Abstract
Objective:
To compare weight and glucose changes of long-term second-generation antipsychotic (SGA) monotherapy versus polytherapy (switching or combining SGAs) in children and adolescents.
Methods:
This is a 24-month retrospective study conducted between November 2005 and June 2013. From 147 antipsychotic-naive patients selected (mean age, 12.8 years; 95% confidence interval [CI], 9.8-15.9), 116 (78.9%) received SGA monotherapy and 31 (21.1%) SGA polytherapy for up to 24 months. Height, weight, and fasting glucose (FG) were measured at baseline and 1, 3, 6, 12, and 24 months. Linear mixed-model analysis was used to compare weight, body mass index z score (BMI z score), and glucose changes between the 2 SGA treatment groups, with the repeated factor being the time relative to baseline at 1, 3, 6, 12, and 24 months.
Results:
Overall, after 24 months of SGA treatment, mean weight increased significantly by 12.8 kg (95% CI, 10.4-15.0), BMI z score by 0.44 (95% CI, 0.21-0.68), and FG levels by 0.29 mmol/L (95% CI, 0.11-0.47). Incidence of overweight/obesity was 22.6%, BMI z score increase over 0.5 was 9.4%, impaired fasting glucose was 9.4%, and type 2 diabetes mellitus was 3.1%. Regarding metabolic effects, no significant difference was found between the subjects taking a single SGA and those exposed to an SGA polytherapy.
Conclusion:
Our study confirms the significant increase of metabolic complications during 24 months of SGA treatment without excluding or confirming a difference between the 2 groups of treatment (mono vs. poly).
During the past decade, increasing use of second-generation antipsychotics (SGAs) for diverse mental health disorders in children and adolescents has been widely reported in international studies. 1 –4 Canadian studies show an increase of 3- to 18-fold in the number of SGA prescriptions, which is disproportionally seen in adolescent boys but also in girls and younger children. 1,5 This increase in prevalence is due to an increased frequency and a longer duration of use. 6
Multiple studies have demonstrated adverse metabolic effects of SGAs in children and adolescents as well as in adult populations, 7 with increasing evidence suggesting a greater orexigenic effect of SGAs in youth than in adults. 8 –10
In a clinical setting, switching or combining SGAs is frequent. A Canadian study reported 31% of antipsychotic switches per year of therapy in youth, 11 similar to the results of a US adult study (29.5%). 12 In a recent systematic review, the average prevalence of antipsychotic polypharmacy (APP), defined as 2 or more antipsychotics, among antipsychotic-treated youths was 9.6% (5.9% and 12.0% among child and adolescent studies, respectively), with 77.9% of antipsychotics being SGAs. 13
This observation is a source of considerable concern because of the increased adverse effects of APP reported both in youth 14 and adult 15 populations compared to antipsychotic monotherapy and the lack of established efficacy of APP in the pediatric and adult studies. 13,15,16 Moreover, higher rates of metabolic syndrome and insulin resistance have been reported in adult patients receiving APP compared with those receiving monotherapy. 17
In pediatric populations, the metabolic side effects of SGAs in polytherapy (switching or combining SGAs) have received little attention, and no previous study has directly studied the comparison between the metabolic complications of SGA polytherapy versus monotherapy.
The aim of this study was to compare the metabolic effects of SGA monotherapy with those of SGA polytherapy in antipsychotic-naive children and adolescents in the ‘real-life’ clinical setting, while collecting descriptive data on metabolic changes occurring in children treated with SGAs for such a prolonged period. We hypothesised that the incidence of metabolic complications would be different in the SGA polytherapy group than in the SGA monotherapy group.
Methods
Study Design
The medical charts from patients in the inpatient and outpatient child, adolescent psychiatric ‘Second-Generation Antipsychotic Monitoring Program’ at Hôtel-Dieu de Lévis Hospital (Quebec, Canada) between November 2005 and June 2013 were retrospectively reviewed. This program was started in 2005 to monitor the occurrence of metabolic effects in patients using SGA treatment for the first time. The population in the geographic area served by the Department of Psychiatry of Hôtel Dieu de Lévis hospital is almost exclusively of French-Canadian origin. All study procedures were reviewed and approved by the local ethics and research committee (CER-1213-018). Patients were not required to give informed consent because this was a retrospective study using data recorded in the medical charts.
Subjects
The inclusion criteria for the study subjects were as follows: age under 18 years, no history of antipsychotic treatment, and received treatment with an SGA for at least 1 month during the study. The exclusion criteria included preexisting dyslipidemia, diabetes, or any treatment for metabolic conditions before starting the SGA.
Data Collection
For each patient, we collected the following data: age, sex, psychiatric diagnoses, SGA treatment (total dose/24 hours, duration of use), other medications (total dose/24 hours, duration of use), height, weight, and biochemical assessments (fasting blood glucose). The height, weight, and blood samples were collected at baseline and 1, 3, 6, 12, and 24 months by a clinical nurse who did the follow-up with the patient, the family, and the child psychiatrist.
Diagnoses were made by a child psychiatrist in accordance with the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM IV-TR) criteria. Clinical data were collected from the chart notes. The main diagnosis was considered the one for which the SGA was prescribed. Other diagnoses were listed as comorbid disorders. In many cases, more than one main diagnosis was recorded because SGAs were used to treat symptoms common to many diagnostics, such as disruptive behavior.
The other medications associated with SGA were grouped in 4 categories: psychostimulants (amphetamine based, methylphenidate based), atomoxetine (to more accurately discriminate the potential metabolic impact of psychostimulants), antidepressants (bupropion, citalopram, fluvoxamine, sertraline, venlafaxine), and mood stabilisers (carbamazepine, lithium, valproic acid). The short- and long-action forms of the same medication were considered together. Any medication taken in a single dose or for less than 1 day was not included.
Treatment
The patients received an SGA (olanzapine, risperidone, quetiapine, or aripiprazole) in monotherapy (the same SGA was prescribed from the beginning to the end of the period of the follow-up) or an SGA in polytherapy (patients switched from one SGA to another or used a combination of 2 SGAs, with the second SGA added between 0.5 and 24 months). The SGA switch was defined as a period of 30 days or less between the first and the second SGAs and the SGA combination as a period of more than 30 days of concomitant SGA treatment.
Measures
The variables studied for metabolic effects were weight; body mass index (BMI), calculated as weight (kg)/height squared (m2); standardised BMI z score, calculated using the children’s BMI percentile-for-age calculator from the Children’s Nutrition Research Center, Baylor College of medicine website (http://www.bcm.edu/cnrc-apps/bodycomp/bmiz2.html); and fasting glucose (FG). The exact day of weight measurement was not always available in the charts; therefore, we calculated the BMI z scores as if the patients were always weighed on the first day of the month to minimise the measurement bias. The BMI z score represents the BMI adjusted for age and sex, thus taking into account the weight gain secondary to the growth process. It allows the comparison between the BMI of children and adolescents of the same age and sex. 18
Overweight was defined as BMI z scores between the 85th and 95th percentiles and obesity as BMI z scores greater than or equal to the 95th percentile. 19 The doses of each SGA were converted in chlorpromazine equivalents. 20
Impaired fasting glucose (IFG) or hyperglycemia is defined as FG values of 5.6 to 6.9 mmol/L, and diabetes mellitus (DM) is defined as FG ≥7.0 mmol/L. 21
Statistical Analysis
The statistical analysis included all the patients with data available at baseline and at least at 1 follow-up assessment time. The data for the characteristics of the sample are presented as means (95% confidence interval [CI]) for the continuous variables and as frequencies and percentages for the categorical variables. All the characteristics of the patients were compared across the 2 SGA treatment groups using the chi-squared test and Fisher’s exact test for the categorical variables and t tests for the continuous variables. We assessed changes in continuous outcomes over time between the 2 SGA treatment groups using the linear mixed model with repeated measures. The repeated factor was the time relative to baseline at 1, 3, 6, 12, and 24 months. For each outcome, we adjusted for age and sex, other medications associated with SGA, and main diagnosis or comorbidities, thus yielding 3 different analyses.
Summary statistics are presented as the adjusted means of change over time (95% CI). Because these data reflect ‘real-life’ clinical situations, the time frame of the measures did not strictly respect the time interval. We averaged data for plus or minus the month interval (e.g., the third month corresponds to data from the second to the fourth months). Incidence was calculated as the proportion of new-onset metabolic complications at each time point divided by the number of patients with available data. All analyses were made using SAS 9.3 (SAS Institute, Cary, NC). The threshold for statistical significance was set at P < 0.05.
Results
A total of 147 patients treated for the first time with an SGA between November 2005 and June 2013 were included in the study. In total 116 (78.9%) received an SGA monotherapy and 31 (21.1%) received an SGA polytherapy. In the polytherapy group, 20 (64.5%) subjects switched SGAs and 11 (35.5%) received a combination of 2 SGAs. The mean duration of the SGA combination was 7.5 months (minimum, 1.5 months; maximum, 24 months). Data were missing at different time points during the follow-up for either weight or FG measures for various clinical reasons as missed appointments or failure to respect the calendar for the blood samples. The number of patients in each treatment group, per each specific molecule and who have data available at each time point, is presented in Figure 1.
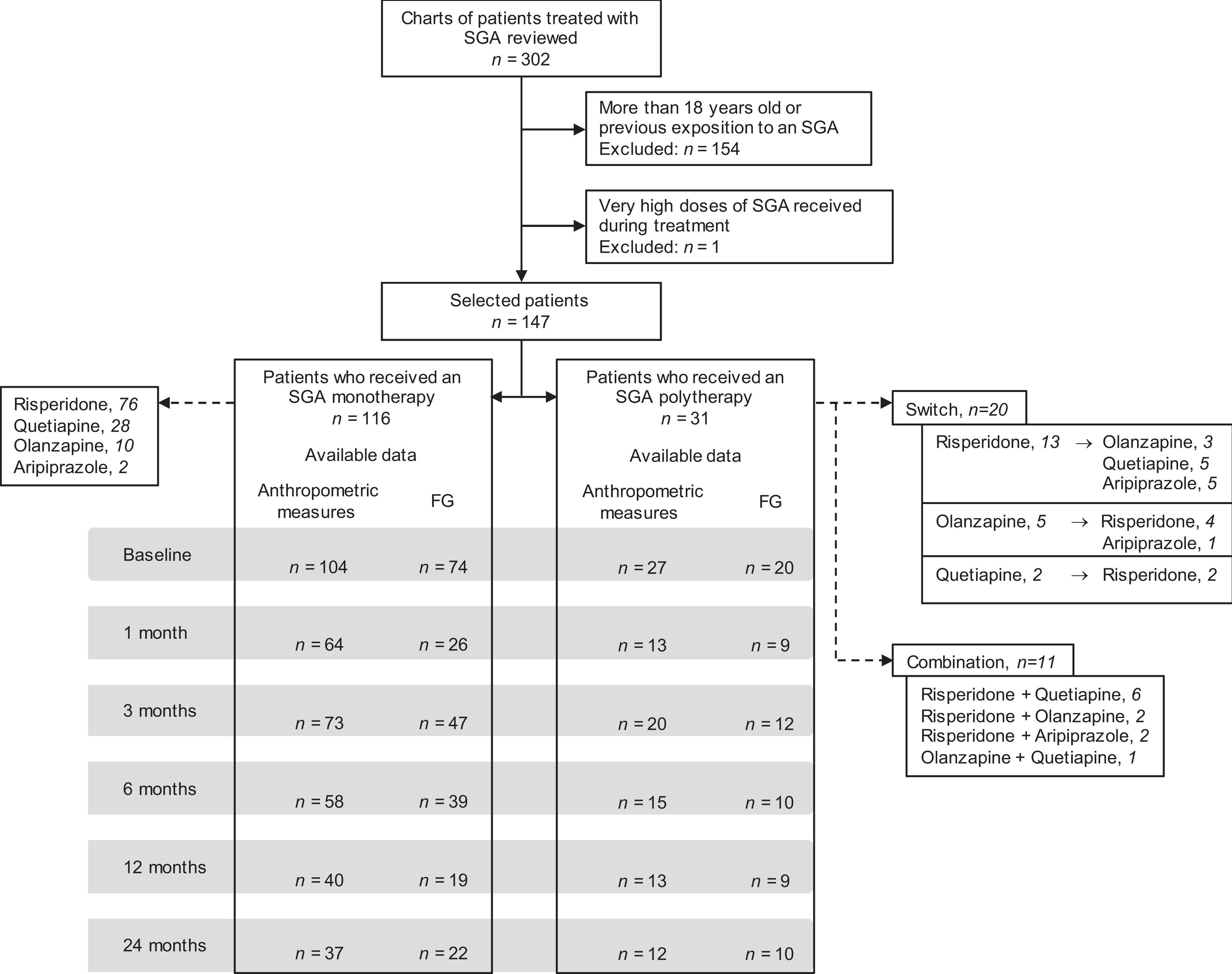
Number of subjects and available data flowchart.
The characteristics of the participants stratified by SGA therapy are presented in Table 1. Subjects were almost exclusively French Canadian. Most subjects were males of similar age in the monotherapy (12.7 years; 95% confidence interval [CI], 9.4-15.9) and polytherapy (13.0 years; 95% CI, 10.0-15.9) groups. Among diagnoses, the only difference was the proportion of psychotic disorders, which was higher in the polytherapy group than in the monotherapy group (45.2% vs. 26.7%; P = 0.047). The category ‘other’ regarding the main psychiatric diagnosis included personality disorder, mental retardation, pervasive developmental disorder, and obsessive-compulsive disorder. At baseline, there was no significant difference between the 2 groups in anthropometric, weight status, and FG levels.
Characteristics of Participants Stratified by SGA Therapy.
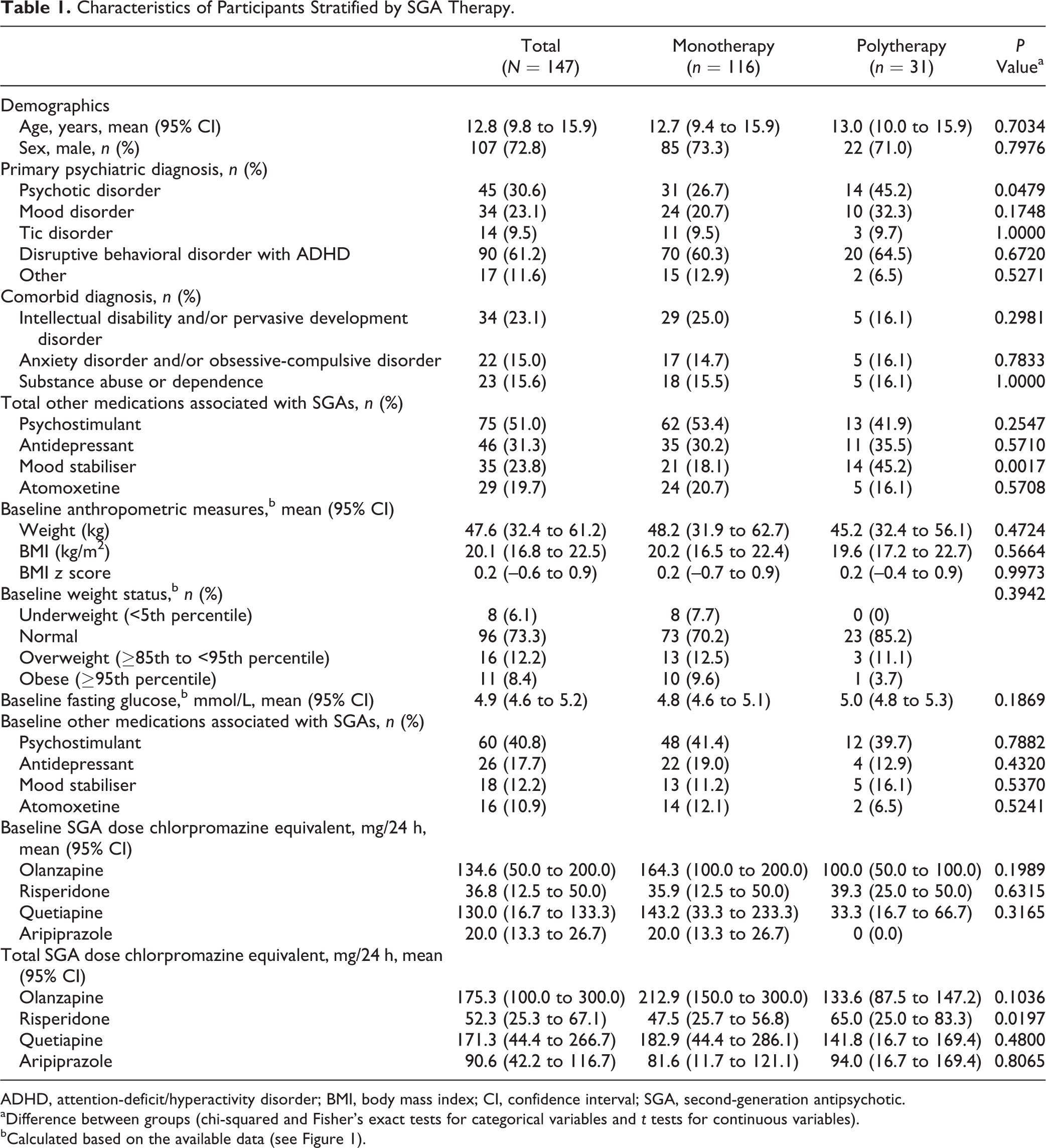
ADHD, attention-deficit/hyperactivity disorder; BMI, body mass index; CI, confidence interval; SGA, second-generation antipsychotic.
aDifference between groups (chi-squared and Fisher’s exact tests for categorical variables and t tests for continuous variables).
bCalculated based on the available data (see Figure 1).
No differences between the 2 groups were registered at baseline in the mean SGA doses and the percentage of other medications associated with the SGAs. The mean total SGA dose (in chlorpromazine equivalent) was greater in the polytherapy group than in the monotherapy group (65.0 [95% CI, 25.0-83.3] vs. 47.5 [95% CI, 25.7-56.8]; P = 0.019) for risperidone only. For the other medications associated with the SGAs, more patients were treated with mood stabilisers in the polytherapy group than in the monotherapy group (45.2% vs. 18.1%; P = 0.001).
The type of SGA therapy did not show a significant difference on the metabolic changes between the two groups.
Changes in anthropometric measures and FG in the 2 groups are presented in Table 2. For clarity, all the results are from the analysis adjusting for the age, sex, and other medications associated with the SGAs. The 2 other analyses produced similar results.
Change in Anthropometric and FG Measurements Stratified by SGA Therapy.
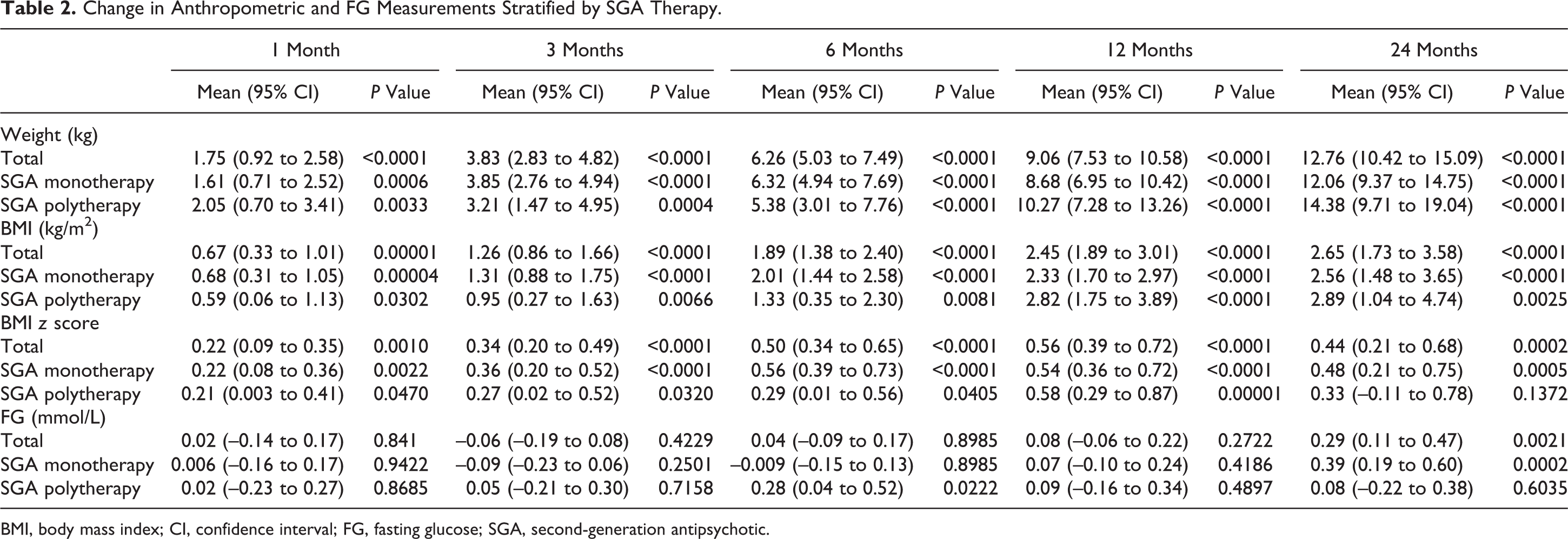
BMI, body mass index; CI, confidence interval; FG, fasting glucose; SGA, second-generation antipsychotic.
Overall, after 24 months of SGA treatment, mean weight increased significantly by 12.8 kg (95% CI, 10.4-15.0), BMI z score by 0.44 (95% CI, 0.21-0.68), and FG levels by 0.29 mmol/L (95% CI, 0.11-0.47).
Newly developed metabolic complications after up to 24 months of SGA treatment are presented in Table 3. Overall, at 24 months, 22.6% of subjects became overweight or obese, 42.9% had a BMI z score increase over 0.5, 9.4% developed IFG, and 1 subject (3.1%) developed DM, with similar incidence in the 2 groups, except for DM.
Incidence of Metabolic Complications Stratified by SGA Therapy.
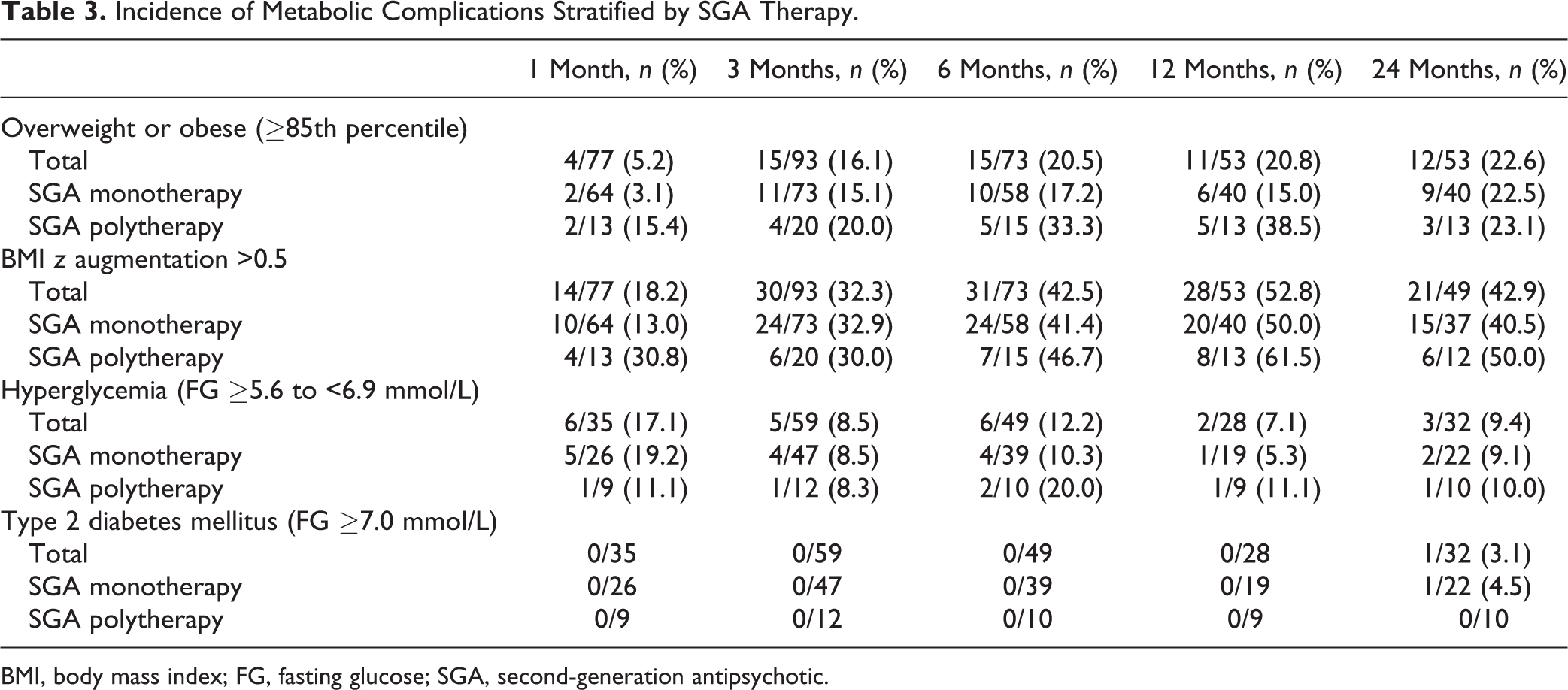
BMI, body mass index; FG, fasting glucose; SGA, second-generation antipsychotic.
Discussion
This is a descriptive study conducted in a clinical naturalistic setting with antipsychotic-naive children and adolescents reporting on long-term metabolic changes secondary to SGA therapy. We compared long-term metabolic changes between SGA monotherapy and polytherapy (switching or combining SGAs). Although the results showed continuing metabolic changes over time, no significant conclusion could be drawn from the mono- versus polytherapy comparison on weight gain, BMI z scores, and FG changes.
The results of our study are concordant with the recent systematic review and exploratory meta-analysis of randomised controlled studies comparing APP versus antipsychotic monotherapy in adult patients with schizophrenia, which revealed no additional adverse effect burden with APP. 22 Similarly, in adolescents and young adults treated with antipsychotics for the first episode psychosis, a retrospective naturalistic cross-sectional study examining the metabolic abnormalities showed that APP (prescribed to 25% of the cohort study) was not a significant predictor of obesity/weight gain. 23
However, 2 previous retrospective cohort studies 14,24 (both with the same cohort), evaluating factors associated with metabolic adverse events of antipsychotic use in antipsychotic-naive youth, reported a significantly higher risk for incident obesity/weight gain and DM in subjects treated with antipsychotic polytherapy. The study by McIntyre and Jerrell 22 has several differences regarding antipsychotic treatment and population characteristics. Our study counted only SGA treatment, whereas in their study, patients received second- or first-generation antipsychotics. Illness characteristics are also different, as indicated by fewer subjects in our study (53.7% vs. 91%) suffering from psychotic disorder and affective disorder/mood disorder, as increased risk of obesity was associated with untreated psychosis (2.8- to 3.5-fold) or bipolar disorder (1.2- to 1.5-fold).25,26 In addition, compared to our study, McIntyre and Jerrell’s 24 cohort had a higher percentage of subjects treated with antidepressants (79.5% vs. 31.3%) and mood stabilisers (45.8% vs. 23.8%). As antidepressants and mood stabilisers were also associated with an increased risk of obesity when associated with antipsychotics in the aforementioned study, although not in ours, one cannot rule out the possibility that this particular comedication is a confounding factor in the estimated obesity risk.
Moreover, 25.8% of the patients in our SGA polytherapy group were treated with aripiprazole (only 1.7% in monotherapy), while in McIntyre and Jerrell’s 24 cohort, SGAs with a lower metabolic impact, such as aripiprazole or ziprasidone, were only marginally used (less than 5% of the subjects). 25,27,28
Our results overall show that, after 24 months of SGA therapy, there is a significant increase in mean weight, BMI z score, and FG level; in addition, a notable number of subjects developed obesity or a clinically significant increase in their BMI z score. When compared with the results of a recent prospective study in youth treated for 12 months with risperidone or quetiapine, 29 our results show similar weight gain but less of an increase in FG, as well as fewer newly developed metabolic complications, suggesting a more favourable 24-month evolution of the metabolic complications than at 12 months. It is possible that the combination of antipsychotics that have low and high impacts on metabolic effects, other than weight gain, in our study has decreased the general effects compared to the study with only risperidone and quetiapine.
Regarding the glucose homeostasis, after 24 months of SGA treatment, the FG levels increased significantly, and we registered a notable incidence of hyperglycemia (9.4%). Consistently, a recent review analysing 506 SGA-treated subjects reported an incidence rate of 7.5% of IFG. 30 The incidence of DM in our study was 3.1%, which is consistent with a greater cumulative antipsychotic dose and treatment duration 29 and with the incidence rate of DM found in other studies. 31,32 In addition, Jerrell and McIntyre 14 found a DM incidence rate of 3.1%. Even if it is relatively rare, the occurrence of DM remains of great clinical importance, as studies 33,34 showed increased morbidity and mortality associated with an earlier onset. 35
The results of this study should be interpreted in consideration of its limitations. Due to the naturalistic design and the retrospective data collection, the metabolic monitoring at 12 and 24 months was relatively low, which limited the data available for the analyses and influenced the statistical power of our results. Because of the small sample size, we cannot exclude or confirm a difference between the 2 SGA therapy groups.
In addition, in the polytherapy group, the SGA combination was present for a variable and sometimes short period, thus possibly limiting the metabolic impact of the second SGA. Furthermore, for half the patients who switched antipsychotics, the switch occurred during the first month of SGA treatment. In this case, the first SGA taken for a short period might have less influence on the weight gain and glucose abnormalities.
The reasons for switching or combining SGAs were not clearly described in the medical charts. Clinically, this could reflect a more severe pathology, supported as well by a significantly higher percentage of patients treated for psychotic disorder and receiving a mood stabiliser in the polytherapy group. Also the homogeneity of the population sample analysed in our study can limit the generalisability of the results. Finally, given the variability of SGA doses in the clinical sample, we were unable to adjust in the statistical analyses for the SGA chlorpromazine-equivalent dose. Despite the mean dose of risperidone being significantly higher in the SGA polytherapy than in the monotherapy group, more subjects received risperidone in the monotherapy group than in the polytherapy group (65.5% vs. 51.6%).
Strengths
One strength of our study is the genetic homogeneity of our sample. The clinical population was almost exclusively French Canadian, limiting the differences attributable to ethnic genetic diversity. Also, unlike many studies of metabolic effects of SGAs, our recruitment was restricted to an antipsychotic-naive population, leading to more accurate conclusions about the first exposure to SGAs. Finally, this is one of the few long-term studies of metabolic complications in child and adolescent populations that can be informative regarding metabolic complications of single, sequential, or combined SGA use in drug-naive populations.
Conclusion
Our study confirms a significant increase in weight, BMI z score, and FG associated with long-term SGA treatment in antipsychotic-naive children and adolescents without excluding or confirming a difference existing between the 2 groups. Long-term prospective studies with larger sample sizes and inclusion of a control group are needed to investigate the metabolic effects of SGAs in single, sequential, or combined use.
Clinical Significance
Our 24-month descriptive retrospective naturalistic study was not sufficiently powered to determine if there is a significant difference between the metabolic complications of SGA treatment in monotherapy or polytherapy in youth. However, despite its naturalistic limitations, it reflects the clinical reality, with ‘real’ unbiased selected patients having complex diagnoses and treatments, and provides clinical information on long-term evolution of the weight gain and FG changes secondary to SGA monotherapy or polytherapy. This is important given that in clinical situations, when an SGA is recommended, the average number of SGAs trials for a given patient is between 2 and 3. 36
Also, in our study, the most frequent diagnosis for the SGA prescription was the disruptive behavioral disorder with attention-deficit hyperactivity disorder, which surpassed those of psychotic and mood disorders in both groups. This contrasts with the American Academy of Child and Adolescent Psychiatry guidelines, which do not recommend SGA use in children and adolescents for the disruptive behavior disorders. 37 Clinicians should consider psychosocial or other alternative pharmacological interventions to the SGAs, particularly when treating disruptive behaviors. 37
Moreover, the increase in weight, BMI z score, and FG seen regardless of the type of SGA treatment used (mono- or polytherapy) remains of great concern given that childhood obesity can adversely affect nearly every organ system. Serious consequences include hypertension, dyslipidemia, diabetes, fatty liver disease, 38,39 an increased risk of cardiovascular disease from early childhood onwards, 40 and psychosocial problems such as low self-esteem, development of eating disorders and depression, 41 –43 and increased morbidity and mortality associated with an earlier onset of DM. 35
These findings emphasise that clinical benefits must outweigh the risks when prescribing SGAs and that the length of the SGA treatment should be carefully planned. It also highlights the need for practitioners to follow monitoring guidelines from the beginning to the end of the SGA prescription to treat the metabolic complications as early as they are identified. Clinically, the combination of 2 or more antipsychotics should be avoided as its efficacy and safety are not clearly established.
Footnotes
Acknowledgements
We thank Radhia Ben Amor and Marie-Eve Brodeur, research assistants, for assistance with the data collection and organisation of the database.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was founded by the Daoussis Bourse of the Department of Psychiatry, University of Montreal.
