Abstract
Objective:
To review the current evidence for efficacy of cannabidiol in the treatment of mood disorders.
Methods:
We systematically searched PubMed, Embase, Web of Science, PsychInfo, Scielo, ClinicalTrials.gov, and The Cochrane Central Register of Controlled Trials for studies published up to July 31, 2019. The inclusion criteria were clinical trials, observational studies, or case reports evaluating the effect of pure cannabidiol or cannabidiol mixed with other cannabinoids on mood symptoms related to either mood disorders or other health conditions. The review was reported in accordance with guidelines from Preferred Reporting Items for Systematic reviews and Meta-Analyses protocol.
Results:
Of the 924 records initially yielded by the search, 16 were included in the final sample. Among them, six were clinical studies that used cannabidiol to treat other health conditions but assessed mood symptoms as an additional outcome. Similarly, four tested cannabidiol blended with Δ-9-tetrahydrocannabinol in the treatment of general health conditions and assessed affective symptoms as secondary outcomes. Two were case reports testing cannabidiol. Four studies were observational studies that evaluated the cannabidiol use and its clinical correlates. However, there were no clinical trials investigating the efficacy of cannabidiol, specifically in mood disorders or assessing affective symptoms as the primary outcome. Although some articles point in the direction of benefits of cannabidiol to treat depressive symptoms, the methodology varied in several aspects and the level of evidence is not enough to support its indication as a treatment for mood disorders.
Conclusions:
There is a lack of evidence to recommend cannabidiol as a treatment for mood disorders. However, considering the preclinical and clinical evidence related to other diseases, cannabidiol might have a role as a treatment for mood disorders. Therefore, there is an urgent need for well-designed clinical trials investigating the efficacy of cannabidiol in mood disorders.
Introduction
Mood disorders are common psychiatric conditions, with major depressive disorder (MDD) reaching a lifetime prevalence of up to 14.6%, 1 affecting more than 300 million people worldwide, 2 while bipolar disorder reaches a prevalence of 4.4%, including subthreshold cases. 3 Both are chronic mental disorders that usually start early in life and follow a chronic course, commonly resulting in significant functional impairment and are associated with premature death due to comorbid medical illness and suicide. 4 –6 Both conditions are also among the leading causes of years lived with disability directly and of years of life lost due to suicide. 7 –9 Long-term treatment strategies are, therefore, the mainstay of management. However, especially in the case of bipolar disorder, the most efficacious medications are associated with adverse effects, which can lead to noncompliance with treatment and poor clinical and functional outcomes. 10,11 In this context, new treatments with novel mechanisms of action and good tolerability profile which are suitable for long-term use are urgently needed.
The plant
Briefly, the endocannabinoid system consists of the CB1 and CB2 receptors and their endogenous ligands. While the CB1 receptors are primarily present in the cortex, basal ganglia, hippocampus, and cerebellum, 26 the CB2 receptors are less prevalent in the central nervous system but can be found in microglia and vascular elements. 27,28 The endocannabinoid system is a neuromodulator in both the central and peripheral nervous systems influencing several biological functions including mood, anxiety, appetite, sleep, memory processing, pain sensation, and immune functions. 29,30 There is evidence that schizophrenia is associated with alterations in the endocannabinoid system, 17,31,32 which could explain favorable results with cannabidiol in some clinical trials in patients with schizophrenia. 17,18 Furthermore, with receptors and ligands present in neurons and immune cells, the endocannabinoid system is especially interesting in the context of mood disorders since both MDD and bipolar disorder are illnesses whose pathophysiology involves changes in the brain 33 –37 and the immune system. 36 –39
Cannabidiol has been studied in several animal models of depression such as forced swimming test,
23,40,41
learned helplessness paradigm,
25
olfactory bulbectomy mouse model,
22
and genetic models
24,42
showing promising results in improving the depressive-like behaviors. Also, it shows antidepressant-like effects in models that are interesting for clinical purposes, for example, models of chronic stress
22
and anhedonia.
24,42
Its antidepressant effects have been shown not only when compared with vehicle solutions but also with active and established antidepressant medications, such as imipramine and fluoxetine.
23,41,43
Moreover, these studies have shown that cannabidiol exerts acute and subchronic and chronic effects and can maintain antidepressant-like effects.
22,25,40,43
In contrast, the only published study investigating the effects of cannabidiol in an animal model of mania induced by
Regarding its psychophysiological basis, the preclinical studies have suggested that the cannabidiol antidepressant-like effect is probably mediated by indirect activation of CB1 receptors, 45 activation of 5-HT1A receptors, 22,23,45 and increased serotonin levels in the central nervous system but are not related to noradrenergic levels. 22,41 For example, in one study, the administration of cannabidiol into the ventral medial prefrontal cortex (vmPFC) induced antidepressant-like effects possibly through indirect activation of CB1 and 5-HT1A receptors. 45 Another study with in vivo microdialysis revealed that the administration of cannabidiol significantly enhanced serotonin and glutamate levels in vmPFC, with the increase in neurotransmitters levels occurring immediately after the first injection of cannabidiol. 22 Moreover, adaptive changes in pre- and postsynaptic 5-HT1A receptor functionality were also found after chronic cannabidiol exposure. 22 Some studies have also shown effects on neuroplasticity. For instance, a study showed that chronic administration of cannabidiol increased BDNF levels in the amygdala of rats treated for depression-like symptoms. 43 Another study showed that the acute antidepressant effects were associated with increased BDNF levels in both medial prefrontal cortex (mPFC) and the hippocampus, as well as increased spine density in the mPFC. 25
In spite of the clinical and preclinical evidence described earlier, a literature search indicated that there is no systematic review on clinical studies of cannabidiol and mood disorders to date. Therefore, the objective of this study is to systematically review the available literature and clarify the current level of evidence for efficacy of cannabidiol in the treatment of mood disorders.
Methods
Search Strategy and Identification of Eligible Articles
This systematic review has been reported following guidance from Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement 46 and was registered at International Prospective Register of Systematic Reviews (PROSPERO) (CRD: 42019119766). The PRISMA flowchart is given in Figure 1, and the PRISMA checklist can be found in the Supplementary Materials. The authors searched PubMed, Web of Science, Embase, and PsychInfo databases for articles published in any language up to July 31, 2019, using the following search terms: cannabidiol AND (mania OR hypomania OR depression OR euphoria OR irritability OR irritable mood OR dysphoria OR anhedonia OR bipolar disorder OR bipolar affective disorder OR bipolar I disorder OR bipolar II disorder OR manic depressive OR manic depression OR major depression OR major depressive disorder OR depressive disorders OR depressive episode OR recurrent depressive disorder OR dysthymia OR dysthymic disorder OR cyclothymia OR cyclothymic disorder OR mood disorder OR mood disorders OR affective disorder OR affective disorders OR affective symptoms OR emotions). A complementary search using the same terms was performed on ClinicalTrials.gov and The Cochrane Central Register of Controlled Trials. The Medical Subject Headings terms were used in PubMed. The reference lists of the included studies were also examined in order to find more results.
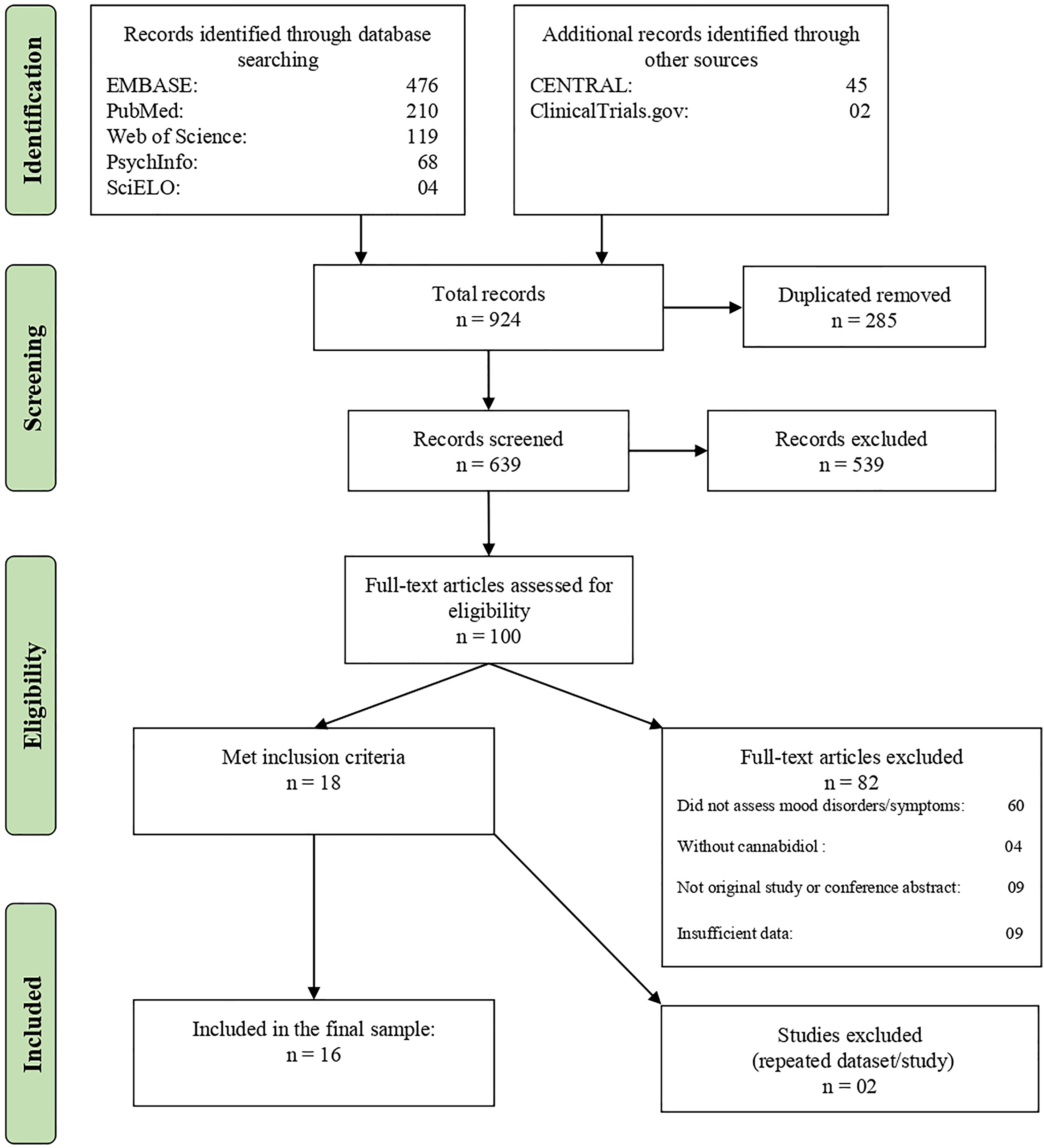
Flowchart of the review process and study selection.
Selection Criteria
Two reviewers (JVP and GS) independently screened and selected the studies, and a third investigator (LNY) made the final decision in cases of disagreement. The inclusion criteria were double-blind randomized clinical trials (RCTs) with and without placebo conditions, open-label clinical trials, observational studies (longitudinal or cross-sectional), or case series that evaluated the effect of pure cannabidiol or cannabidiol mixed with other cannabinoids on affective symptoms. These symptoms could be discrete hypomanic, manic, mixed, or depressive episodes in mood disorders or mood symptoms associated with other psychiatric illnesses and neuropsychiatric disorders. The exclusion criteria were studies not reporting mood symptoms, not using cannabidiol and nonoriginal studies (reviews, editorials, book chapters). The authors did not specifically search gray literature. The methodological quality appraisal of included clinical studies was carried out using the Cochrane Collaboration’s tool for assessing the risk of bias in randomized trials, while the quality of the observational studies was evaluated using the Newcastle-Ottawa Quality Assessment Scale. 47
Results
Figure 1 summarizes the selection process according to the PRISMA protocol. The literature search identified a total of 924 records, but, after the exclusion of duplicates, a final sample of 639 remained. Then, the abstracts and their titles were screened independently, and a total of 539 abstracts were excluded after initial screening. Finally, 100 full texts were reviewed, and 84 were excluded. A final sample of 18 full texts met the inclusion criteria. However, only 16 were included in the final sample because two papers were from the same study or shared the same data set of other included articles. In these cases, the papers with the largest sample size were included. The characteristics of the studies included are described in Tables 1 and 2. Seven papers were clinical studies that used cannabidiol to treat other medical and psychiatric conditions but assessed mood symptoms as an additional outcome. Five studies tested medical cannabis, nabiximols, or other blends of cannabinoids that included cannabidiol in the treatment of medical or psychiatric conditions and assessed affective symptoms as secondary outcomes. Among the clinical studies with cannabidiol, two were case reports or case series using cannabidiol to treat psychiatric disorders. Four were surveys or observational studies that assessed the cannabidiol use and its clinical correlates.
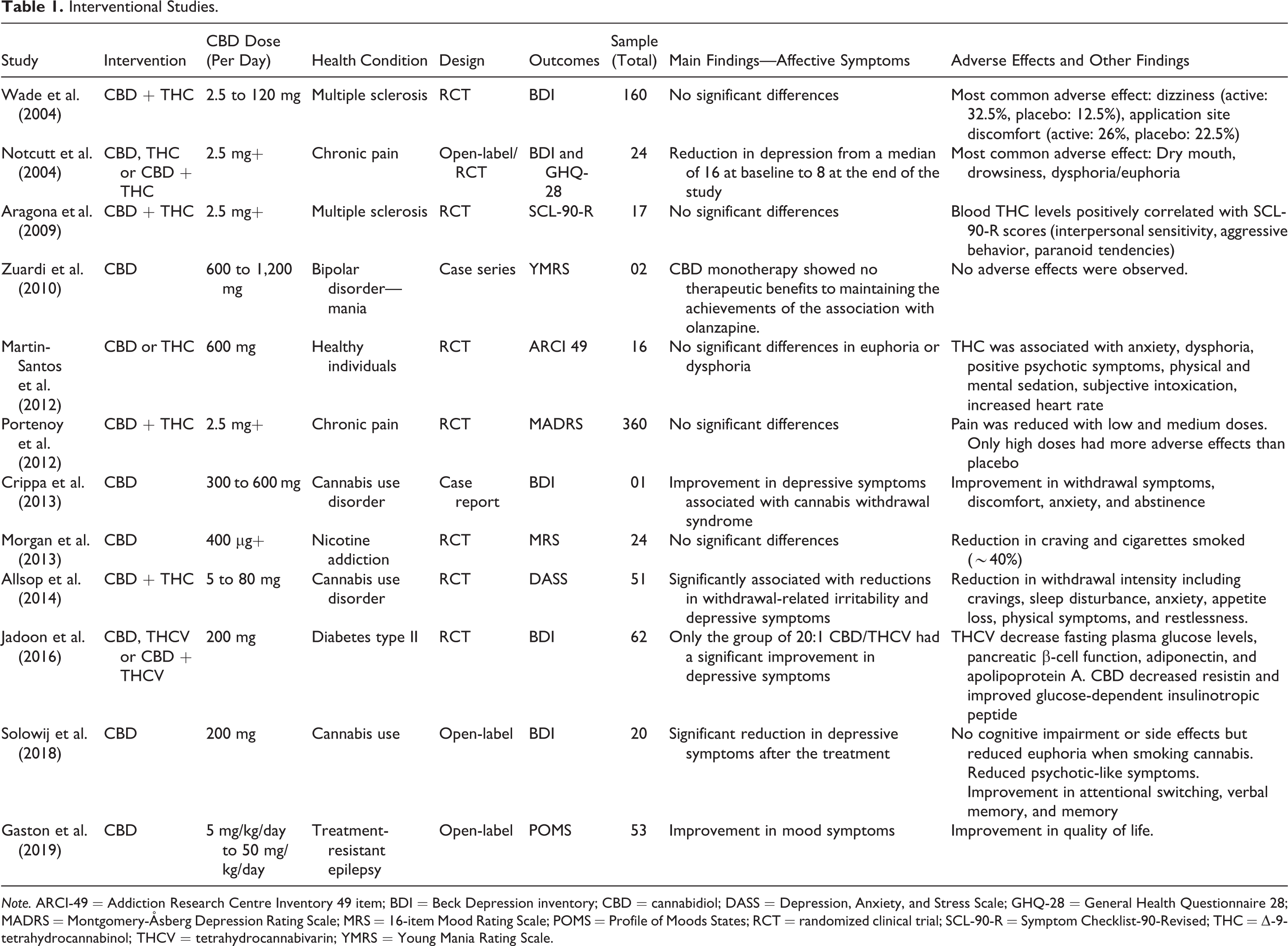
Interventional Studies.
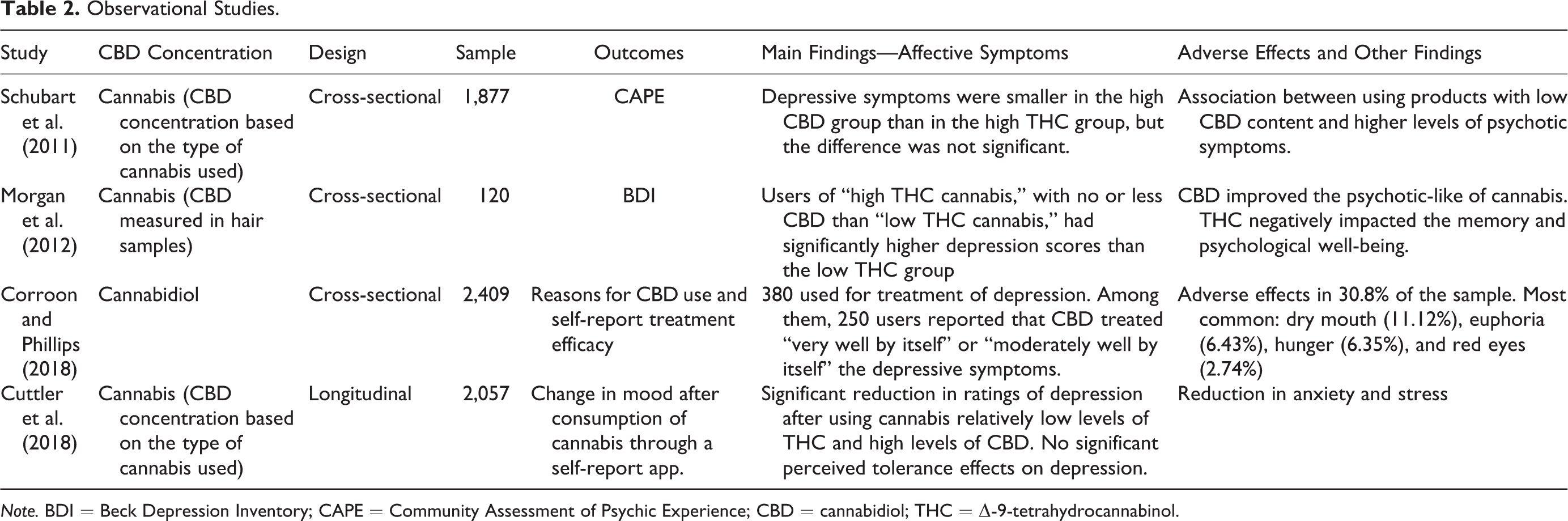
Observational Studies.
Interventional Studies With Cannabidiol
Placebo-controlled trials
A randomized, double-blind, cross-over, placebo-controlled trial with 16 healthy male participants observed the pharmacological effects of both THC and cannabidiol separately in the same subjects. They all received THC 10 mg, cannabidiol 600 mg, and placebo in three individual sessions at 1 month intervals. Physiological parameters, as well as symptoms ratings, were collected before and at 1, 2, and 3 hr after drug administration. Cannabidiol was not different from placebo in physiological measurements and clinical rating scales such as Visual Analogue Mood Scale, 48 State Anxiety Inventory (STAI), 49 Positive and Negative Syndrome Scale, 50 and Addiction Research Centre Inventory-49. 51 In contrast, THC increased anxiety, sedation, dysphoria, positive psychotic symptoms, negative psychotic symptoms, general psychopathology, and heart rate. 52
A 13-week randomized, double-blind, placebo-controlled, parallel-group study examined the effect of cannabidiol and/or D9-tetrahydrocannabivarin (THCV) on lipid and glucose metabolism in 62 patients with type II diabetes. Secondary measurements included the Beck Depression Inventory-II (BDI-II),
53
which showed no difference from the placebo group except for the 20:1 cannabidiol/THCV compound, which had a statistically significant, though mild, increase in depressive symptoms, with an estimated mean difference on the BDI-II of 4.77 (
A pilot randomized double-blind placebo-controlled study examined the effect of inhaled cannabidiol 400 μg dissolved in absolute ethanol (≈5%) or placebo (ethanol alone) in 24 tobacco smokers wanting to quit. The 16-item Mood Rating Scale 55 was used at baseline and after the 7-day trial period. No significant difference was measured in depressive symptoms or sedation. Both conditions showed a marked decrease in anxiety scores over time, but there was no significant effect of condition. 56
Open-label trials and case series
A pragmatic open-label clinical trial assessed the effects of cannabidiol 200 mg/day for 12 weeks on psychiatric symptoms and cognitive functioning in 20 chronic cannabis users. The scores on BDI,
57
STAI, Community Assessment of Psychic Experiences (CAPE),
58
global and social functioning (Global Assessment of Functioning [GAF], Social and Occupational Functioning Assessment Scale [SOFAS]),
59
verbal learning (Rey Auditory Verbal Learning Test),
60
and attention switching scales (from the Cambridge Neuropsychological Test Automated Battery)
61
were compared between post-treatment and baseline. The severity of depressive symptoms as assessed by BDI was significantly lower after treatment, compared to the baseline scores (
A 1-year follow-up open-label study included 53 participants with treatment-resistant epilepsy and assessed the effect of cannabidiol (starting at 5 mg/kg/day and titrated to 50 mg/kg/day) on the quality of life using the Quality of Life in Epilepsy–89 (QOLIE-89).
63,64
As an additional outcome, the Profile of Moods States (POMS) was included.
65
The POMS is a 65-item validated questionnaire commonly used to assess mood symptoms in epilepsy clinical trials. It consists of six subscales, among which the Depression/Dejection subscale correlates with BDI.
66
The results showed improvement in mood symptoms with a significant reduction in POMS scores (9.7,
A case study described two patients with bipolar I disorder without comorbidity, hospitalized for a manic episode with psychotic symptoms, who were treated with cannabidiol (initially 600 mg/day, then 900 mg/day, and 1200 mg/day). 67 The first patient received cannabidiol plus olanzapine for 2 weeks. After this period, olanzapine was withdrawn to observe whether the cannabidiol alone would maintain the improvement but cannabidiol alone did not maintain the improvements achieved through the combination with olanzapine. The second patient received monotherapy with cannabidiol but had no symptoms improvement with any dose. In both cases, the symptoms were evaluated through the Young Mania Rating Scale 68 and Brief Psychiatric Rating Scale, 69 and adverse effects were evaluated with the Udvalg for Kliniske Undersogelser Scale. 70 Tolerability was very good, and no side effects were reported, even with doses as high as 1200 mg/day. 67
In another case study, a 19-year-old woman with cannabis dependence (according to
Interventional Studies With Medical Cannabis, Nabiximols, and Other Blends of Cannabinoids
Placebo-Controlled Trials
A double-blind RCT randomized 51 inpatients treated for cannabis dependence with nabiximols (86.4 mg THC and 80 mg cannabidiol) or placebo for 6 days to alleviate withdrawal symptoms. The symptoms were measured with the Cannabis Withdrawal Scale 74 ; with a subscale from the Depression, Anxiety, and Stress Scale 75 ; and with the Anxiety Sensitivity Index–Revised, 76 as well as retention from withdrawal and adverse events. Adjusting for covariates, nabiximols use was significantly associated with reductions in withdrawal-related irritability and depressive symptoms. Positive effects on sleep, anxiety, appetite loss, physical symptoms, and restlessness were also observed but less pronounced and nonsignificant. Treatment participation was longer in the nabiximols group but self-reported cannabis use, cannabis-related problems, cannabis dependence, and adverse events frequency and severity did not differ from placebo. 77
An initially open-label “N of 1” trial, which then converted to a randomized, double-blind, placebo-controlled, crossover study of 34 patients, examined the effects of sublingually applied medicinal cannabis extracts for chronic pain. Patients received either THC 2.5 mg, cannabidiol 2.5 mg, or a 1:1 mixture (cannabidiol 2.5 mg and THC 2.5 mg) over a 12-week period. Each extract contained >95% of the specified cannabinoid, the remainder being a mixture of other plant compounds. Twenty-four patients completed the crossover period without cannabinoid rescue medication and, in this group, there was an improvement in depression severity from a median BDI score of 16 at baseline to 8 at the end of the study. Furthermore, there were significant improvements in pain symptoms measured with the visual analogue scale with THC alone and a mixture of THC and cannabidiol groups. In addition, these patients showed improvements in the total scores of the General Health Questionnaire 28 78 as well as in each subdomain of this instrument (Severe Depression, Anxiety, and Insomnia, Somatic Symptoms and Social Dysfunction). 79
In an 8-week crossover, double-blind, placebo-controlled, randomized controlled trial, 17 patients with multiple sclerosis were given nabiximols (THC 2.7 mg and cannabidiol 2.5 mg) to evaluate psychopathological and cognitive effects. 80 The Symptom Checklist–90 Revised 81 showed no differences in depressive symptoms between the active and the placebo period. In addition, no correlations were found between the plasma levels of cannabidiol and the depressive symptoms. 80
Another randomized, double-blind, placebo-controlled, parallel-group trial examined 160 patients with multiple sclerosis who were given cannabis-based medicinal extracts with a THC/cannabidiol ratio of 1:1 (max dose 120 mg/day each) over a 6-week period. Among other outcome measures, the BDI showed no significant difference between the placebo and the cannabis-based medicinal extracts group after the trial. 82
A large randomized, double-blind, placebo-controlled, graded-dose, parallel-group trial with 360 opioid-treated cancer patients with poorly controlled chronic pain observed the effects of nabiximols (cannabis extract containing 2.7 mg of THC and 2.5 mg of cannabidiol) on their pain. 83 Patients were excluded from study participation if they had a major psychiatric disorder. The patients were split into three dosage groups and assessed after 4 weeks. There were no significant differences between groups regarding depressive symptoms assessed with the Montgomery-Åsberg Depression Rating Scale (MADRS). 83,84
Surveys and Observational Studies
An Internet survey aimed to investigate the reported reasons for self-described cannabidiol use in 2,490 cannabis users. A questionnaire was developed to assess broad characteristics of self-described cannabidiol users and consisted of structured questions answered by either yes/no or multiple-choice responses. Questions included reasons for use, duration and frequency of use, method of administration, perceived clinical efficacy, and adverse effects. Both anxiety and depression were commonly self-reported reasons for cannabidiol use, with around 380 participants reporting use of cannabidiol for treatment of depression. In addition, the survey investigated the self-report treatment efficacy asking how well the user felt cannabidiol treated their medical conditions. In this regard, around 250 participants reported that cannabidiol treated their symptoms “very well by itself” or “moderately well by itself” (the other two possible responses were “well in combination with conventional medicine” and “not very well”). 85
In an observational study, 120 cannabis smokers (recreational and daily users) were examined with regard to the THC and cannabidiol content in their consumed cannabis (measured in hair samples) in relation to cognition, psychotic-like symptoms, and psychological well-being. All participants were tested twice, when intoxicated and when nonintoxicated. Depressive symptoms were examined with the BDI and anxiety symptoms with the STAI. Overall, users of high THC cannabis, which tended to contain no or less cannabidiol than low THC cannabis, had significantly higher depression and anxiety scores than the low THC group, independent of daily or recreational use. 86
A Web-based cross-sectional study evaluated 1,877 cannabis users with respect to the type of cannabis (including THC/cannabidiol ratios) consumed and their subclinical psychiatric experiences. This was achieved through the use of the CAPE. High cannabidiol contents in the consumed cannabis were statistically associated with fewer positive psychotic symptoms. The scores of negative psychotic symptoms and depressive symptoms were smaller in the high-cannabidiol group than in the high-THC group, but the difference was not significant. 87
A large longitudinal naturalistic study examined 1,399 medical cannabis users with regard to the change in mood after consumption through a self-report app called Strainprint™, in which the user could rate the severity of their symptom on a scale of 0 (none) to 10 (extreme). Participants had to inform the reasons they were using cannabis through a list of health conditions or symptoms. There was a significant reduction in ratings of depression from before to after using cannabis, with the greatest reduction in ratings of depression being reported after using cannabis with relatively low levels of THC and high levels of cannabidiol. Furthermore, analyses examining changes in perceived efficacy of cannabis across time revealed no significant perceived tolerance effects on depression. 88
Discussion
Current Evidence
To the best of our knowledge, this is the first study to systematically review the current evidence of cannabidiol as a treatment for mood disorders and affective symptoms associated with other health conditions. We did not find any published clinical trial investigating the efficacy of cannabidiol in patients with mood disorders. In other health conditions, there were no studies that examined affective symptoms as a primary outcome, although 10 clinical studies evaluated affective symptoms, mainly depressive, as secondary outcomes. In general, these studies showed a good safety profile of cannabidiol, without inducing dysphoria, anxiety, or psychotic symptoms and with few adverse effects. 52,77,79,82 However, regarding the mood symptoms, the results were conflicting, showing both significant improvements in depressive symptoms and absence of significant differences. 54,56,64,77,79,80,82,83 These results are probably due to the methodological differences among the studies.
Our review revealed considerable heterogeneity and methodological variations among the trials in several aspects. Among the eight studies including at least a phase of randomization and blinding, one evaluated the effects of pure cannabidiol or THC in healthy participants and the affective symptoms assessed were dysphoria and euphoria. 52 All the others assessed depressive symptoms, but in patients with different health conditions. 54,56,77,79,80,82,83 Furthermore, some studies investigated pure cannabidiol, 52,54,56,79 and others evaluated combinations of cannabidiol and THC. 77,80,82,83 The daily doses of cannabidiol also varied among the studies, ranging from 2.5 mg to 600 mg/day. In terms of clinical rating scales, the clinician-rated MADRS, 83 a rating scale widely used as an outcome measure in RCTs of mood disorders, was used in only one study while the self-rated BDI was used in three studies, 54,79,82 and different measures were used in remaining studies. 52,56,77,80
The methodology also varied among studies with other levels of evidence. For example, an open-label trial and a case report assessed depressive symptoms in cannabis use disorder through BDI. 62,73 Only one of the open-label studies testing purified cannabidiol assessed symptoms with the POMS. 64 A cross-sectional study that assessed the concentration of cannabidiol in cannabis used by evaluating hair samples included BDI as an outcome. 86 Similarly, in another survey investigating depressive symptoms, the cannabidiol content was estimated by getting information from the subjects about the type of cannabis consumed instead of an objective measurement. 87 A cross-sectional study assessed the reasons for use and self-reported perception of improvement of depressive symptoms in people using only cannabidiol, but the symptoms were assessed without a validated scale. 85 The only longitudinal study investigated the concentration of cannabidiol based on the type of cannabis consumed and rated the depressive symptoms through a self-report app, without any validated scale, although it showed improvement in symptoms. 88 The only study done in patients with mania was a case series that included two patients with bipolar disorder and assessed whether cannabidiol monotherapy could maintain the improvement achieved with an initial combination of cannabidiol and olanzapine in the first patient and whether cannabidiol alone could improve the manic symptoms in the second patient. 67 Considering all these aspects, the results were, as expected, diverse. Some papers showed no difference while others demonstrated improvements in depressive symptoms. Because of this conflicting evidence, it is essential to discuss the studies investigating the relationship between the endocannabinoid system and mood disorders to try to understand the possible role of cannabidiol as a treatment for bipolar disorder and MDD.
Clinical evidence linking the endocannabinoid system and mood disorders
As presented before, the endocannabinoid system has its receptors and ligands present in neurons and immune cells, an interesting fact in the context of mood disorders considering the interplay between the immune and the nervous system in their pathophysiological basis. Apart from that, studies evaluating specifically the relations of endocannabinoid system and affective symptoms and emotional functions are of interest too. For instance, both preclinical and clinical studies have shown the role of endocannabinoid system components on reactions to traumatic events 89,90 as well as its relation with emotional processing. 91 In addition, some clinical studies also showed the endocannabinoids as markers of improvement in schizophrenia and mood disorders. 17,92 Besides, it is well known that cannabis use affects the endocannabinoid system, leading to a wide variety of neurobiological processes, including brain development and emotions as well as other neuropsychiatric functions. 93,94
Notwithstanding these theoretical digressions and experimental evidence, the clinical impact of cannabis use on mood disorders is well-known and indirectly shows the relationship of these illnesses and the endocannabinoid system. For example, longitudinal studies have associated cannabis use during adolescence with later development of MDD, depressive symptoms,
95,96
and suicidal ideation.
97
–100
In addition, a prospective study also suggested that adolescent cannabis use may be an independent risk factor for the development of hypomania in young adulthood.
101
Although there is uncertainty about causal effects, in general, recent cannabis use is associated with more severe symptoms (manic, depressive, and especially anhedonia) and long-term cannabis use is associated with negative outcomes such as higher rates of recurrence in mood disorders.
102,103
Particularly, cannabis use in bipolar disorder is associated with earlier onset of illness, psychotic symptoms, suicide, and misuse of other substances.
104
Although these can be considered outcomes related to the use of cannabis, it is essential to note that these results are derived from cross-sectional studies which are not conducive to causal inference. In addition, it important to note that, as mentioned earlier, the plant
Potential use of cannabidiol in mood disorders
Considering the evidence discussed so far, what would be the potential benefits of further testing the efficacy of cannabidiol as a treatment in mood disorders? Cannabidiol represents a potentially novel therapeutic strategy for depression, with mechanism of action distinct from the traditional antidepressant medication which target the monoaminergic pathways. 13,14,45 It is well established that about a third of patients with MDD are treatment resistant to such medications; cannabidiol could be a new therapeutic approach for this population either as monotherapy or an augmentation strategy. 105,106 Regarding bipolar disorder, a medication with anxiolytic, 19 –21 anticonvulsant, 15,16 and antipsychotic 17,18 properties could help reduce the polypharmacy so common in this illness. 107,108 Furthermore, the neuroprotective effects of cannabidiol, widely described in the literature, 12,44,109 –112 could help arrest the neuroprogression that occurs over the long-term in bipolar disorder. 113,114 Given the excellent tolerability of cannabidiol, this might be more acceptable for bipolar disorder in the early stages of the illness and in those at high-risk for BD, where tolerability and treatment acceptability are very important considerations. 115 –117
An important potential advantage of testing cannabidiol in mood disorders is its well-known effect on anxiety, including physiological anxiety, 52,118,119 social phobia, 19,120 and even anxiety associated with heroin abstinence. 121 It can be useful for populations with mood disorders, since 45.7% of patients with MDD and 34.7% of patients with bipolar disorder have a comorbid diagnosis of anxiety disorder and many more have anxiety symptoms associated with depressive episodes. 122,123 In addition, comorbid anxiety disorders have been associated with pernicious outcomes in mood disorders, such as lower remission rates of depressive symptoms, increased suicidal ideation, and occupational disabilities. 123 –126 However, treatment of anxiety in mood disorders challenging and therapeutic options are limited, particularly in the light of the safety concerns such as the risk of dependence and cognitive impairment associated with benzodiazepines 127 –130 and the risk of manic switches in the case of antidepressants when prescribed to patients with bipolar disorder. 131,132
Conclusions and Future Directions
A strength of this systematic review was the search strategy employed, including a broad range of search terms and databases. However, the review also has limitations. The synthesis of existing studies did not lend itself to a meta-analysis due to the lack of consistent outcome measures and rating scales used across studies. This highlights the fact that there is a lack of clinical evidence about the potential role of cannabidiol as a treatment of mood disorders. Given this lack of evidence, it is not surprising that cannabidiol is not recommended as a treatment for mood disorders according to the treatment guidelines, for example, the Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines for treatment of MDD 133 and the CANMAT and International Society for Bipolar Disorders guidelines for the management of patients with bipolar disorder. 134
Nevertheless, considering that evidence from preclinical studies suggests antidepressants effects and that well-conducted RCTs in other health conditions have shown antipsychotic, anticonvulsant, and anxiolytic effects, as well as a favorable adverse effects profile, cannabidiol may still have a role in the treatment of mood disorders. Hence, there is an urgent need for well-conducted RCTs of cannabidiol in the treatment of acute episodes of MDD and bipolar depression. In this sense, future studies in this field could start with studies testing cannabidiol as add-on drug, similar to what was done in schizophrenia, 18 and then, depending on the results, this could be tested further for its utility as monotherapy for mood disorders.
Another important point for future studies is defining a dose window for mood disorders since studies have shown an inverted U-curve profile of cannabidiol 20,21 and the doses used in clinical trials vary between anxiety disorders, 19,120 schizophrenia, 17,18 and epilepsy. 15,16 In addition, despite its general safety and good tolerability profile, the studies testing cannabidiol in epilepsy have shown critical drug–drug interactions with medications commonly used to treat mood disorders, such as benzodiazepines, valproate, and other anticonvulsants. 135,136 This fact highlights the necessity for careful drug monitoring when testing cannabidiol in mood disorders, especially bipolar disorder. In this regard, it is noteworthy that, given the risks associated with THC in particular in the induction of psychosis 93,137 and mania, 101,103 future studies should focus primarily on pure cannabidiol produced according to Good Manufacturing Practices rather than medicinal cannabis or other products containing blends of cannabinoids. However, even products purportedly containing only cannabidiol should be carefully evaluated, as there are many artisanal cannabidiol oil extracts that may contain unknown amounts of this substance as well as other cannabinoids, especially THC, which may worsen the outcomes. 138 –140 It is critically important that clinicians be aware of this fact when providing education to patients.
Supplemental Material
Supplemental Material, 19039-d-CJP-2019-218-SR.R1-_abstract_for_translation - Cannabidiol as a Treatment for Mood Disorders: A Systematic Review
Supplemental Material, 19039-d-CJP-2019-218-SR.R1-_abstract_for_translation for Cannabidiol as a Treatment for Mood Disorders: A Systematic Review by Jairo Vinícius Pinto, Gayatri Saraf, Christian Frysch, Daniel Vigo, Kamyar Keramatian, Trisha Chakrabarty, Raymond W. Lam, Márcia Kauer-Sant’Anna and Lakshmi N. Yatham in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Data available upon request.
Acknowledgments
JVP acknowledges the financial support by the National Council for Scientific and Technological Development, Ministry of Science and Technology, Brazil (
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LNY has been on speaker/advisory boards for or has received research grants from Alkermes, AstraZeneca, Bristol Myers Squibb, Canadian Network for Mood and Anxiety Treatments (CANMAT), Canadian Institutes of Health Research (CIHR), DSP, Eli Lilly, GlaxoSmithKline, Janssen, the Michael Smith Foundation for Health Research, Pfizer, Servier, Sunovion, and the Stanley Foundation. MK-S has received consulting or speaking fees from Daichii Sankyo and Shire. RWL has received ad hoc speaking/consulting fees or research grants from Akili, Allergan, Asia-Pacific Economic Cooperation, BC Leading Edge Foundation, CANMAT, Canadian Psychiatric Association, CIHR, CME Institute, Hansoh, Healthy Minds Canada, Janssen, Lundbeck, Lundbeck Institute, Medscape, Mind.Me, MITACS, Ontario Brain Institute, Otsuka, Pfizer, St. Jude Medical, University Health Network Foundation, and VGH-UBCH Foundation. DV, GS, JVP, KK, and TC report no biomedical financial interests or potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JVP has received a scholarship from CNPq, Brazil. MK-S has received research grants from CNPq and CAPES, Brazil.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
