Abstract
Objectives
Cannabis use is common in people with early-phase psychosis (EP) and is associated with worse treatment outcomes. Few targeted interventions for cannabis use behaviour in this population exist, most focusing on abstinence, none focusing on harm reduction. Many people with EP will not seek treatment for their cannabis use with current therapeutic options. Understanding preferences for cannabis-focused harm reduction interventions may be key to improving outcomes. This study aimed to determine preferences of young adults with EP who use cannabis for cannabis-focused harm reduction interventions.
Methods
Eighty-nine young adults across Canada with EP interested in reducing cannabis-related harms were recruited. An online questionnaire combining conventional survey methodology and two unique discrete choice experiments (DCEs) was administered. One DCE focused on attributes of core harm reduction interventions (DCE 1) and the second on attributes of boosters (DCE 2). We analysed these using mixed ranked-ordered logistic regression models. Preference questions using conventional survey methodology were analysed using summary statistics.
Results
Preferred characteristics for cannabis-focused harm reduction interventions (DCE 1) were: shorter sessions (60 min vs. 10 min, odds ratio (OR): 0.72; P < 0.001); less frequent sessions (daily vs. monthly, OR: 0.68; P < 0.001); shorter interventions (3 months vs. 1 month, OR: 0.80; P < 0.01); technology-based interventions (vs. in-person, OR: 1.17; P < 0.05). Preferences for post-intervention boosters (DCE 2) included opting into boosters (vs. opting out, OR: 3.53; P < 0.001) and having shorter boosters (3 months vs. 1 month, OR: 0.79; P < 0.01). Nearly half of the participants preferred to reduce cannabis use as a principal intervention goal (vs. using in less harmful ways or avoiding risky situations).
Conclusions
Further research is required to see if technology-based harm reduction interventions for cannabis featuring these preferences translate into greater engagement and improved outcomes in EP patients.
Background
Cannabis use is common in young people with early-phase psychosis (EP), with the majority admitted into specialized clinics reporting lifetime cannabis use at admission (62%) 1 and almost half (43%) having a cannabis use disorder (CUD). 2 Research shows a strong association between cannabis use in this population and worse clinical outcomes, including more positive psychotic and depressive symptoms, decreased quality of life and worse psychosocial functioning compared to people not using cannabis.2–4
Limited intervention options exist to address cannabis use for young people with EP, including prevention and CUD treatments targeting cessation or reduction; none specifically address cannabis harms.5–7 Various approaches, including cognitive behavioural therapy, contingency management, family-based therapy, motivational interviewing (MI), pharmacology or psychoeducation were applied in substance use interventions for people with psychosis, with none demonstrating clear, robust superiority in decreasing substance use.6–8 Among potential strategies to improve cannabis use-related outcomes in that population, alternative, more accessible modes of administration and approaches to extend the short-term benefits of interventions may be promising. Mobile health (e.g., web-based) psychosocial interventions have shown limited support for reducing cannabis use, yet are considered safe, accessible and low-cost intervention options.9,10 Some interventions, both for people with psychosis and for people with substance use disorders, have also integrated post-intervention booster sessions with the aim of extending intervention benefits. These have shown promising results, such as improvements in long-term outcomes relating to psychiatric symptomatology, 11 engagement12,13 and substance use.14,15
Beyond the aforementioned adaptations to existing interventions, another strategy may include exploring alternative outcomes, like harm reduction, to assess intervention effectiveness in addressing problematic cannabis use. 5 Harm reduction refers to decreasing negative consequences of a substance on the health, social and economic well-being of an individual, while not requiring abstinence; this constitutes a central pillar of the Canadian Drugs and Substances Strategy.16,17 This approach can be applied through different techniques like psychoeducation to decrease risks of cannabis use-related adverse psychosis outcomes 18 or MI to reduce risky drug use behaviours. 19 Further, decades of research support harm reduction-based interventions given their potential to improve well-being of people with persistent substance use. 16 While this approach may appeal to a wide spectrum of people with psychosis using cannabis, further research is required to establish its utility in this population.
Altogether, the existing evidence points towards the potential utility of developing new interventions for cannabis use in young people with psychosis, focusing on alternative administration modes, the addition of boosters to extend benefits and the use of more realistic, pragmatic goals centred around decreasing harms. Given limited efficacy data, guidelines recommend that individual preferences be prioritized to guide such intervention selection and design 20 ; however, preference data for cannabis-specific interventions is also scarce in this population. Preference studies have primarily focused on antipsychotic treatments or specialized early intervention services (EIS; mainstay treatment for EP in Canada).21–23 Our team conducted a qualitative study of clinicians and individuals with EP and CUD, revealing that (a) harm reduction interventions were recommended for those unwilling to stop use and (b) intervention preferences varied among individuals. 24 Clinicians suggested tailoring interventions to individuals’ needs and motivations and recommended boosters to ensure sustained benefits. Further, this study emphasized a need for thorough assessment of intervention preferences in this population, including specific parameters of intervention modalities. This knowledge gap led to the present study, which aims to determine preferences of people with EP who use cannabis for cannabis-specific harm reduction interventions, using the Survey of Patient Interests for Cannabis E-interventions (SPICE).
Materials and methods
Design
The SPICE survey was for people with EP who used cannabis and wanted to reduce their cannabis-related harms. It was administered online using Research Electronic Data Capture (REDCap). 25 It included two unique discrete choice experiments (DCEs) and a questionnaire. DCEs, standard in preference assessment studies, can quantify the relative importance of intervention characteristics and provide preference information while protecting against social desirability bias. DCEs are feasible for people with psychosis,21–23 though they have not been conducted for cannabis-focused harm reduction interventions.
Survey
The SPICE survey collected information on sociodemographic characteristics; clinical and substance use history; acceptability and preferences for cannabis-specific harm reduction interventions (non-DCE); and intervention and booster preferences (DCEs) (Supplemental Table S1).
Survey development was informed through: (a) our systematic review of cannabis-focused preventive interventions in youth with EP 5 ; (b) our qualitative study on cannabis-focused treatments 24 ; and (c) consultation with experts in cannabis-related harms and psychosis (e.g., clinicians, people with lived experience).
Typically, DCEs ask respondents to choose their preference among hypothetical alternatives presented in choice sets and described by pre-determined characteristics (i.e., attributes) with specific sub-categories (i.e., levels). Initially, a list of characteristics was created based on the aforementioned studies, and the most relevant were selected as DCE attributes. We used experimental design techniques to combine DCE attributes into choice sets. A fractional factorial design was generated using the SAS software, 26 encompassing all attribute level combinations for testing within a choice set. We established our experimental design based on the D-efficiency approach, which retains optimal orthogonality in a fractional design and excludes unrealistic level combinations. 27 This design facilitated estimation of main effects and interaction effects between the attributes.
DCE 1 tested intervention preferences for session length; session frequency; intervention duration; delivery mode. DCE 2 evaluated preferences for booster participation and booster delivery mode; frequency; duration (Supplemental Figures S1 and S2). Each DCE had 18 choice sets, divided into 2 blocks of 9 uncorrelated sets. For DCE 1, choice sets had three alternatives. DCE 2 choice sets also had three alternatives – two hypothetical alternatives and one opt-out alternative. These numbers were chosen to balance information gain and participant cognitive burden of assessing multiple alternatives. 28 The DCEs employed the “best-best” approach, where participants chose their most and second most preferred alternative in the choice set, yielding additional preference information from reduced choice sets and resulting in statistical efficiency gains. 29 This is advantageous for small populations. 30
To ensure comprehension, survey questions were reviewed by patient partners and adjusted accordingly; it was then pilot-tested with four individuals with EP. Items were revised based on feedback; data was not analysed. Technical term definitions (e.g., “MI”) were provided throughout the survey via pop-up boxes and visual representations for DCEs were provided before starting DCEs (Supplemental Figure S3).
Participants
Participants were recruited from eight EIS across diverse clinical and sociogeographic realities (e.g., big cities, rural areas), in three Canadian provinces: Quebec (6 sites), Nova Scotia (1) and Alberta (1). The study was approved by the Research Ethics Boards at the Centre Hospitalier de l’Université de Montréal (#19.233), Nova Scotia Health (#1026417) and University of Calgary (#REB20-2227). Participants were included if they were aged 18–35 years; had a psychotic disorder (e.g., mood disorders with psychotic features, substance-induced psychotic disorders) diagnosed by a psychiatrist; were attending a first-episode psychosis clinic for minimum 2 months; used cannabis in the last 30 days; and wanted to change their cannabis use-related practices. They had to be able to complete a computer-based survey; provide informed consent; comply with study procedures and read French or English. Participants were excluded if there was a presence of psychiatric, physical or cognitive conditions that would preclude participation (e.g., acute suicidal ideation, intellectual disability, severe visual impairment). While reducing use is a harm reduction strategy, we hypothesized that intervention preferences would differ between those having CUD (diagnosed by psychiatrist using DSM-V criteria) and being open to CUD treatment and those unwilling to treat their CUD. Participants were thus excluded if they had CUD and were seeking CUD treatment, and were redirected to a related CUD treatment preference survey.
Procedures
Recruitment and data collection occurred between March 2020 and August 2022. Sites employed recruitment methods compliant with local ethics standards (i.e., poster advertisements, physician referrals). Clinicians screened and referred potentially interested individuals to research staff, who approached them, explained the study and requested written informed consent from interested individuals. To minimize respondent burden and promote response efficiency, participants were randomized to one of two blocks of pre-specified survey sequences using real-time randomization. 31 A computer-generated randomization schedule stratified by site used random block sizes of two to enforce balance between groups. 32 Participants completed surveys at the clinic with study staff assistance or outside the clinic using their own devices.
Sample Size
Sample size estimations were based on the parametric approach for DCE designs. 33 For DCE 1, the required minimum sample size was 92 respondents, with a statistical power of 80% and assuming (a) two sides α = 0.05 and (b) the design matrix described above, where all levels were significantly different from the reference levels with the initial beta parameter belief values of at least Cohen's d = 0.4 for all attributes. For DCE 2, the minimum sample size was 90 respondents with a statistical power of 80% and assuming (a) two sides α = 0.05 for the main effect session duration, (b) two sides α = 0.10 for the interaction of delivery mode × session frequency, and (c) the design matrix described above, where all levels were significantly different from the reference levels with initial beta parameter belief values of at least Cohen's d = 0.3 and d = 0.5 for the main attribute levels and the interaction levels, respectively. Expecting 5% attrition rate, we aimed to enrol approximately 97 participants; this was based on surveys of people with psychosis that reported 0% attrition rates post-consent.34,35
Statistical Analysis
The primary study outcomes were (a) patient preferences for the four DCE 1 attributes and (b) patient preferences for the three DCE 2 attributes or opt-out option. Non-DCE sections were analysed using frequencies for categorical variables and mean and standard deviation for continuous variables. Mixed rank-ordered logit (MROL) models were estimated for DCEs.36,37 These models accounted for the first- and second-best alternatives as the outcome and main preference attributes (both DCEs) and opt-out option (DCE 2 only) as the independent variables. We explored predefined interactions between preference attributes: session length × session frequency, delivery mode × session frequency, and intervention duration × session frequency for DCE 1 and delivery mode × booster frequency, and booster frequency × booster duration for DCE 2. We also explored predefined interactions between attributes and participant characteristics (CUD status, recent cannabis use, gender, education level, occupation status). For simplicity, we categorized as follows: CUD status: has CUD, no CUD; recent cannabis use frequency: high-risk use (i.e., using minimum five days per week), low-risk use (i.e., using maximum four days per week); gender: men, women, gender-diverse people; education level: high school diploma or less, more than high school diploma; occupation status: full-time occupation, no full-time occupation.
Sensitivity analyses using stratification verified whether speeders, language (i.e., English vs. French), or location (i.e., Quebec vs. other provinces) affected responses. Speeders were defined as participants taking 50% or less of the median time spent on DCE 1, 38 measured using REDCap's response duration tracking. All data with at least one answer on either DCE was included in the analysis. Imputation was not performed. DCE results were presented in tabulated (odds ratios (OR) and 95% confidence intervals (CI)) and graphical forms (predictive margins plots with 95% CI for interactions). 36 DCE tests were performed at two sided 0.05 significance level and used Stata version 16.0 39 for calculations.
Results
Sample
Clinicians approached 493 participants and 89 were analysed (Figure 1); 69 were from Quebec, 17 from Nova Scotia, and 3 from Alberta. One participant did not answer three DCE 1 questions or any DCE 2 questions, resulting in 2394 DCE 1 and 2376 DCE 2 observations. Table 1 presents sample characteristics.
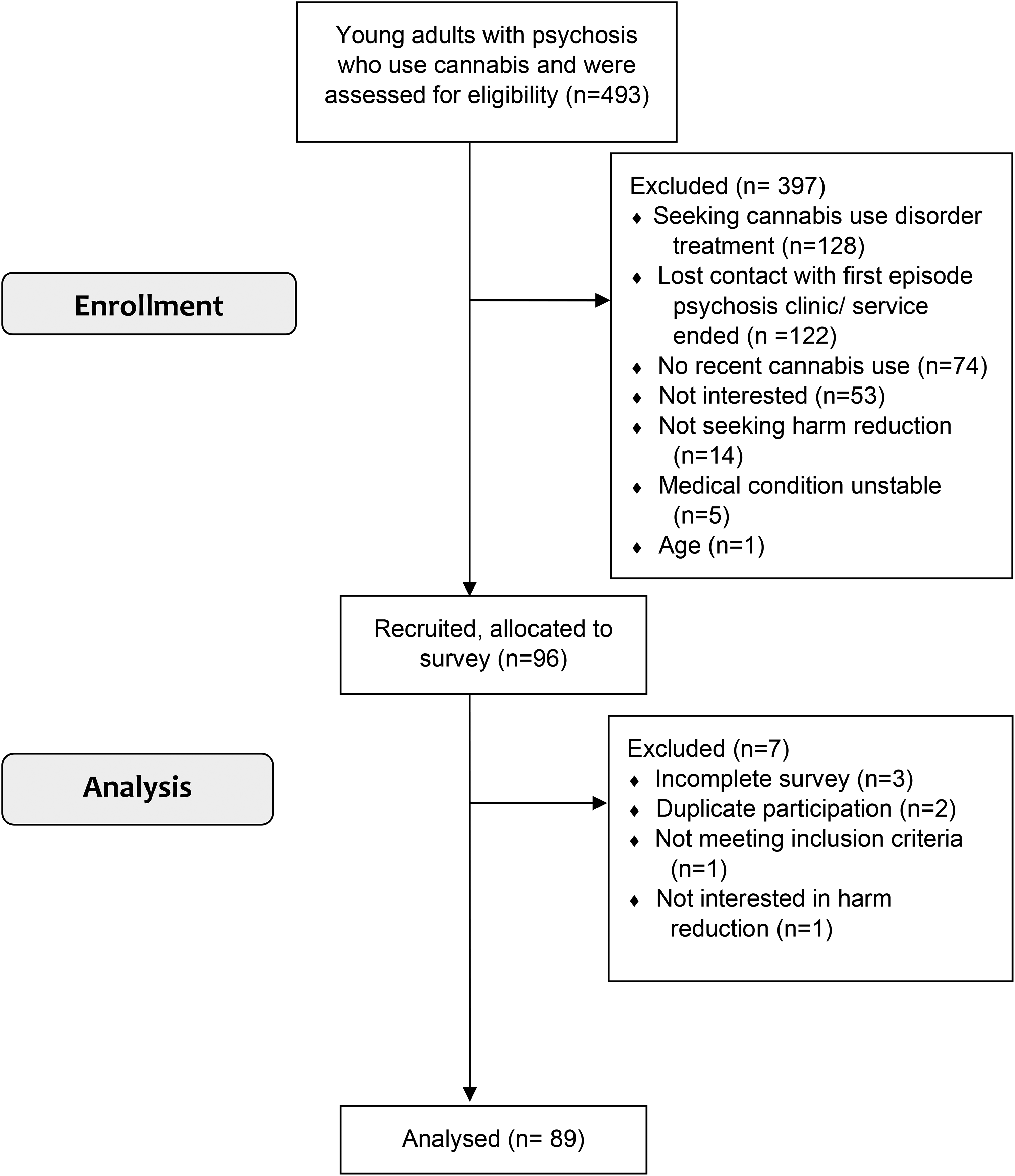
Flow diagram of SPICE recruitment.
Participant Characteristics.

Note. n = number; SD = standard deviation.
Some participants did not answer question.
Acceptability and Preferences
Table 2 presents the results on acceptability and preferences for cannabis harm reduction interventions (non-DCE survey). Almost half of the participants (44%) preferred to reduce cannabis use as a principal intervention aim. Participants preferred an intervention using cognitive-behavioural therapy (29%), MI (28%) or brief psychoeducation (21%). Most (83%) viewed technology-based interventions as acceptable, with 62% preferring smartphone-based interventions.
Acceptability and Preferences of Participants.

Primary Outcomes
Table 3 presents the results of the MROL models for DCEs 1 and 2.
Estimated Rank-ordered Logistic Regression Models for DCE 1 (Intervention) and DCE 2 (Booster session).
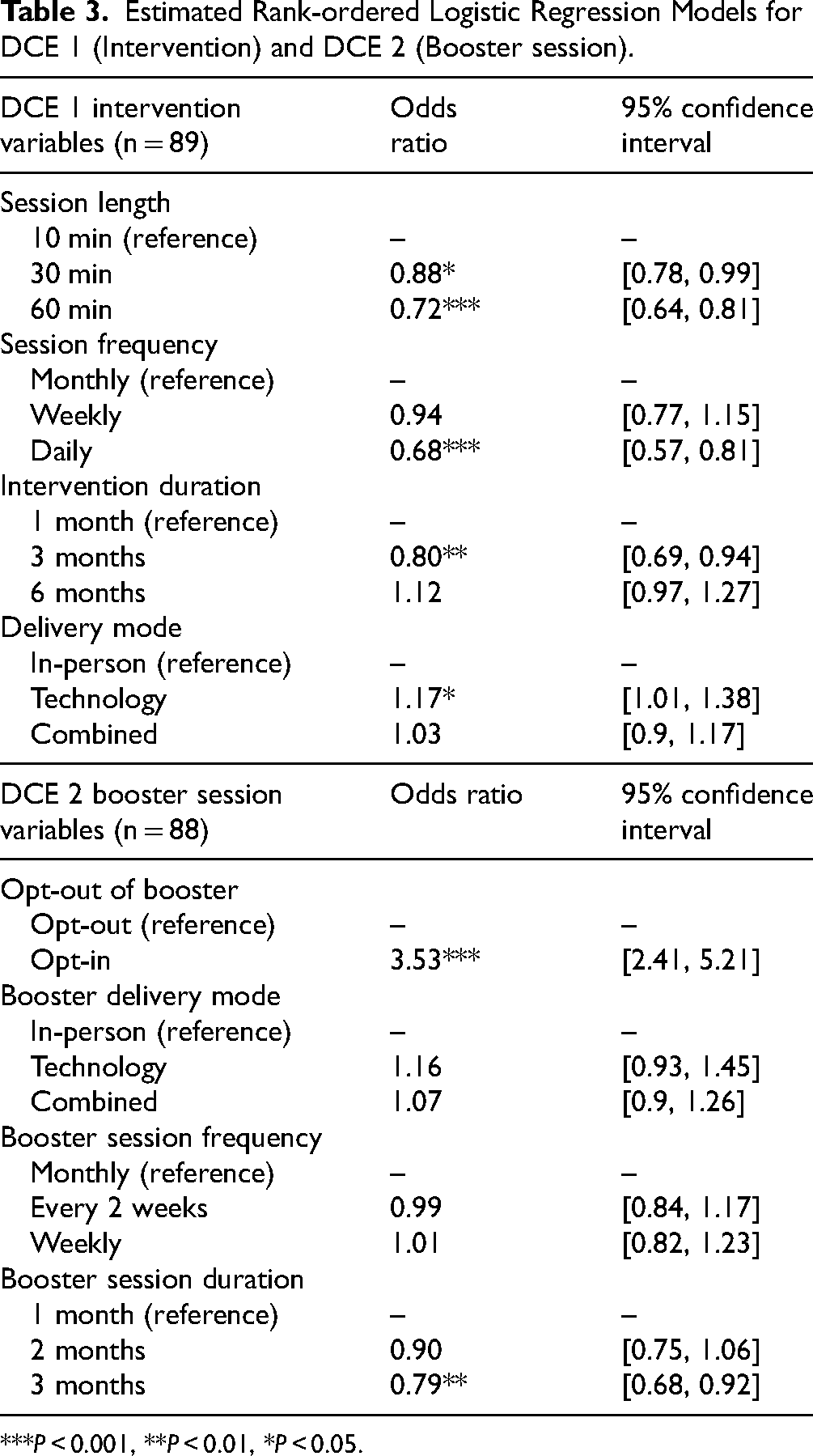
***P < 0.001, **P < 0.01, *P < 0.05.
Interventions
Participants had less preference for (a) daily over monthly sessions (OR = 0.68; 95% CI: 0.57, 0.81), (b) 30 or 60-min sessions compared to 10-min sessions (OR = 0.88; 95% CI: 0.78, 0.99 and OR = 0.72; 95% CI: 0.64, 0.81 respectively), and (c) 3-month over 1-month intervention duration (OR = 0.80; 95% CI: 0.69, 0.94). Participants had greater preference for technology-based interventions compared to in-person interventions (OR = 1.17; 95% CI:1.01, 1.38).
Boosters
Two attributes were significantly preferred by participants: opt-in and booster duration. Individuals preferred participating rather than opting out of boosters (OR = 3.53; 95% CI:2.41, 5.21) and they had less preference for 3-month compared to 1-month boosters (OR = 0.79; 95% CI:0.68, 0.92).
Exploratory Outcomes
Interventions
Two interactions resulted in significant differences in preferences: session length × session frequency and session frequency × delivery mode. Visual inspection of predictive margins plots suggested that the most preferred combinations were interventions with weekly 10-min sessions or monthly 30-min sessions, and the least preferred option included daily 60-min sessions (Supplemental Figure S4A). The predictive margins plot suggested the most preferred combinations were weekly or monthly technology-based interventions (Supplemental Figure S4B). The least preferred options were those with daily sessions, whether technology-based or in-person. Other interactions were not significant (data not shown).
Gender significantly influenced attribute preferences. The predictive margins plot suggested that men had no delivery mode preferences, while women and gender-diverse people had stronger preferences for technology-based interventions (Supplemental Figure S4C). The predictive margins plot (Supplemental Figure S4D) suggested that regardless of gender, participants preferred monthly to daily sessions, though gender-diverse people had greater preference for this. Other interactions of participant characteristics were not significant (data not shown).
Boosters
Interacting booster frequency with duration resulted in significantly different preferences. Visual inspection of predictive margins plot (Supplemental Figure S5A) suggested that the most preferred combination was a booster occurring bi-weekly for 1 month. The least preferred combinations were boosters occurring bi-weekly and lasting 2 months or occurring monthly and lasting 3 months. Education significantly influenced delivery mode preferences. Visual inspection of predictive margins plot (Supplemental Figure S5B) suggested that participants with higher education levels preferred technology-based interventions. Other interactions of participant characteristics were not significant (data not shown).
Sensitivity Analyses
The median time spent on DCEs 1 and 2 was 6 and 3 min, respectively. Removing speeders (n = 11) from DCEs did not change preferences. Following visual inspections of DCE sensitivity analyses, we ran additional analyses that did not find significant preference differences based on interaction of attributes and province or language in either DCE (data not shown).
Discussion
This study is the first to assess preferences for cannabis-specific harm reduction interventions in people with EP who use cannabis. Results suggested that participants were more likely to choose an intervention if it: featured shorter sessions (e.g., 10 min), less frequent sessions (e.g., monthly), shorter intervention durations (e.g., 1 month) and was technology-based. Participants strongly preferred participating in a shorter booster (e.g., 1 month). Individual preference differences were identified, with women, gender-diverse people and people with higher education levels preferring technology-based interventions.
The findings from the DCEs and the non-DCE questionnaire were congruent, indicating a proclivity for shorter interventions. While patients preferred the briefest intervention options, it may be that interventions that effectively promote change may require more than a single 10-min session. Likewise, an intervention of low efficacy which integrates population preferences into its design may be moderately effective due to other factors (e.g., appeal, engagement). The evidence regarding optimal duration and its relationship to treatment efficacy remains unclear. There is evidence supporting long- over short-duration cannabis interventions (i.e., >4 sessions vs. <4 sessions) in people with CUD. 40 Preliminary evidence supports brief psychosocial interventions (e.g., 7 weekly sessions) for people with psychosis on other clinical outcomes41,42 and for brief cannabis-specific interventions (e.g., single session) in the general population. 43 A review of MI interventions for individuals with dual diagnoses did not find an association between treatment length and efficacy for substance use. 44 Our results raise the question of whether shorter interventions would translate into clinical benefits (e.g., efficacy/effectiveness), underscoring the need for thorough evaluation (including acceptability, feasibility and clinical effectiveness) before implementation. It also highlights the challenge of striking a careful balance between patient preferences (which may contribute to intervention acceptability) and what is recommended based on efficacy data.
This study highlighted a strong preference for technology-based harm reduction interventions, consistent with prior research indicating openness to technology-based services among people with EP.34,45 The potential of technology-based interventions as an alternative to in-person interventions has been documented. 46 Whether this translates to intervention adherence or efficiency remains to be studied. Considering the scarcity of cannabis-specific interventions, the challenge of engagement, and the potential for technology-based interventions to enhance well-being in individuals with EP, 47 developing cannabis-focused, technology-based interventions offer an opportunity to enhance health services. While over 60 technology-based interventions exist for individuals with psychosis, none focus on managing cannabis use, and high-quality clinical trials evaluating efficacy are lacking. 47 More research on technology-based interventions specifically addressing cannabis use and their implementation in clinical settings is needed.
Nearly half of the participants indicated their preferred primary goal for a harm reduction intervention as reducing use, rather than using cannabis in less harmful ways or avoiding risky cannabis-related situations. It is worth noting that half of our sample had a CUD but did not want CUD treatment, instead preferring harm reduction interventions. This seeming discrepancy is also documented in the MI literature, recognizing that a participant's motivation to change (e.g., their stage of change) may not directly translate into actionable change. Consequently, motivational interventions are recommended to bridge the gap between contemplation and commitment. 48 Our findings underscore the pertinence of developing harm reduction interventions that meet participants at their stage of change, and that provide built-in flexibility regarding cannabis goals, including proposing diverse cannabis-related objectives. As the question on preferred primary goal was non-DCE data, another possible explanation may relate to social desirability bias: participants may have chosen the more “appropriate” response, which aligned with the commonly-emphasized goal in clinical care of cannabis cessation or reduction.
Previously, DCEs have been conducted to quantify preferences and identify segments of people expressing distinct preferences.23,49 While not designed for this purpose, our results too suggest differences in preferences based on certain characteristics; future research could explore whether specific profiles correspond to particular preferences. DCE 1 suggested that technology-based interventions were specifically preferred by women and gender-diverse people; DCE 2 suggested education-based differences in preferences. The non-DCE questionnaire results suggested varied preferences for therapeutic approaches, with participants split between cognitive-behavioral therapy, MI and brief psychoeducation interventions. Another study also noted gender differences in technology use, finding women with EP more likely to use smartphones, while men preferred phones without internet. 50 Collectively, findings of preference differences highlight the need for further research to explore factors influencing the receptiveness and effectiveness of harm reduction interventions among subgroups of participants and/or the need to offer diverse interventions to better personalize treatment.
Limitations
There are several limitations to consider. First, despite consulting relevant literature and experts when selecting attributes, we may have missed the evaluation of important intervention features (e.g., clinician involvement). Second, the absence of a DCE 1 opt-in option precluded quantitative analysis of preference to participate in a harm reduction intervention. However, our eligibility criteria included a desire to modify cannabis-related practices, indicating participant interest in such an intervention. This, and our aim to simplify the DCE, is why we excluded the opt-in option from DCE 1. Third, our DCEs may reflect preferences beyond harm reduction interventions, given broadly labelled attributes. However, explicit instructions and preceding survey questions aimed to frame responses within the context of harm reduction interventions and to guide participants to express preferences specifically for such interventions. Fourth, preference differences relating to CUD status may have gone undetected in the non-DCE sections, potentially influencing our findings. While we conducted sensitivity analysis based on CUD status in both DCEs and did not find significant differences within these two groups, we were not powered to do this for non-DCE sections. Fifth, visual representations in our DCEs may have inadvertently affected stated preferences. For example, portraying “in person” (DCE 1) with a person wearing a white coat might have triggered anxiogenic responses, considering the white-coat effect research indicating elevated blood pressure in reaction to medical settings. 51 Finally, the study was conducted during the pandemic and in Canada, where cannabis is legalized, and recruitment difficulties leading to a primarily Quebec-based sample; this limits generalization of results to other populations.
Conclusion
Our study highlights preferences for brief, technology-based cannabis harm reduction interventions. These findings may provide key information to guide the development of well-adapted interventions for adults with psychosis using cannabis. In line with the harm reduction principles of meeting participants where they are at, it is important that future cannabis harm reduction interventions remain flexible and tailorable to the participants’ needs and desired goals, whether that involves use reduction or harm reduction. An accelerated research effort is needed to develop, evaluate and implement such approaches for young people with EP.
Supplemental Material
sj-docx-1-cpa-10.1177_07067437241242395 - Supplemental material for Preferences of Young Adults With Psychosis for Cannabis-Focused Harm Reduction Interventions: A Cross-Sectional Study: Préférences des jeunes adultes souffrant de psychose pour les interventions de réduction des méfaits axées sur le cannabis : une étude transversale
Supplemental material, sj-docx-1-cpa-10.1177_07067437241242395 for Preferences of Young Adults With Psychosis for Cannabis-Focused Harm Reduction Interventions: A Cross-Sectional Study: Préférences des jeunes adultes souffrant de psychose pour les interventions de réduction des méfaits axées sur le cannabis : une étude transversale by Stephanie Coronado-Montoya, Amal Abdel-Baki, David Crockford, José Côté, Simon Dubreucq, Alina Dyachenko, Benedikt Fischer, Tania Lecomte, Sophie L’Heureux, Clairélaine Ouellet-Plamondon, Marc-André Roy, Philip Tibbo, Marie Villeneuve and Didier Jutras-Aswad in The Canadian Journal of Psychiatry
Footnotes
Author Contributions
SCM wrote the initial manuscript draft, tables and figures. SCM, AAB, DC, JC, SD, AD, BF, TL, SH, COP, MR, PT, MV and DJA contributed to the study conceptualization, critically reviewed and edited the manuscript. AAB, DC, SD, TL, SH, COP, MR, PT, MV and DJA contributed to the data acquisition. SCM and AD conducted formal analyses. DJA obtained funding and supervised the study development and conduct. All authors approved the manuscript.
Data Availability
The data that support the findings of this study as well as the analytic code and research material are available from the corresponding author, DJA, upon reasonable request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BF has held research grants and contracts in the areas of substance use, health and policy from public funding and government-related agency (i.e., public-only) organizations and has consulted as a scientific expert with such entities in different countries. DJA received investigational products (2022-23) from Cardiol Therapeutics for a clinical trial funded by Quebec Ministry of Health and Social Services.
Ethics Statement
This study was performed in accordance with the Declaration of Helsinki. This human study was approved by Comité d’éthique de la recherche du Centre hospitalier de l’Université de Montréal – approval: 19.233; Nova Scotia Health – approval: 1026417 and the University of Calgary – approval REB20-2227. All adult participants provided written informed consent to participate in this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Quebec Ministry of Health and Social Services (Public Health Division). Authors would like to acknowledge support from Fonds de Recherche du Québec en Santé (DJA), University Institute on Addictions (SCM), and Vanier Canada Graduate Scholarship (SCM). The funding sources had no input in the study design, data analysis or interpretation of findings. Further, the authors would like to acknowledge the significant contribution of Marie-Claude Durpès in facilitating the execution of the SPICE study. They would also like to extend gratitude to Jill Fikowski and the Changemark team, as well as Laurie Argourd, Anita Abboud, Alice Gauchet, and Pamela Lachance-Touchette for their ongoing support of this study.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
