Abstract
Background
Neurological soft signs (NSSs), minor physical anomalies (MPAs), and oculomotor abnormalities were plausible biomarkers in bipolar disorder (BD). However, specific impairments in these markers in patients after the first episode mania (FEM), in comparison with first-degree relatives (high risk [HR]) of BD and healthy subjects (health control [HC]) are sparse.
Aim of the study
This study aimed at examining NSSs, MPAs, and oculomotor abnormalities in remitted adult subjects following FEM and HR subjects in comparison with matched healthy controls. Investigated when taken together, could serve as composite endophenotype for BD.
Methods
NSSs, MPAs, and oculomotor abnormalities were evaluated in FEM (n = 31), HR (n = 31), and HC (n = 30) subjects, matched for age (years) (p = 0.44) and sex (p = 0.70) using neurological evaluation scale, Waldrop's physical anomaly scale and eye tracking (SPEM) and antisaccades (AS) paradigms, respectively.
Results
Significant differences were found between groups on NSSs, MPAs, and oculomotor parameters. Abnormalities are higher in FEM subjects compared to HR and HC subjects. Using linear discriminant analysis, all 3 markers combined accurately classified 72% of the original 82 subjects (79·2% BD, 56·70% HR, and 82·1% HC subjects).
Conclusions
AS and SPEM could enhance the utility of NSSs, and MPAs as markers for BD. The presence of these abnormalities in FEM suggests their role in understanding the etiopathogenesis of BD in patients who are in the early course of illness. These have the potential to be composite endophenotypes and have further utility in early identification in BD.
Plain Language Summary Title
Eye movement abnormalities and Atypical Neurodevelopmental markers as Composite Measurable components in the pathway between disease manifestation and genetics in Bipolar I Disorder
Plain Language Summary
Introduction
Bipolar disorder (BD) is a heterogeneous and genetically complex mood disorder. 1 The longitudinal course of BD is progressive and more disabling over time, with mood episodes becoming more severe and refractory to treatment, with less inter-episode recovery, and increased risk of relapse.2,3 Delays in diagnosis and treatment were one of the reasons for the unfavourable outcomes of BD. 4 Early diagnosis helps improve the course of the illness and also has precise value for conceptualizing the underlying neurobiology of BD. There are several studies which have evaluated the various biomarkers of BD, one of the latest reviews has concluded that BD is a heterogeneous disorder with varied etiologic and pathophysiologic connotations a single biomarker may not be identified. In this challenging condition, a panel of tests is expected to be needed to identify and monitor progression, and in therapeutic decision-making. 5 Another review which looked at the candidate biomarkers across psychiatric disorders, has similarly quoted that looking at combinations of available markers may also be useful and this could also provide complementary information. 6
One of the potential biomarkers which have been studied recently is the ocular-related markers. One study which looked at the retinal microvasculature among schizophrenia and BD patients noted that, both these groups had narrower vasculature compared to healthy people. 7 Another systematic review looked at studies which have reported retinal abnormalities using optical coherence tomography and electroretinography among schizophrenia spectrum patients. 8 This review reported that the retinal changes in these patients can be both trait and state markers. Eye-tracking studies offer an opportunity to understand oculomotor functioning which correlates with various aspects of brain functioning. In this context, smooth pursuit eye movement (SPEM) and antisaccades (AS) eye movement abnormalities have been studied in BD. 9 In addition, oculomotor abnormalities have been demonstrated in the subjects at high risk (HR) for BD. 10 There is preliminary evidence to suggest that oculomotor abnormalities are a potential endophenotype for BD. 11 Neurological soft signs (NSSs) are subtle deficits in motor coordination, sensory integration, sequencing of complex motor acts, and primitive reflexes which cannot be decisively limited to localized brain regions. 12 Minor physical anomalies (MPAs) are inconspicuous, clinically and cosmetically insignificant errors in the development of morphological structures, 13 which persevere into adulthood and can be assessed clinically. In addition, oculomotor abnormalities, MPAs, and NSS have been demonstrated in the subjects at HR for BD10,14 Hence there is preliminary evidence to suggest that these are potential endophenotypes for BD.
Simple clinical markers such as MPA, NSS, and oculomotor abnormalities, in combination, maybe a more robust endophenotype in BD. The literature evaluating these markers in individuals at risk for BD is limited. This study aimed at examining NSS, MPA, and oculomotor abnormalities in remitted adult subjects early in the course of BD (first episode mania [FEM]) and HR subjects in comparison with the matched healthy controls (HCs) and evaluating if these taken together could serve as a composite endophenotype for BD.
Materials and Methods
This study was conducted at a tertiary care psychiatry centre in southern India and approved by the Institute Ethics Committee.
Sample Size
For evaluating the objectives mentioned above, the optimal sample size was estimated using G power 3.1. 15 Based on effect size data from previous studies,16,17 it was estimated that a sample size of at least 30 subjects in each group would be required to detect a 2-tailed significant difference of α = 0·016 (to control for multiple comparison corrections) 17 for 2 critical parameters of interest, that is, pursuit velocity gain and AS error percentage with an estimated 80% power.
Subjects
FEM (<6 months of remission; ICD-10 Diagnostic Criteria for Research; n = 31), with/without psychotic symptoms; including those with prior depression were recruited. Remission was defined by the Young Mania Rating Scale (YMRS) and Hamilton Rating Scale for Depression (HDRS) < 8. Individuals aged between 18 and 45 years, of all genders, right-handed and with education of at least 7th standard were included. Those with any other axis I psychiatric disorders including substance dependence (except nicotine dependence), mental retardation, or other clinically recognizable neurological conditions, colour blindness, and history of head injury were excluded. Siblings or offspring of patients with BD (30% were relatives of the FEM group), matched for age, gender, education, and handedness were recruited as HR subjects (n = 31). HC (n = 30) with no family history of psychiatric disorder were also recruited. All participants provided written informed consent.
Clinical Assessments
Mini-International Neuropsychiatric Interview (MINI-version 6) 18 was used to screen all the subjects for diagnosis and comorbidities, Edinburgh handedness inventory (EHI) 19 for handedness, Ishihara colour blindness evaluation 20 charts to screen the subjects for colour blindness. Modified Waldrop MPA Scale 21 (WPA) was used to assess 12 MPAs located in the eyes, ears, oral cavity, hands, and feet. The NSS was evaluated using the modified neurological evaluation scale (NES). 22
The assessments were carried out by a trained rater who established good interrater reliability for MINI, YMRS, HDRS, EHI, NES, and WPA in a sample of 12 independent subjects before the study. The interrater reliability was 0.4–0.6 for all the assessments.
Eye-Tracking Assessments
Monocular eye movement recordings of the dominant eye were conducted in a room with controlled illuminance using Eye-link 1000 (SR Research, Canada) with a desktop mount camera at 1,000 Hz. 23 A 10-point calibration followed by validation was done to ensure an eye-target error of <10 was performed before beginning each task.
AS Paradigm
The AS task is based on the principles and procedures described in the literature. 24 Each participant performed a total of 24 prosaccades (PS) and 48 AS trials. The stimuli for each trial were presented on a screen with a black background. This target appeared at any of the 4 possible locations, at either of the sides ±6 degrees and ±12 degrees of visual angle from the centre. 25 Trials with the following features were discarded: (a) saccades with latencies <80 and >600 ms after target onset as they would have been either anticipations or artefacts of the subject not paying enough attention, respectively. (b) Trials in which though the first saccade is in the correct direction, the eye crossed over the midline beyond the fixation window (i.e., ±28 pixels) and looked close to/at the target during the course of the trial. (3) Trials containing random performance errors such as the subject looking out of screen/distorted corneal reflection (resulting in the start/end position of the saccade taking negative values).
SPEM Paradigm
For SPEM experiments a 0.25 × 0.25-degree moving red-coloured circle was asked to be followed in 20 (10 fast and 10 slow) trials. In each trial, the target started from the centre position using an unpredictable initial target step in the opposite direction of subsequent target motion, which crosses the centre point after 200 ms of motion, then traverses horizontally at a predictable constant speed of 9.4 degrees/s (slow trials) or 18.7 degrees/s (fast trial) at 24 degrees of visual angle (a ramp). Each trial consisted of 6-and-a-half ramps. The position gain and velocity gain for each trial were later evaluated using a custom-made script in the student's edition of LabVIEW 8·6·1 (National Instruments™, USA). The interrater reliability exercise was conducted on independent samples before the analysis of the eye-tracking data with Cronbach's alpha of more than 0.95 for all the eye-tracking parameters. The eye-tracking data were analysed after blinding the raters for the subjects and the group.
Statistics
Statistical analysis was done using IBM SPSS statistics version 26.0 (IBM, USA). The Shapiro–Wilk test was used to assess normality. Parametric (one-way analysis of variance with post hoc Tukey) and nonparametric (Kruskal–Wallis H with post hoc pairwise comparison) statistical tests were used for normally and nonnormally distributed continuous variables, respectively. The chi-square test was used to assess the differences in categorical variables between the groups. The significance of α was set at p < 0·05 (two-tailed) and the p-value was corrected using the Benjamin–Hochberg false discovery rate, a sequential Bonferroni correction for multiple hypothesis tests. 26
Kendall's tau_b correlation was done to evaluate the association between the MPA, NSS, and eye-tracking measures. Finally, linear discriminant functional analysis was carried out to predict group membership (FEM, HR, and HC) based on the predictor endophenotype variables, that is, MPAs total score, NES total score, SPEM, and saccades parameters. No covariates were used in any of the comparison analyses since groups were comparable in terms of age and gender which are the two variables that could influence the results.
Results
The sociodemographic variables across the 3 groups were comparable, a statistically significant difference was found between FEM subjects when compared with HR and HC subjects concerning the total duration of education and marital status (p < 0.05) (Table 1). FEM subjects had a mean age at the onset of illness 25.19 ± 6.19 (in years). The mean age of onset of FEM was 25.35 ± 6.21 years. Five (16%) of the patients had a prior history of depression with onset at 27.60 ± 9.39 years with 1.25 ± 0.5 mean number of depressive episodes each lasting for 1.37 ± 0.53 months. The duration of remission from illness at the time of recruitment was 4.29 ± 1.7 months. Following FEM, 38% of the subjects were treated with antipsychotics only and 62% were treated with a combination of mood stabilizers plus antipsychotics. Lithium was used in 90% (n = 17) FEM as a mood stabilizer.
Sociodemographic Characteristics of the 3 Groups (Total N = 92).
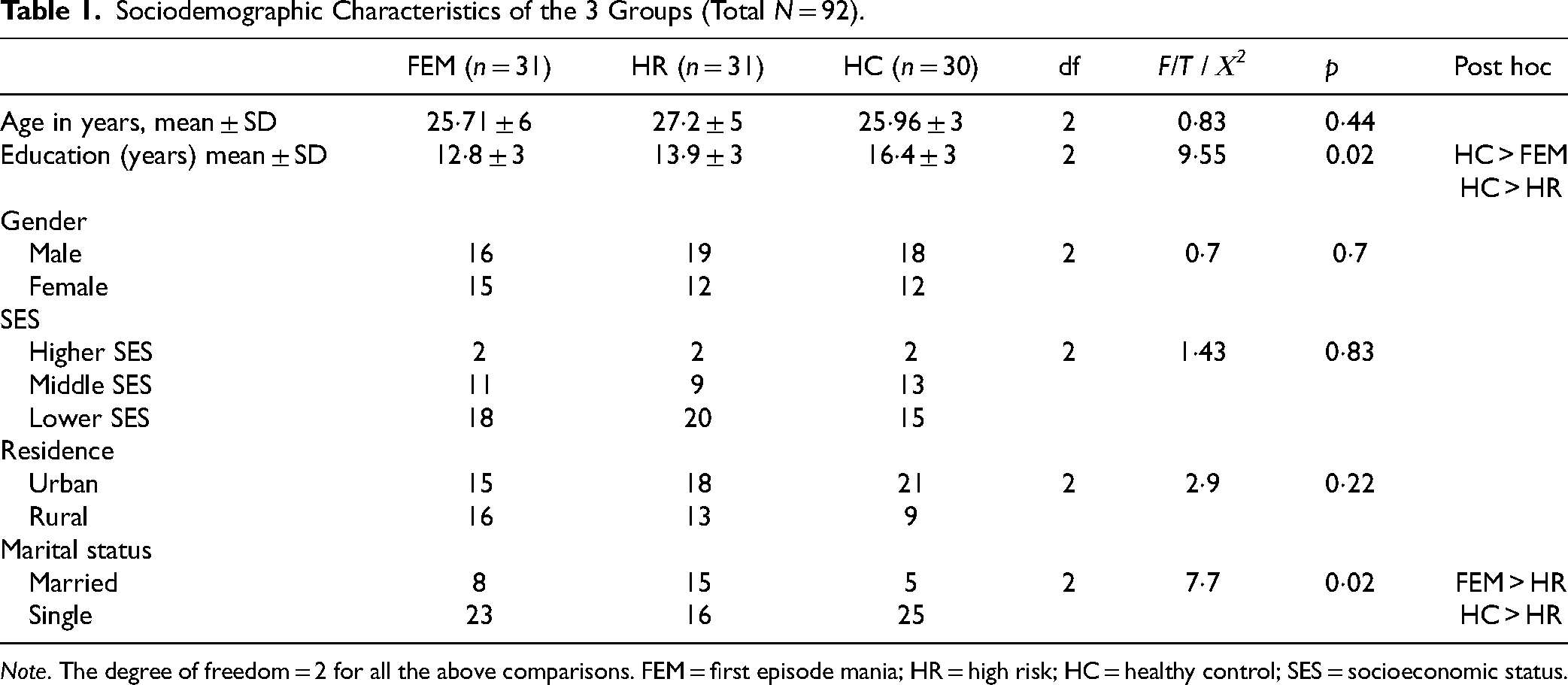
Note. The degree of freedom = 2 for all the above comparisons. FEM = first episode mania; HR = high risk; HC = healthy control; SES = socioeconomic status.
On comparing the NSS scores across the 3 study groups, a significant group difference was noted (Table 2). Post hoc analysis showed the total scores and all the subscores of NSS were highest in FEM compared to HR and HC. Similar results (Table 2) were found by comparing the total and regional MPA scores (except head) between the 3 groups. Though there was no statistically significant difference between HR and HC, there was a general trend of higher mean scores in HR than in HC.
Comparison of Results of NSS and MPAs Scores in 3 Groups (Total n = 92).
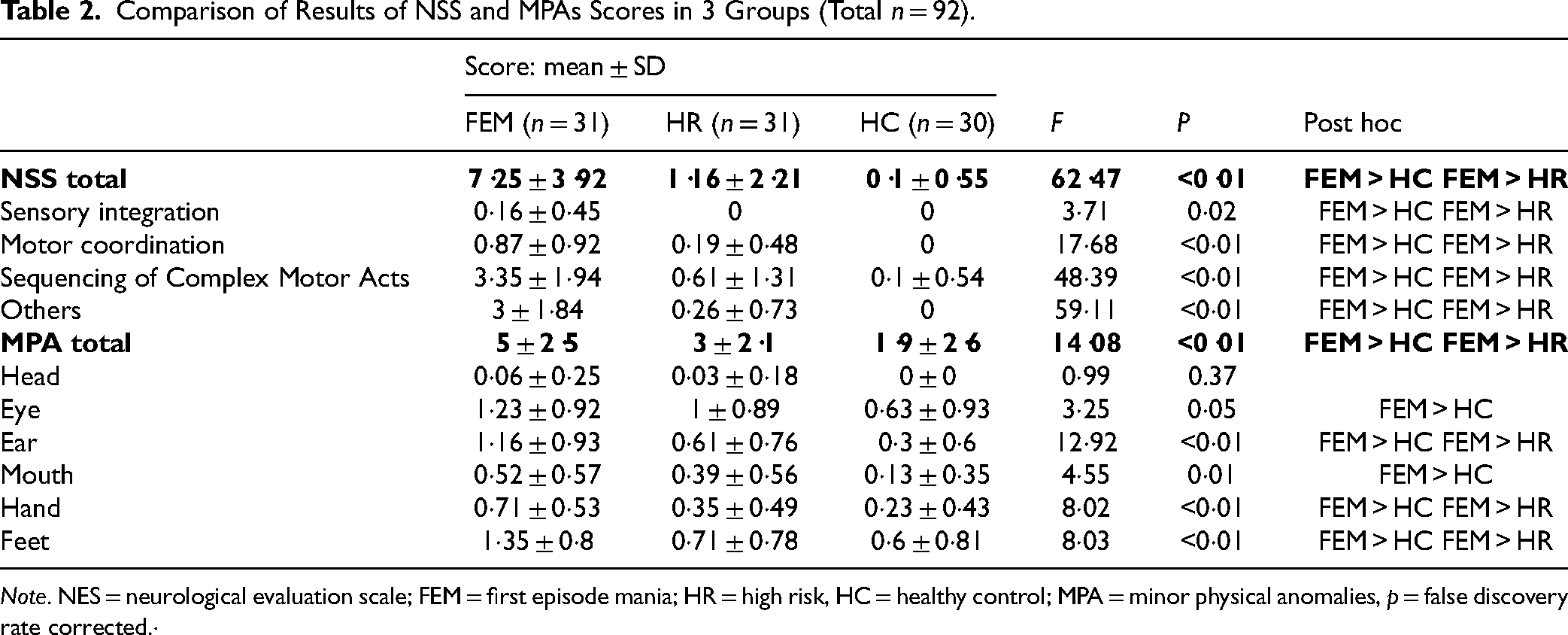
Note. NES = neurological evaluation scale; FEM = first episode mania; HR = high risk, HC = healthy control; MPA = minor physical anomalies, p = false discovery rate corrected.·
Eye-tracking was completed in 90 subjects (1 FEM and 1 HR had epiphora during the task necessitating termination of the session). AS data were excluded for 8 subjects (6 in FEM and 2 in HC group) as <5% of the total trials were analysable in them. As mentioned earlier saccades with latencies <80 ms and >600 ms after target onset were excluded from analysis 27 because these would have been either anticipations or artefacts of the subject not paying enough attention, respectively. A statistically significant difference was noted in the velocity and position gains on a combined evaluation of slow and fast SPEM trials as well as slow trials evaluated independently. Though the difference in median followed a step-ladder pattern with higher deficits in FEM followed by HR compared to HC, the post hoc analyses showed significant deficits in FEM but not HR. The results are postulated in Table 3.
Comparison of SPEM Results in Three Groups.
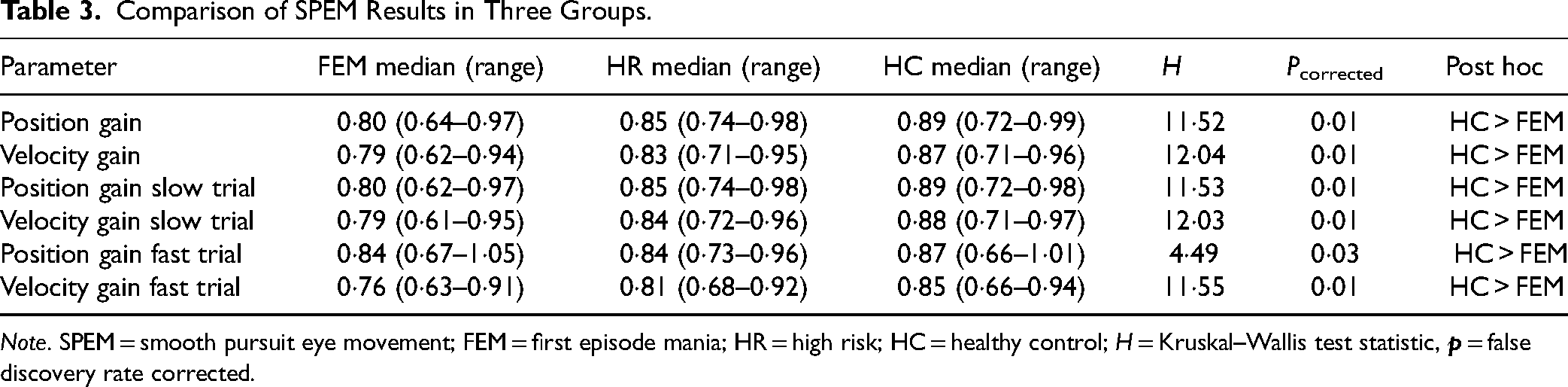
Note. SPEM = smooth pursuit eye movement; FEM = first episode mania; HR = high risk; HC = healthy control; H = Kruskal–Wallis test statistic,
Mean first correct saccade peak velocity was the only parameter in the PS task which was significantly different in the groups (H = 6.44, df = 2, p < 0.05) but post hoc pairwise comparison did not survive for any statistical difference in any of the pairs.
In AS parameters, error percentage (H = 10.66, df = 2, p = 0·05) and position error (H = 8.73, df = 2, p = 0.01) parameters were significantly different among the groups. On post hoc analysis (pairwise comparison) significant difference was found between the groups with FEM significantly higher than HC. HR had a median between the 2 groups but did not differ significantly from either. The results (PS and AS) are postulated in Table 4.
Comparison of Results of PS and AS in 3 Groups.
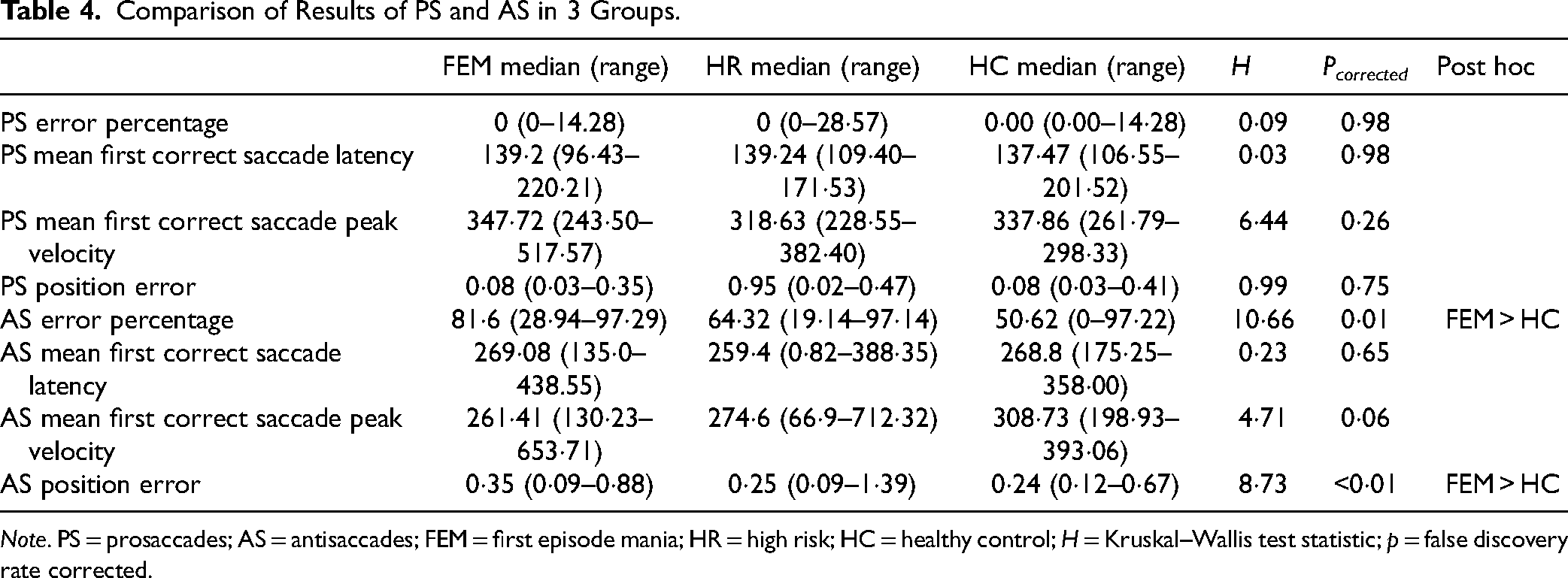
Note. PS = prosaccades; AS = antisaccades; FEM = first episode mania; HR = high risk; HC = healthy control; H = Kruskal–Wallis test statistic; p = false discovery rate corrected.
Correlational analyses using Kendall's tau_b revealed a significant positive relationship between MPA total scores and the NSS total scores (Kendall's tau_b = 0.480; p < 0.01). There were significant correlations between NSS total scores and SPEM abnormalities in overall position gain (Kendall's tau_b = 0.238; p < 0.01) and velocity gain (Kendall's tau_b = 0.248; p < 0.01). This was evident in position (Kendall's tau_b = 0.237; p < 0.01) and velocity (Kendall's tau_b = 0.253; p < 0.01) gains of slow trials but not fast trials. AS error percentage negatively correlated with SPEM measures of both slow and fast trials, that is, position gain (Kendall's tau_b = 0·235 and 0·234; p < 0·01, respectively) and velocity gain (Kendall's tau_b = 0.260 and 0.302; p < 0.01). No significant effect of antipsychotic (olanzapine equivalent dose = 11.62 + 4.32 mg/day) or lithium was noted on eye-tracking measures but a significant positive correlation was noted between olanzapine equivalent antipsychotic daily doses and NSS total score (r = 0.34, p = 0.02).
A linear discriminant function analysis was performed, and when total MPA scores were entered independently in a separate discriminant function analysis only 55.4% of the original cases were correctly classified (Wilks’ λ = 0.776; χ2 = 22.53; df = 2; p < 0·001). When the NSS total score was entered independently in a separate discriminant function analysis only 60.9% of the original cases were correctly classified (Wilks’ λ = 0.387; χ2 = 84.59; df = 2; p < 0.001). On combined NSS and MPA total scores were entered 64.1% of the original cases were correctly classified. When SPEM parameters were entered independently 48.9% of original cases were correctly classified. When AS parameters were entered independently 53.7% of original cases were correctly classified. On combined evaluation, NSS total scores, MPAs total scores, SPEM, and AS parameters accurately classified 72.0% (Wilks’ λ = 0.311; χ2 = 85.734; df = 24; p < 0.001) of the original 82 subjects (79.2% of the BD subjects, 56.70% of the HR subjects, and 82.1% of HC) were correctly classified. On the leave-one-out method of cross-validation, the classifiability remained at 59.8%.
Discussion
This is the first study to evaluate the composite endophenotype markers in BD using NSS, MPA, and oculomotor abnormalities. Impairments in all these measures were observed in FEM. This finding is of importance, particularly as these patients were in the early course of the illness and remained relatively free from confounding effects of progressive pathophysiology and lifetime cumulative treatment effects. The measurements done during remission suggest these markers are not state-dependent.
NSS, though non-specific, has been implicated to have an association with functional abnormalities in specific brain regions. 28 MPA scores were found to have a strong association with NSS scores, favours the neurodevelopmental basis of BD. 29 Previous studies have found higher MPAs in BD, 30 similar to our current findings and further supports this hypothesis. Impairments in various subscores of NSS suggest a more widespread neuroanatomical involvement in BD. Different regional MPAs are also demonstrated to be highly prevalent in BD, implicating that the neurodevelopmental insults are not restricted to any region or embryonic stage.
Eye movements are crucial for vision, attention, and memory; they determine what we see, attend to, and remember about our surroundings. They are thus central to executive functions. 31 SPEM is considered a basic eye movement necessitating a brain-wide functionally intact neural network. Pursuit abnormalities are well-recognized endophenotype markers of schizophrenia, but less well-studied in BD. 17 Deficits in position gain and velocity gain during SPEM suggest neurophysiological impairment. Similarly, the PS task depends on a neural network of saccadic eye movement, which is relatively simpler than SPEM. However, AS includes a top-down regulation of saccadic movements measuring inhibitory control. Impairment of AS with intact PS suggests deficits in inhibitory control on reflexive saccades and motion planning, a domain of a prefrontal executive function that is known to be impaired in BD. 10 Though we could demonstrate the impaired performances in both eye movement tasks, the impairment was not established in HRs. The sample size would have constrained the visibility of effects. However, these oculomotor functions had their measures of central tendencies lying between FEM and HCs, similar to NSS and MPAs. The NSS and MPAs are fairly established as endophenotypes of psychotic disorders including BD. 12 The literature on the endophenotype role of SPEM and AS in BD is inconsistent.9,32,33 The severity of oculomotor deficits had significant correlations with NSS and MPAs in the whole study sample. A shared pathophysiological process could be attributed to this strong association with an underlying neurodevelopmental origin.
In this context, the ability of these markers to discriminate subjects into different groups was evaluated. Composite functions including all 4 parameters had the highest power to classify subjects into their original group than any of these measures modelled independently. However, though there was a statistically significant finding the values came out as moderate in magnitude in terms of sensitivity and specificity in differentiating the groups. But these also increased proportionally when the markers were taken together instead of each individual marker. This suggested that the composite endophenotype may be more robust in phenotyping individuals with BD and may provide a better measure in genetic association analysis. The findings of our study add weight to some of the recent reviews which have pointed out that going ahead with combined or composite markers might give us more answers than individual candidate markers.5,6
Limitations
The study was conducted in a tertiary care institute with most patients having had psychotic symptoms during the episode which might not be reflective of the general population. Education was not matched in this cross-sectional study. However, the study parameters are free from the effects of formal education and were not correlated with the education. Thus, education is unlikely to have influenced the results. The effects of medication, in particular, antipsychotics on NSS is a potentially substantial confounder. But an absence of difference in the oculomotor parameters of FEM from HR, who are not on any medications, implies the deficits could be secondary to illness-related factors independent of medications. We would also want to point out that to diagnose BD in ICD-10, as used by us we need at least 2 mood episodes of opposite polarity whereas, in DSM a person with a single manic episode can be diagnosed with BD. But still, since the patients that are studied here are of FEM and the criteria for diagnosing mania is the same across the diagnostic systems our results are expandable to patients with DSM-based BD as well.
Implications
Investigating these biomarkers can have clinical and research implications. They will help us in the early identification of the ultra-HR individuals within the larger group of HR individuals. A closer clinical monitoring of these individuals would further support the interventions on prevention and early intervention of disease and thereby influence prognosis favourably. Thus, it can be inferred that taken together, MPAs, NSS, SPEM, and AS are robust composite risk markers of BD. It may have clinical implications in the early identification and management of illness.
Conclusion
It can be concluded that the addition of AS and SPEM could enhance the utility of NSS and MPA as the neuronal markers of BD. The presence of these abnormalities in FEM suggests their role in understanding the etiopathogenesis of BD. These composite endophenotypes may be more robust in phenotyping individuals with BD and early identification in HRs, hence need further evaluation in larger samples.
Footnotes
Data Access
This is not available as per the institute policy
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the International Society for Bipolar Disorder (ISBD) by the Samuel Gershon Junior Investigator Award 2020.
