Abstract
Objectives:
The impact of comorbid posttraumatic stress disorder (PTSD) symptoms on the anti-depressive outcomes of repetitive transcranial magnetic stimulation (rTMS) for civilians with major depressive disorder (MDD) is poorly studied. We aimed to understand whether proximal PTSD symptoms would interact with distal traumas in impacting depression outcomes from rTMS treatment.
Methods:
A retrospective analysis was performed on 133 patients with MDD receiving open-label high-frequency rTMS to the left dorsolateral prefrontal cortex for 4 weeks. Probable PTSD was defined as scoring ≥ 4 on the Primary Care PTSD Screen for DSM-5. Distal traumas were quantified using the Adverse Childhood Experiences (ACE-10) questionnaire. Primary outcomes were improvement in Hamilton Rating Scale for Depression 17 item scale (HAMD-17) scores from baseline to 4 weeks as well as remission (HAMD-17 ≤ 7) and response (greater than 50% improvement from baseline).
Results:
29/133 had probable PTSD. Patients with probable PTSD had more ACEs, as well as higher depression, anxiety and medical comorbidity scores. Neither probable PTSD status nor its interaction with ACEs significantly impacted depression outcomes. However, having more ACEs was associated with greater odds of remission and response.
Conclusions:
Our findings suggest neither co-morbid PTSD symptoms nor distal childhood adversities should preclude patients with MDD from receiving rTMS for depression.
Plain Language Summary Title
Repetitive transcranial magnetic stimulation for civilian patients with depression and posttraumatic stress symptoms
Plain Language Summary
To date, most research studies investigating whether posttraumatic stress-related symptoms can impact the effectiveness of repetitive transcranial magnetic stimulation (rTMS) have been conducted on military veterans. Here, we studied depressed civilians with no military experience that received transcranial magnetic stimulation for depression. Over 1 in 5 screened positive for probable post-traumatic stress disorder (PTSD). Probable PTSD was associated with higher symptoms of depression and anxiety prior to the start of treatment. However, probable PTSD did not lead to poorer depression outcomes from rTMS treatment. Probable PTSD and childhood adversity also did not interact to lead to poorer depression outcomes from rTMS treatment. These results suggest depressed adults with current PTSD-related symptoms and/or a history of childhood adversity can benefit from receiving rTMS for depression.
Introduction
Major depressive disorder (MDD) is associated with morbidity and mortality worldwide.1–3 Poor prognostic indicators for MDD include comorbid posttraumatic stress disorder (PTSD) 4 and a history of childhood adversity.5,6 Comorbid PTSD is associated with decreased chances of remission with antidepressant treatment. 7 Childhood adversity is associated with poorer responses to pharmacotherapy, psychotherapy, or their combination. 5 Repetitive transcranial magnetic stimulation (rTMS) is increasingly used to treat MDD and considered a first-line treatment for MDD that has not responded to at least one antidepressant. 8
The impact of comorbid MDD and PTSD on rTMS outcomes has been studied in military veterans with mixed results. A double-blind, randomized trial comparing active and sham rTMS for treatment-resistant MDD in 164 veterans showed higher rates of remission of depressive symptoms among those with co-morbid PTSD compared to those without PTSD. 9 In contrast, a retrospective analysis of 118 veterans treated with rTMS for MDD showed similar response and remission rates regardless of comorbid PTSD status. 10 A chart review of a separate 57 patients with MDD in a Veterans Affairs clinic similarly showed that PTSD diagnosis did not significantly impact improvement in MDD symptoms. 11 However, PTSD diagnosis predicted lower odds of response and remission of depressive symptoms. Another retrospective chart review of 77 patients from a military facility showed reduction in both depression and PTSD symptoms irrespective of PTSD comorbidity. 12
Whether comorbid PTSD negatively impacts rTMS treatment outcomes for MDD among civilians with non-military backgrounds has not been explored. Moreover, the majority of participants in previous studies involving military veterans were men, whereas PTSD is more prevalent in woman in the general population, limiting generalizability.
rTMS has been used to treat PTSD symptoms with mixed results. One network meta-analysis showed significant reductions in PTSD symptoms with high-frequency or intermittent theta burst rTMS with standardized mean differences of −0.97 and −0.93, respectively. 13 Another network meta-analysis concluded that high-frequency rTMS targeting both dorsolateral prefrontal cortices or only the right side showed the greatest reductions in PTSD symptoms. 14 In contrast, a Cochrane review concluded active rTMS does not significantly impact PTSD severity with a certainty of evidence graded as moderate. 15 There is heterogeneity in protocols used 15 and the optimal neuroanatomical target for PTSD symptoms continues to be explored. 16
While not well studied, the impact of distal trauma in the form of childhood adversity on rTMS outcomes for MDD is mixed. One open-label multi-site trial of rTMS in 62 patients showed higher adverse childhood experiences (ACEs) predicted larger decreases in depression scores. 17 A retrospective analysis from our group on 99 patients showed high levels of ACEs was not associated with once-daily rTMS treatment outcomes. 18 By contrast, another group found in a sample of 51 patients that after twice daily rTMS targeting the left dorsolateral prefrontal cortex (DLPFC) for 5 days, greater childhood trauma correlated with less reduction in depression scores in self-reported but not clinician-administered scales in women. 19 In investigating the impact of PTSD on MDD outcomes, it is important to also consider the impact of ACEs as their impact may be pronounced due to occurring at more sensitive periods of development.
The current study had three aims: (1) assess the prevalence of comorbid PTSD symptoms among patients receiving rTMS for MDD; (2) assess whether comorbid PTSD symptoms impact antidepressant outcomes of rTMS, and (3) determine whether a history of ACEs would interact with current PTSD symptoms to impact MDD outcomes. Though the literature reviewed is mixed, we hypothesized that PTSD symptoms and its interaction with more ACEs would be associated with poorer depressive outcomes with rTMS due to known associations between comorbid PTSD and ACEs with poorer outcomes in MDD with other treatment modalities.
Patients & Methods
A retrospective analysis of outcomes was performed on patients who received an acute course of rTMS for MDD at the Harquail Centre for Neuromodulation at Sunnybrook Health Sciences Centre from March 2019 to October 2022. The study was approved by the Sunnybrook Research Ethics Board and conformed to the Declaration of Helsinki. Patients provided informed consent. Physical, administrative, and technical safeguards were followed to maintain confidentiality.
Participants
Inclusion and exclusion criteria were previously reported on.18,20 Inclusion criteria included: 1) patients ≥16 years of age; 2) patients who are able and willing to give consent and adhere to treatment schedule; 3) DSM-5 diagnosis of MDD confirmed in clinical assessment; 4) current major depressive episode at time of inclusion based on DSM-5 criteria, of moderate or greater severity, as determined by a Hamilton Depression Rating Scale (HAMD-17) of ≥16; 5) on stable psychotropic medication doses in the 4 weeks prior to screening; 6) pass safety screening. Exclusion criteria included: 1) active substance abuse in the last 3 months, except nicotine or caffeine; 2) history of or current psychosis; 3) diagnosis of obsessive-compulsive disorder or personality disorder, considered primary and causing greater impairment than MDD; 4) non-response to ECT; 5) if participating in psychotherapy, must have been in stable treatment for at least 3 months, with no anticipation of a change over the duration of the study; 6) clinically significant laboratory abnormality; 7) unstable medical illness; 8) contraindication to rTMS; 9) on >2 mg of lorazepam or equivalent; 10) history of seizures. A diagnosis of bipolar disorder was not excluded. Patients on anticonvulsant medication were not excluded but patients were advised to taper and/or discontinue ahead of starting rTMS.
rTMS Treatment
rTMS treatment was previously described.18,20 Patients received Health Canada-approved open-label high-frequency rTMS to the left DLPFC five times a week for 4 weeks. 20 treatments delivered over a 4-week period is considered a minimally adequate acute trial. 8 High-frequency stimulation with a deep TMS (dTMS) coil or intermittent theta burst stimulation with a figure-8 coil (iTBS) was provided based on patient preference and machine availability. For each patient, only their first course of rTMS treatment at the clinic was included in the analysis. Those who had previously received rTMS at other centers were not excluded. For iTBS, the left DLPFC was localized using the 5.5 cm method for iTBS and used: 120% resting motor threshold, triplet 50 Hz bursts, repeated at 5 Hz, 2 s on and 8 s off, 600 pulses per session for at least 20 sessions (5 sessions per week over 4 weeks). 21 For dTMS, the Brainsway H1 coil targets the dorsolateral and ventrolateral prefrontal cortices bilaterally but has greater intensity and penetration of the left hemisphere. 22 High-frequency dTMS was delivered using: 18 Hz stimulation, 120% resting motor threshold, 55 trains of 2 s duration, inter-train interval 20 s, and 1980 pulses per session for at least 20 sessions (5 sessions per week over 4 weeks). 22
Outcomes and Variables
The main predictor variables of interest were current trauma-related symptoms, history of childhood adversity, and their interaction. As an index of current trauma-related symptoms, probable PTSD status was defined as scoring ≥4 out of 5 on the Primary Care PTSD Screen for DSM-5 (PC-PTSD-5) 23 completed in the week before starting an acute course of rTMS. The PC-PTSD-5 is a self-report scale that asks about the presence or absence of criterion A traumatic experiences (as defined in the DSM) and various trauma-related symptoms within the last month. 23 In primary analyses, probable PTSD status was treated as a binary variable, as binary diagnostic status is commonly used clinically. In secondary analyses, probable PTSD status was treated as a continuous variable to preserve more information; these analyses were conducted to see if results converged with results obtained by using the binary variable. As an index of traumatic experiences in early life, the Adverse Childhood Experiences Questionnaire (ACE-10) was used. The ACE-10 is a self-report scale with scores ranging from 0 to 10 quantifying presence of abuse (physical, emotional, sexual), neglect, and household dysfunction during childhood.24,25 ACE-10 was treated as a continuous variable.
Depression symptoms were assessed using the GRID HAMD-17. 26 The primary outcomes were improvement in HAMD-17 from baseline to end-of-acute treatment at 4 weeks as well as remission (HAMD-17 ≤ 7) and response (decrease in HAMD-17 by over 50% from baseline). Improvement in HAMD-17 was calculated as the difference between 4-week HAMD-17 score and baseline HAMD-17 multiplied by −1. A secondary outcome included the HAMD-17 scores measured longitudinally (at baseline, 2 weeks and 4 weeks). All patients received 20 sessions. In a clinical sample, patients sometimes miss scheduled sessions. Any missed sessions were made up on subsequent days; patients did not receive more than one session daily.
Covariates included in statistical analyses were age, gender, studying/working or not, baseline depression scores, baseline anxiety scores, medical comorbidity, treatment resistance, 27 rTMS type (dTMS or iTBS), and perceived social support. Baseline anxiety was defined as the sum of the anxiety-related questions (items 6 to 8) of the DSM-5 Self-Rated Level 1 Cross-Cutting Symptom Measure (DSM-XC). 28 Medical comorbidity was assessed using the Cumulative Illness Rating Scale-Geriatric (CIRS-G). 29 Despite its name, CIRS-G has excellent psychometric properties for patients across the lifespan. 30 The number of adequate antidepressant medication trials was recorded using the Antidepressant Treatment History Form—Short Form (ATHF-SF). 31 Treatment resistance was calculated based on the Maudsley Staging Method; it accounts for antidepressant medication trials, augmentation, and ECT trials. 27 Perceived social status was included as it may buffer the impact of ACEs. 32
Statistical Analyses
Baseline patient characteristics were stratified by probable PTSD status (PC-PTSD-5 score ≥ 4). Differences between groups at baseline were compared using t-tests or chi-squared tests. Tests for multicollinearity were conducted with inclusion of the variables used in multivariate models; the mean variance inflation factor was 1.35, suggesting minimal multicollinearity. Multiple linear regression was used to model the impact of probable PTSD, ACE-10, and their interaction on HAMD-17 improvement while adjusting for covariates. Multiple logistic regression was used to model the impact of probable PTSD, ACE-10, and their interaction on remission at end-of-acute treatment while adjusting for covariates. Linear mixed-effects models with random intercepts and slopes on time by subject were used for longitudinal analyses of HAMD-17 as the outcome variable with probable PTSD, ACE-10 and their interaction as the main predictors of interest while controlling for other covariates. Two-tailed tests were used with alpha set at 0.05. The above statistical analyses were implemented using Stata 17.0 software (College Station, Texas).
Results
Baseline Characteristics
Data were collected from March 2019 to October 2022. There were 133 unique patients receiving high-frequency stimulation to the left DLPFC for the first time at our center, for which we had PC-PTSD-5 scores, ACE-10, HAMD-17, and other covariates of interest.
Table 1a displays baseline patient characteristics. Participants were on average 41.4 years of age. The majority were women. 29/133 (21.8%) were defined as having probable PTSD (PC-PTSD-5 scores of ≥4). Baseline HAMD-17 was 21.6 (SD = 4.7) and on average, patients had 4.4 (SD = 2.9) antidepressant trials prior to rTMS treatment.
Baseline Characteristics.
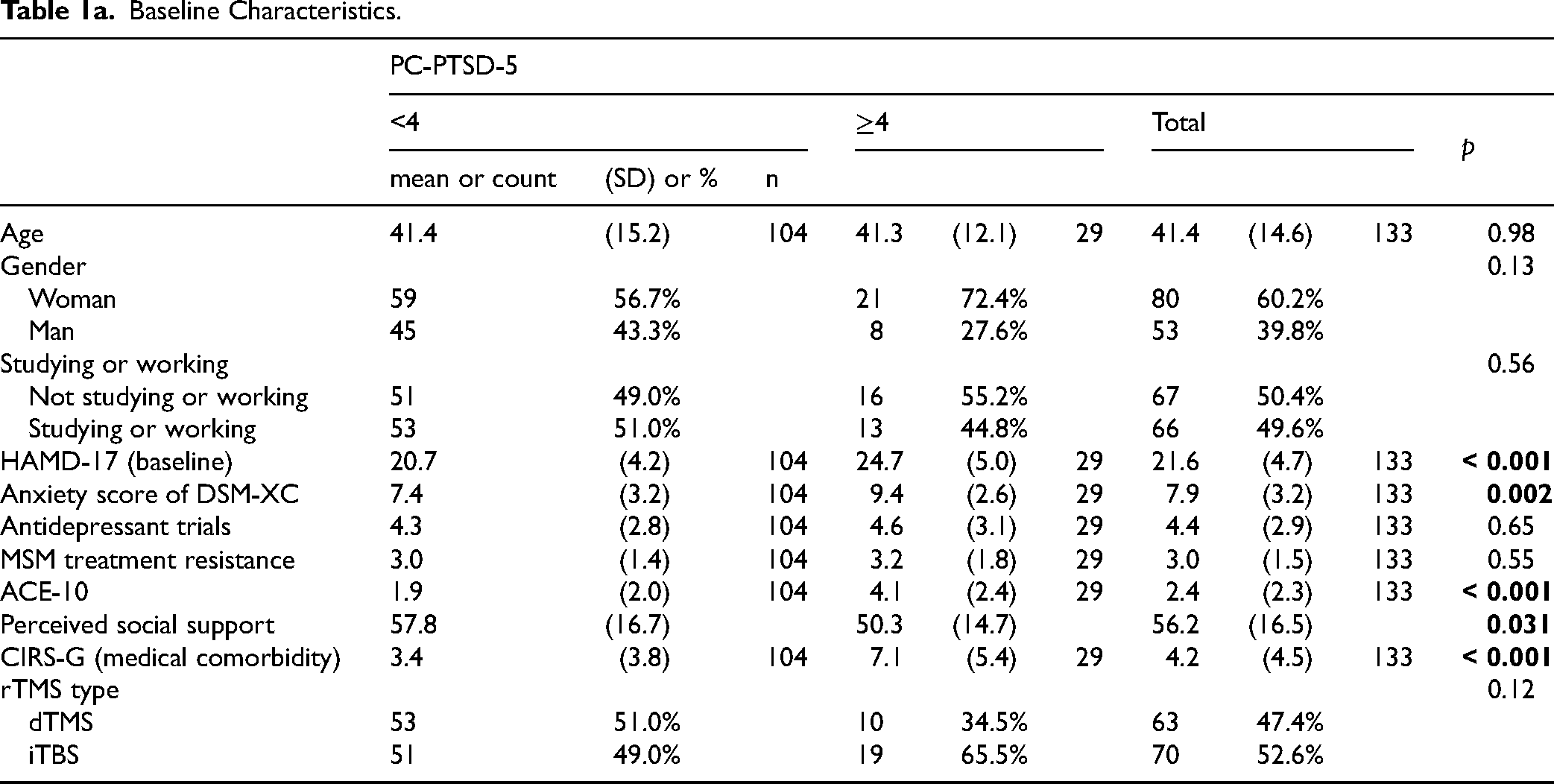
There were significant differences in various characteristics at baseline when stratified by probable PTSD status. Those with probable PTSD had higher mean baseline depression (t = −4.34; p < 0.001) and anxiety scores (t = −3.14; p = 0.002). Those with probable PTSD also had higher mean levels of ACEs (t = −4.87; p < 0.001), higher mean medical comorbidity scores (t = −4.29; p = 0.001), and lower mean perceived social support (t = 2.18, p = 0.031). While the ratio of women: men was higher in the probable PTSD group (21:8), there was no statistically significant difference relative to those without probable PTSD (59:45) (chi-squared = 2.3, p = 0.13).
Outcomes Using the Dichotomous PC-PTSD-5 variable
The mean improvement in HAMD-17 was 8.4 (SD = 6.1) from baseline to 4 weeks (Table 1b). Mean improvement was not significantly different based on probable PTSD status (t = −2.40; p = 0.018). Table 2 shows the results of a multiple linear regression model for HAMD-17 improvement. The interaction of probable PTSD and ACE-10 did not significantly predict improvement (Beta = −0.73; t = −1.29, p = 0.20), neither did probable PTSD (Beta = 4.96; t = 1.94; p = 0.055) or ACE-10 (Beta = 0.47; t = 1.41, p = 0.16). Studying/working (versus not studying/working) (Beta = 2.65; t = 1.12; p = 0.020) and higher baseline HAMD-17 (Beta = 0.37; t = 2.71; p = 0.008) were associated with greater improvement. Neither age, gender, baseline anxiety, treatment resistance, medical comorbidity, rTMS type, nor perceived social support significantly impacted improvement (see Table 2).
Outcomes.

Note. ACE-10= Adverse Childhood Experiences Questionnaire; CIRS-G= Cumulative Illness Rating Scale-Geriatric; DSM-XC= DSM-5 Self-Rated Level 1 Cross-Cutting Symptom Measure; dTMS= deep TMS; HAMD-17= Hamilton Depression Rating Scale; MSM= Maudsley Staging Method; rTMS= repetitive transcranial magnetic stimulation; iTBS= intermittent theta burst stimulation; PC-PTSD-5= Primary Care PTSD Screen for DSM-5; PTSD= posttraumatic stress disorder; rTMS= repetitive transcranial magnetic stimulation.
Bold value indicates statistical significance of p < 0.05.
Regression Models Using Dichotomous PC-PTSD-5 Variable.

*p < 0.05, **p < 0.01, ***p < 0.001.
Note. ACE-10= Adverse Childhood Experiences Questionnaire; CIRS-G= Cumulative Illness Rating Scale-Geriatric; dTMS= deep TMS; iTBS= intermittent theta burst stimulation; HAMD-17= Hamilton Depression Rating Scale; MSM= Maudsley Staging Method; PTSD= posttraumatic stress disorder
By 4 weeks, 22.1% (29/132) achieved remission of depression symptoms (Table 1b). Remission rates were similar between those with and without probable PTSD (chi2 = 1.50; p = 0.22). In a multiple logistic regression model with remission as the outcome, the interaction of probable PTSD and ACE-10 did not significantly impact the odds of remission (Table 2). Higher levels of childhood adversity were associated with higher odds of remission (OR 1.45; 95%CI 1.07–1.96; z = 2.42; p = 0.015). Studying or working (OR 5.8; 95%CI 1.8–18.5; z = 2.96; p = 0.003) was associated with greater odds of remission, whereas higher baseline HAMD-17 was associated with lower odds (OR 0.84; 95%CI 0.73–0.98; z = −2.30; p = 0.022). Other covariates accounted for did not significantly impact odds of remission (see Table 2).
The overall antidepressant response was 42.9% at 4 weeks (57/133) and was similar between the groups (chi2 = 0.059; p = 0.81). Multiple logistic regression with response as the outcome showed no significant impact for the interaction of probable PTSD and ACE-10 (Table 2). Higher ACE-10 scores was associated with higher odds of response (OR 1.44; 95%CI 1.11–1.87; z = 2.76; p = 0.006) as was studying or working (OR 2.50; 95%CI 1.09–5.71; z = 2.17; p = 0.030). Greater medical comorbidity predicted lower odds of response (OR 0.87; 95%CI 0.78–0.98; z = −2.29, p = 0.020). Other covariates accounted for did not significantly impact odds of response (see Table 2).
Longitudinal HAMD-17 scores over the course of treatment were modeled using linear mixed effects (Figure 1a). Probable PTSD status was associated with higher HAMD-17 scores overall during the treatment course (Beta = 5.11; 95%CI 1.46–9.75; z = 2.75; p = 0.006). Overall, HAMD-17 scores decreased significantly by week 2 relative to baseline (Beta = −5.59; z = −8.29; p < 0.001) and continued to decrease into week 4. A contrast of linear predictions showed those with probable PTSD status had a larger change in HAMD-17 going from baseline to 2 weeks compared to those without probable PTSD (chi-squared = 9.32; p = 0.0023). There was no significant ACE-10 by time interaction. There was no significant probable PTSD status by ACE-10 by time interaction.
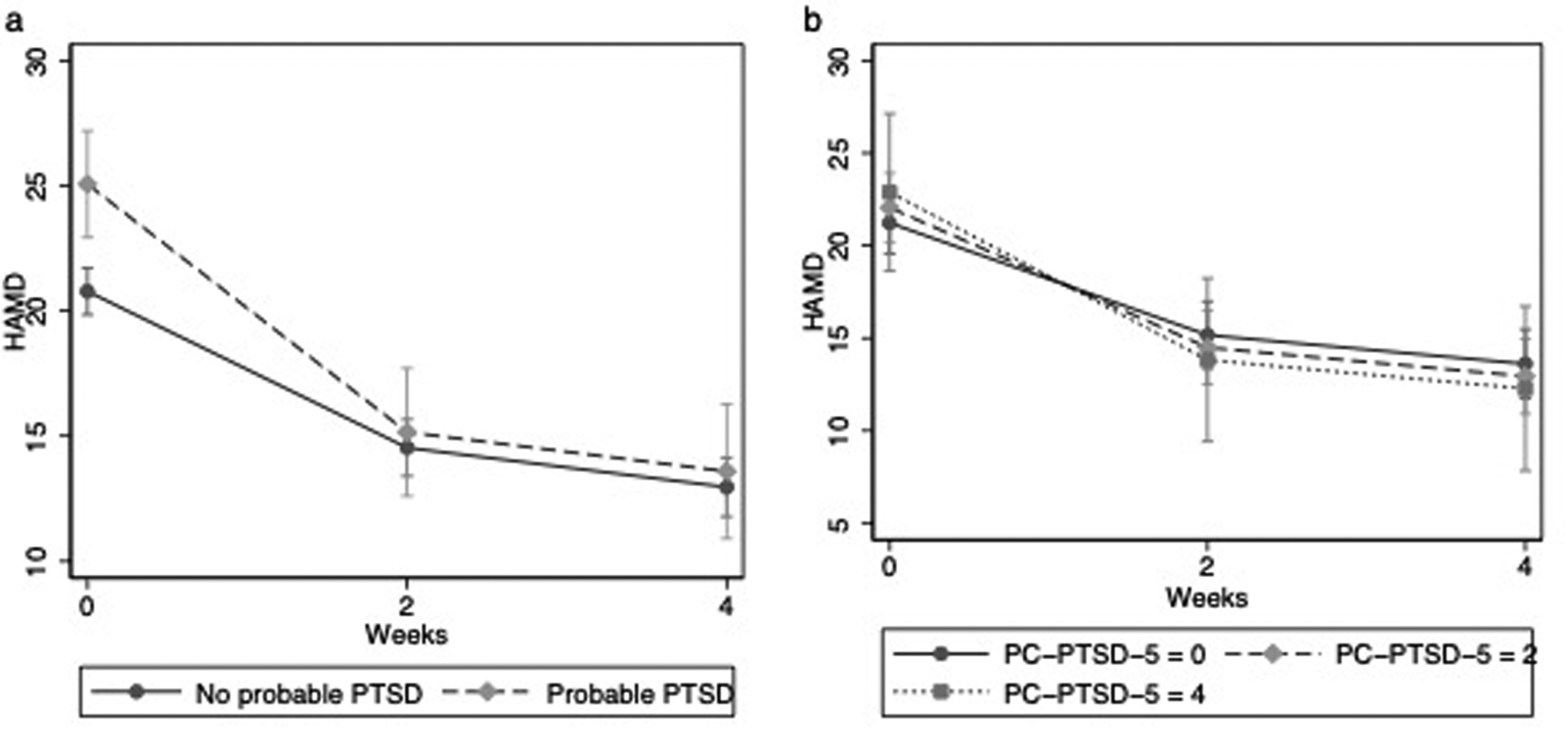
Models of HAMD-17 scores over time with (a) the dichotomous PC-PTSD-5 variable and (b) a continuous PC-PTSD-5 variable (with predicted HAMD-17 scores shown for three levels of PC-PTSD-5 score for illustrative purposes). PTSD, posttraumatic stress disorder.
Outcomes Using PC-PTSD-5 as a Continuous variable
Similar results were obtained when baseline PC-PTSD-5 was treated as a continuous variable in models (Table 3). In a multiple linear regression model for HAMD-17 improvement, the major predictors were studying/working and baseline HAMD-17 (Table 3). PC-PTSD-5 score and its interaction with ACE-10 were not significant predictors of improvement. In a multiple logistic regression model, ACE-10 and studying/working were associated with significantly increased odds of remission, while baseline HAMD-17 was associated with lower odds of remission. Again, PC-PTSD-5 score and its interaction with ACE-10 did not significantly impact odds of remission. A logistic model of response showed ACE-10 and studying/working was associated with greater odds of response, whereas higher medical comorbidity was associated with lower odds of response. Figure 1b shows the predicted trajectories of HAMD-17 scores over time at various levels of PC-PTSD-5 score modeled using linear mixed effects.
Regression Models Using Continuous PC-PTSD-5 variable.

*p < 0.05, **p < 0.01, ***p < 0.001.
Note. ACE-10= Adverse Childhood Experiences Questionnaire; CIRS-G= Cumulative Illness Rating Scale-Geriatric; dTMS= deep TMS; iTBS= intermittent theta burst stimulation; HAMD-17= Hamilton Depression Rating Scale; MSM= Maudsley Staging Method; PTSD= posttraumatic stress disorder.
Results from these regression models did not change substantially when subjects with a bipolar I or II disorder or who had previously received rTMS at another center were excluded leading to a sample size of 111 (see Supplementary Materials).
Discussion
Our study investigated how current PTSD symptoms impact depression outcomes among civilians with no military experience receiving rTMS for MDD. This study advances the field by providing data on civilians (whereas most data previously have been from military veterans) and also considering interaction between current PTSD symptoms and a history of ACEs. Contrary to what might be expected based on comorbid PTSD being associated with poorer depression outcomes, neither current PTSD symptoms nor its interaction with childhood adversity led to poorer treatment response to rTMS for MDD.
Prevalence of Concurrent Posttraumatic Stress-Related Symptoms
In our cohort of adult patients with MDD referred for rTMS treatment at an academic center in Canada, over one in five (21.8%, 29/133) screened positive for probable PTSD. This level of PTSD comorbidity is lower than seen in military veterans with MDD receiving rTMS (between 38 and 49%)9,10,12 and higher than in another retrospective chart review of civilians with MDD receiving rTMS (13%). 33 Those with probable PTSD had greater childhood adversity (on average 4 compared to 2 ACEs) and also higher baseline depression and anxiety as well as medical comorbidity. This concords with the literature showing childhood adversity predicts PTSD development in later life and worsened mental and physical health.24,34 It also fits with known associations between PTSD, childhood adversity, and depressive symptoms.4,5,7
Impact of Proximal and Distal Trauma on Depression Outcomes
We did not find a significant interaction between proximal posttraumatic stress symptoms in the month before rTMS treatment and distal experiences of adversity occurring in childhood and adolescence, on antidepressant outcomes from rTMS. Though probable PTSD status was associated with higher baseline symptom scores, it did not significantly impact improvement in depressive symptoms, remission, or response rates. In contrast, higher ACEs were associated with greater odds of remission and response. Though a trend was noted in our previous study, the current larger sample showed stronger effects and the direction of the effect is in-keeping with another cohort of 62 patients that found childhood adversity was associated with improved outcomes from rTMS treatment. 17 This finding may be consistent with certain hypotheses regarding the mechanism of action of rTMS.
rTMS may have its therapeutic effects by normalizing connectivity and activity between and within brain networks such as the salience network, default mode network, and central executive network.35–37 The salience network is proposed to be involved in cognitive control, detection of rewarding stimuli, and helping to switch between the default mode network (associated with introspective rumination) and the central executive network (associated with goal-directed tasks and sustained attention and working memory).35,37 In depression, there is aberrant connectivity between the default mode network and the salience network which may be associated with increased negative rumination. rTMS may work by normalizing this aberrant connectivity.37,38 Overall, changes in network connectivity are proposed to be associated not just with anti-depressive effect but via improvements in cognitive control and emotional resilience which could have transdiagnostic therapeutic potential. 35
Interestingly, in studies of the impact of 10 Hz or iTBS rTMS on these brain networks in healthy subjects, harm avoidance is associated with connectivity between these brain networks and how much connectivity changes with stimulation.39,40 For example, higher harm avoidance (more vigilance toward potential external threats) is associated with greater change in the functional connectivity between the default mode network and the rostral anterior cinculate cortex with iTBS. 39 Posttraumatic stress symptoms and childhood adversity are associated with greater harm avoidance,41,42 and this may be one explanation for why there may be equal or greater anti-depressive response to rTMS among patients with these comorbidities. Further work would be required to test this conjecture, especially as the relationship between harm avoidance and connectivity changes differs based on stimulation pattern (10 Hz vs iTBS). 40
Strengths and Limitations
The current study has multiple strengths. First, we provide data on the impact of comorbid posttraumatic stress symptoms on rTMS treatment for depression in civilians, the majority of whom were women. Prior to this, much of the data derived from military veterans and men. Second, the data is from real-world practice of rTMS delivery that together with the first strength make it more generalizable. Third, the study not only assessed for posttraumatic stress symptoms but also accounted for childhood adversity and their interaction. The study has a number of limitations, including being retrospective and lacking a sham control. Additionally, the index used for probable PTSD status was a screening measure rather than from a semi-structured interview or a comprehensive scale such as the PTSD Checklist for DSM-5. The PC-PTSD-5 does not capture nature of the trauma(s) experienced. Therefore, we are not able to ascertain whether certain types of trauma have differential effects. Severity of PTSD symptoms were not measured longitudinally.
Conclusion
Over one in five patients with MDD receiving high-frequency rTMS to the left DLPFC screened positive for probable PTSD within the last month, prior to starting rTMS. Though those with comorbid posttraumatic stress symptoms had higher baseline depression and anxiety scores, they had similar antidepressant outcomes in response to rTMS to those without posttraumatic stress symptoms. Higher levels of childhood adversity did not interact with probable PTSD status in worsening response to rTMS for depression. Rather, childhood adversity seemed to predict better outcomes. These findings suggest that neither comorbid posttraumatic stress symptoms nor childhood adversities should preclude patients with MDD from being treated with rTMS. An analysis of the neurobiological effect of rTMS on brain substrates involved in resilience to trauma merits further exploration.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437251342277 - Supplemental material for Impact of Comorbid Posttraumatic Stress-Related Symptoms on Repetitive Transcranial Magnetic Stimulation for Depression in Civilians: Incidence des symptômes du trouble de stress post-traumatique (TSPT) comorbide sur la stimulation magnétique transcrânienne répétitive pour traiter la dépression
Supplemental material, sj-docx-1-cpa-10.1177_07067437251342277 for Impact of Comorbid Posttraumatic Stress-Related Symptoms on Repetitive Transcranial Magnetic Stimulation for Depression in Civilians: Incidence des symptômes du trouble de stress post-traumatique (TSPT) comorbide sur la stimulation magnétique transcrânienne répétitive pour traiter la dépression by Enoch Ng, Sean M. Nestor, Jennifer S. Rabin, Clement Hamani, Nir Lipsman and Peter Giacobbe in The Canadian Journal of Psychiatry
Footnotes
Acknowledgments
The Harquail family through its Midas Touch Foundation provided funding for the establishment of the Harquail Centre for Neuromodulation. The University of Toronto Academic Scholars Award supports the research time of Peter Giacobbe
Data Access
Data access is not available
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
