Abstract
Objective
Electroconvulsive therapy (ECT) is an evidence-based treatment for schizophrenia when anti-psychotic medications do not sufficiently control symptoms of psychosis or rapid response is required. Little is known about how it is used in routine clinical practice. The aim of this study was to identify the association of demographic and clinical characteristics with administration of ECT for schizophrenia spectrum disorders (SSD).
Methods
Among psychiatric inpatients with a diagnosis of SSD in Ontario, Canada (2006–2023), patient-level socio-demographic and clinical characteristics were described in those who did and did not receive ECT. We used multi-variable logistic regression to assess the association between patient-level characteristics and administration of ECT during index hospitalization.
Results
From 164,632 admissions, 2,168 (1.3%) involved exposure to ≥1 inpatient ECT procedure. Compared to those not receiving ECT, those receiving ECT were older, had higher rates of pre-admission medication use, medical and psychiatric comorbidities, outpatient mental health service use, but lower rates of substance use disorders. In the multi-variable logistic regression model, patient-level characteristics most strongly associated with receiving inpatient ECT were the presence of catatonia (odds ratio [OR]: 5.83; 95% confidence interval [95% CI]: 4.01–8.46), comorbid depression (OR: 2.49; 95% CI: 2.07–2.98), obsessive-compulsive disorder (OR: 2.16; 95% CI: 1.55–3.00), while characteristics most strongly associated with not receiving inpatient ECT were myocardial infarction (OR: 0.44; 95% CI: 0.20–0.95) and family conflict towards patient (OR: 0.47; 95% CI: 0.31–0.71). Neither severity of psychotic symptoms, non-command auditory hallucinations nor delusions were associated with administration of ECT.
Conclusions
While characteristics associated with the use of ECT are generally consistent with the indications for ECT (e.g., catatonia, mood disorders), ECT is rarely used amongst individuals with SSD. Severity of psychotic symptoms was not associated with the use of inpatient ECT suggesting an opportunity to increase the use of ECT in this population.
Plain Language Summary Title
Patient characteristics associated with receiving electroconvulsive therapy in schizophrenia and other psychotic illnesses
Plain Language Summary
Electroconvulsive therapy (ECT) is occasionally used for very severe schizophrenia to treat psychosis. However, it is not known how often it is used or what characteristics predict whether someone with schizophrenia will receive ECT. Therefore, we used data from the province of Ontario to better understand the use of ECT amongst individuals with schizophrenia. For this work we used data from the province of Ontario to examine all patients with schizophrenia who were hospitalized. We then examined which of these individuals received ECT and compared them to those who did not. We also used statistical methods to determine which characteristics most strongly predicted whether an individual would receive ECT. Overall, we found that about 1% of individuals with schizophrenia received ECT as an inpatient. There were many differences between those who did and did not receive ECT. Patients who received ECT were were older, less likely to use substances, and had higher rates of medical and psychiatric issues. They also tended to be higher users of mental health services with more outpatient psychiatrist visits, mental health emergency department visits, and mental health hospitalizations. When we looked at which characteristics most strongly predicted whether someone would receive ECT, we found the five strongest predictors of receiving ECT were: (1) a diagnosis of catatonia (where someone stops eating and drinking), (2) diagnosis of depression, (3) not having a history of myocardial infarction (heart attack), (4) being diagnosed with obsessive compulsive disorder, and (5) lack of family conflict towards the patient. Overall, our results suggest that ECT is rarely used amongst inpatients with schizophrenia. There are many characteristics that predict the use of ECT; however, the severity of psychosis does not seem to predict the use of ECT. This suggests there may be opportunities to increase the use of ECT in schizophrenia for severe psychosis.
This visual abstract was created by Michael Wong.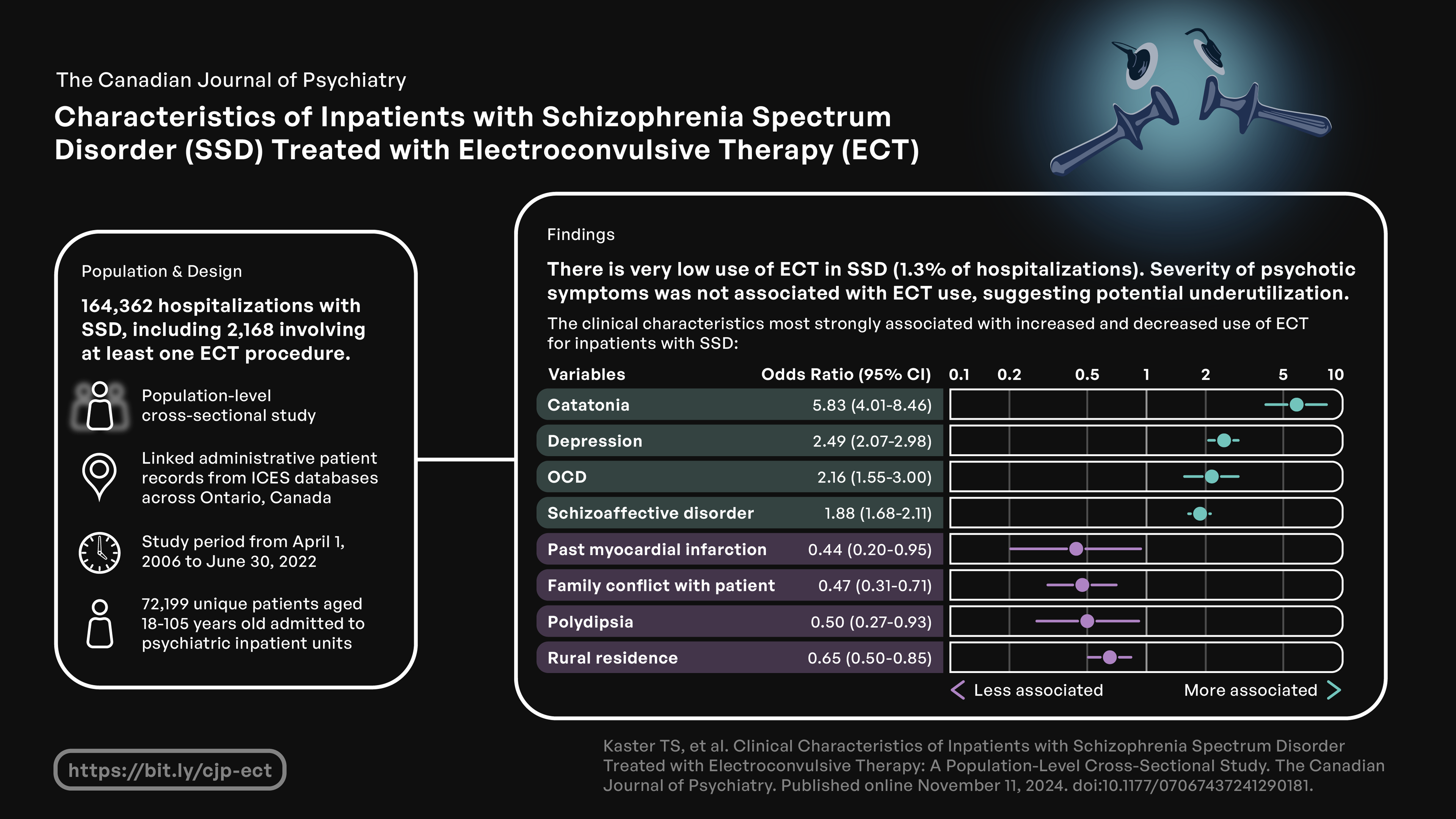
Introduction
Schizophrenia is a severe mental illness affecting approximately 370,000 Canadians. 1 Inadequately treated schizophrenia is linked to lower quality of life, 2 longer hospitalization, 3 and early mortality.4–6 Anti-psychotic medications are a mainstay of treatment but unfortunately are ineffective in >20% of individuals with schizophrenia. 7 One anti-psychotic medication – clozapine – demonstrates superior efficacy for individuals not responding to other anti-psychotics. 8 However, while clozapine is more effective than other anti-psychotics, there are many barriers to its use including side effects and monitoring requirements. 9 Furthermore, approximately 60% of individuals do not respond to clozapine treatment, 10 and of those treated with clozapine approximately 45% will discontinue clozapine within 2 years. 11 Furthermore, anti-psychotic medications require weeks to take effect and individuals with severe symptoms at risk of life-threatening complications – such as suicide or catatonia12,13 – require more rapid improvement than what medications offer.
Electroconvulsive therapy (ECT) is a well-established treatment for severe psychiatric disorders. It consists of an electrical stimulus that causes a brief generalized seizure in patients under general anaesthesia who have also received a paralytic agent. While the use of ECT is well established in depression, 14 its use is less established in schizophrenia. Despite this, the most recent Cochrane Systematic Review on ECT for treatment-resistant schizophrenia concluded that ‘adding ECT to standard care has a positive effect on clinical response when compared with standard care’. 15 The best available evidence supporting ECT for treatment-resistant schizophrenia comes from a prospective randomized clinical trial of clozapine non-responders and randomized them to continued clozapine treatment or ECT. 16 This trial found a 51% response rate for psychosis amongst those receiving ECT initially and a 49% response rate amongst the individuals who crossed over from clozapine treatment to ECT. 16 It is not clear whether ECT is advantageous or not for other outcomes, such as cognitive functioning, general functioning or adverse effects. There is also evidence that ECT can alleviate depressive symptoms and suicidality amongst those with schizophrenia. 17 As a result, the American Psychiatric Association guidelines for the treatment of schizophrenia recommend considering ECT for individuals with treatment-refractory illness (including clozapine-refractory illness) or those requiring a rapid response due to significant suicide risk, catatonia or other indication of significant medical or psychiatric severity. 18
Despite guideline recommendations and evidence from randomized clinical trials, it has been suggested that ECT is underused in developed countries and there is even some suggestion its use is declining.19,20 To our knowledge, how ECT is implemented in routine clinical practice for schizophrenia spectrum disorders (SSD) is largely unknown. Our prior work studying ECT for depression suggests there is likely to be inequitable access to ECT that may be adversely impacted by non-clinical factors such as marital status, education level and socio-economic status that reflect the logistical aspects of obtaining care as opposed to clinical need. 21 Understanding the role of both clinical and non-clinical factors that are associated with administration of ECT treatment in SSD is particularly important given the disproportionate socio-economic disadvantages in this population.22–24
To address these knowledge gaps, we used a clinically representative population-level sample of individuals with SSD to examine the extent to which ECT is used for inpatients with SSD and identify patient characteristics associated with administration of ECT. By improving the knowledge of which characteristics are associated with ECT in SSD, this work will help guide improvements in care for these individuals.
Methods
Study Design and Data Sources
This was a population-based cross-sectional study using linked health administrative datasets in Ontario, Canada, housed at ICES, an independent, non-profit research institute, whose legal status under Ontario's health information privacy law allows it to collect and analyse health care and demographic data, without consent, for health system evaluation and quality improvement purposes. The study cohort was created using diagnostic codes from patient records across databases at ICES using linked unique encoded identifiers. A complete list of data sources used is available in Supplementary Appendix 1.
Study Population
The Ontario Mental Health Reporting System (OMHRS) dataset was used to identify adults (aged 18–105 years old) in Ontario from 1 April 2006 to 30 June 2022 admitted to a psychiatric inpatient unit with a primary discharge diagnosis of an SSD, including schizophrenia, schizoaffective disorder and psychosis not otherwise specified (NOS) using a validated algorithm (positive predictive value: 0.93, 95% CI: 0.84–0.98; sensitivity: 0.95, 95% CI: 0.87–0.99; kappa: 0.94, 95% CI: 0.90–0.98; diagnostic codes in Supplementary Appendix 2).25,26 An individual could have multiple index admissions unless it occurred within 30 days of an earlier admission, which is typically conceptualized as a readmission or continuation of their previous hospitalization rather than a new episode of illness. 27 We excluded those with missing sex, non-Ontario residents, individuals with short-stay admissions (≤3 days), those who died in-hospital and those who were discharged from a non-designated psychiatric unit. Short stay admissions were excluded because these patients receive assessments that do not capture details required to differentiate between ECT-exposed and unexposed admissions.21,28,29
Administration of an ECT Procedure
The main exposure in this study was administration of an ECT procedure during an eligible psychiatric hospitalization. This was determined using Ontario Health Insurance Plan (OHIP) physician billing codes for inpatient ECT (G478) and defined as a binary variable (yes or no) for each index admission. Physician billing codes have high validity and have been used in previous ECT administrative health studies.25,30,31
Covariates
We identified seven categories of characteristics for exploration of their association with administration of ECT: (1) socio-demographics, (2) clinical characteristics, (3) psychiatric comorbidities, (4) medical comorbidities, (5) psychometric and functional domains, (6) health service use prior to admission and (7) pre-hospitalization medication information. For pre-hospitalization medication information, each medication contributed information to multiple covariates based on (i) its route of administration (oral vs. injectable), (ii) its class (typical vs. atypical; list in Supplementary Appendix 3) and (iii) whether it was prescribed as part of anti-psychotic polypharmacy (present vs. absent). For a full list of covariates within each category, refer to Supplementary Appendix 3.
Statistical Analyses
We described the characteristics of individuals who did and did not receive ECT during the study period. The characteristics of an individual who received ECT in an index admission were assessed at their first index admission in which they received ECT (ECT-exposed). If an individual did not receive ECT during any index admission, their characteristics were assessed at their first index admission in the observation period (ECT-unexposed). The characteristics of individuals with multiple index admissions involved both ECT and not ECT were assessed at the time of their first index admission involving ECT (ECT-exposed). We summarized covariates using descriptive statistics and compared groups using standardized differences, where an absolute difference of >0.10 is considered clinically meaningful. 32 Psychotropic medication use 120 days prior to index admission was compared between groups among those individuals eligible for public drug coverage.
To identify the association of patient-level characteristics associated with receiving ECT, we used hospital admission data – which allows multiple admissions per patient and applied a multi-variable logistic regression model with generalized estimating equations, including an exchangeable correlation structure to account for clustering occurring within individuals experiencing repeated hospitalizations. Covariates were presented as either binary variables, continuous variables, or categorical variables with clinically meaningful categories. Sparse data was assessed for all categorical variables. The main multi-variable model included all variables, except those related to prescription medication use prior to index admission as this data was only available for those eligible for public drug coverage. We conducted a complete case analysis. We checked for multi-collinearity in the model to ensure variance inflation factor was <10 for all included variables. Using the same methodology as the primary analysis, an additional model was generated among all individuals who were eligible for public drug coverage, which was determined by (1) dispensing of publicly funded medication within 365 days prior to admission, (2) patient ≥65 years old, or (3) patient indicated as receiving social assistance in OMHRS during index admission. In this model, psychotropic prescription use 120 days prior to index admission was included, in addition to all variables from the main model.
All analyses were conducted using SAS Enterprise Guide version 8.3 1 (SAS Institute, Cary, North Carolina, United States). Referent categories for multi-level categorical variables were used where reference groups were selected to be the ‘ideal’ outcome. For example, the referent group for the ‘unstable living environment’ variable was ‘stable living situation’. As this was a descriptive study, our focus was on effect size and not statistical significance such that we a priori did not adjust for multiple comparisons. 33 This study was reported in accordance with the STROBE guidelines (Supplementary Appendix 4). 34
Results
Descriptive Characteristics
Of 164,362 hospitalizations – which included 72,199 unique individuals – there were 2,168 hospitalizations (1.3%) that involved at least one ECT procedure. There were 1,862 unique individuals who received ECT at least once during a hospitalization (2.6% of all unique individuals). Amongst individuals who received ECT during hospitalization, the median number of ECT procedures was 10 (interquartile range [IQR]: 6–14) and most of these individuals did not receive ECT post-discharge (72%) (Table 1).
Characteristics of Individuals Who Received versus Did Not Receive Electroconvulsive Therapy (ECT).
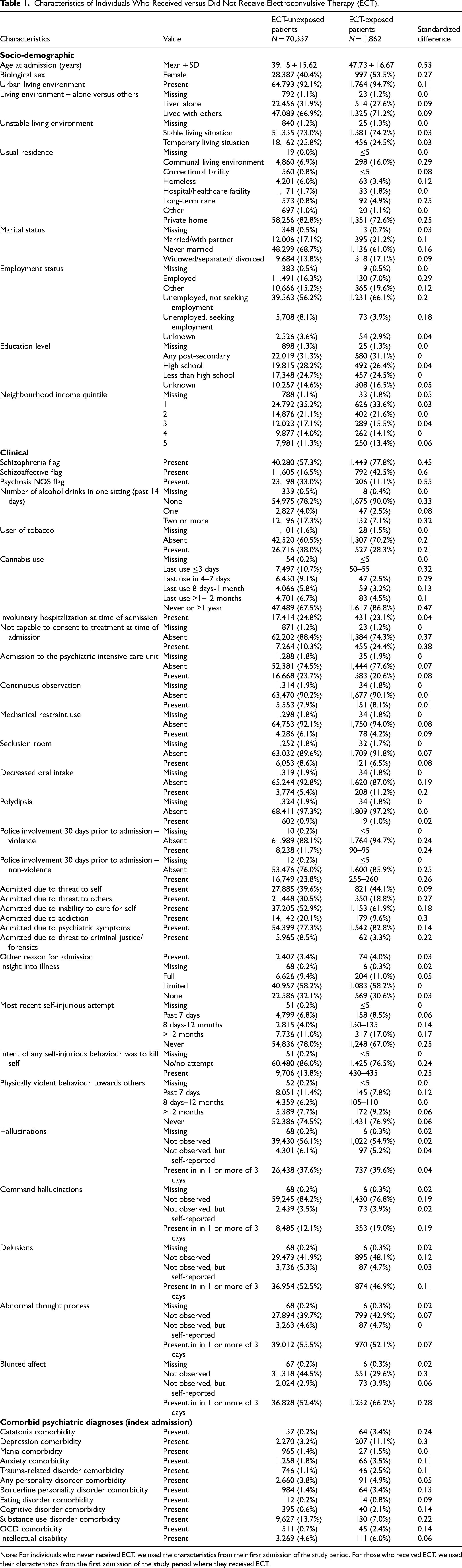
Note: For individuals who never received ECT, we used the characteristics from their first admission of the study period. For those who received ECT, we used their characteristics from the first admission of the study period where they received ECT.
Individuals who received ECT, compared to those who did not, were older (47.73 vs. 39.15 years old), more likely to be female (53.5% vs. 40.4%), living in an urban environment (94.7% vs. 92.1%) in a communal living environment (16.0% vs. 6.9%). ECT-exposed individuals were more likely to be married (21.2% vs. 17.1%) or widowed/separated/divorced (17.1% vs. 13.8%). In terms of clinical characteristics, ECT-exposed individuals compared to ECT-unexposed were more likely to have schizophrenia (77.8% vs. 57.3%) or schizoaffective disorder (42.5% vs. 16.5%) as opposed to an unspecified psychotic disorder (11.1% vs. 33.0%). They consumed less alcohol per sitting in the past 14 days (7.1% vs. 17.3% consumed ≥2 standard drinks per sitting), were less frequent users of cannabis (86.8% vs. 67.5% never used) and were less likely to be admitted due to substance use (9.6% vs. 20.1%). ECT-exposed individuals had a more frequent history of self-injurious attempts (33% vs. 22% any history). ECT-exposed individuals had higher (worse) scores on ADL, Anhedonia, Cognitive Performance, Depressive Severity, Self-Care, Severity of Self-Harm and IADL Impairment Scales (Supplementary Appendix 5). They had higher frequencies for most psychiatric and medical comorbidities and were more likely to have publicly funded drug coverage (Supplementary Appendix 5). Individuals who received ECT treatment were also more likely to have outpatient psychiatrist visits (6.65 vs. 2.89), mental health-related hospitalizations (1.05 vs. 0.36) and mental health-related emergency department visits (1.80 vs. 1.15) in the year prior to admission (Supplementary Appendix 5). Among those with information on prescription medication use, individuals who received ECT treatment had more medication prescriptions across all medications (Table 2).
Presence of Psychotropic Medication Prescriptions for Individuals Who Were Eligible for Public Drug Coverage, Comparing Those who did versus Did Not Receive Electroconvulsive Therapy (ECT).
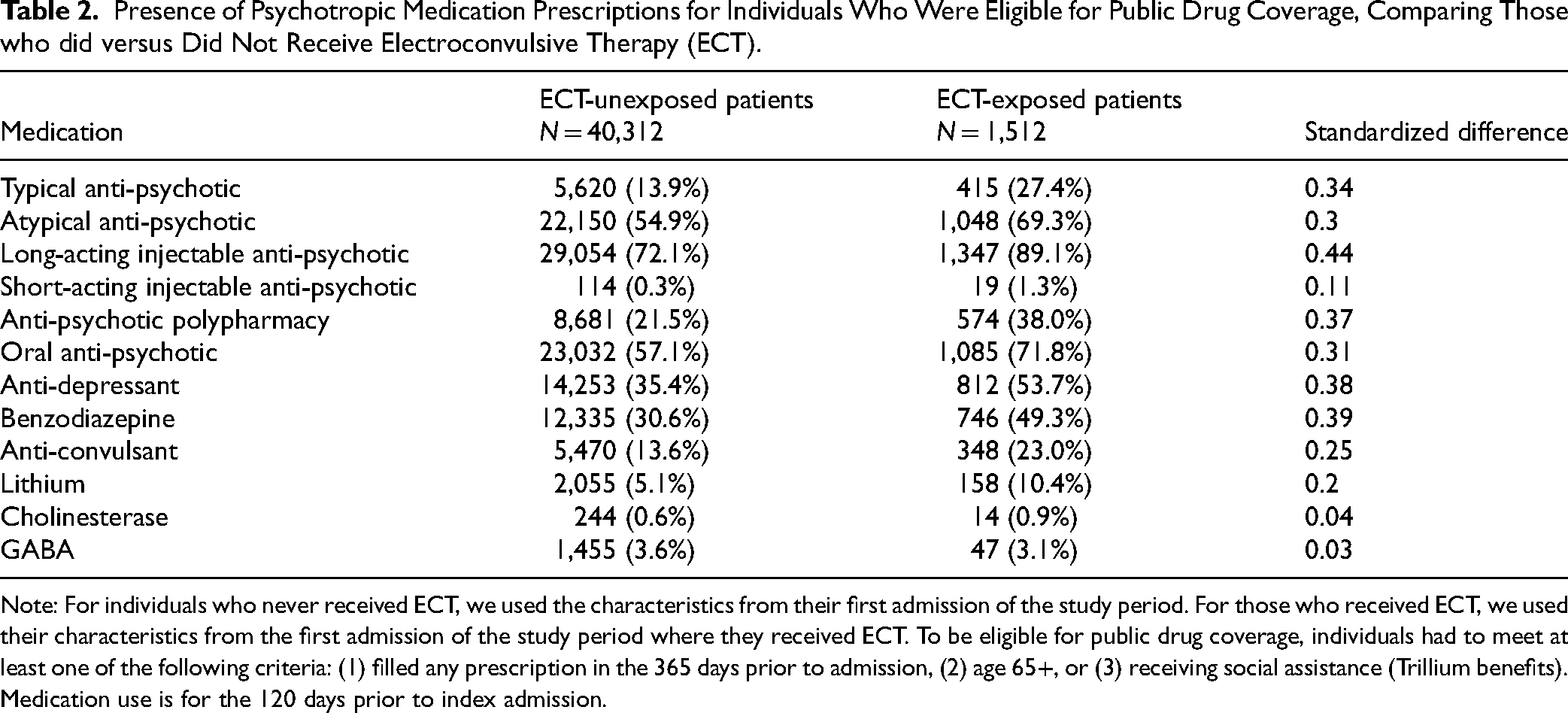
Note: For individuals who never received ECT, we used the characteristics from their first admission of the study period. For those who received ECT, we used their characteristics from the first admission of the study period where they received ECT. To be eligible for public drug coverage, individuals had to meet at least one of the following criteria: (1) filled any prescription in the 365 days prior to admission, (2) age 65+, or (3) receiving social assistance (Trillium benefits). Medication use is for the 120 days prior to index admission.
Multi-variable Models
Socio-demographic Factors
Compared to adults 40–49 years old, younger age was associated with reduced odds of receiving ECT (18–29 years old, OR: 0.77; 95% CI: 0.63–0.95) and older age was associated with increased odds (65+ years old, OR: 1.78; 95% CI: 1.41–2.25) (Figure 1). Individuals who lived in the highest neighbourhood income quintile also had significantly increased odds of receiving ECT (OR: 1.29; 95% CI: 1.06–1.56) relative to the middle-income quintile. Individuals living in a rural (vs. urban) environment were significantly less likely to receive ECT (OR: 0.65; 95% CI: 0.50–0.85) and compared to those currently married, those who were either never married (OR: 0.78; 95% CI: 0.67–0.92) or widowed/separated/divorced (OR: 0.74; 95% CI: 0.62–0.89) were significantly less likely to receive ECT. The presence of family conflict towards the patient significantly reduced odds of receiving ECT and had one of the largest effect sizes of all covariates assessed (OR: 0.47; 95% CI: 0.31–0.71).
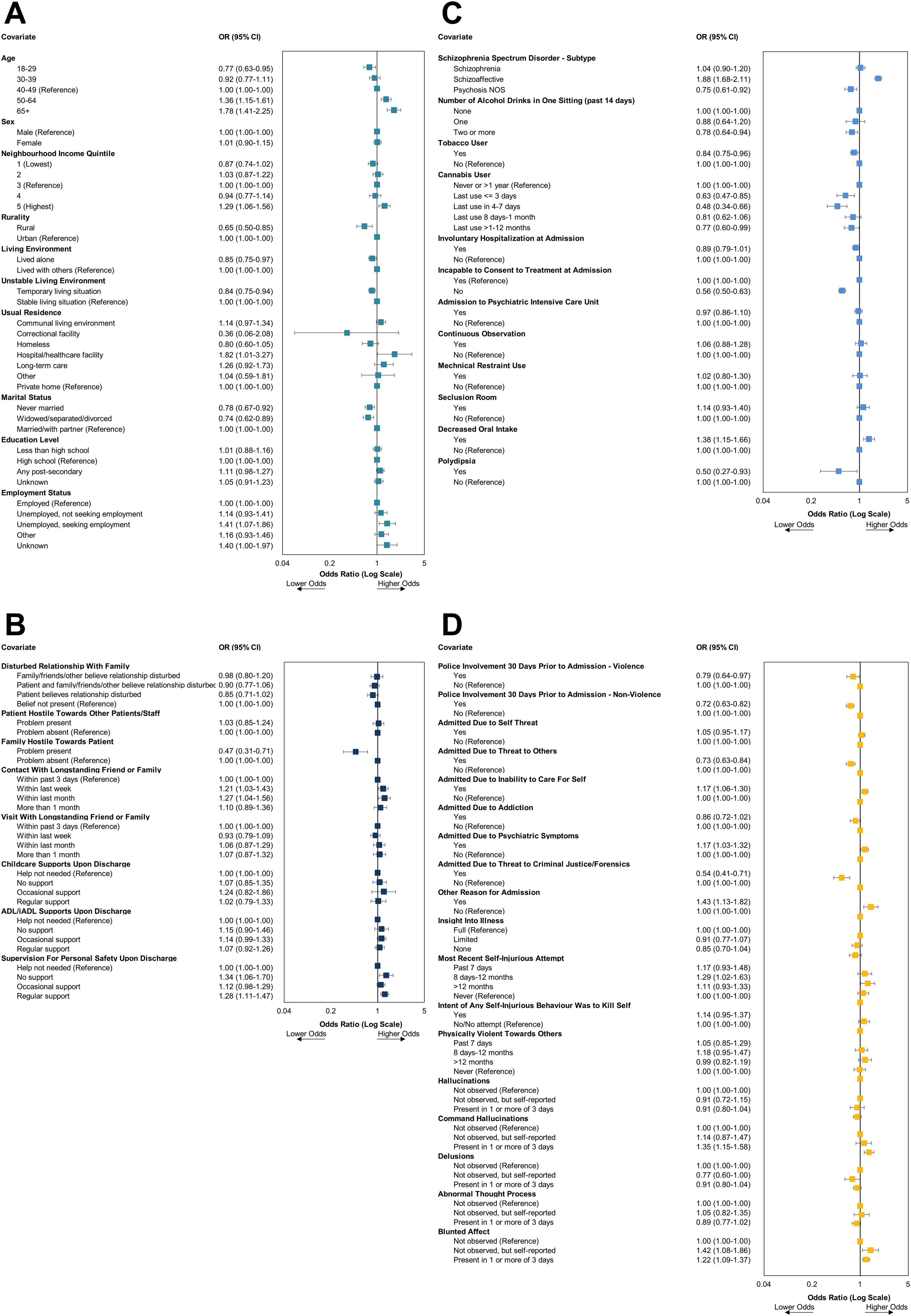
Forrest plot of the association of patient-level characteristics at admission and the administration of inpatient electroconvulsive therapy for (a) socio-demographic, (b) social supports, (c) clinical characteristics and (d) symptom characteristics.
Clinical Factors
The presence of schizoaffective disorder diagnosis was associated with significantly increased odds of receiving ECT (OR: 1.88; 95% CI: 1.68–2.11), while unspecified psychosis was associated with reduced odds (OR: 0.75; 95% CI: 0.61–0.92). Consuming ≥2 alcohol drinks per sitting in the past two weeks (OR: 0.78; 95% CI: 0.64–0.94), using tobacco (OR: 0.84; 95% CI: 0.75–0.96) and using cannabis ≤3 days prior to admission (OR: 0.63; 95% CI: 0.47–0.85) were associated with reduced odds of receiving ECT. Decreased oral intake was associated with increased odds of receiving ECT (OR: 1.38; 95% CI: 1.15–1.66) and polydipsia was associated with reduced odds of receiving ECT (OR: 0.50; 95% CI: 0.27–0.93). A self-injurious attempt within 8 days to 12 months prior to index admission (vs. no history of attempt) was associated with increased odds of ECT (OR: 1.29; 95% CI: 1.02–1.63) as were command auditory hallucinations being present in ≥1 out of past 3 days (vs. not present) (OR: 1.35; 95% CI: 1.15–1.58). In contrast, the presence of hallucinations ≥1 out of past 3 days (OR: 0.91; 95% CI: 0.80–1.04) and the presence of delusions ≥1 out of past 3 days (OR: 0.91; 95% CI: 0.80–1.04) were not associated with receiving ECT.
Psychiatric and Functional Domains
Higher scores on the Activity of Daily Living scale were associated with greater odds of receiving ECT (OR: 1.10; 95% CI: 1.04–1.16) as were greater scores on the Anhedonia scale (OR: 1.04; 95% CI: 1.03–1.06), Depressive Severity Index (OR: 1.04; 95% CI: 1.02–1.06), Self-Care Index (OR: 1.07; 95% CI: 1.02–1.11) (Supplementary Appendix 6). Both the Positive Symptom Scale (OR: 0.99; 95% CI: 0.97–1.01) and the Severity of Self-Harm Index (1.03; 95% CI: 0.98–1.08) were not associated with administration of ECT.
Comorbidities
The presence of catatonia was strongly associated with receiving ECT (OR: 5.83; 95% CI: 4.01–8.46) and was the covariate with the largest effect size (Figure 2). Depression (OR: 2.49; 95% CI: 2.07–2.98) and OCD (OR: 2.16; 95% CI: 1.55–3.00) were also strongly associated with ECT and were two of the largest effect sizes. The presence of mania was also significantly associated with receiving ECT (OR: 1.48; 95% CI: 1.01–2.16), while borderline personality disorder (OR: 1.09; 95% CI: 0.79–1.51) and all other personality disorders (OR: 1.10; 95% CI: 0.86–1.40) were not. For medical comorbidities, a history of myocardial infarction (OR: 0.44; 95% CI: 0.20–0.95) was associated with reduced odds of receiving ECT and was one of the covariates with the largest effect sizes. No other medical comorbidities were associated with receiving ECT (Supplementary Appendix 6).
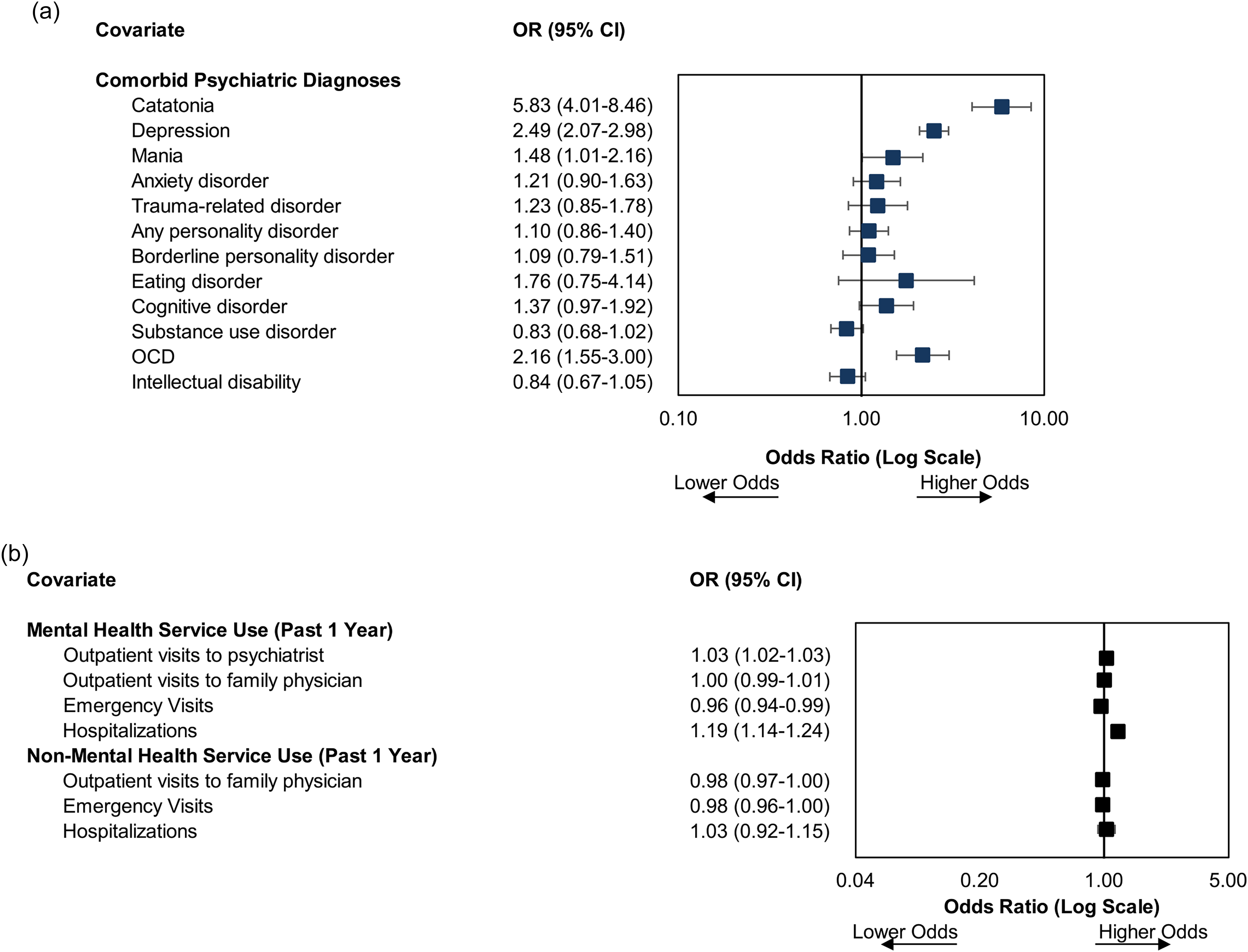
Forrest plot of the association of patient-level characteristics and the administration of inpatient electroconvulsive therapy for (a) psychiatric comorbidities, and (b) health service use.
Medication Information
Individuals eligible for public drug coverage had increased odds of receiving ECT versus those who were ineligible (OR: 1.43; 95% CI: 1.22–1.68) (Supplementary Appendix 6). Individuals who were non-adherent to their medications versus those who were always adherent had reduced odds of receiving ECT (OR: 0.75; 95% CI: 0.65–0.85).
Health Service Use
Outpatient visits to a psychiatrist increasing the odds of receiving ECT (OR: 1.03; 95% CI: 1.02–1.03) and more psychiatric hospitalizations increasing the odds of receiving ECT (OR: 1.19; 95% CI: 1.14–1.24) (Figure 2). In contrast, more mental health emergency visits reduced the odds of receiving ECT (OR: 0.96; 95% CI: 0.94–0.99). Non-mental health service use was not associated with receiving ECT.
Additional Analyses
For individuals with information on prescription medication use, the use of typical anti-psychotics (OR: 1.24; 95% CI: 1.08–1.42), anti-depressants (OR: 1.32; 95% CI: 1.18–1.48) and benzodiazepines (OR: 1.20; 95% CI: 1.08–1.34) were associated with increased odds of ECT, while anti-convulsants (OR: 0.79; 95% CI: 0.70–0.90), oral anti-psychotic medication (OR: 0.79; 95% CI: 0.67–0.93) and cholinesterase inhibitors (OR: 0.47; 95% CI: 0.27–0.81), were associated with reduced odds of receiving ECT (Figure 3).
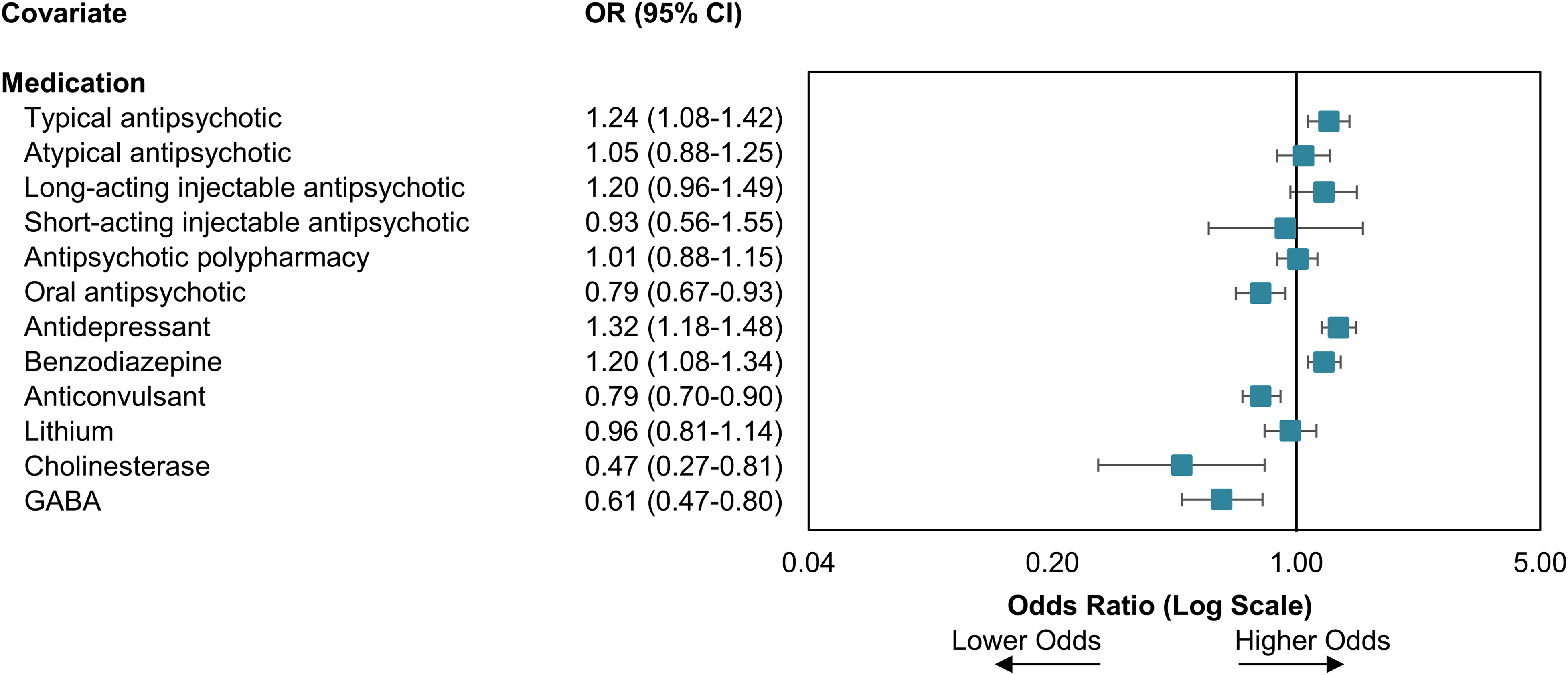
Forrest plot of the odds ratio regarding association of medication information and the administration of inpatient electroconvulsive therapy for those with medication information available.
Discussion
Using a population-level dataset of 164,632 psychiatric admissions among individuals with SSD, we found that less than 2% of admissions involved the delivery of ECT. Patient-level characteristics differed dramatically between individuals who did versus did not receive ECT. When we examined the association between patient-level characteristics and administration of ECT, we identified three key themes. First, our analysis identified a number of clinically sensible patient characteristics associated with receiving ECT. The presence of catatonia, decreased oral intake, recent self-injurious behaviour and command auditory hallucinations were all associated with an increased likelihood of receiving ECT. Similarly, a history of myocardial infarction, anti-convulsant medication prescriptions, recent consumption of alcohol, cannabis or tobacco were all associated with reduced odds of receiving ECT. Second, there were several socio-demographic and non-clinical characteristics that were found to be associated with ECT including age, neighbourhood income quintile, living in an urban setting, family conflict and marital/partnered status that were all significantly associated with receiving ECT. Third, patient-level characteristics related to mood disorders were more often associated with receiving ECT compared to characteristics related to psychotic symptoms. For example, comorbid mood disorders, the presence of schizoaffective disorder, anti-depressant medication use, psychometric scales of anhedonia and depression were all associated with the increased use of ECT. In contrast, a diagnosis of schizophrenia, presence of delusions or non-command auditory hallucinations and psychometric scales of psychosis were not associated with ECT use.
The fact that our results suggest that many clinically sensible factors are being considered in the delivery of ECT for patients with SSD is reassuring. For example, individuals with a prior myocardial infarction (indicating cardiac disease) may be at greater risk of cardiovascular complications from ECT (e.g., cardiac arrhythmias). 35 Similarly, individuals with significant substance use may be at greater risk of complications from general anaesthesia as well as have reduced efficacy from this intervention. 36 After accounting for other clinical characteristics, our findings indicate that these factors independently reduce the likelihood of an individual receiving ECT, which is consistent with best practices in ECT care. 37 Our results indicate that clinicians are also appropriately considering patients who require rapid response to treatment. For example, individuals with catatonia may be at risk of life-threatening complications and require rapid response. 12 Similarly, individuals with reduced oral intake, command auditory hallucinations or recent intentional self-injury all require rapid symptomatic improvement to mitigate the risk of adverse outcomes. 38 These findings are concordant with the American Psychiatric Association Practice Guidelines for schizophrenia which suggest that individuals – treated with clozapine or not – who have ‘catatonia or significant suicide risk or who require a rapid response because of the severity of their psychiatric or medical condition’ be considered for ECT. 18 The findings are also concordant with the Royal Australian and New Zealand College of Psychiatrists guideline for schizophrenia which states that ‘ECT for the treatment of schizophrenia may be considered in combination with anti-psychotic medication when a rapid clinical response is an urgent priority’. 39
In terms of the socio-demographic and non-clinical characteristics associated with ECT, similar to our prior work on the use of ECT in depression, 21 it appears there is inequitable access to ECT in SSD. We found several indicators that logistical aspects of care, known as ‘enabling resources’ influence the likelihood of someone receiving ECT. 40 Examples of enabling resources identified in this work that increased the likelihood of receiving inpatient ECT included higher neighbourhood income quintile, being married, living in an urban location, absence of family conflict towards patient and larger number of outpatient psychiatrist visits prior to hospitalization. These results suggest that outpatient supports – in particular, an individual's family support system – play an important role in determining whether an individual with SSD will receive ECT during a hospitalization. This is an important finding because social disability and disconnection are often significantly impaired amongst those with SSD.41,42
The fact that mood or affective symptoms were strongly associated with the use of ECT in SSD is surprising given the sparse evidence for this indication in schizophrenia.17,43 It is possible the use of ECT may therefore reflect the difficulty of treating comorbid depression in schizophrenia with small effect sizes being observed with anti-depressant treatment. 44 In contrast, our results found that indicators of psychosis severity (e.g., positive symptom scale, presence delusions) were not associated with the use of ECT. This is despite evidence indicating that ECT can effectively treat psychotic symptoms coming from observational studies, 45 randomized controlled trials, 16 meta-analyses, 15 and also being recommended for consideration by the American Psychiatric Association. 18 It is possible that this may be due to a lack of awareness about the use of ECT for SSD in Canada, or perhaps reflect ongoing uncertainty about the effectiveness for this indication. As a result, there may be opportunities to increase the use of ECT for the treatment of psychotic symptoms amongst hospitalized patients.
Though these findings have relevance at both the individual clinician and healthcare system level, there are important limitations to acknowledge. Our study lacked information on clozapine, which is a particularly important anti-psychotic medication for schizophrenia. 18 This is due to clozapine being funded by a different mechanism in Ontario, Canada compared to all other medications such that it is not available in the administrative health databases used for this study. We also did not have information available on prior treatment response. That being said, we accounted for numerous other clinical factors that may indirectly account or correlate with these covariates such as number of mental health hospitalizations, psychosis severity and anti-psychotic polypharmacy. 46 Furthermore, we only considered inpatient ECT and therefore our results are not generalizable to individuals receiving outpatient ECT. However, given the comprehensive data available in the OMHRS dataset and the fact that >80% of ECT procedures in Ontario are delivered to inpatients, these limits on generalizability are likely minimal. 47
Overall, our study provides important information about the use of ECT for SSD. Our results indicate that ECT is infrequently used amongst hospitalized patients with SSD in Ontario, Canada. Despite this, our findings suggest that many clinically sensible and appropriate patient characteristics influence whether an individual receives ECT as an inpatient. Unfortunately, there is indication that non-need-based characteristics may influence the administration of ECT and that ECT may be primarily used in this population for mood disorders as opposed to psychotic symptoms. Future work can be directed to better understand the reasons for inequitable access to ECT and develop evidence to address the uncertainty around the use of ECT in this population. For example, conducting studies that assess the impact of ECT on clinically relevant real-world outcomes such as hospitalization or suicide death. Ultimately, improving the availability of ECT for those with severe psychotic symptoms has the potential to significantly improve outcomes for those suffering from this serious and persistent illness.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437241290181 - Supplemental material for Clinical Characteristics of Inpatients with Schizophrenia Spectrum Disorder Treated with Electroconvulsive Therapy: A Population-Level Cross-Sectional Study: Titre: Caractéristiques cliniques des patients hospitalisés présentant un trouble du spectre de la schizophrénie et traités par électrochocs : Une étude de population transversale
Supplemental material, sj-docx-1-cpa-10.1177_07067437241290181 for Clinical Characteristics of Inpatients with Schizophrenia Spectrum Disorder Treated with Electroconvulsive Therapy: A Population-Level Cross-Sectional Study: Titre: Caractéristiques cliniques des patients hospitalisés présentant un trouble du spectre de la schizophrénie et traités par électrochocs : Une étude de population transversale by Tyler S. Kaster, Amreen Babujee, Isobel Sharpe, Taeho Greg Rhee, Tara Gomes, Paul Kurdyak, George Foussias, Duminda Wijeysundera, Daniel M. Blumberger and Simone N. Vigod in The Canadian Journal of Psychiatry
Footnotes
Acknowledgements
We would like to thank Kinwah Fung for her assistance and contributions to this work. This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). Parts of this material are based on data and/or information compiled and provided by CIHI and the Ontario Ministry of Health. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred. We thank IQVIA Solutions Canada Inc. for use of their Drug Information File. Parts of this material are based on data and information compiled and provided by the Ontario Ministry of Health. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred. This document used data adapted from the Statistics Canada Postal Code OM Conversion File, which is based on data licensed from Canada Post Corporation and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from ©Canada Post Corporation and Statistics Canada.
Declaration of Conflicting Interests
TSK receives research support from the Canadian Institutes of Health Research (CIHR), the Patient-Centered Outcomes Research Institute (PCORI) and the AFP Innovation Fund. GR was supported in part by the National Institute on Aging (NIA) (#R21AG070666; R21AG078972), the National Institute of Mental Health (#R21MH117438), the National Institute on Drug Abuse (#R21DA057540), and the Institute for Collaboration on Health, Intervention, and Policy (InCHIP) of the University of Connecticut. TGR serves as a review committee member for Patient-Centered Outcomes Research Institute (PCORI) and Substance Abuse and Mental Health Services Administration (SAMHSA) and has received honoraria payments from PCORI and SAMHSA. TGR has also served as a stakeholder/consultant for PCORI and received consulting fees from PCORI. TGR serves as an advisory committee member for International Alliance of Mental Health Research Funders (IAMHRF). TG was supported by a Canada Research Chair in Drug Policy Research and Evaluation and has received grant support for a research programme outside of the present manuscript from the Ontario Ministry of Health. GF has been involved in research sponsored by Medicure Inc., and Neurocrine Bioscience, has received consultant fees from Roche and has received speaker fees from Roche, Lundbeck and Novartis. DMB receives research support from CIHR, NIMH (R01MH112815; R21MH128815 and R01MH1192850), Brain Canada and the Temerty Family through the CAMH Foundation and the Campbell Family Research Institute. He received research support and in-kind equipment support for an investigator-initiated study from Brainsway Ltd. He was the site principal investigator for three sponsor-initiated studies for Brainsway Ltd. He also received in-kind equipment support from Magventure for two investigator-initiated studies. He received medication supplies for an investigator-initiated trial from Indivior. He is a scientific advisor for Sooma Medical. He is the Co-Chair of the Clinical Standards Committee of the Clinical TMS Society (unpaid). SNV receives royalties from Up To Date for authorship of materials on depression and pregnancy. AB, IS, PK, and DW declare that they have no conflicts of interest.
Data Availability
The dataset from this study is held securely in coded form at ICES. Although data-sharing agreements prohibit ICES from making the dataset publicly available, access might be granted to those who meet pre-specified criteria for confidential access (![]() ). The full dataset creation plan and underlying analytic code are available from the corresponding author on request, understanding that the computer programs might rely on coding templates or macros that are unique to ICES and are therefore either inaccessible or might require modification.
). The full dataset creation plan and underlying analytic code are available from the corresponding author on request, understanding that the computer programs might rely on coding templates or macros that are unique to ICES and are therefore either inaccessible or might require modification.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was funded by the Canadian Institutes for Health Research. The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
