Abstract
Background:
Individuals with intellectual disability (ID) and autism spectrum disorder (ASD) often receive psychotropic medications such as antipsychotics and antidepressants to treat aberrant behaviors and mood symptoms, frequently resulting in polypharmacy and drug-related adverse effects. Pharmacogenomic (PGx) studies with ASD and/or ID (ASD/ID) have been scarce despite the promise of optimizing treatment outcomes. We reviewed the literature on PGx studies with antipsychotics and antidepressants (e.g., treatment response and adverse effects) in ASD/ID.
Methods:
We performed a systematic review using MEDLINE, Embase, and PsycINFO, including peer-reviewed original articles in English referring to PGx in the treatment of ASD/ID in any age groups (e.g., treatment response and adverse effects).
Results:
A total of 28 PGx studies using mostly candidate gene approaches were identified across age groups. Notably, only 3 studies included adults with ASD/ID while the other 25 studies focused specifically on children/adolescents with ASD/ID. Twelve studies primarily investigated treatment response, of which 5 and 6 studies included patients treated with antipsychotics and antidepressants, respectively. Most interesting results for response were reported for 2 sets of candidate gene studies, namely: (1) The DRD3 Ser9Gly (rs6280) polymorphism was examined in patients treated with risperidone in 3 studies, 2 of which reported an association with risperidone treatment response and (2) the SLC6A4 5-HTTLPR polymorphism and treatment response to antidepressants which was investigated in 4 studies, 3 of which reported significant associations. In regard to side effects, 9 of 15 studies focused on hyperprolactinemia in patients treated with risperidone. Among them, 7 and 5 studies examined the impact of CYP2D6 and DRD2 Taq1A polymorphisms, respectively, yielding mostly negative study findings.
Conclusions:
There is limited data available on PGx in individuals with ASD/ID and in particular in adults. Given the potential for PGx testing in improving treatment outcomes, additional PGx studies for psychotropic treatment in ASD/ID across age groups are warranted.
Introduction
It is estimated that 1% to 2% of the population are affected by either intellectual disability (ID) or autism spectrum disorder (ASD), 1 –3 which are neurodevelopmental disorders according to the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders. 4 Children/adolescents with ASD and/or ID (i.e., ASD/ID for short) often present with problem behaviors including aggression toward others, self-injurious behavior, and disruptive behavior. 5,6 Such challenging behaviors often persist into adulthood. 7 –9 In addition, individuals with ASD/ID have higher rates of comorbid psychiatric disorders than other individuals; approximately 30% of individuals with ID 10 and 70% with ASD have comorbid psychiatric disorders. 11,12
High rates of use of psychotropic medications for children/adolescents and adults with ASD/ID have been reported in many countries. 13 –21 Notably, psychotropic medication use increases with age 22,23 and is highest in individuals with both ASD and ID. 24 Furthermore, polypharmacy and excessive dosages are common in children/adolescents and adults with ASD/ID. 22,25,26 Polypharmacy is frequent in these populations, and rates among individuals with ID have been reported between 11% and 60%, depending on the study design and sample size. 22 Likewise, a recent systematic review reported that the rate of psychotropic polypharmacy in individuals with ASD was estimated between 5.4% and 54%. 25 In general, polypharmacy and high doses are commonly associated with increased adverse effects, medication nonadherence, functional decline, and cognitive impairment, in addition to increased health-care costs. 27 Furthermore, polypharmacy is associated with a highly increased risk for drug–drug interactions typically occurring at the pharmacokinetic level, that is, the Phase-I cytochrome P450 enzymes. 28 Individuals with ID have been reported to be more susceptible to movement side effects of antipsychotic medications than those without ID. 29
Pharmacogenomics (PGx) enables us the opportunity to remedy these treatment inadequacies in individuals with ASD/ID. In general, PGx represents a decision support tool based on well-established gene–drug interactions. 30 Such gene–drug interactions depend on interindividual variability in human DNA sequence, which can determine plasma levels of medications and metabolites and thereby tolerance and response to medications. For antidepressants and antipsychotics medications, which are predominantly metabolized by CYP2C19 and CYP2D6, assessing the genetic variation of these enzymes has enabled researchers and clinicians to estimate their activities; this strongly correlates with exposure to medications (i.e., parent compound and metabolites) and affects treatment outcome for depression and psychotic disorders, respectively. 31,32 For various nonpsychiatric medications, clinical utility of PGx testing compared to treatment as usual has been demonstrated resulting in a reduction of hospitalization rates, health-care costs, and polypharmacy. 33 –35 In psychiatry, favorable treatment outcomes (e.g., higher remission rates) have also been observed in patients receiving PGx-guided antidepressant and antipsychotic treatments compared to those receiving treatment as usual. 36 –41 Therefore, PGx testing is globally becoming increasingly implemented, which is further encouraged by expert recommendation guidelines for psychiatric medications. 31,42 –44
Taken together, in addition to avoiding polypharmacy, PGx testing could be extremely useful for optimizing pharmacological treatment in individuals with ASD/ID by optimizing likelihood for treatment response and minimizing risk for adverse events. However, to the best of our knowledge, no study has systematically reviewed the clinical utility of PGx testing in individuals with ASD or ID, and no reviews have focused specifically on particular in adults. While there is a literature review using only 1 search engine (i.e., PubMed) that focused on PGx studies in ASD, there was no discussion regarding the age of the participants. 45 We aimed to review the literature on PGx studies with psychotropic drugs including antipsychotics and antidepressants (e.g., treatment response and adverse effects) in individuals with ASD/ID across all age groups (i.e., adults and children/adolescents).
Methods
Literature Search
We have followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 46 The search was performed with MEDLINE, Embase, and PsycINFO until February 29, 2020. The following search terms were applied: (neuroleptic* OR antipsychotic* OR amisulpride OR aripiprazole OR chlorpromazine OR fluphenazine OR haloperidol OR olanzapine OR quetiapine OR risperidone OR thioridazine OR ziprasidone OR antidepressant* OR SNRI OR SSRI OR citalopram OR duloxetine OR escitalopram OR fluoxetine OR fluvoxamine OR mirtazapine OR paroxetine OR sertraline OR venlafaxine OR “alpha agonist” OR stimulant* OR atomoxetine OR clonidine OR guanfacine OR methylphenidate OR benzodiazepine* OR “mood stabilizer*” OR valproate) and (variant* OR polymorphism* OR gene OR genetic OR genetics OR pharmacogenetic OR pharmacogenetics OR pharmacogenomic OR pharmacogenomics) and (autism OR ASD OR “Intellectual* disab*” OR “Intellectual* impair*” OR “Intellectual* retard*” OR “Intellectual* handicap*” OR “Intellectual* subnormal*” OR “Intellectual* deficien*” OR “Learning disab*” OR “Learning impair*” OR “Learning retard*” OR “Learning handicap*” OR “Learning subnormal*” OR “Learning deficien*” OR “Mental* disab*” OR “Mental* impair*” OR “Mental* retard*” OR “Mental* handicap*” OR “Mental* subnormal*” OR “Mental* deficien*” OR “Developmental* disab*” OR “Developmental* impair*” OR “Developmental* retard*” OR “Developmental* handicap*” OR “Developmental* subnormal*” OR “Developmental* deficien*” OR “Neurodevelopmental* disab*” OR “Neurodevelopmental* impair*” OR “Neurodevelopmental* retard*” OR “Neurodevelopmental* handicap*” OR “Neurodevelopmental* subnormal*” OR “Neurodevelopmental* deficien*” OR “down syndrome” OR “Fragile X Syndrome” OR “Prader-Willi Syndrome” OR “Smith-Magenis Syndrome” OR “22q11.2 Deletion Syndrome” OR “15q13.3 Deletion Syndrome”). Limit was set for “English language” and “humans.” References of relevant articles were manually searched and an additional hand search was performed using available citations by 2 authors (K.Y. and E.K.) independently. Candidate articles were independently screened and scrutinized by these authors. Discrepancies in study selection were resolved by discussion between them.
Inclusion Criteria
Studies were included if (1) they were peer-reviewed original articles; (2) they investigated the association between any gene variants and serum/plasma concentrations or dosages of any psychotropics, treatment response to any psychotropics, and adverse effects of any psychotropics; and (3) they were published in English until February 29, 2020. In addition, we included PGx studies meeting the inclusion criteria above across all age groups in order to perform the literature search as comprehensively as possible.
Data Extraction
The following data were extracted by the 2 authors (K.Y. and E.K.) independently for each study: author name, year of publication, diagnosis, age, presence/absence of ID, gene(s) and polymorphism(s), outcomes, study design, sample size, ethnicity or nationality, treatment duration, treatment medication, and main findings.
Results
Twenty-eight studies were identified through the literature search (Figure 1). Identified studies were summarized in Table 1. We summarized those studies based on age category (i.e., adults and children/adolescents) and treatment outcomes, respectively.
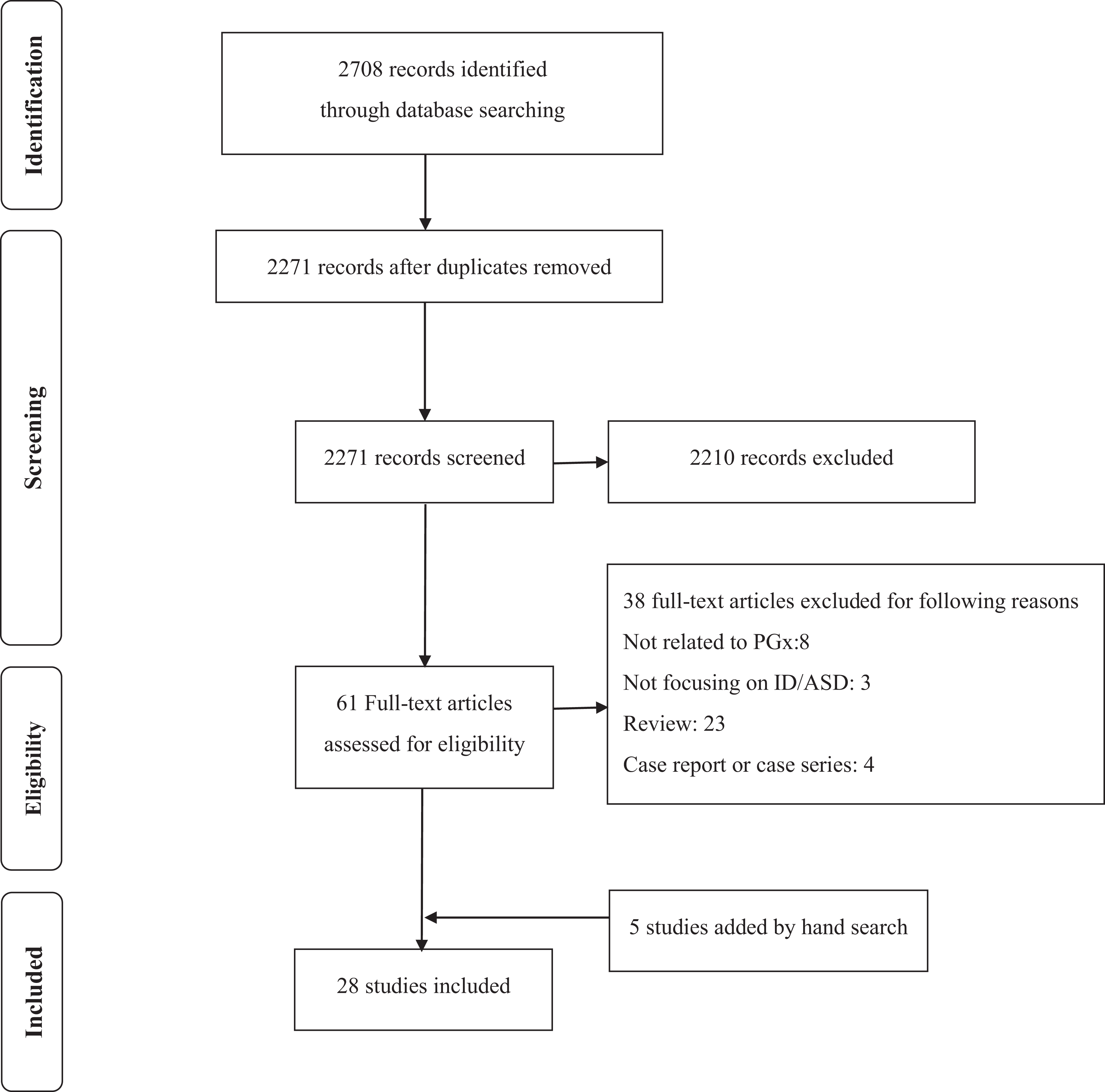
Preferred Reporting Items for Systematic Reviews and Meta-Analyses diagram for review eligibility and inclusion. Note. ASD = autism spectrum disorder; ID = intellectual disabilities; PGx = pharmacogenomics.
Association between Genetic Polymorphisms and Treatment Outcomes.
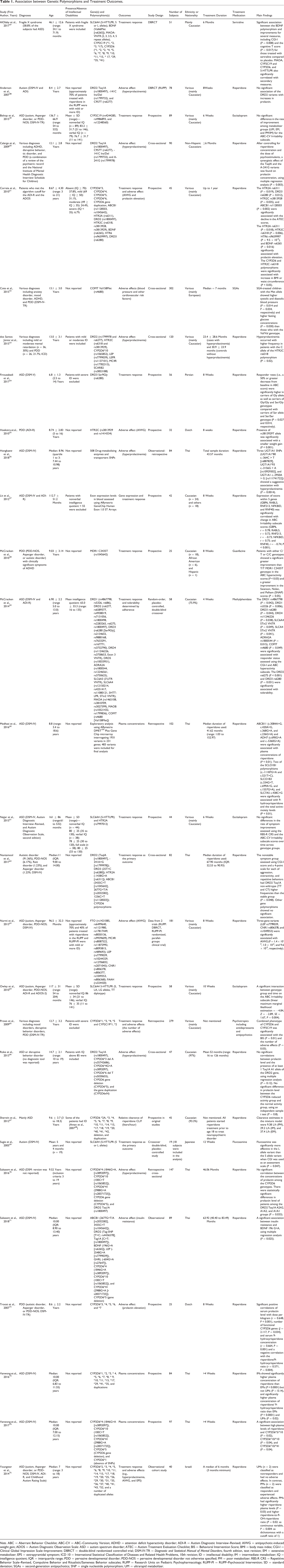
Note. ABC = Aberrant Behavior Checklist; ABC-CV = ABC–Community Version; ADHD = attention deficit hyperactivity disorder; ADI-R = Autism Diagnostic Interview–Revised; AIWG = antipsychotic-induced weight gain; ADOS = Autism Diagnostic Observation Scale: ASD = autism spectrum disorder; ATEC = Autism Treatment Evaluation Checklist; BIS = Behavioral Intervention Score; BMI = body mass index; CGI-I = Clinical Global Impression Scale–Improvement; DBRCT = double-blind randomized controlled trial; DSM-IV-TR = Diagnostic and Statistical Manual of Mental Disorders, fourth edition, text revision; EM = extensive metabolizer; EPS = extrapyramidal symptom; ICD 10 = International Statistical Classification of Diseases and Related Health Problems, 10th revision; ID = intellectual disability; IM = intermediate metabolizer; IQ = intelligence quotient; IQR = interquartile range; PDD = pervasive developmental disorder; PDD-NOS = pervasive developmental disorder not otherwise specified; PM = poor metabolizer; RBS-R CRS = Repetitive Behavior Scale–Revised, Compulsive Behavior and Ritualistic/Sameness Behavior subscales; RUPP = Research Units on Pediatric Psychopharmacology; RUPP-PI = RUPP–Psychosocial Intervention; SD = standard deviation; SGAs = second-generation antipsychotics; SNP = single nucleotide polymorphism; UM = ultrarapid metabolizer.
PGx Studies Based on Age Groups
PGx studies in adults with ASD/ID
There were no PGx studies exclusively focusing on adults with ASD/ID although 3 PGx studies included adults with ASD/ID in their analyses. 49,51,61 One PGx study by Bishop et al. examined the association between metabolizer status for the CYP2C19 gene (i.e., ultrarapid metabolizer [UM; n = 26], extensive metabolizer [EM; n = 40], and poor [PM]/intermediate [IM] metabolizer; n = 23) and assessed treatment response to escitalopram using the Aberrant Behavior Checklist–Community Version (ABC-CV) in individuals with ASD. 49 Although adults were included, no breakdown by age was provided (mean ± standard deviation [SD] [range]: 136.7 ± 66.9 [54 to 532] months, N = 89), but this study included at least 1 adult patient (44.3 years old). A subgroup of individuals in this study had ID in addition to ASD, but no individuals were exclusively diagnosed with ID (nonverbal intelligence quotient [IQ]: 83.2 ± 31.7 [21 to 146], N = 89; verbal IQ: 76.7 ± 31.7 [11 to 141], N = 79). However, no information on response in adults versus children/adolescents or in those with or without concurrent ID was provided. Another study by Najjar et al. examined whether the SLC6A4 (5-HTTLPR) and HTR2A (rs7997012) polymorphisms were associated with response to escitalopram, using the Repetitive Behavior Scale–Revised, Compulsive Behavior and Ritualistic/Sameness Behavior Subscales (RBS-R-CRS) and ABC-CV Irritability subscale (ABC-CV-IRR) scores (N = 44). 61 Similar to the previous study, 49 this study also included adults but did not provide a breakdown by age (mean ± SD [range]: 161 ± 86 [61 to 532] months) while this study included at least 1 adult patient aged 44 years. 61 A subgroup of individuals had ASD with ID, but no individuals were exclusively affected with ID (nonverbal IQ: 80 ± 25 [35 to 130], N = 44; verbal IQ: 78 ± 25 [30 to 120], N = 38). A study by Correia et al. examined the relationship between treatment response to risperidone, which was assessed by the Autism Treatment Evaluation Checklist (ATEC), and 15 variants across 8 genes in autistic children and young adults who were receiving risperidone up to 1 year (N = 45). 51 Similar to the other 2 studies, 49,61 this study also included at least 1 adult with an age of 21 years (mean ± SD [range]: 8.67 ± 4.30 [3 to 21]). It reported patients’ IQ levels and included individuals with ID; however, it was not clear whether adults with ID were included or not (IQ ≥ 70, 37.8% of the patients; 69 ≥ IQ ≥ 50, 31.1%; 49 ≥ IQ ≥ 35, 24.4%; and IQ < 35, 6.7%). 51
PGx studies in children/adolescents with ASD/ID
In contrast to the 3 studies that included adults and children/adolescents, 49,51,61 the other 25 studies focused specifically on children/adolescents with ASD/ID. Among them, 1 PGx study by AlOlaby et al. included only children/adolescents with fragile X syndrome which is the most common inherited cause of ID. 47 In addition, 6 additional studies were conducted in children/adolescents with ASD, some of whom also had ID. 48,53,59,63,64,67 Three of the 6 studies clearly described that they included ASD with ID. 53,59,64 For the other 3 studies, 48,63,67 it was likely that they also included children/adolescents with ASD and ID, given their inclusion criteria. More specifically, Nurmi et al. investigated the association of key energy balance genes (i.e., FTO, MC4R, LEP, CNR1, FAAH) with antipsychotic-induced weight gain (AIWG) in children/adolescents with ASD treated with risperidone in the 2 National Institute of Mental Health Research Units on Pediatric Psychopharmacology (RUPP) Autism Network trials. 63 It was reported that approximately 70% and 40% of patients in the 2 trials had intellectual disabilities. 68,76 Likewise, Anderson et al. included individuals with ASD treated with risperidone from the RUPP trial, in which approximately 70% of the individuals were affected with mild or more ID. 48 The study by Sherwin et al. investigated the effect of the CYP2D6 phenotype on pharmacokinetic variability of risperidone in children and adolescents (majority of ASD; N = 45). 67 This study did not specify diagnoses but reported that the majority of individuals were affected by ASD. Forty-one of the 45 patients included in this study were from other studies, 1 of which included some individuals with ASD who had co-occurring ID. 68 Thus, it was possible that some individuals had ASD and possibly ID in this study. 67
There were 18 studies of children/adolescents with ASD, which either did not describe whether any of the participants also had ID or specifically mentioned including only individuals without ID. 50,52,54 –58,60,62,65,66,69,70 –75 For example, Roke et al. listed IQ above 85 in their inclusion criteria. 66 Three other studies specified excluding children/adolescents with IQs below 55 but did not provide information indicating whether individuals with IQs ranging from 56 to 75 participated in the studies. 54,57,65 The other 14 studies did not report if they included children/adolescents with ASD who also had ID.
PGx Studies Based on Treatment Outcomes
Response to psychotropics
Among the 28 studies included, twelve studies primarily investigated the association between treatment response and specific gene polymorphisms. 47,49,51,54,58,59,61,62,64,65,69,75
Response to antipsychotics
Five of the 12 studies focused on patients treated with antipsychotics. 51,54,62,65,75 Among them, 4 studies 51,54,62,75 included only patients treated with risperidone monotherapy and the other one used various antipsychotics. 65
Pharmacokinetic Genes: Three studies investigated the CYP polymorphisms, 51,62,75 among which 1 study 75 suggested an association of CYP2D6 metabolizer status with treatment response to risperidone. More specifically, an observational cohort study of 40 Israeli children by Youngster et al. evaluated the association between CYP2D6 genotypes (up to 34 CYP2D6 alleles and allele duplications) and treatment response to risperidone determined by parents and the treating neurologist, using a simple 3-point scale (i.e., improvement in disruptive behaviors, no change, or worsening). 75 This study reported that PMs (n = 2) were classified as responders whereas UMs (n = 2) were classified as nonresponders, while no serum levels of risperidone were taken. However, other studies reported no association of the CYP2D6 polymorphisms with treatment response. 51,62 Two studies examined the impact of the ABCB1 1236C>T polymorphism on treatment response to risperidone. 51,62 Correia et al. reported that the ABCB1 1236C>T (rs1128503) was significantly associated with clinical improvement assessed by the ATEC (P = 0.002; see also the “PGx Studies in Adults with ASD/ID” section) 51 whereas a cross-sectional study by Nuntamool et al., 62 where 82 Thai children/adolescents treated with risperidone for more than 1 year were included, reported no significant association with treatment response determined by the Clinical Global Impression Scale–Improvement (CGI-I) score and a 4-point scale for each of aggression, overactivity, and repetitive behaviors.
Pharmacodynamic Genes: Among the 4 studies investigating gene variants associated with treatment response to risperidone, 51,54,62,75 the DRD3 Ser9Gly (rs6280) polymorphism was examined in 3 studies, 51,54,62 2 of which reported significant findings. 51,54 More specifically, Correia et al. reported carriers of Gly allele showed greater treatment response to risperidone than noncarriers of Gly allele (i.e., Ser/Ser genotype; see also the “PGx Studies in Adults with ASD/ID” section). 51 An 8-week prospective study by Firouzabadi et al. reported that responder rates (i.e., a 50% or greater decrease of the ABC scores from baseline) were significantly higher in carriers of Gly allele as well as carriers of Gly/Gly and Ser/Gly genotypes compared with carriers of Ser allele and Ser/Ser genotype in Iranian children (N = 56; P = 0.027 and 0.014, respectively). 54 In contrast, a cross-sectional study by Nuntamool et al. 62 reported no significant association of the DRD3 rs6280 polymorphism with treatment response. The following other gene variants were also significantly associated with treatment response to risperidone in 1 study: the HTR2A c.-1438G>A (rs6311; P = 0.019), 51 HTR2C c.995G>A (rs3813928; P = 0.035), 51 and DRD2 Taq1A (rs1800497; P = 0.048). 62
Response to antidepressants
The association between response to antidepressants and gene variants was examined in 6 studies. 47,49,61,64,65,69 Among them, escitalopram was used in 3 studies (see also the “PGx Studies in Adults with ASD/ID” section), 49,61,64 sertraline in 1 study (see also the “Pharmacogenomic Studies in Children/Adolescents with ASD/ID” section), 47 fluvoxamine in 1 study, 69 and various antidepressants in 1 study. 65
Pharmacokinetic Genes: Bishop et al. found that there were no differences in the rate of improvement assessed using the ABC-CV across metabolizer groups for the CYP2C19 gene (i.e., UM [n = 26], EM [n = 40], and PM/IM metabolizer [n = 23]) in individuals with ASD treated with escitalopram in 6 weeks (P = 0.39). 49 However, the UM group exhibited a slower rate of dosing change compared to other groups when looking at titration trajectories, contrary to expectations. On the other hand, AlOlaby et al. reported that subjects with the PM/IM genotypes for the CYP2C19 gene showed a significant percentage in the very much improved/much improved CGI-I if they were treated with sertraline (n = 6) compared to those who were with placebo (n = 5) in patients with fragile X syndrome (P = 0.007). 47
Pharmacodynamic Genes: The association of the SLC6A4 (5-HTTLPR) gene and treatment response to antidepressants was investigated in 4 studies. 47,61,64,69 AlOlaby et al. reported that sertraline was associated with a significantly different change (i.e., symptom improvement) from baseline in the social participation raw score on the active arm compared to placebo in those with the L/L genotype (P = 0.005) whereas no significant difference was observed for the S/L (P = 0.422) or S/S (P = 0.997) genotypes (N = 51). 47 Likewise, Sugie et al. 69 found that the L allele conferred better response to fluvoxamine than the S allele in a 12-week double-blind crossover trial of fluvoxamine and placebo, in which treatment response was determined by CGI scores in Japanese patients (N = 18; P = 0.047). In addition, Owley et al. reported a significant interaction between genotype group of the 5-HTTLPR and time on the ABC-CV-IRR in patients treated with escitalopram for 10 weeks (N = 58; P = 0.004). 64 In contrast, Najjar et al. reported no significant differences in the rate of symptom improvement assessed using the RBS-R-CRS and ABC-CV-IRR across genotype groups in ASD treated with escitalopram over the 6 weeks (N = 44; P = 0.273 for RBS-R-CRS and P = 0.122 for ABC-CV-IRR). 61 Other results were summarized in Table 1.
Response to other medications
An 8-week open-label trial examined the effect of the MDR1 (ABCB1) C3435T polymorphism on treatment response to guanfacine in PDD patients with clinically significant symptoms of attention deficit hyperactivity disorder (ADHD; N = 25). 58 Patients with either C/T or C/C genotypes showed a significantly greater improvement than T/T genotype in the ABC Hyperactivity scores (P < 0.03) and Swanson, Nolan, and Pelham (SNAP) scores (P = 0.05). Another study reported a 4-week, placebo-controlled, double-blind crossover study with 58 children to evaluate the association between 36 variants across 10 genes and treatment response to methylphenidate defined by CGI and ABC hyperactivity subscale. 59 This study reported that the DRD1 rs4867798 (P = 0.042) and rs5326 (P = 0.006), DRD3 rs6280 (P = 0.044), DRD4 rs11246226 (P = 0.038), SLC6A3 VNTR (P = 0.049), SLC6A4 STin2 VNTR (P = 0.041), ADRA2A rs1800544 (P = 0.015), and COMT rs4680 (P = 0.049) among 36 variants tested were significantly associated with responder status; however, this significance in each variant did not remain after correction for multiple testing.
Adverse effects
Fifteen studies primarily investigated the association of side effects with gene polymorphisms. 48,50 –53,55,56,59,63,65,66,70 –72,75 Among them, 9 studies focused on prolactin elevation or hyperprolactinemia as primary outcome in patients treated with risperidone. 48,50,51,53,56,66,70,72,75 Other adverse effects were also investigated in 8 studies (e.g., AIWG, blood pressure, and insulin resistance). 51,52,55,59,63,65,71,75
Prolactin elevation or hyperprolactinemia
The most commonly investigated gene variant associated with prolactin elevation or hyperprolactinemia was CYP2D6 polymorphisms, 51,53,56,66,70,72,75 followed by the DRD2 Taq1A (rs1800497) polymorphism. 48,50,51,66,70
Pharmacokinetic Genes
Seven studies examined the impact of CYP2D6 polymorphisms on prolactin elevation or hyperprolactinemia. 51,53,56,66,70,72,75 Although 1 prospective study by Troost et al. reported a positive correlation of the number of functional CYP2D6 genes and serum prolactin level in 8 weeks (P = 0.034), 72 other studies reported no significant association of CYP2D6 polymorphisms, genotypes, or predicted phenotypes with prolactin elevation or hyperprolactinemia. 51,53,56,66,70,75
Pharmacodynamic Genes
Five studies investigated the association of the DRD2 Taq1A (rs1800497) polymorphism with hyperprolactinemia in patients treated with risperidone, 48,50,51,66,70 and 3 of them reported nonsignificant findings 48,51,66 whereas the others reported a synergistic effect of the DRD2 TaqIA and DRD2 A-241G variants on prolactin concentration using multiple regression analysis (P = 0.003) 50 and significant differences in prolactin level of patients among the DRD2 Taq1A A2A2, A1A2, and A1A1 groups (P = 0.033). 70
The presence of the C allele of HTR2C rs6318 polymorphism was significantly associated with prolactin elevation or hyperprolactinemia in 2 studies (P = 0.006 and 0.02, respectively). 51,53 Other results were summarized in Table 1.
Other adverse effects
Four studies investigated the association of gene variants with AIWG in patients treated with risperidone. 51,55,63,75 The HTR2C rs6318 51 and rs3813929, 55 LEP rs7799039, 63 and CNR1 rs806378 and rs1049353 polymorphisms 63 were significantly associated with AIWG (P < 0.05) whereas the findings of the association between the HTR2C rs3813929 polymorphism and AIWG yielded opposite findings in 2 studies. 51,55 In addition, UMs of CYP2D6 showed a 4.8% and 5.8% lower increase in body mass index and waist circumference compared to EMs. 51
Drug concentrations
Four studies investigated the association of drug concentrations with gene polymorphisms in patients treated with risperidone. 60,67,73,74 PMs/IMs of CYP2D6 showed significantly higher plasma concentration of risperidone and risperidone/9-hydroxyrisperidone ratio than EMs in 2 studies focusing on Thai patients (P < 0.05). 73,74 Other results were summarized in Table 1.
Discussion
The aim of this systematic review was to identify and review publications investigating the association between selected gene variants and treatment outcomes (e.g., treatment response and adverse effects) in individuals with ASD/ID and to review the clinical validity and utility of PGx. To achieve this aim, we included each identifiable study of individuals with ASD/ID regardless of age in our research. We found that although there were several PGx studies in children/adolescents with ASD, there were only very limited studies reported in adults with ASD/ID while not a single study focused exclusively on adults.
Similar to a recent review on PGx studies in ASD by Brown, 45 several gene variants associated with treatment response, and adverse effects of antipsychotics and antidepressants were reported exclusively in children/adolescents with ASD/ID. However, the number of PGx studies in ASD/ID across age groups, especially in adults with ASD/ID, was still very few. Also, it should be kept in mind that identified studies had relatively limited sample sizes and a variety of ethnicities and study designs. Furthermore, almost all studies applied a classic “candidate gene” approach. No genome-wide association studies were reported while there were 1 study investigating exonic expression levels using Affymetrix GeneChip Human Exon 1.0 ST Arrays (Affymetrix, Santa Clara, CA) 57 and 2 studies investigating several genetic variants in drug-metabolizing enzyme and transporter (DMET) genes using Affymetrix DMET arrays (Affymetrix Inc., Santa Clara, CA). 56,60
The majority of studies identified in this review have examined PGx associations with treatment response and/or adverse effects in patients treated with risperidone. The possible reasons for this finding are as follows: (1) Risperidone is one of the FDA-approved drugs for the treatment of challenging behavior in children/adolescents with ASD, 77 and (2) risperidone is similarly one of the most commonly studied medications for challenging behavior (i.e., repetitive, self-injurious, and aggressive behaviors) in adults with ASD/ID. 78 Although the DRD3 rs6280 polymorphism was the most investigated gene variant in regard to treatment response to risperidone in the 3 studies, 51,54,62 the results were still inconclusive. This inconclusive finding is consistent with a recent systematic review on the association between dopamine receptor gene polymorphisms and treatment response to risperidone assessed using the Positive and Negative Syndrome Scale, Brief Psychiatric Rating Scale, or CGI in schizophrenia. 79 Likewise, the association between pharmacokinetic gene variants (e.g., CYP and ABCB1 gene variants) and response to risperidone also remains controversial in ASD/ID, which is consistent with mixed findings in a recent review in patients with schizophrenia. 80
In regard to adverse effects, prolactin elevation or hyperprolactinemia was the most investigated adverse effect as a primary outcome in patients treated with risperidone in 9 studies, 48,50,51,53,56,66,70,72,75 examining the impact of several gene variants (e.g., CYP2D6 polymorphisms and DRD2 rs1800497). Most of the studies examining the impact of CYP2D6 polymorphisms on prolactin elevation or hyperprolactinemia reported negative findings. However, most of them were based on a cross-sectional or observational study design in relatively small sample sizes (n = 40 to 147). In regard to the DRD2 rs1800497, a recent meta-analysis that included 772 patients with schizophrenia, ASD, or disruptive behavior disorder from 8 studies showed no significant difference between the DRD2 Taq1A (rs1800497) A1 carriers and non-A1 carriers in risperidone-related prolactin level (P = 0.423); 81 this meta-analysis included 5 studies identified in our review. 48,50,51,66,70
Similar to PGx studies on antipsychotics, published PGx studies using antidepressants (e.g., selective serotonin reuptake inhibitors [SSRI]) are still limited in individuals with ASD/ID across age groups. We identified only 6 studies with relatively small sample sizes (n = 19 to 279) for whom the majority of individuals were of European ancestry. 47,49,61,64,65,69 For instance, although 3 studies reported a significant association of the 5-HTTLPR with treatment response to antidepressants (escitalopram, sertraline, and fluvoxamine) assessed using the ABC-CV-IRR or CGI, the small sample size in each study is a major limitation in each study (N = 58, 51, and 18, respectively). 47,64,69 It is of interest that this finding in individuals with ASD/ID is consistent with a meta-analysis of the association between 5-HTTLPR and treatment response to SSRIs (i.e., remission and response rates) in patients with major depressive disorder (MDD) and bipolar disorder (28 studies and 3,866 subjects). 82 In line with the findings of this meta-analysis, 82 the associations between 5-HTTLPR and treatment response to SSRIs were reported in patients with anxiety disorder in several studies although the findings were mixed. 83 However, it should be kept in mind that treatment response is assessed by depression and anxiety scales but not the ABC or CGI in the studies in patients with mood disorders and anxiety disorder. 82,83 Likewise, the findings of the association between CYP2C19 gene variants and response to antidepressants are mixed in ASD/ID, which is in line with previous studies showing inconsistent linking the CYP2D6 and CYP2C19 gene variants to antidepressant treatment response in patients with MDD. 84,85 Although antipsychotics are the most commonly prescribed medications in adults with ID in the United States, 24 a population-based study in UK (N = 33,016) reported the most common class of drugs to be prescribed was anxiolytics/hypnotics, followed by antidepressants in adults with ID between 1999 and 2013 and that the incidence rate of new antidepressants over the follow-up period was approximately 350 per 10,000 person years in 2013. 15 Nevertheless, the evidence for antidepressant use in individuals with ID across all age groups is sparse, and generally, low response rates and high rates of adverse events have been reported. 86 Although studies in ASD suggested that SSRIs might be better tolerated in adults than in children, most studies of SSRIs in individuals with ID included both adults and children/adolescents, and age-specific data were still limited. 86 Therefore, further investigation on PGx studies on antidepressants as well as antipsychotics is warranted.
While specific PGx guidelines have not been established for individuals with ASD/ID, several established PGx guidelines provided useful gene–drug information for medications used in ASD/ID across age groups. 87,88 For instance, the Clinical Pharmacogenetics Implementation Consortium Dosing Guidelines recommend SSRI dosing adjustment based on the metabolizer status of the CYP2C19 for citalopram, escitalopram, and sertraline and the CYP2D6 for fluvoxamine. 87 Likewise, the Royal Dutch Association for the Advancement of Pharmacy–Pharmacogenetics Working Group has also recommended the dosing adjustment of antipsychotics based on the CYP2D6 genotypes for 6 antipsychotics: aripiprazole, brexpiprazole, clozapine, haloperidol, olanzapine, risperidone, and zuclopenthixol. 89 These guidelines are also well summarized on the Pharmacogenomics Knowledgebase website, 88 which also provides levels of evidence for gene–drug associations. Given that those gene–drug pairs should be also relevant in ASD/ID across all age groups as regardless of diagnoses, minimal or excessive serum/plasma levels of antipsychotics and antidepressants are likely to affect treatment response and side effects in this population.
To the best of our knowledge, this is the first systematic review of PGx studies with psychotropic drugs including antipsychotics and antidepressants (i.e., treatment response, adverse effects, and drug concentrations) in adults and children/adolescents with ASD/ID. Nevertheless, the results of our study must be interpreted with caution, given several limitations. First, on a systematic level, despite that articles were systematically investigated through MEDLINE, Embase, and PsycINFO, some references may still have been missed, nonsignificant findings might not have been published (i.e. “publication bias”), and only articles written in English were included in this review. Second, although we included some studies that might have partly included adults with ASD/ID, the actual number of the adults included in each study was unclear. Third, although several studies reported significant associations between several gene variants and treatment outcomes (e.g., treatment response to antipsychotics and antidepressants, and adverse effects) prior to multiple testing, there were few studies that reported gene variants surviving correction for multiple testing (e.g., Nurmi et al. 63 ).
In conclusion, there is a limited number of PGx studies in individuals with ASD/ID in particular in adults. Given that psychotropic medication use increases with age, 22,23 accumulating evidence on PGx studies and further investigation focusing on the clinical validity and efficacy of PGx testing for psychotropic treatment in individuals with ASD/ID across age groups are warranted.
Footnotes
Author Contributions
These authors Pushpal Desarkar and Daniel J. Müller equally contributed to this work. Kazunari Yoshida and Emiko Koyama did the literature search, extracted the data, and wrote the first draft of the manuscript. All authors interpreted the data, wrote the report, and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Pushpal Desarkar is currently supported by Scottish Rite Charitable Foundation of Canada Research Grant, CAMH Discovery Fund, and the Academic Scholars Award from the Department of Psychiatry, University of Toronto. Kazunari Yoshida has received manuscript fees from Sumitomo Dainippon Pharma, fellowship grants from the Japan Research Foundation for Clinical Pharmacology and Azrieli Adult Neurodevelopmental Centre Postdoctoral Fellowship at CAMH, and consultant fees from Signant Health and VeraSci within the past 3 years. Emiko Koyama has received fellowship grants from the Discovery Fund Postdoctoral Fellowship at CAMH within the past 3 years. James L. Kennedy is a member of the Scientific Advisory Board of Myriad Neuroscience (unpaid) and holds several patents relating to pharmacogenetic tests for psychiatric medications. Daniel J. Müller was funded by the Canadian Institutes of Health Research (CIHR) and holds the Joanne Murphy Chair at CAMH. The other authors have nothing to disclose.
