Abstract
Objectives:
To evaluate the cost-effectiveness of repetitive transcranial magnetic stimulation (rTMS) and electroconvulsive therapy (ECT), and combining both treatments in a stepped care pathway for patients with treatment-resistant depression (TRD) in Ontario.
Methods:
A cost–utility analysis evaluated the lifetime costs and benefits to society of rTMS and ECT as first-line treatments for TRD using a Markov model, which simulates the costs and health benefits of patients over their lifetime. Health states included acute treatment, maintenance treatment, remission, and severe depression. Treatment efficacy and health utility data were extracted and synthesized from randomized controlled trials and meta-analyses evaluating these techniques. Direct costing data were obtained from national and provincial costing databases. Indirect costs were derived from government records. Scenario, threshold, and probabilistic sensitivity analyses were performed to test robustness of the results.
Results:
rTMS dominated ECT, as it was less costly and produced better health outcomes, measured in quality-adjusted life years (QALYs), in the base case scenario. rTMS patients gained an average of 0.96 additional QALYs (equivalent to approximately 1 year in perfect health) over their lifetime with costs that were $46,094 less than ECT. rTMS remained dominant in the majority of scenario and threshold analyses. However, results from scenarios in which the model’s maximum lifetime allowance of rTMS treatment courses was substantially limited, the dominance of rTMS over ECT was attenuated. The scenario that showed the highest QALY gain (1.19) and the greatest cost-savings ($46,614) was when rTMS nonresponders switched to ECT.
Conclusion:
From a societal perspective utilizing a lifetime horizon, rTMS is a cost-effective first-line treatment option for TRD relative to ECT, as it is less expensive and produces better health outcomes. The reduced side effect profile and greater patient acceptability of rTMS that allow it to be administered more times than ECT in a patient’s lifetime may contribute to its cost-effectiveness.
Introduction
Major depressive disorder (MDD) is the most prevalent mental illness in Canada, estimated to be as high as 17%. 1 More than 50% of MDD patients fail to remit after first-line therapy (e.g., psychotherapy, pharmacotherapy, or both). 1 After two failed treatments, patients are classified as having treatment-resistant depression (TRD). 2 While this is the most widely accepted definition of TRD, there is variation in the definition and clinicians may refer to TRD as “chronic” or “complex” depression. 3 Consequences of TRD range from decreased quality of life, decreased labor force participation, and increased medical resource utilization. 1,4,5 TRD represents at least 21% of all Ontario MDD cases, 6 with some estimates as high as 30–60%. 6 American studies suggest TRD could present additional annual societal costs of $29-48 billion in addition to costs associated with MDD. 1
There are myriad pharmacologic and nonpharmacologic treatment options for depression that patients and clinicians must navigate, and there is a need for clear evidence to guide these decisions. Given pharmacologic treatments have limited effectiveness for TRD, 7 clinicians must explore other options.
Electroconvulsive therapy (ECT) is the most effective treatment for TRD, but it requires 6–18 treatments under general anesthesia. 8 ECT reduces psychiatric hospitalization rates, long-term suicide risk, and all-cause mortality. 9 However, ECT is also associated with significant side effects and adverse events including nausea, headache, muscle pain, oral lacerations, dental injuries, and persistent myalgia. 6 The neurocognitive side effect profile includes disorientation and impaired short-term memory function during an ECT acute course. 10 Because of these side effects and the persistent stigma associated with ECT, many patients who qualify for the treatment refuse it. 6
Another recently emerging treatment option for TRD is repetitive transcranial magnetic stimulation (rTMS). rTMS is a noninvasive treatment that delivers magnetic stimulation to specific regions in the brain. rTMS has been shown to induce clinically meaningful reductions in depression in >50% of patients. 10 rTMS is associated with reduced side effects and reduced costs for the health-care system, as it is completed almost entirely on an ambulatory basis without the need for anesthesia. 11 rTMS is not associated with cognitive impairment and may actually improve cognitive functions including both short- and long-term memory. 12 Studies demonstrate rTMS reduces health-care resource utilization following treatment. 13
Many previously published cost–utility analyses of ECT and rTMS have found ECT to be more cost-effective. 6,14 –17 However, these studies failed to consider costs associated with disability or unemployment and only considered direct health-care costs. Moreover, costs and treatment outcomes beyond 1 year were not evaluated. One exception is a study from Singapore by Zhao and colleagues, which took a societal perspective wherein all costs irrespective of payer are considered, and found rTMS to be more cost-effective. 18 Currently, there are no published North American cost–utility analyses of rTMS and ECT that account for societal costs.
This study sought to evaluate the cost-effectiveness of rTMS relative to ECT as a first-line treatment for TRD from a societal perspective. Due to the chronic and relapsing nature of TRD, a lifetime horizon is most appropriate as it tracks patients across their lifetime, evaluating the multiple iterations of treatment that represent clinical reality for TRD patients. Determining the most cost-effective treatment pathway for TRD patients can assist decision makers in efficiently distributing scarce health-care resources while maximizing patient outcomes.
Methods
A decision analytic model compared the cost-effectiveness of rTMS to ECT using TreeAge Pro (Version 2018) software. 19 We used a Markov model, which is a state-transition model that uses probabilities to transition patients from one health state to another at set time intervals. Our model was a microsimulation, which tracks individual patient characteristics including their age and number of treatments received, contrasting with cohort models that only track aggregate cohort changes.
Model Structure
The simulated cohort consisted of 10,000 individuals whose age when entering the model was distributed according to the 2017 adult age distribution of the Ontario population. 20 The model ran in 6-month intervals for a maximum of 164 cycles (82 years), when the entire cohort had died. Modeled health states included remission, acute treatment, maintenance treatment, severe depression, and death. ECT acute treatment consisted of 15 sessions, including tapering, and ECT maintenance treatment consisted of 1 session per month, for a total of 6 sessions. rTMS acute treatment consisted of 42 sessions, including tapering, and rTMS maintenance consisted of 1 session every 2 weeks, for a total of 12 sessions (Figure 1). 6 Current rTMS maintenance therapy protocols vary with respect to frequency and duration, and solid evidence to guide practice is lacking. 21
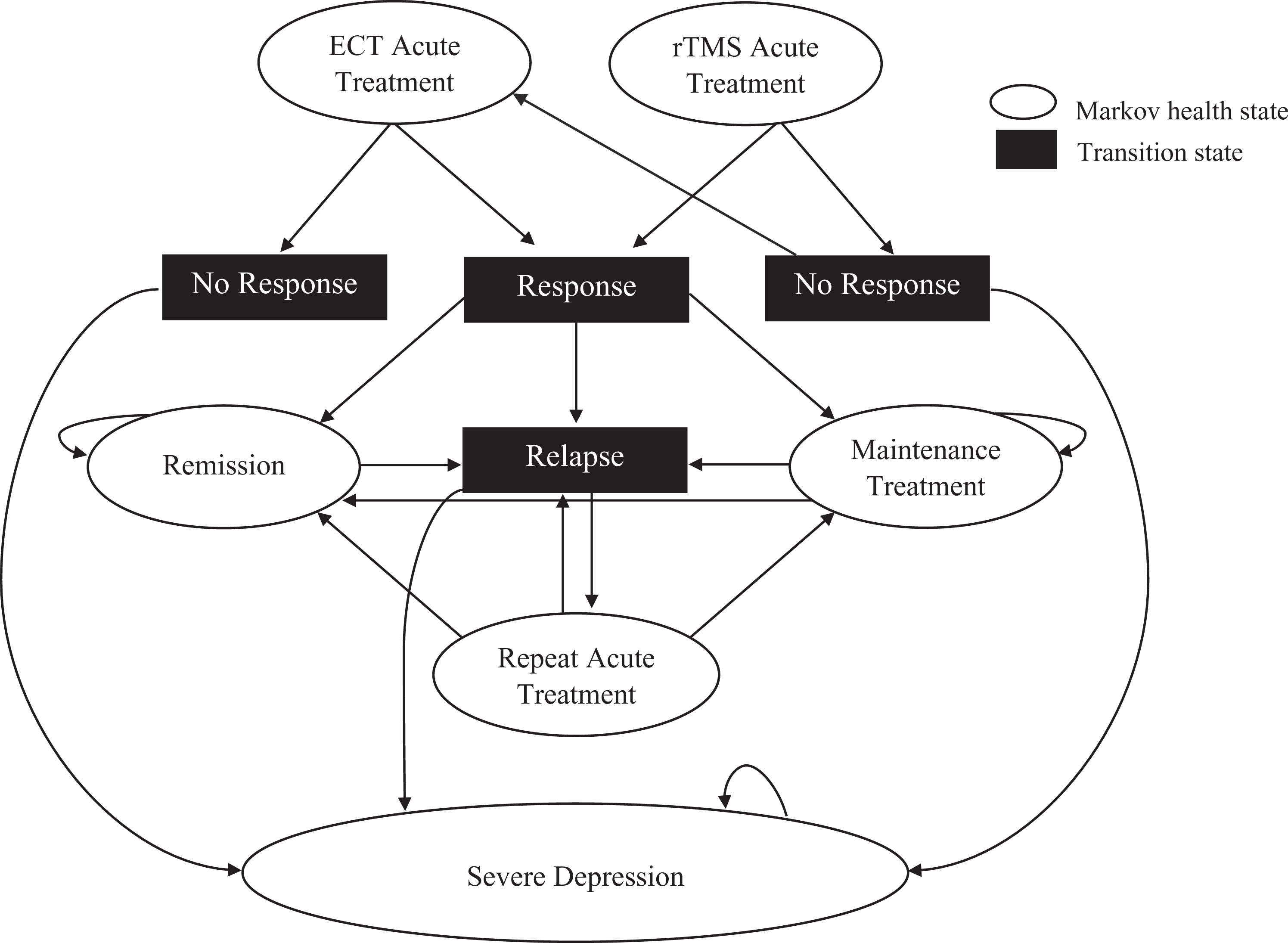
Simplified depiction of the Markov decision analytic model. Costs and health utilities are only calculated in Markov health states. The pathway from relapse to severe depression is contingent on patients reaching their maximum lifetime treatments. Death is not depicted. ECT = electroconvulsive therapy; rTMS = repetitive transcranial magnetic stimulation.
The same cohort of patients was run through both ECT and rTMS as first-line treatment options. Those who failed to respond initially to ECT did not receive further stimulation therapy (as evidence-based treatment options are lacking for these individuals) and remained in a state of severe depression (which included pharmacologic treatment) for the remainder of the simulation. Those who failed to respond initially to rTMS could switch to ECT, representing a stepwise clinical trajectory from less to more invasive treatments.
Patients transitioned between health states at 6-month intervals according to transition probabilities in the model. Within each health state, the patient incurred the costs associated with that state and a quality-of-life measure that reflects the patient’s health and functionality, called a health utility. A half cycle correction was applied to account for bias associated with health state costs and outcomes not being applied randomly during health state transitions. 22 Our microsimulation imposed a maximum limit of 12 courses of acute treatment for rTMS and 4 courses of acute treatment for ECT over a patient’s lifetime. This 3:1 ratio is based on greater side effects and lower patient acceptability 23 of ECT compared to rTMS. This estimate’s impact on the results was tested during scenario analyses where adjustments to the limits were investigated. Upon reaching this limit, treatment was presumed to have failed, and patients remained in the severe depression state for the remainder of the simulation.
Model Inputs Overview
The model used three types of data: transition probabilities, health utilities, and costs. Transition probabilities were derived from clinical trial efficacy data and determined the movement between health states at the end of each 6-month Markov cycle. Health utilities were derived from recently published cost–utility analyses and represented the health-related quality-of-life (HRQoL) associated with each health state. Costs were derived from a variety of sources including recently published cost–utility analyses 16,18 and government costing databases. 24 –28 These included direct treatment costs, transportation costs, unemployment costs (e.g., lost wages, unemployment insurance), and disability insurance. A global discount rate of 1.5% annually was applied to all costs and health outcomes (measured in quality-adjusted life years [QALYs]) in the model, as recommended by the Canadian Agency for Drugs and Technology in Health guidelines (Table 1). 29
Model Input Parameters.
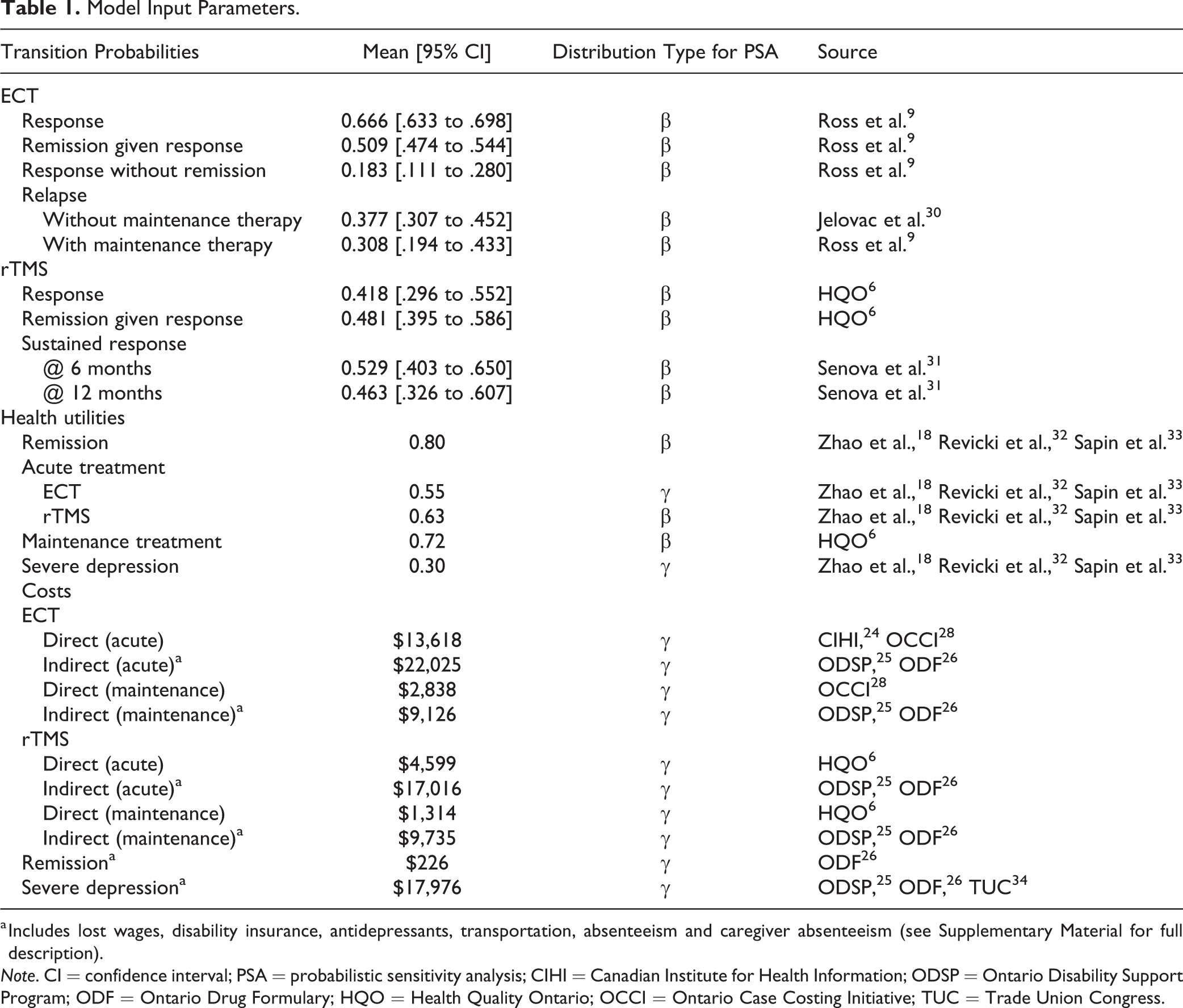
a Includes lost wages, disability insurance, antidepressants, transportation, absenteeism and caregiver absenteeism (see Supplementary Material for full description).
Note. CI = confidence interval; PSA = probabilistic sensitivity analysis; CIHI = Canadian Institute for Health Information; ODSP = Ontario Disability Support Program; ODF = Ontario Drug Formulary; HQO = Health Quality Ontario; OCCI = Ontario Case Costing Initiative; TUC = Trade Union Congress.
Transition Probabilities
Transition probabilities used were remission, response without remission, relapse, and death. Our model assumed all patients who responded to treatment without attaining remission would receive maintenance therapy. We assumed all patients who relapsed would receive another acute treatment (until their lifetime maximum was reached). ECT response, remission, and relapse probabilities following maintenance therapy were obtained from a study by Ross and colleagues. 9 Relapse rates for those not receiving maintenance therapy came from a meta-analysis by Jelovac. 30 Response and remission rates for rTMS were taken from a health technology assessment (HTA) completed by Health Quality Ontario (HQO), 2014. 6 A study by Senova and colleagues reported rTMS sustained response rates. 31 We assumed patients who did not maintain sustained response (i.e., relapsed) would receive another course of acute treatment.
Mortality risk was applied to all patients at the end of each cycle and was age-adjusted based on Canadian mortality data. 20 This mortality risk was increased by 29% for all individuals not in remission to account for the increased all-cause mortality risk associated with TRD. 35 –37
Costs
ECT treatment costs were derived from the Canadian Institute for Health Information 24 and the Ontario Case Costing Initiative 28 and reported in 2018 Canadian dollars ($1.00CAN = $1.296US) 38 using the general Consumer Price Index to ensure all costs were reported in common units. rTMS treatment costs were obtained from HQO’s 2014 report, 6 which estimated the per session cost at $109.52, including nursing time, equipment costs, and psychiatric expertise, and was validated through comparison with a University of Calgary HTA of rTMS. 39 No inpatient costs were included for rTMS. With 42 treatments during an acute phase (including tapering), the total direct costs were $4,599.84.
Additional indirect costs incurred by the patient, government, or society include absenteeism, transportation, lost wages, long-term disability (LTD) payments, and antidepressant medication costs. An estimated 54% of TRD patients are unemployed because of their condition, 40 and many are receiving disability benefits from the Ontario Disability Support Program (ODSP) and/or relying on support from friends or family. To account for these costs, lost wages associated with unemployment and benefits paid by ODSP were built into the model, based on the average Ontario salary 20 and average ODSP benefits received by those with depression. 25,41 Patients in more functional health states, including remission and maintenance therapy, incurred reduced productivity costs due to increased labor force participation. 40 Patients receiving acute treatment and patients who remained severely depressed after unsuccessful treatment continued to incur significant productivity costs due to low labor force participation rates for these patients (26.2%). 34 Antidepressant medication costs were estimated as the average of all antidepressants listed in the Ontario Drug Formulary 26 and were factored into all health states, assuming all TRD patients, regardless of their health state, would continue lifelong pharmacotherapy. Transportation costs were estimated at $25 per person per trip to and from the hospital or clinic. For ECT outpatient treatments, transportation costs included two people per session, since use of anesthesia requires ECT patients to bring a companion. The cost for the companion to take a half day off work to accompany the patient was also factored in for 50% of ECT outpatients. For rTMS, only transportation costs for the patient were included, as no companion is required. See Supplementary Material for full description of costs.
Health Outcomes
Health utility scores reflected the HRQoL of patients in various health states and were used to estimate treatment efficacy. Our model used the standard measure for health utilities, the QALY. QALYs range from 1 (perfect health) to 0 (death) and are determined according to patient preferences. Health utility scores were derived from previously published cost–utility analyses comparing ECT and rTMS. 6,18 The severe depression health state had the lowest utility score in our model (0.30), which occurred after treatment options were exhausted. Based on the side effects, 6 decreased cognitive performance experienced during and immediately after acute ECT treatment, 42 and low patient acceptability 6,23 suggesting a patient preference to remain depressed rather than receive ECT, those receiving ECT also had a health utility of 0.30 during the 6-week period of the acute phase treatment. For the remainder of the 6-month Markov cycle for the ECT acute phase health state, the health utility was equal to that of rTMS acute treatment, 0.63, giving the ECT acute phase health state a weighted average health utility of 0.55.
Threshold Analyses
Threshold analyses were conducted to determine how much each input parameter needed to change to alter the base case results. To determine the sensitivity of the model to the costs and health utilities for each health state, the model was run with variations on these values.
Probabilistic Sensitivity Analysis (PSA)
A PSA was used to model uncertainty around input parameters. Each parameter was assigned a range of values based on reported confidence intervals and assigned a distribution for the model to randomly sample from. Transition probabilities and health utilities used a β distribution based on confidence intervals reported in the source literature. Costs varied according to a γ distribution using a standard deviation of 10% of the mean value for each health state.
Scenario Analyses
Scenario analyses tested model assumptions and their impact on overall results. Previous research demonstrated that an rTMS to ECT pathway may be most cost-effective, 43 so we evaluated the impact of removing ECT as a secondary treatment option for rTMS nonresponders. Current policy aims to conduct more ECT on an outpatient basis, so we adjusted ECT costing to have all treatments conducted as outpatient. Recent research suggests rTMS maintenance therapy efficacy may improve to eventually match that of ECT maintenance therapy, 21,44 so we tested our results with this adjustment. The model was also run with adjustments to the time horizon (1- and 2-year time horizons instead of lifetime), global discount rate (3.5% 45 and 5% 29 ), the removal of all indirect costs, and changing the maximum number of lifetime treatments allowed for rTMS from 12 to 4, equal to that of ECT.
Results
The rTMS pathway was less expensive and more effective as a first-line treatment than the ECT pathway in the base case scenario. rTMS patients gained an average of 0.96 additional lifetime QALYs (equivalent to about 1 year in perfect health) and had lifetime costs (in 2018 Canadian dollars) that were $46,094 lower than ECT.
Threshold analyses results found that when costs and health utilities were adjusted within a range that exceeded realistic values (based on previous studies and expert consultation), rTMS continued to dominate as the more cost-effective option (see Table 2). The model showed some sensitivity to the costs of acute phase treatment. However, for ECT to no longer be dominated, its cost of acute treatment needed to be reduced from $35,644 to $13,515. Even if all ECT were conducted on an outpatient basis, productivity costs associated with ECT alone exceed this value. ECT was also no longer dominated when the cost of rTMS acute treatment increased 1.5 times from $21,616 to $30,992. In both cases, even though ECT was no longer dominated, rTMS remained the more cost-effective option. PSA results demonstrated that when the estimates for all transition probabilities, health utilities, and costs were sampled from a distribution around a mean value, rTMS remained dominant in all cases (Figure 2).
Threshold Sensitivity Analysis for Health State Costs and QALYs.
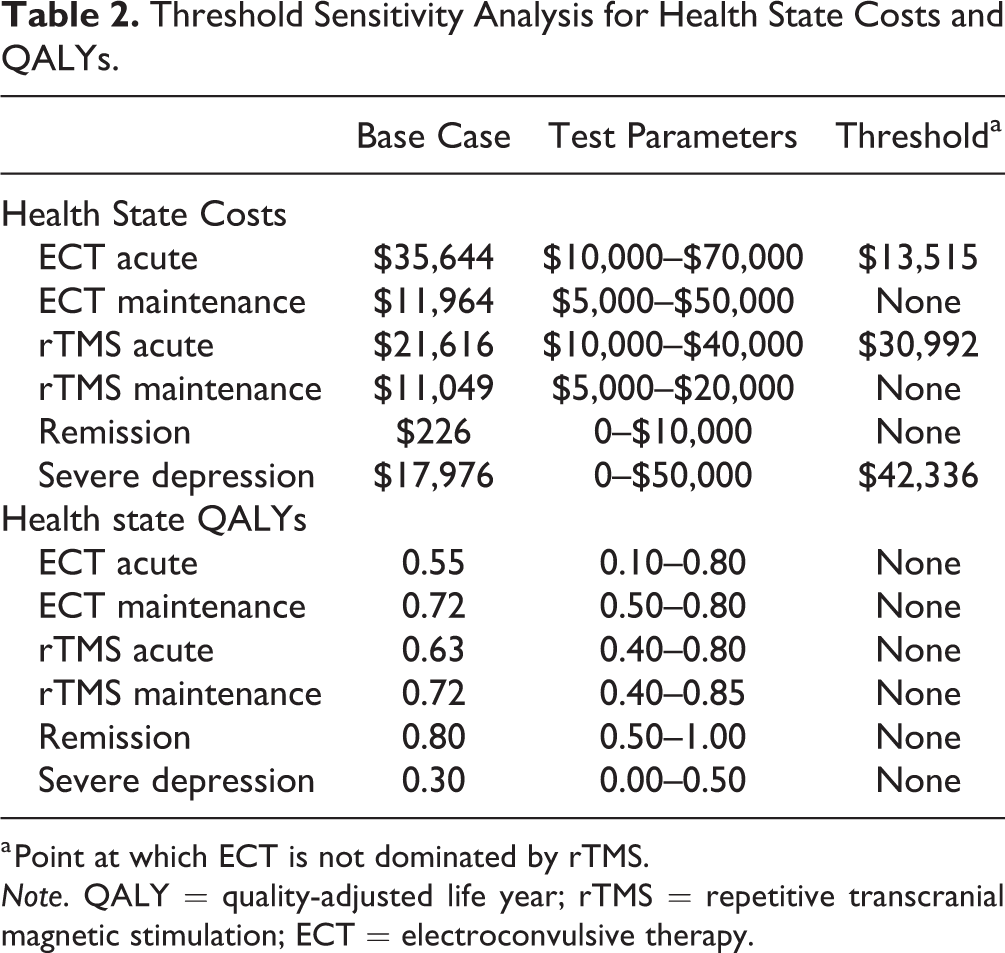
a Point at which ECT is not dominated by rTMS.
Note. QALY = quality-adjusted life year; rTMS = repetitive transcranial magnetic stimulation; ECT = electroconvulsive therapy.
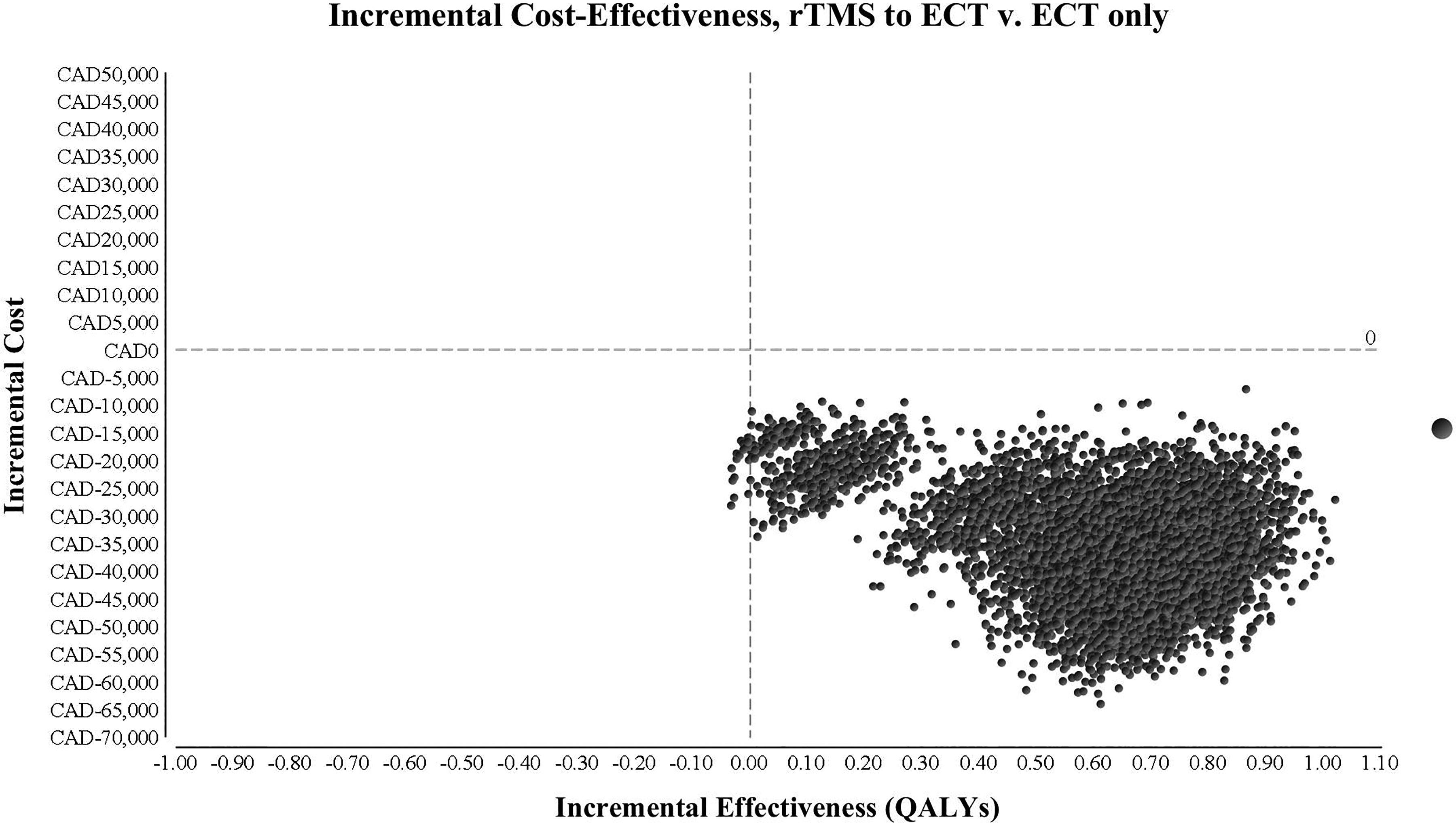
Incremental cost of rTMS, results from probabilistic sensitivity analysis (10,000 iterations). Plot shows that rTMS is less costly and more effective than ECT. ECT = electroconvulsive therapy; rTMS = repetitive transcranial magnetic stimulation; QALY = quality-adjusted life year.
Scenario analyses demonstrated that rTMS remained the dominant treatment in all cases, except where the maximum number of lifetime acute phase rTMS treatments was reduced to equal that of ECT. When the model was set to only allow four lifetime treatments for both ECT and rTMS, ECT had an incremental cost-effectiveness ratio of $69,886/QALY. The scenario where the highest number of QALYs were gained (1.19) and the greatest cost-savings ($46,614) were obtained was when 80% of rTMS nonresponders transitioned to ECT (in contrast to 35% in the base case; Table 3).
Scenario Analysis.
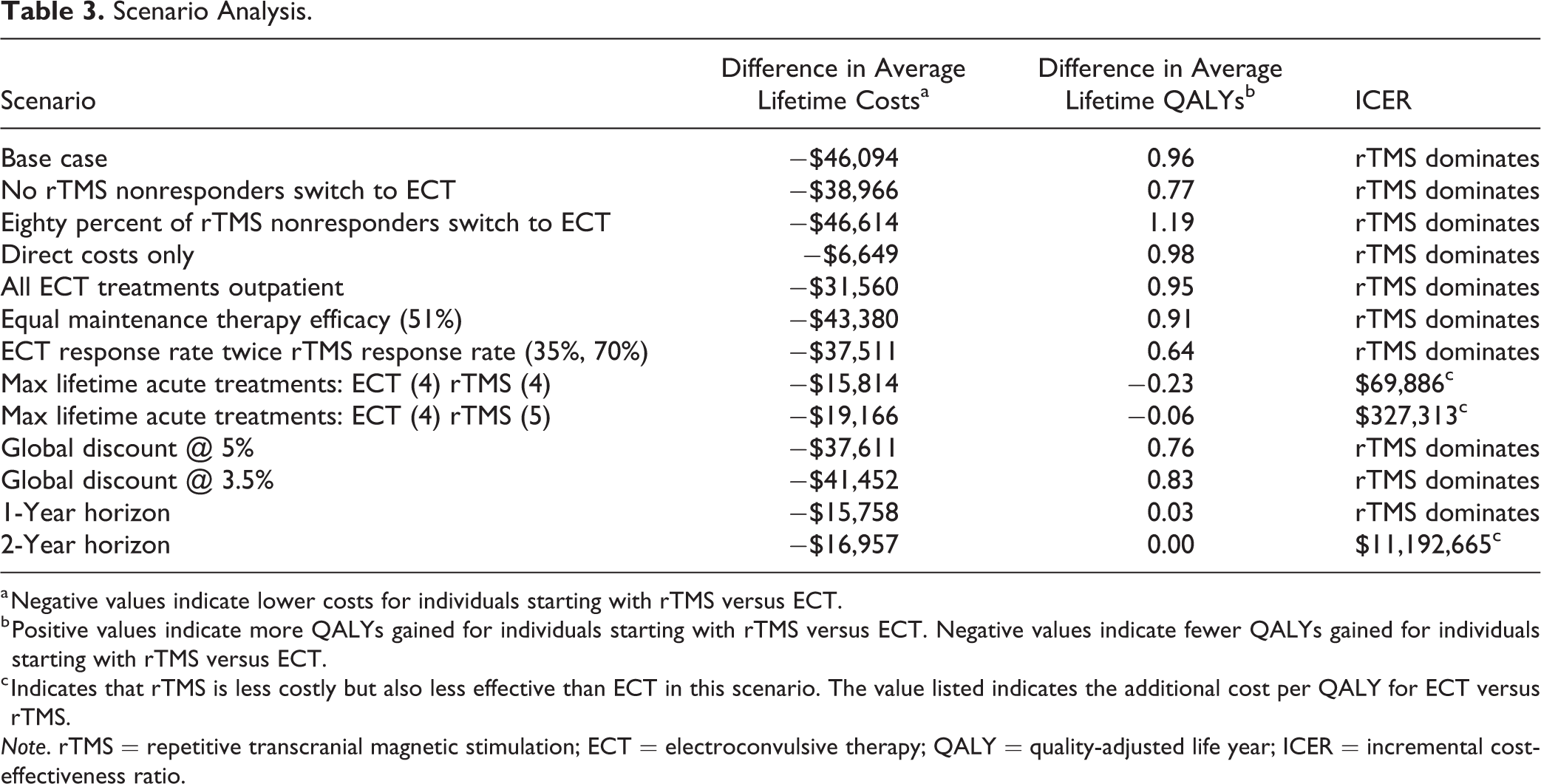
a Negative values indicate lower costs for individuals starting with rTMS versus ECT.
b Positive values indicate more QALYs gained for individuals starting with rTMS versus ECT. Negative values indicate fewer QALYs gained for individuals starting with rTMS versus ECT.
c Indicates that rTMS is less costly but also less effective than ECT in this scenario. The value listed indicates the additional cost per QALY for ECT versus rTMS.
Note. rTMS = repetitive transcranial magnetic stimulation; ECT = electroconvulsive therapy; QALY = quality-adjusted life year; ICER = incremental cost-effectiveness ratio.
Discussion
This analysis sought to determine whether rTMS was a cost-effective first-line treatment for TRD relative to ECT from a societal perspective using a lifetime horizon. Our base case results demonstrated the rTMS pathway had lower costs and produced superior health outcomes (more QALYs) than ECT over a patient’s lifetime. Results from the PSA and threshold analyses demonstrated the robustness of these findings. The scenario analyses demonstrated that the relative cost-effectiveness of rTMS is influenced by how many times it can be administered in a patient’s lifetime. The best outcomes are produced when all patients start with rTMS and then transition to ECT if they fail to respond to rTMS, suggesting the most cost-effective treatment pathway uses rTMS as a first step in treatment and ECT as the next step in a treatment pathway. However, individual patient scenarios with high symptom burden and suicidality may warrant pursuing ECT first because of the greater effectiveness and need to ameliorate these symptoms quickly.
The results of this study are notably distinct from other studies that compared these treatments using similar methodology. Vallejo-Torres et al. found ECT to be more cost-effective in Spain using a 12-month time horizon, and two UK-based studies came to the same conclusion using a 6-month time horizon. 14,15 A cost–utility analysis conducted by HQO in 2015 also found ECT to be more cost-effective using a 6-month time horizon. 6 The difference in results can be attributed to the time horizon of our model. First, given the nature of TRD as a chronic illness with very high relapse rates, limiting the model to 1 year or less does not account for the long-term costs and benefits associated with both treatments.
A second design feature unique to our model is the addition of societal costs. When a model only considers direct treatment costs, there is no accounting for the substantial differences in the economic benefits that stand to be gained when patients achieve remission or ongoing indirect costs that result from patients’ failure to remit. Patients with TRD often require disability benefits or support from friends/family to sustain day-to-day life, but in remission, they cease to incur these costs. A recent study by Zhao et al. used a societal cost perspective and found results similar to ours—that rTMS is more cost-effective. 18 Given that rTMS does not have cognitive adverse effects and does not require anesthesia, 46 it is reasonable to see how models that fail to incorporate these impacts may not represent clinical reality and may be biased in favor of ECT.
Scenario analyses demonstrated that although ECT is more efficacious than rTMS in obtaining and sustaining remission, 47 the greater acceptability of rTMS and the associated possibility of administering more courses of rTMS acute treatment in a patient’s lifetime can postpone or prevent the state of chronic unremitting depression. ECT results in patients exhausting treatment options earlier for two reasons: First, rTMS nonresponders can switch to ECT rather than entering directly into severe depression, which represents the stepwise clinical trajectory moving from less to more invasive treatments; second, patients receiving ECT can undergo fewer lifetime courses of acute treatment than rTMS due to lack of accessibility and reduced acceptability of treatment. Our sensitivity analyses demonstrated that even if the number of ECT treatments was equivalent to rTMS, the cost effectiveness of rTMS remains superior due to its reduced side effect profile and associated increased labor force participation.
Health Canada approved rTMS in 2002, but it is currently only publicly funded in Alberta, Quebec, and Saskatchewan. 6 In light of our findings, policy makers in other provinces may wish to explore the budget impact of funding rTMS as a first-line treatment option for TRD. Our analysis suggests rTMS could be used on a wider scale across Canada as it dominated ECT as a first-line treatment. 48 rTMS has the additional benefit of not requiring a speciality care setting such as a hospital, as it does not use anesthesia and is conducted entirely on an outpatient basis. This means rTMS can be conducted in community clinics, making it possible to set up clinics in rural areas that lack well-resourced hospitals. Reduced geographical barriers could make rTMS more accessible to patients, which in turn could increase the number of patients treated. The community setting also creates the potential for more rapid scalability. These factors suggest rTMS will continue to represent a cost-effective treatment option for managing TRD in Canada in the future.
Limitations of our model included the assumption that patients who respond initially to rTMS treatment cannot later switch to ECT. In clinical reality, some patients who receive multiple iterations of rTMS may switch to ECT later. 43 Our model assumed all ECT patients who either did not respond initially to ECT and those who reached their maximum number of acute treatments would remain in a state of severe depression. Clinically, patients who receive care in speciality care settings may be enrolled in clinical trials investigating new treatments for TRD. 49 The efficacy data for both treatments in the model were based on clinical trial data, which is collected from patients who lack complex comorbidities and receive treatment following protocols exactly. This may bias the data toward both treatments appearing more efficacious than they are under real-world conditions, where patients have multimorbidities and poor treatment adherence. Moreover, these studies used a strict definition for TRD, which may not map directly onto the vocabulary used by clinicians, potentially reducing the generalizability of our results to real-world practice. 3 Costing data in the model relied on point estimates from previous analyses and databases, many of which lacked associated confidence intervals, reducing our ability to accurately model the uncertainty associated with those costs. Finally, our model assumed the efficacy for both rTMS and ECT did not decrease with successive exposure to treatment. Clinical reports have shown that rTMS has a minor decrease in efficacy for each successive exposure. 50 Generalization of our results beyond Ontario should be undertaken with caution as the treatment and population data in our model were Ontario-specific.
Conclusion
Our results demonstrated that rTMS dominated ECT as a first-line treatment option for patients with TRD. Future research should investigate and measure the long-term treatment outcomes and costs for TRD patients to better model the lifetime impacts of treatment, including long-term utilization of other health-care resources by patients following each treatment, as this represented an area of uncertainty in our model. Moreover, further effectiveness trials are needed to better understand health outcomes for real-world patients, who have multimorbidities and poor treatment adherence, to better model the population-wide cost effectiveness of treatment. As evidence emerges, future research should also investigate the impact of evidence-based rTMS maintenance therapy protocols on overall cost-effectiveness.
Supplemental Material
Supplemental Material, SupplementaryData_CostingInformation_CLEAN - Cost–Utility Analysis of Electroconvulsive Therapy and Repetitive Transcranial Magnetic Stimulation for Treatment-Resistant Depression in Ontario
Supplemental Material, SupplementaryData_CostingInformation_CLEAN for Cost–Utility Analysis of Electroconvulsive Therapy and Repetitive Transcranial Magnetic Stimulation for Treatment-Resistant Depression in Ontario by Kyle P. Fitzgibbon, Donna Plett, Brian C. F. Chan, Rebecca Hancock-Howard, Peter C. Coyte and Daniel M. Blumberger in The Canadian Journal of Psychiatry
Footnotes
Authors’ Note
Kyle P. Fitzgibbon and Donna Plett are joint first authors. Additional Supplementary Material can be found in the online version of this article at the publisher’s website.
Declaration of Conflicting Interests
DMB has received research support from the CIHR, NIH, Brain Canada and the Temerty Family through the CAMH Foundation and the Campbell Research Institute. He received research support and in-kind equipment support for an investigator-initiated study from Brainsway Ltd., and he is the principal site investigator for three sponsor-initiated studies for Brainsway Ltd. He received in-kind equipment support from Magventure for investigator-initiated research. He received medication supplies for an investigator-initiated trial from Indivior. He has participated in an advisory board for Janssen. All other authors declare they do not have any conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
