analysis, we conducted tetrachoric correlations, 2 × 2 contingency tables, and a GLMM evaluating the concordance between self-reported use and UDS for any hydrophilic opioids (i.e. hydromorphone, heroin and oxycodone, excluding fentanyl) rather than looking at specific opioids.
Results
Participant Characteristics
A participant flow chart, sociodemographic, and baseline characteristics have already been published elsewhere. 18 In brief, most participants were middle-aged (mean ± standard deviation [SD] = 38.9 ± 10.5 years), white (n = 183, 67.3%), and male (n = 179, 65.8%). About a third of the sample self-reported recent very unstable housing (n = 85, 31.3%). Most participants had completed high school (n = 222, 81.6%). Of the 3523 total possible observations, both UDS and self-reported data were missing for 1377 observations (39.1%); UDS data only was missing for 119 observations (3.4%) and self-reported data only was missing for two observations (0.1%).
Sensitivity, Specificity, Positive Predictive Value, and Negative Predictive Value
Concordance measures by substance type for the overall study period are reported in Figure 1. As a reminder, UDS results were used as the reference (‘true’) value. Sensitivity ranged from 0.185 for hydromorphone to 0.786 for fentanyl. Specificity was high for all substances, ranging from 0.925 for cannabis to 0.987 for fentanyl. Positive predictive value was the highest for fentanyl (0.975), followed by cannabis (0.898), amphetamines (0.895), cocaine (0.880), hydromorphone (0.798), oxycodone (0.618), benzodiazepines (0.561), and heroin (0.505). Negative predictive value ranged from 0.587 (hydromorphone) to 0.971 (oxycodone).
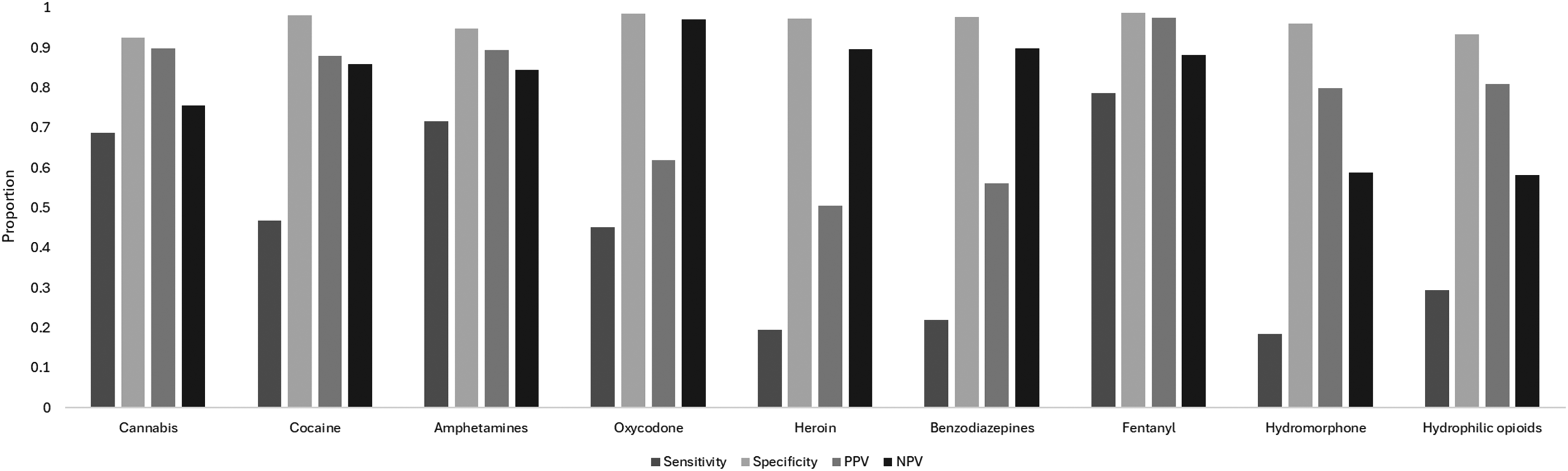
Sensitivity, specificity, positive predictive value, and negative predictive value of self-reported use by substance type. Sensitivity was the highest for fentanyl, followed by amphetamines, cannabis, cocaine, oxycodone, benzodiazepines, heroin, and hydromorphone. Specificity was high for all substances. Positive predictive value (PPV) was the highest for fentanyl, followed by cannabis, amphetamines, cocaine, hydromorphone, oxycodone, benzodiazepines, and heroin. Negative predictive value (NPV) was the highest for oxycodone, followed by benzodiazepines, heroin, fentanyl, cocaine, amphetamines, cannabis, and hydromorphone. When considering hydrophilic opioids (excluding fentanyl), sensitivity was low, specificity was high, PPV was moderate and NPV was moderate. Note. PPV, positive predictive value; NPV, negative predictive value.
Concordance Between Urine Drug Screening and Self-Reported use by Substance Type, Visit Number, Treatment Assignment, and Study Site
Results from the GLMM looking at the moderating effects of substance type, time in the study, assigned treatment, and study site can be found in Table 2. There was a significant moderating effect of substance type on the likelihood of self-reporting substance use (p < 0.001). When a UDS was positive, the probability that a participant self-reported their substance use was the highest for fentanyl, followed by amphetamines, cannabis, oxycodone, cocaine, heroin, benzodiazepines, and hydromorphone (see Figure 2 and Supplemental Table S2 for pairwise comparisons between substances). All differences between substance types were significant (all p < 0.050), except for between benzodiazepines and heroin (p = 0.998), benzodiazepines and hydromorphone (p = 1.000), cocaine and oxycodone (p = 1.000), heroin and hydromorphone (p = 0.610), and heroin and oxycodone (p = 0.058). There was no significant moderating effect of treatment assignment (p = 0.174), visit number (p = 0.221), unstable housing (p = 0.733), or sex (p = 0.321). There was a significant moderating effect of study site (p < 0.001). When a UDS was positive, participants from British Columbia, Alberta, and Ontario had a significantly lower likelihood of self-reporting substance use compared to individuals from Québec (p < 0.001; see Figure 3 and Supplemental Table S3 for comparisons between provinces).
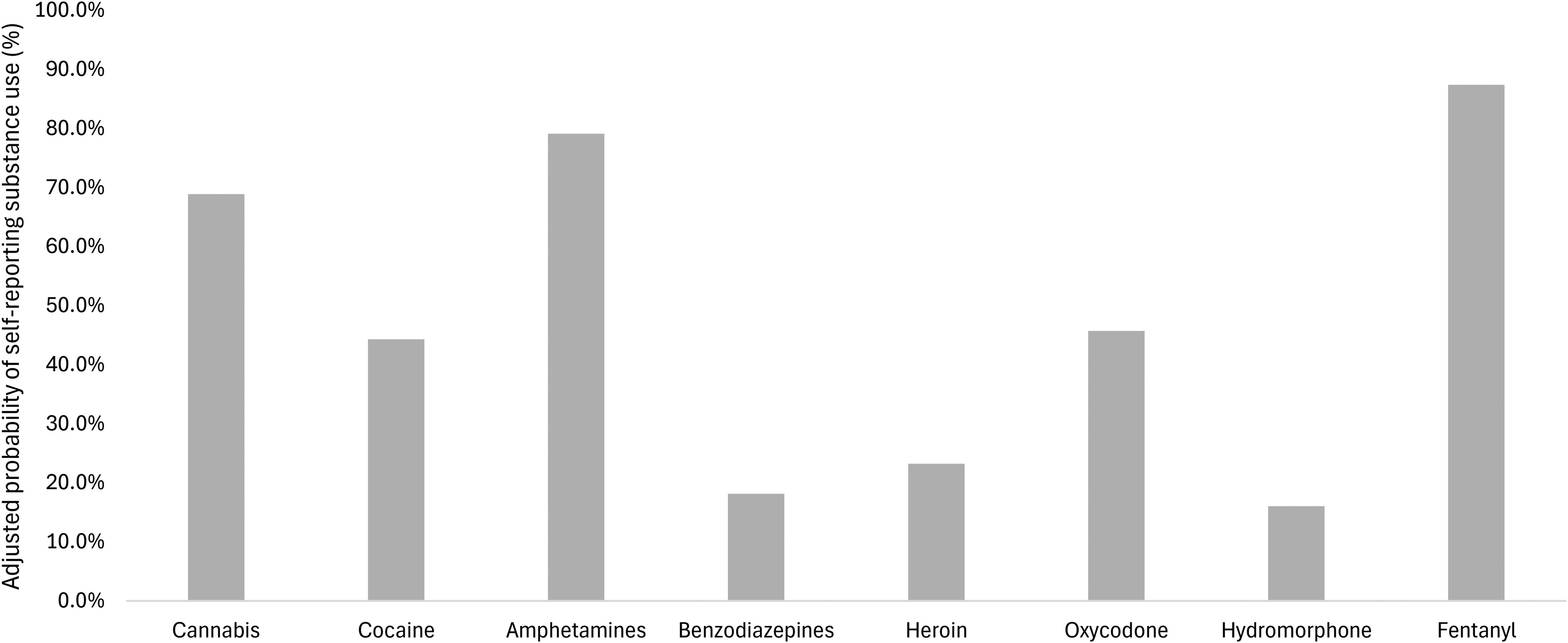
Adjusted probabilities of self-reporting substance use by substance type. When a urine drug screening was positive, the probability of self-reporting use of that substance was the highest for fentanyl, followed by amphetamines, cannabis, oxycodone, cocaine, heroin, benzodiazepines, and hydromorphone. Differences between substance types were all highly significant (all p < 0.050), except for between benzodiazepines and heroin (p = 0.998), benzodiazepines and hydromorphone (p = 1.000), cocaine and oxycodone (p = 1.000), heroin and hydromorphone (p = 0.610), and heroin and oxycodone (p = 0.058). Probabilities were adjusted for time in the study, treatment assignment, study site, unstable housing, sex, and participant identification number.
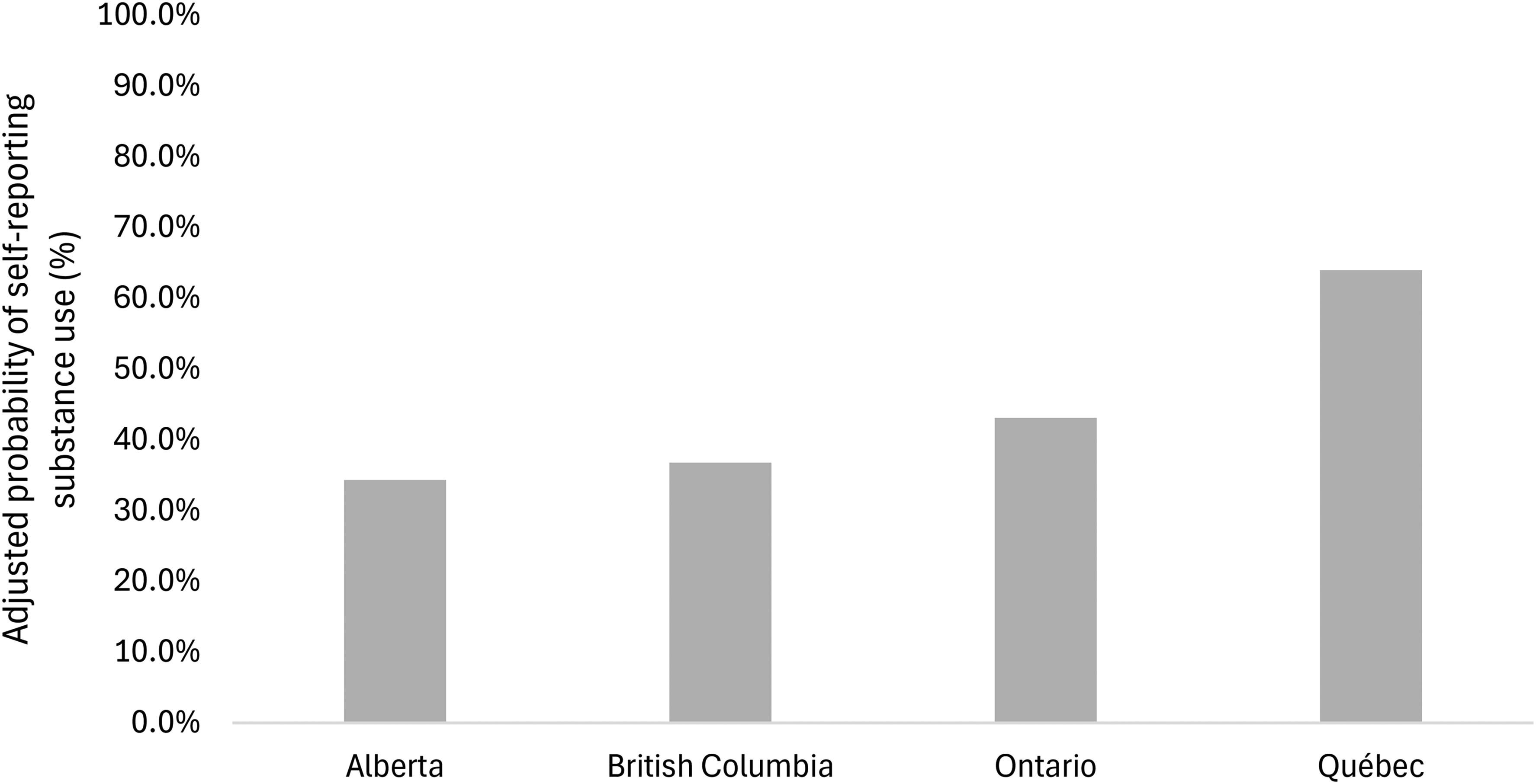
Adjusted probabilities of self-reporting substance use by study site. When a urine drug screening was positive, the probability of self-reporting use of that substance was significantly greater for Québec compared to Alberta (p < 0.001), British Columbia (p < 0.001), and Ontario (p < 0.001). There were no other significant differences between study sites. Probabilities were adjusted for substance type, time in the study, treatment assignment, unstable housing, sex, and participant identification number.
Results from the Generalized Linear Mixed Model for the Effects of Substance Type, Time in the Study, Treatment Assignment, and Study Site (n = 271).
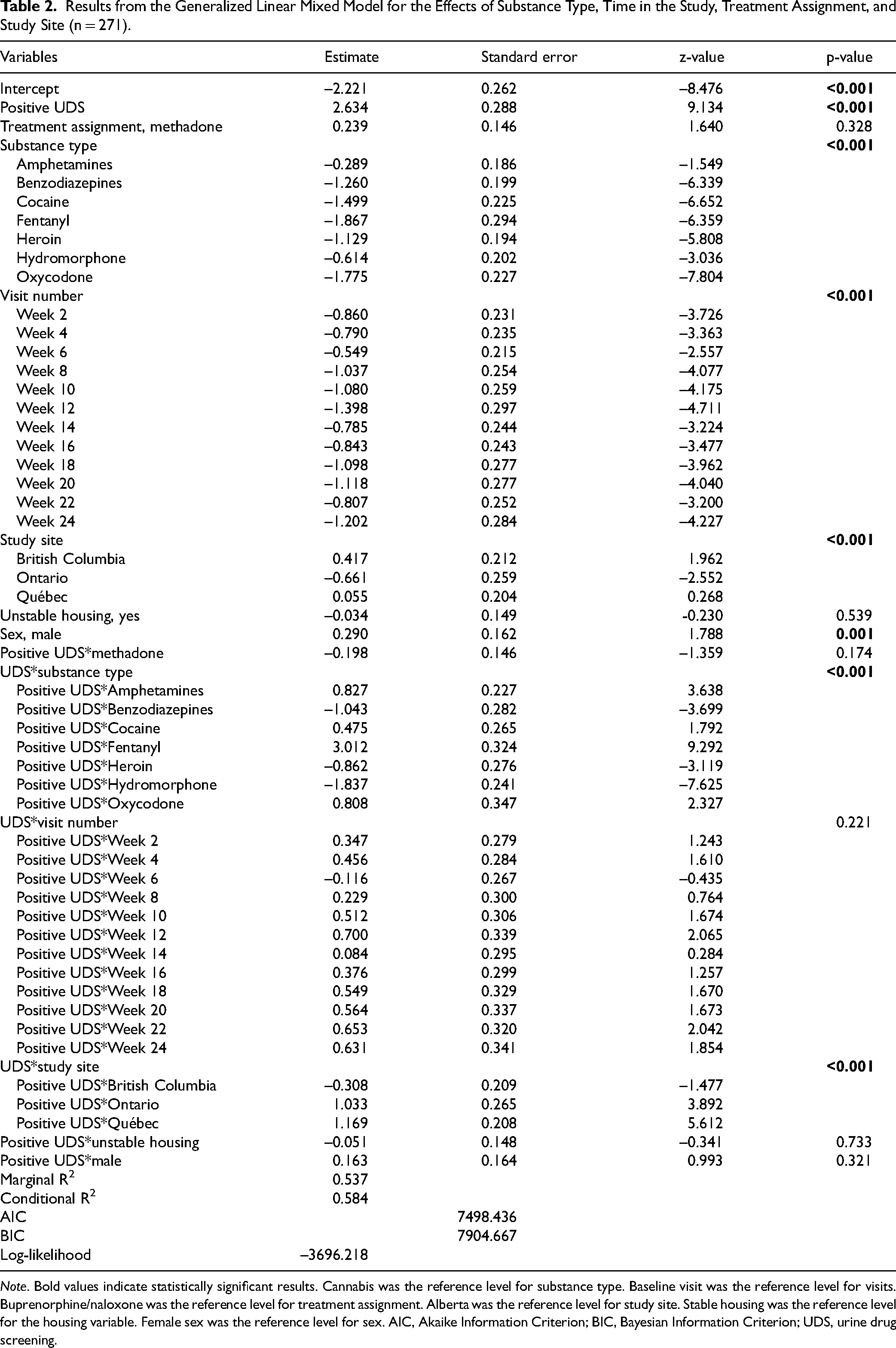
Note. Bold values indicate statistically significant results. Cannabis was the reference level for substance type. Baseline visit was the reference level for visits. Buprenorphine/naloxone was the reference level for treatment assignment. Alberta was the reference level for study site. Stable housing was the reference level for the housing variable. Female sex was the reference level for sex. AIC, Akaike Information Criterion; BIC, Bayesian Information Criterion; UDS, urine drug screening.
Sensitivity Analysis
Results from the GLMM that considered hydrophilic opioids together (i.e. excluding fentanyl) are presented in Supplemental Table S4. The results of the sensitivity analysis were the same as the main analysis. Although, when UDS was positive, the probability of self-reporting use of that substance was the highest for fentanyl, followed by amphetamines, cannabis, cocaine, hydrophilic opioids, and benzodiazepines (all p < 0.050; see Supplemental Figure S1 and Supplemental Table S5 for pairwise comparisons).
Discussion
In our study, we evaluated the concordance between UDS and self-reported substance use using data from a pragmatic randomized-controlled trial evaluating the non-inferiority of flexible take-home dosing of buprenorphine/naloxone compared to the methadone standard model of care for the treatment of prescription-type OUD. Fentanyl, amphetamines, and cannabis consistently showed the highest concordance between UDS and self-reported use. Use of cocaine was moderately self-reported in people with positive UDS results. Overall, use of hydrophilic opioids i.e. heroin, hydromorphone and oxycodone, was poorly reported; however, considering these substances as a group increased the concordance between UDS and self-reported use, especially the positive predictive value. There was no moderating effect of treatment assignment, study visit, unstable housing, and sex. We also found that participants from Alberta, British Columbia and Ontario had significantly lower concordance between UDS and self-reported use compared to those from Québec.
Interpretation
Many factors might explain the variability in concordance between UDS and self-reported use between substances and study sites. First, there is evidence that individuals might not be aware of what they are, in fact, using. Among people self-reporting use of a substance, between 2.5% and 49.5% of participants had a negative UDS to that substance, with oxycodone, heroin, and benzodiazepines showing the lowest positive predictive values. In contrast, between 21.4% and 81.5% of individuals with a positive UDS did not disclose using that substance, mostly hydromorphone, heroin, oxycodone, and benzodiazepines. These results are in line with drug checking reports showing that, between April 2018 and August 2019, about 13% of opioids samples, 11% of methamphetamine samples, 5% of cocaine samples, and 36% of fentanyl samples contained at least one other unexpected psychoactive substance. 34 A 2017 report from the U.S.-China Economic and Security Review Commission showed that fentanyl was often mixed with heroin. 35 Another more recent report showed that between 20% and 50% of opioid samples from British Columbia were contaminated with benzodiazepines. 36 Therefore, data from drug checking services indicate that individuals who use illicit substances may be more likely to be unknowingly exposed to opioids and benzodiazepines, in line with our results showing lower accuracy of self-reported use for these substances. This hypothesis is further supported by our results showing significant regional differences – participants from British Columbia, Alberta, and Ontario were more prone to inaccurately self-report their substance use compared to participants from Québec. For instance, between 2018 and 2019, 15% of opioids samples from British Columbia contained at least one other non-opioid psychoactive substance. 34 In contrast, in Québec, that percentage was only 7%. 34 This report also showed that 91% of opioids from British Columbia contained fentanyl, compared to only 14% in Québec. 34 This volatility of the unregulated drug market probably had a substantial impact on the accuracy of self-reporting.
Second, the discrepancy between UDS and self-reported use may be explained by some limitations of the immunoassay technique used to analyse urine samples. Cross-reactivity of immunoassay UDS may arise when screened substances have similar metabolites. For example, opioids from the epoxymorphinans family (i.e. oxycodone, heroin and hydromorphone) are mostly metabolized into morphine and hydromorphone, which is usually what UDS are detecting in urine samples. 5 Therefore, UDS might have a very low ability to differentiate specific opioids from this family. 5 This hypothesis is supported by our findings showing an increased concordance between UDS and TLFB when considering hydrophilic opioids together. Drug–drug interactions might also impact UDS validity. Both methadone and buprenorphine are metabolized by cytochromes P450 3A4 and 2D6.37–40 These enzymes are also involved in the metabolism and excretion of oxycodone, hydromorphone, heroin, and fentanyl, as well as commonly prescribed medications such as antipsychotics, antidepressants, and antiretroviral medications.37,38,41 Therefore, using opioids while being on opioid agonist treatment or other medications can lead to a prolongation of the effects of opioids and an increase in opioid blood concentrations.38,42,43 These drug–drug interactions, combined with interindividual variability in drug metabolism, likely impacted the duration during which opioids were detected in the urine, potentially leading to a mismatch between self-reported use and UDS. These limitations can significantly impact the quality of the treatments provided, as false positives can lead to an inappropriate adjustment of opioid agonist treatment doses, contribute to an untrusting relationship between patients and clinicians, and impact treatment retention by limiting access to take-home doses.2,3,44 However, opioids false negatives could also greatly impact safety by increasing the risk of overdose, especially for individuals prescribed methadone. 11
Several factors could also have impacted the reliability of self-reported use. First, since the TLFB retrospectively measures substance use, it is susceptible to recall bias. 45 This might be especially relevant for people with OUD, who may have cognitive impairments in executive functioning and memory processing.46,47 Individuals with OUD often present with comorbid psychiatric disorders, 48 which are also likely to impair cognitive functioning.49,50 Participants presenting memory deficits could inaccurately self-report their substance use, with no deceptive intent. Social desirability can also impact the accuracy of self-reported measures, especially considering that people who use drugs are often stigmatized. 3 Some individuals might be inclined to underreport their substance use for fear of being denied treatment, loosing take-home doses of opioid agonist treatment or being judged or mistreated by the study and clinical staff.2,3,44 To improve the accuracy of self-reported measurements, research staff and clinicians should address these fears prior to data collection, in order to reassure participants that self-reporting their substance use will not impact their access to quality health services.
In summary, oxycodone, heroin, hydromorphone, and benzodiazepines showed lower concordance between self-reported use and UDS results, likely due to the limitations of the immunoassay technique and the adulteration of the unregulated drug market. Individuals using cannabis, amphetamines, and fentanyl on a voluntary basis accurately self-reported their substance use. For these substances, self-report may be sufficient to measure drug use exposure in clinical care and research. However, an ‘objective’ measure might be necessary to measure the exposure to other opioids and benzodiazepines to properly adapt opioid agonist treatment modalities such as doses to limit the risk of overdose. The use of mass spectrometry or gas chromatography is highly encouraged to avoid some of the limitations of immunoassay UDS. To ensure acceptability of such strategies, reasons for conducting UDS and consequences of a positive result should be properly explained to patients to build a trusting relationship. Combining both UDS and self-reported use can help to differentiate between undesired drug exposure and substance use patterns. From a research perspective, the use of one method over the other should be driven by the outcome of interest. When researchers are interested in measuring changes in drug-seeking behaviours, self-report of substance use might be sufficient. However, laboratory-confirmed UDS may be better at measuring actual drug use exposure.
Limitations
Some limitations must be noted. First, data were missing for 42.6% of the 3523 total observations. This might undermine the reliability and validity of the findings. Also, individual variations in drug metabolism might have led to an overestimation of substance use in some, as it can significantly impact UDS detection windows. Lipophilic substances, like cannabis and fentanyl, can be detected in urine for up to 30 days when used chronically; however, these subgroup analyses were beyond the scope of our article. Considering chronic users in our analysis may have led to a higher sensitivity and negative predictive value for cannabis and fentanyl, which already had the highest values. Finally, UDS results were not laboratory-confirmed for logistical and financial reasons, limiting our ability to confirm the presence of specific opioids in the urine. This might lead to false positives due to cross-reactivity of the immunoassay technique. However, laboratory methods such as mass spectrometry or gas chromatography are rarely used in clinical settings – our results reflect the reality of clinical care in this population.
Conclusion
In this study, we evaluated the concordance between UDS and self-reported use in a pragmatic randomized-controlled trial treating individuals with OUD with flexible take-home dosing of buprenorphine or with methadone standard model of care. The concordance between these two measurements varied by substance type and location. Drug screening tools, such as UDS, can provide valuable insight into what people who use drugs are exposed to, especially for highly adulterated substances such as opioids and benzodiazepines. However, self-report measures offer more precise information, like frequency of use, modes of administration and behavioural changes related to drug use. Self-reported use might be sufficient to measure use of cannabis, amphetamines and fentanyl. Combining UDS with self-reported use may significantly improve treatment outcomes by allowing clinicians to identify targeted strategies for treatment-seeking individuals who are unwittingly exposed to psychoactive substances.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251367180 - Supplemental material for Concordance Between Urine Drug Screening and Self-Reported Use in the Context of a Pragmatic Randomized-Controlled Trial in People with Prescription-Type Opioid Use Disorder: Concordance entre le dépistage de drogues dans l’urine et l’usage autodéclaré dans le contexte d’un essai pragmatique contrôlé à répartition aléatoire chez des personnes présentant un trouble lié à l'usage d’opioïdes vendus sur ordonnance
Supplemental material, sj-docx-1-cpa-10.1177_07067437251367180 for Concordance Between Urine Drug Screening and Self-Reported Use in the Context of a Pragmatic Randomized-Controlled Trial in People with Prescription-Type Opioid Use Disorder: Concordance entre le dépistage de drogues dans l’urine et l’usage autodéclaré dans le contexte d’un essai pragmatique contrôlé à répartition aléatoire chez des personnes présentant un trouble lié à l'usage d’opioïdes vendus sur ordonnance by Gabriel Bastien, Anita Abboud, Christina McAnulty, Amani Mahroug, Bernard Le Foll, M. Eugenia Socias, Louis-Christophe Juteau, Simon Dubreucq and Didier Jutras-Aswad in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
The authors would like to acknowledge the work of Denise Adams, Oluwadamilola Akinyemi, Benoit Masse, Jill Fikowski, Aïssata Sako, Katrina Blommaert, Emma Garrod, José Trigo, Amel Zertal, Nirupa Goel, Farihah Ali, Wendy Mauro-Allard, Kristen Morin, Benita Okacha, Eve Poirier, Geneviève St-Onge, Angela Wallace and the OPTIMA Research Group for assisting in the conduct and administration of the trial. Our team expresses gratitude to Kim Brochu for her clinical input. Finally, our team would like to thank all participants and clinical staff who took part in this research.
CRediT Authorship Contribution Statement
Conceptualization: GB, AA, CM, AM, DJA; Methodology: GB, AA, CM, AM, DJA; Data curation: GB, AA, CM; Formal analysis: GB, AA; Validation: GB, AA; Visualization: GB, AA; Investigation: BLF, MES, LCJ, SB, DJA; Writing – Original draft: GB, AA, CM, AM; Writing – Review & editing: GB, AA, CM, AM, BLF, MES, LCJ, SB, DJA; Supervision: DJA; Project administration GB, BLF, MES, DJA; Funding acquisition: BLF, MES, DJA.
Data Availability Statement
Declaration of Competing Interest
GB, AA, CM, AM, LCJ, and SD report no conflict of interests. BLF has obtained funding from Pfizer Inc. (GRAND Awards, including salary support) for investigator-initiated projects. BLF has obtained funding from Indivior for a clinical trial sponsored by Indivior. BLF has in-kind donations of cannabis products from Aurora Cannabis Enterprises Inc., placebo edibles from Indiva and study medication donations from Pfizer Inc. (varenicline for smoking cessation) and Bioprojet Pharma. He was also provided a coil for a transcranial magnetic stimulation study from Brainsway. BLF has obtained industry funding from Canopy Growth Corporation (through research grants handled by the Centre for Addiction and Mental Health and the University of Toronto), Bioprojet Pharma, Alcohol Countermeasure Systems (ACS), Alkermes and Universal Ibogaine. BLF has received in kind donations of nabiximols from GW Pharmaceuticals for past studies funded by CIHR and NIH. He has participated in a session of a National Advisory Board Meeting (Emerging Trends BUP-XR) for Indivior Canada and is part of Steering Board for a clinical trial for Indivior. He has been consultant for Shinogi, ThirdBridge, and Changemark. He is part of a scientific advisory board for NFL Biosciences. He got travel support to attend an event by Bioprojet. He is supported by CAMH, Waypoint Centre for Mental Health Care, a clinician-scientist award from the department of Family and Community Medicine of the University of Toronto and a Chair in Addiction Psychiatry from the department of Psychiatry of University of Toronto. M.E.S. has received partial support from Indivior's investigator-initiated study program for work outside this study. DJA received investigational products from Cardiol Therapeutics for a clinical trial funded by the Québec Ministry of Health and Social Services (2022–2023).
Funding
This work was supported by funds received from Health Canada and the Canadian Institutes of Health Research (CIHR) through the Canadian Research Initiative in Substance Misuse (CRISM; grant numbers CIS-144301, CIS-144302, CIS-144303, CIS-144304). The four nodes of CRISM received independent funding through a CIHR priority-driven initiative (grant numbers SMN-139148, SMN-139149, SMN-139150, SMN-139151).
GB holds a doctoral training scholarship from the Fonds de recherche du Québec – Santé (https://doi.org/10.69777/330941). BLF is supported by CAMH, Waypoint Centre for Mental Health Care, a clinician-scientist award from the department of Family and Community Medicine of the University of Toronto, and a Chair in Addiction Psychiatry from the department of Psychiatry of University of Toronto. DJA is supported by a Fonds de recherche Québec – Santé clinical scientist career award (![]() ). The funding bodies had no role in the study design, collection, analysis and interpretation of data, writing of the report, and decision to submit the article for publication.
). The funding bodies had no role in the study design, collection, analysis and interpretation of data, writing of the report, and decision to submit the article for publication.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
