Abstract
Objective:
Combining antidepressants (ADs) for therapy of acute depression is frequently employed, but randomized studies have yielded conflicting results. We conducted a systematic review and meta-analysis aimed at determining efficacy and tolerability of combination therapy.
Methods:
MEDLINE, Embase, PsycINFO, and CENTRAL databases were systematically searched through March 2014 for controlled studies comparing combinations of ADs with AD monotherapy in adult patients suffering from acute depression. The prespecified primary outcome was standardized mean difference (SMD), secondary outcomes were response, remission, and dropouts.
Results:
Among 8688 articles screened, 38 studies were eligible, including 4511 patients. Combination treatment was statistically, significantly superior to monotherapy (SMD 0.29; 95% CI 0.16 to 0.42). During monotherapy, slightly fewer patients dropped out due to adverse events (OR 0.90; 95% CI 0.53 to 1.53). Studies were heterogeneous (I2 = 63%), and there was indication of moderate publication bias (fail-safe N for an effect of 0.1:44), but results remained robust across prespecified secondary outcomes and subgroups, including analyses restricted to randomized controlled trials and low risk of bias studies. Meta-regression revealed an association of SMD with difference in imipramine-equivalent dose. Combining a reuptake inhibitor with an antagonist of presynaptic α2-autoreceptors was superior to other combinations.
Conclusion:
Combining ADs seems to be superior to monotherapy with only slightly more patients dropping out. Combining a reuptake inhibitor with an antagonist of presynaptic α2-autoreceptors seems to be significantly more effective than other combinations. Overall, our search revealed a dearth of well-designed studies.
Depressive disorders are major challenges. Twelve-month prevalence estimates differ depending on diagnostic methods but are consistently high ranging between 1% and 10% for major depressive episodes. 1 The global burden of disease study ranks major depressive disorder (MDD) as fourth and fifth of the most burdensome disorders in Western Europe and North America, respectively, and globally ranked eleventh, with an increase of illness burden of 37% over the last 2 decades. 2
International guidelines by the American Psychiatric Association 3 and the Canadian Network for Mood and Anxiety Treatments, 4 as well as the German National Clinical Practice Guideline 5 recommend use of a single, non-monoamine oxidase inhibitor antidepressant (AD) as initial treatment in severe depression. However, despite the quantity of ADs and despite continuing development of new antidepressive agents, responder rates to initial AD monotherapy remain unsatisfactory, ranging from 40% to 60%, 6 –8 and remissions occur in only 20% to 30%. 9,10
In nonresponders, several second-step treatments are advocated in guidelines, 11 especially switching to a different monotherapy, high-dose treatment, augmentation (for example, with lithium), or combining 2 ADs. 12 As a consequence, combination treatment is frequent. In a recent naturalistic study 13 of nonresponders to initial monotherapy, 19% of patients received combined AD pharmacotherapy. Also, in Veterans Health Administration settings, 11% of all patients with depression were treated with the combination of 2 ADs. 14 However, studies have yielded conflicting results regarding the efficacy of combination treatment. For example, Blier et al 15 and Maes et al 16,17 published results in favour of combining ADs, but studies by Fava et al 18 and Leuchter et al 19 were negative. Despite a considerable number of trials comparing combination treatment to AD monotherapy, to our knowledge, no comprehensive systematic review and meta-analysis of available controlled trials exists. The authors of 1 earlier meta-analysis of 5 combination studies in treatment-naive patients concluded that there were too few studies to draw definitive conclusions. 20 In another systematic review of 5 studies, combination treatment, compared with monotherapy among incomplete responders, was summarized but not quantified, and a lack of evidence was emphasized. 21 Both studies applied restrictive selection criteria resulting in the exclusion of many studies carried out on the subject. Moreover, both studies did not contain a meta-analysis of dropouts, a critical aspect of combination treatment. In this vein, Thase, 22 in a recent qualitative review, emphasized the lack of adequate research on AD combinations. Accordingly, we carried out a systematic review and meta-analysis of controlled studies comparing combinations of 2 ADs and AD monotherapy in adult patients with acute depression. As data from randomized controlled trials (RCTs) were expected to be sparse, we intended to evaluate all accessible evidence by including controlled trials of different methodological rigour and to present studies both in subgroups according to methodological similarity, as well as in a global assessment across all studies. We hypothesized that combination therapy is superior to monotherapy regarding efficacy but at the price of higher dropout rates.
Methods
This is a systematic literature review and meta-analysis. The protocol has been published on Prospero (Prospero record registration no: CRD42013004407). Methods followed the PRISMA guidelines for systematic reviews and are described in detail in an online supplement (available at cpa.sagepub.com/supplemental). In brief, employing the Cochrane Highly Sensitive Search Strategy we searched PubMed, PsycINFO, Embase, and CENTRAL databases. Trials were included when they met the following criteria: existence of a control group of AD monotherapy (including open label and nonrandomized trials), inclusion of participants aged 18 years or older, of both sexes with depressive disorder, and diagnosis according to standard operationalized criteria. Diagnoses of other psychiatric disorders, as well as comorbid medical conditions, were no exclusion criteria. Studies specifically on bipolar disorder (BD) were excluded. Irrespective of dosage, we included all pharmacological interventions using a combination of 2 ADs, that is, initial combination therapy, as well as adjunctive administration of a second to a first AD. Both trials on first-line treatment and trials among patients with resistance to previous AD treatment(s) were included. We excluded trials on maintenance therapy.
All full texts included were screened independently by 2 reviewers. Study evaluation, data extraction, meta-analyses, tests for heterogeneity, and publication bias, followed the Cochrane Collaboration Handbook. 23 Primary outcome criterion was standardized mean difference (SMD) between treatments on an intention-to-treat basis. Secondary outcome criteria were rates of remission, response, and dropouts. Prespecified subgroup analyses included double-blind RCTs, studies in nonresponders to previous treatment trials, and studies with a low risk of bias. Trials were evaluated according to Cochrane’s risk of bias tool 23 : random sequence generation, allocation concealment, blinding of participants, personnel and outcome assessors, incomplete outcome data, selective reporting, sponsorship, and other potential sources of bias. An overall assessment of risk of bias (low, or unknown or high) was added. Post hoc analyses referred to samples restricted to continuation of monotherapy, compared with switch to another monotherapy, to combining monoamine reuptake inhibitors (selective serotonin reuptake inhibitor [SSRI], serotonin-norepinephrine reuptake inhibitor [SNRI], and tricyclic antidepressant [TCA]) with antagonists of presynaptic α2-autoreceptors (mianserin, mirtazapine, and trazodone), to MDD and to unipolar depressive disorder.
Results
Our literature search retrieved 8688 different articles. After screening titles and abstracts full texts of 172 articles were read, and 38 studies included (Figure 1).
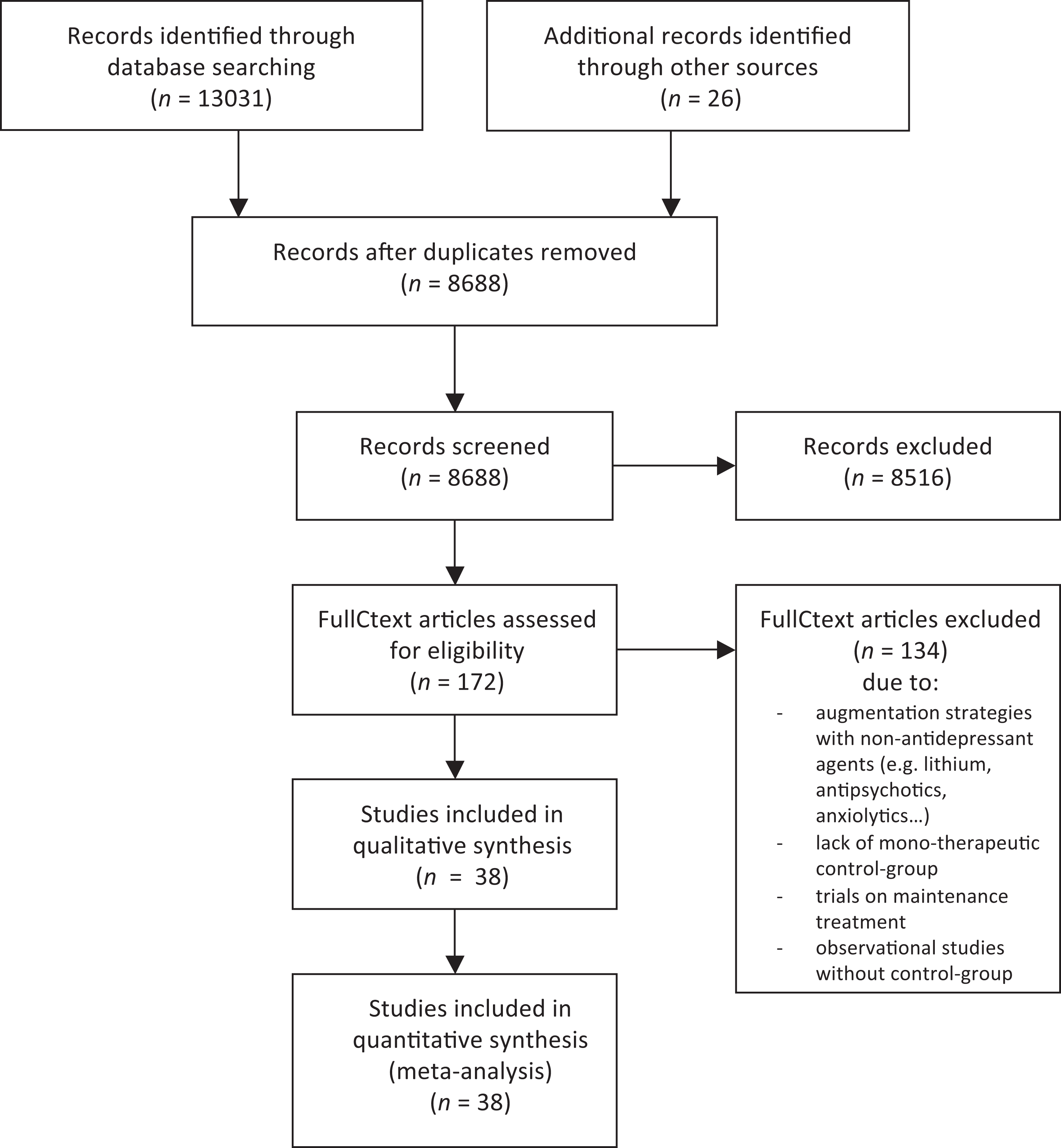
Flow chart of trials considered, eliminated, and included in study (adapted from PRISMA).
In total, trials included 4511 patients, with 1857 receiving combinations and 2654 receiving monotherapy. Publication dates ranged from 1977 to 2013. Articles were published in English, Chinese (4 articles), and Korean (1 article). Thirty-two studies (84%) were randomized, 27 (71%) used blinding measures, and 20 (53%) were double-blind. Seventeen trials (45%) recruited nonresponders to initial AD treatment only (Table 1).
Characteristics of trials.
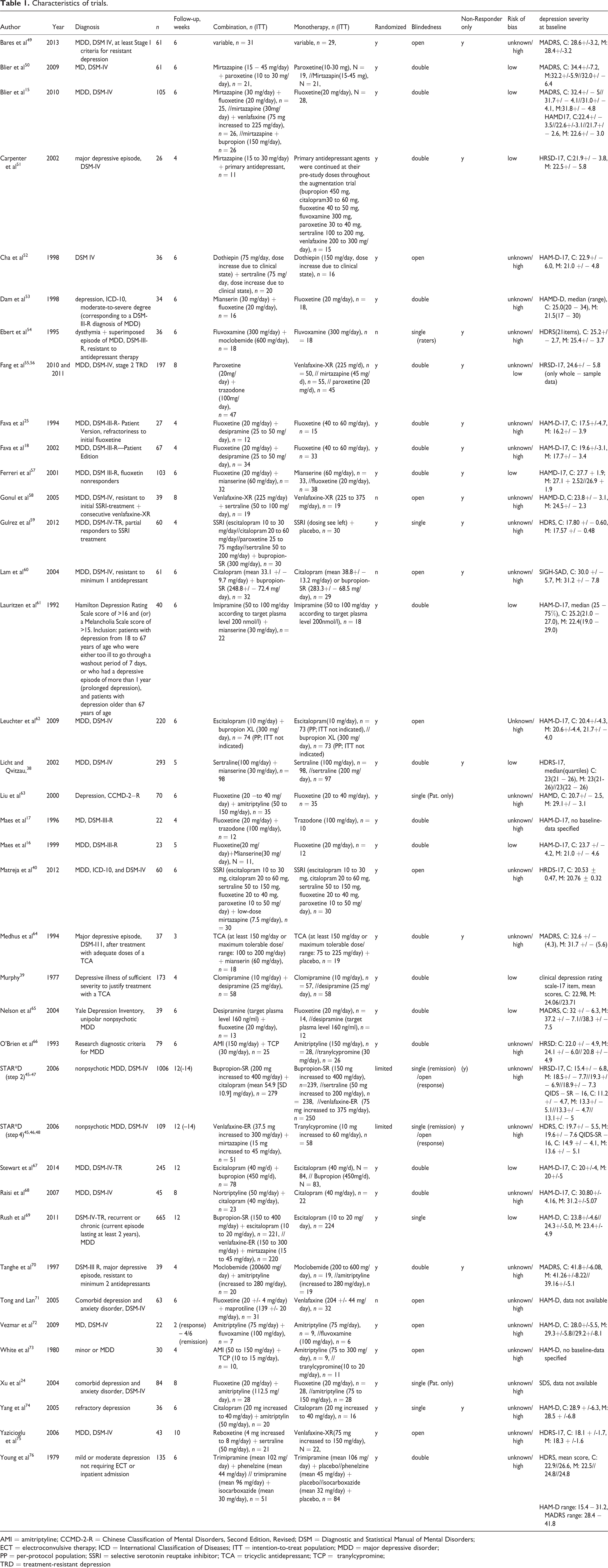
AMI = amitriptyline; CCMD-2-R = Chinese Classification of Mental Disorders, Second Edition, Revised; DSM = Diagnostic and Statistical Manual of Mental Disorders;
ECT = electroconvulsive therapy; ICD = International Classification of Diseases; ITT = intention-to-treat population; MDD = major depressive disorder;
PP = per-protocol population; SSRI = selective serotonin reuptake inhibitor; TCA = tricyclic antidepressant; TCP = tranylcypromine;
TRD = treatment-resistant depression
Efficacy
Primary Outcome
The analysis sample regarding our primary outcome criterion—efficacy as measured in SMD—consisted of 36 studies with 4342 patients. The SMD was 0.29 (95% CI 0.16 to 0.42) in favour of combination treatment (P < 0.001; random effects model) (Figure 2). Between-study heterogeneity was substantial (I2 = 63%, τ2 = 0.08).
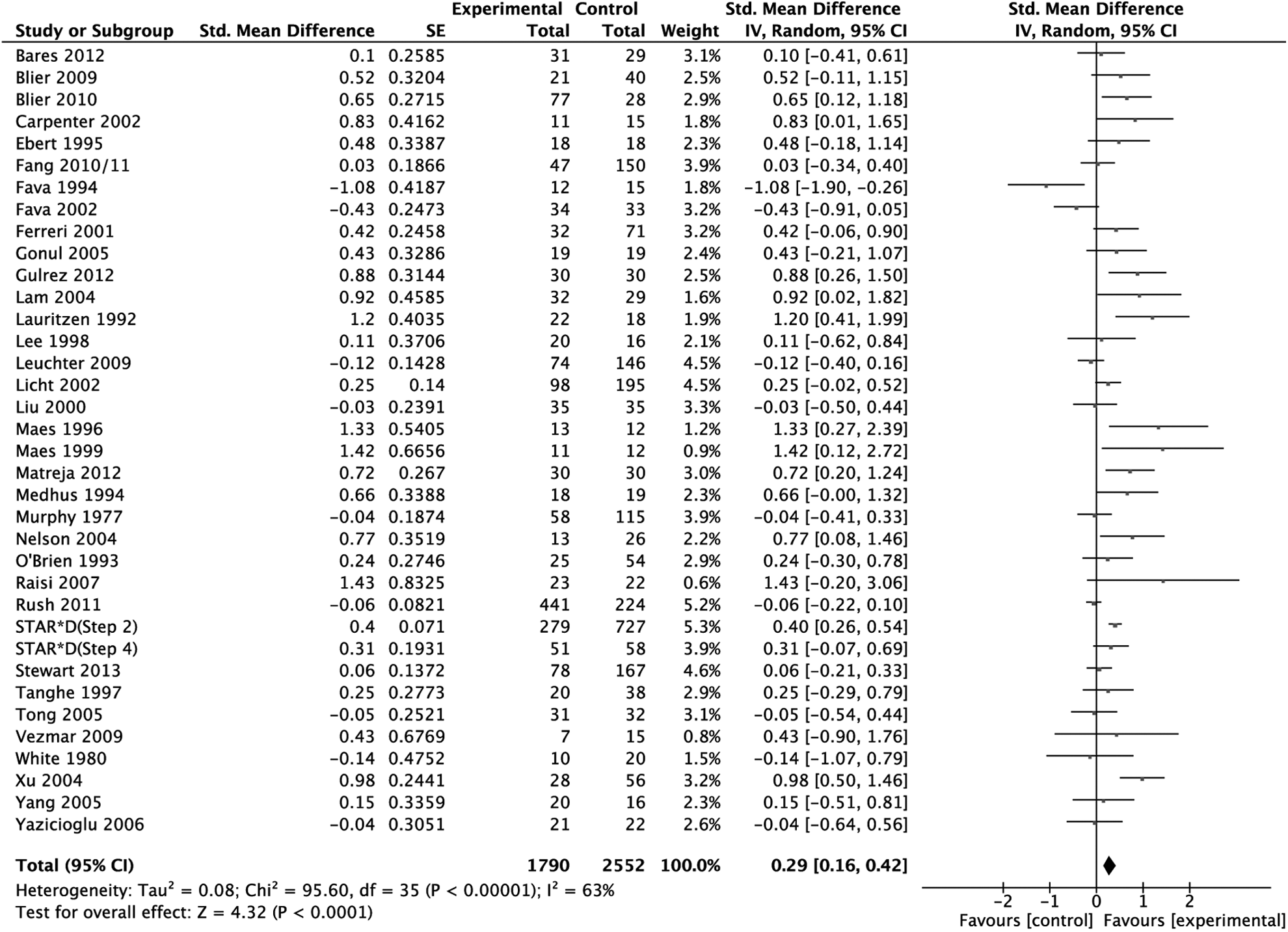
Primary outcome: treatment effect as measured in standardized mean difference weighted according to random effects analysis.
To avoid undue reliance on single studies, we removed studies one by one from the analysis. None of the 36 rounds resulted in a substantial change of point estimate or significance. Effect sizes varied between 0.26 (after elimination of Xu et al 24 ) and 0.31 (when Fava et al 18,25 were removed).
Sensitivity and Subgroup Analyses
The effect remained robust across subgroup analyses restricted to randomized, double-blind trials (SMD 0.33; 95% CI 0.11 to 0.54, P = 0.003) to low risk of bias-trials (SMD 0.36; 95% CI 0.13 to 0.59, P = 0.002), to trials excluding patients with BD (SMD 0.30; 95% CI 0.07 to 0.53, P = 0.01), and to RCTs of MDD patients treated with standard doses (SMD 0.25; 95% CI 0.04 to 0.46, P = 0.02). In trials limited to nonresponders, the direction of effect remained but effect sizes were lower (SMD 0.13; 95% CI –0.18 to 0.44, P = 0.42).
Secondary Outcomes
Secondary outcome analyses based on remission and response supported the superiority of combination treatment over monotherapy: odds ratios of 1.63 (95% CI 1.25 to 2.12, P < 0.001), and of 1.68 (95% CI 1.32 to 2.14, P < 0.001), respectively. In a meta-analysis confined to continuous data (rating scale differences) a similar effect emerged (SMD –0.20; 95% CI –0.37 to –0.03, P = 0.02). Primary and secondary outcomes, as well as subgroup analyses, are presented in Table 2.
Results and outcomes across subgroup analyses
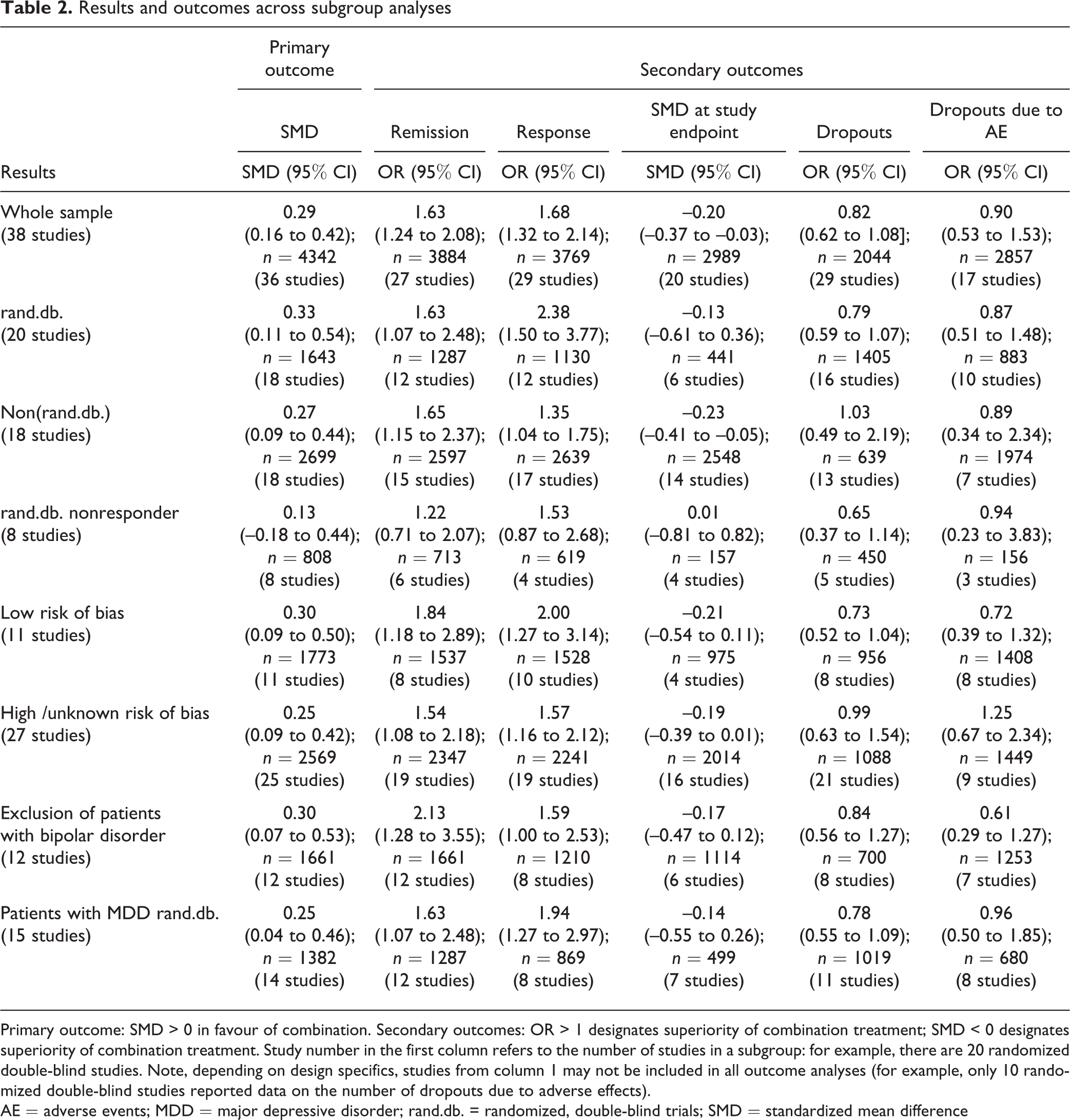
Primary outcome: SMD > 0 in favour of combination. Secondary outcomes: OR > 1 designates superiority of combination treatment; SMD < 0 designates superiority of combination treatment. Study number in the first column refers to the number of studies in a subgroup: for example, there are 20 randomized double-blind studies. Note, depending on design specifics, studies from column 1 may not be included in all outcome analyses (for example, only 10 randomized double-blind studies reported data on the number of dropouts due to adverse effects).
AE = adverse events; MDD = major depressive disorder; rand.db. = randomized, double-blind trials; SMD = standardized mean difference
Tolerability
Regarding both dropouts, due to any reason and to dropouts due to adverse effects, fewer patients dropped out in monotherapy arms (OR 0.82; 95% CI 0.62 to 1.08 and OR 0.90; 95% CI 0.53 to 1.53, respectively). Results were not significant and high P values indicate a possible role of chance (0.16 and 0.70, respectively).
Heterogeneity
Between-study heterogeneity was substantial in the primary outcome meta-analysis (I2 = 63%, τ2 = 0.08, Figure 2) and in most of the subgroup analyses. In some subgroup analyses and secondary outcomes I2 indicated lower heterogeneity between studies, especially in dropout analyses (I2 = 0%) and analyses restricted to studies of patients with MDD only (I2 = 49%). Heterogeneity as measured by tau-squared varied from 0.07 to 0.12 in primary outcome and primary outcome subgroup analyses. Accordingly, Tau ranged from 0.26 to 0.35, indicating that the standard deviation of the weighted SMD estimate was about equal to the effect size.
Publication Bias
The funnel plot of studies included in the primary outcome analysis did not rule out publication bias. Eggers test was borderline negative ( df = 34, P = 0.07), a trim and fill procedure (Duval and Tweedie) with 9 studies trimmed to the left of the mean resulted in a reduced effect size (0.16; 95% CI 0.02 to 0.29). Forty-four studies with an effect size of 0 would be necessary to reduce the overall effect to 0.1 (Orwin’s fail-safe N).
Trials selected had an average power of 26.4% (9.3% to 98.6%) to detect an effect size of 0.29 (SMD)–the primary outcome effect estimate.
Post Hoc Analyses
Two different modalities of monotherapy were compared with add-on combination treatment: continuation of monotherapy (10 studies) and switch to another monotherapy (2 studies). Both favoured add-on combination SMD (0.39; 95% CI 0.25 to 0.52, compared with 0.35; 95% CI 0.01 to 0.69).
Another post hoc analysis revealed similar effects in samples of only patients with MDD (SMD 0.30; 95% CI 0.15 to 0.44) (23 studies). The difference between combination and monotherapy was more pronounced in treatment-naive patients than in patients with treatment-resistant depression (TRD) (SMD 0.41; 95% CI 0.10 to 0.72) [6 studies], compared with 0.13; 95% CI –0.18 to 0.44 [8 studies]; randomized, double-blind studies, patients with MDD only).
Combination of a monoamine reuptake inhibitor (SSRI, SNRI, and TCA) with an antagonist of the presynaptic α2-autoreceptor (mianserin, mirtazapine, and trazodone) (13 studies) resulted in a statistically, significantly higher effect than other combinations (24 studies) (SMD 0.47; 95% CI 0.24 to 0.71, compared with SMD 0.17; 95% CI 0.02 to 0.32, P = 0.03). Restriction of this analysis to randomized, double-blind studies only consolidated the finding (SMD 0.54; 95% CI 0.29 to 0.79 [10 studies], compared with SMD 0.04; 95% CI –0.27 to 0.35 [8 studies]; P = 0.01); I2 = 47%) (Figure 3).
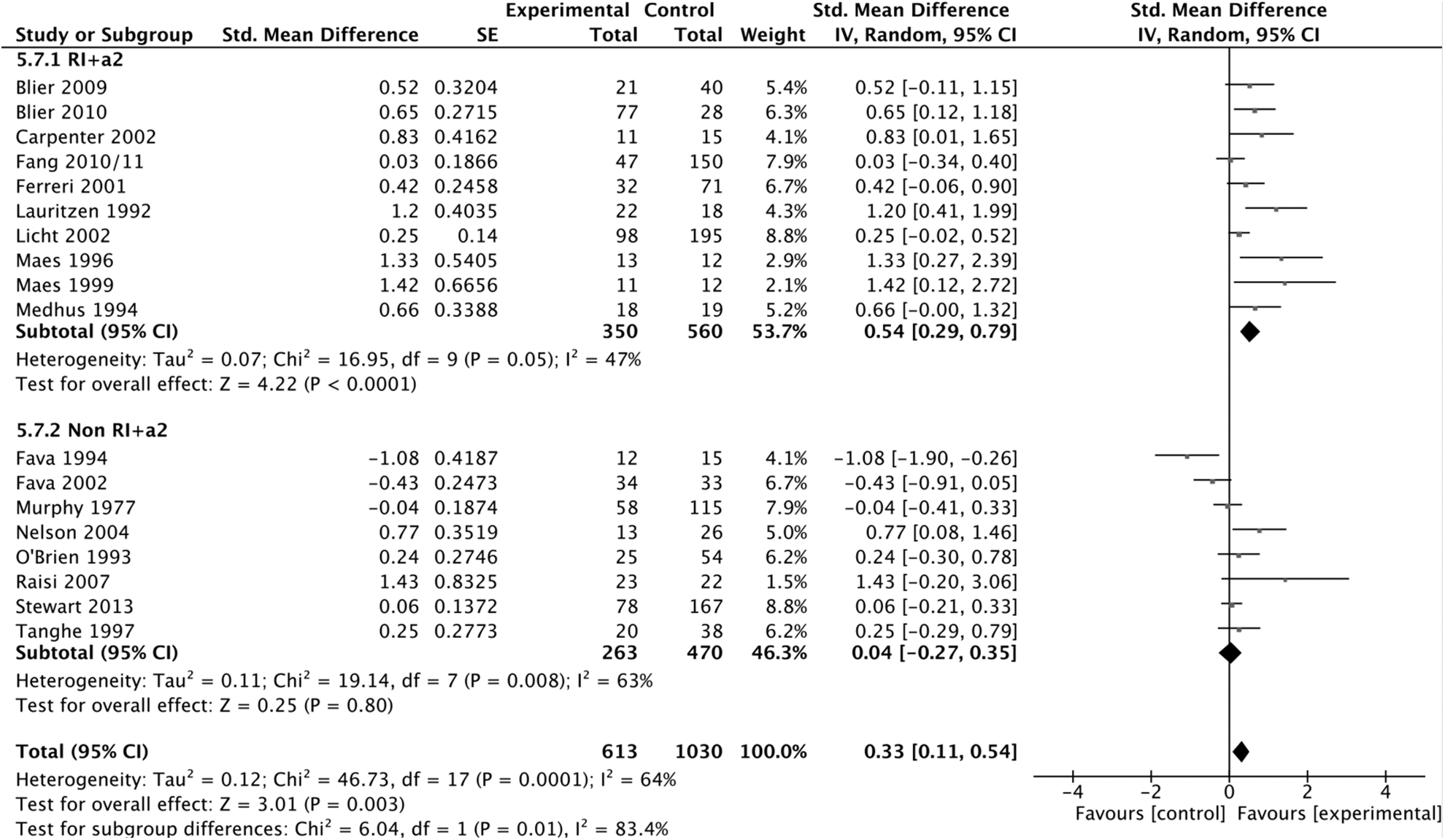
Primary outcome: treatment effect as measured in standardized mean difference (SMD) subgroup analysis: RI+α2 versus other pharmacologic combinations. Randomized, double-blind trials only weighted according to random effects analysis.
The fourth post hoc analysis confirmed an advantage of combination over monotherapy in patients with BD as well (SMD 0.30; 95% CI 0.07 to 0.53).
Meta-Regressions and Moderator Analyses
Average imipramine-equivalent doses were 269.8 mg (SD 134.1 mg) in combination arms and 170.1 mg (SD 72.9 mg) in monotherapy arms, a ratio of 1.6:1 (n = 35 studies). In meta-regression, dosage difference was substantially associated with SMD (slope 0.0014; 95% CI 0.00003 to 0.0028, P = 0.045, n = 34). A similar result emerged for dosage ratio (slope 0.286; 95% CI 0.0285 to 0.543, P = 0.029, n = 35).
Meta-regression revealed no association of duration of follow-up in weeks with SMD (slope –0.01; 95% CI –0.06 to 0.04, P = 0.60, n = 36).
Moderator analyses of dichotomized variables did not result in strong signals: randomized, compared with nonrandomized trials (P = 0.66); low, compared with non-low risk of bias studies (P = 0.46); and treatment resistant, compared with nontreatment-resistant patient samples (P = 0.78) (Table 2).
Discussion
Our study yielded 3 main results. First, combination treatment seems to be superior to monotherapy. Second, combination treatment is not associated with substantially more dropouts. Third, the combination of monoamine reuptake inhibitors and α2-adrenergic receptor antagonists seems to be superior to other AD combinations.
In this area of research an SMD of about 0.3, as estimated in this meta-analysis, is substantial, particularly because it emerged from comparisons between active treatments. Even when compared with placebo, AD monotherapy has effect sizes of no more than about 0.3. 26 Thus, even the lower end of the effect size confidence interval of the primary outcome, 0.16, indicates a non-negligible effect in AD pharmacotherapy. In the same vein, the subgroup analysis regarding remission lead to number needed to treat estimate of 9 (6 to 21) for combination treatment in comparison with monotherapy. In contrast, regarding dropouts, the point estimate of number needed to harm amounts to 50 (not significant).
Some, but not all, previous reviews judged combination therapy to be superior. 20,27 –31 Nevertheless, owing to small numbers of studies, or in the absence of meta-analyses, differences were not quantified or the role of chance was not estimated. Unfortunately, previous reviews did not quantify adverse effects but some, largely on theoretical grounds, highlighted increased risks of adverse events during combination therapy. 20,28,29 Thus, our tolerability analyses are of clinical importance: while monotherapy resulted in fewer dropouts due to any reason to adverse events, both effects were small and P values were high. The results refute our hypothesis of lower tolerability during combination treatment.
Regarding other treatment strategies in TRD, studies support lithium augmentation and augmentation with atypical antipsychotics. 32,33 However, switching to another AD cannot be considered evidence based. Bschor and Baethge, 34 in a systematic review and meta-analysis found switching not to be superior to continuation of initial ADs. Similarly, our subgroup analyses showed the advantage of add-on combination treatment to be more pronounced when compared with switching, than to continuation of monotherapy. As a result, it seems justified to particularly prefer combining ADs, compared with switching to another AD. It is important to note that, in our analysis, combination treatment in TRD was only marginally and not statistically, significantly effective in comparison with monotherapy. The finding may prove clinically useful anyway for 2 reasons. First, patients with TRD are a difficult-to-treat subgroup—and one that is probably frequently subjected to combination treatment—and any approach that shows some potential may have clinical merit. Second, the effect estimated in our study could be real even in the absence of statistical significance, mainly because the positive results in our various comparisons between combination and monotherapy increase the a priori probability of superiority in patients with TRD as well. In any event, additional methodologically rigorous research on this topic is essential.
Combinations of monoamine reuptake inhibitors and α2-adrenergic receptor antagonists turned out to be particularly effective. On pharmacodynamic grounds, monoamine reuptake inhibitors may act synergistically with α2-adrenergic receptor antagonists. Reuptake inhibitors are thought to be limited in effect, because increased levels of monoamines in the synaptic cleft result in increased activation of inverse feedback by stimulation of presynaptic α2-adrenergic receptors. 35,36 However, this finding has to be viewed with caution as it resulted from a post hoc analysis.
In our regression model, dosage difference accounted for about one-half of the total difference in treatment effect. Still, it has to be borne in mind that several studies did not provide detailed data on dosages. However, if the dose difference is correct it is all the more remarkable that tolerability, as measured in dropouts, seems to be sufficient when ADs are combined. Additionally, in a systematic review, a dose–effect relation could be demonstrated for some, but not for all ADs. 37 Licht and Qvitzau, 38 comparing combination therapy to standard- and high-dose sertraline, found the latter to be the least effective regimen. However, more 3-armed studies including a standard-dose monotherapy arm, a high-dose monotherapy arm, and a combination arm are needed to shed light on the significance of dosage.
Murphy et al 39 examined application of ADs at subtherapeutic doses. Matreja et al 40 observed application of low-dose mirtazapine in combination treatment. Both were not part of the MDD subgroup analyses. These may simultaneously represent subgroup analyses of therapeutic AD dosing.
Limitations
First, this meta-analysis included studies covering different patient populations, ADs, dosages, or durations of treatment, as well as studies of different methodological rigour. However, as earlier studies were contradictory and many underpowered, we aimed at including as many publications as possible. Ioannidis et al 41 argue that in most cases, even in the presence of heterogeneity, quantitative synthesis may be preferable to approaches frequently found in narrative reviews, as long as limitations of meta-analyses are acknowledged. In this vein, had we limited our calculations to meta-analyses restricted to a small group of studies we would not have found preliminary evidence for the superiority of combining reuptake inhibitors with α2-autoreceptor antagonists.
I2 values indicated substantial heterogeneity of effects. However, they are known to increase with accumulating N, additional tau-squared statistics were calculated, indicating a spread of data not unfamiliar in medical studies. The standard deviation had the same order of magnitude as the effect size. Nevertheless, heterogeneity was carefully considered in various sensitivity and subgroup analyses of more homogeneous study samples, in using random effects models, in testing whether results remained robust after each study was left out, and in considering possible effects of publication bias. Finally, 2 meta-regressions and 3 additional moderator analyses addressed the role of possible confounders. Therefore, our study does not represent an analysis of broad, instead of narrow, inclusion criteria but rather several analyses of different grades of homogeneity regarding trials selected, narrow and broad. Although we acknowledge that different confounding factors are interconnected, unfortunately, we could not calculate a single meta-analysis adjusting for all covariates simultaneously.
The meta-analyses of double-blind randomized trials and double-blind randomized trials among patients with MDD supported our findings. In fact, the advantage of combination treatment became stronger. Similar results apply to the more homogeneous subgroup populations. Some factors causing variability in the group of studies included likely have made the sample as a whole more conservative (for example, selection of studies employing low dose combinations, or including a subgroup of patients with BD). Still, rather than to find an exact point estimate for the efficacy of combined ADs relative to monotherapy it was the primary aim of our study to determine whether there is a difference at all between these 2 treatment principles and whether such a difference would be outweighed by an increase in dropouts. Note, secondary outcome analyses using different effect estimates aimed at confirming or falsifying primary results and not at finding significant results. Every effect estimate has to be viewed as what it is—an estimate that makes sense only with its confidence interval.
Some of the trials (N = 10) required nonresponse to an initial period of AD monotherapy. Trials consisted of AD monotherapy of this agent (in continuation), compared with add-on therapy with an additional AD. Therefore, dropouts due to adverse effects may have been decreased in the monotherapy group, as patients may have dropped out during the pretrial period. In these studies, dropout rates may be biased against combination treatment—a methodological problem that strengthens our findings.
Further, it has been shown that the results of some meta-analyses are inflated. While Pereira and Ioannidis 42 confirmed that results of most meta-analyses represent true effects, they calculated that there may occur effect inflation, particularly in meta-analyses with sparse data. However, our study benefitted from many studies and events. With a sample size of 4300 patients for our primary outcome, there seems reason to believe that the results of our meta-analysis will be stable over time. 43
It is possible that we did not include all relevant studies, but with MEDLINE, Embase, PsycINFO, and CENTRAL, 4 different large international databases were searched. It is doubtful that, in the field of combination treatment, publication bias is as important as it is in placebo studies of AD monotherapies. Nevertheless, adjusting for possible publication bias resulted in a weakened but still positive effect (SMD 0.16).
Finally, the limitations of our study will always be those of the included trials. For example, the trials that were published as blinded studies did not report on measures to ensure blinding. Therefore, it is difficult to judge the quality of blinding—a problem that has been shown to be prevalent in psychiatric research. 44 In addition, some of the study arms (step 2 in the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) Study 45 –47 ) were not meant to be directly compared, and step 2 and step 4 of the STAR*D trial 45 –48 were sequential treatments among the same study population. It is, therefore, reassuring that results were supported by the analysis of trials with low risk of bias.
Conclusions
AD combination seems to be superior to monotherapy and this advantage does not come at the cost of higher rates of severe adverse events. Possible areas of application might be in particularly severe cases of depression and treatment-resistant populations (although in this population the effect estimate was low). Our results point to a possibly important role of higher dosage in combination therapy and to promising combinations of reuptake inhibitors with α2-autoreceptor antagonists. Still, even with our systematic search of ample and broad inclusion criteria we find a dearth of evidence in this field of research. Future research should focus on the clinically important field of TRD.
Footnotes
Acknowledgements
We gratefully acknowledge the generous help by Ming Deng, Dr Sun-Hee Lee, He-Loung Jeong, Stanislav Bleer, and Ulla Beck in translating articles in Chinese, Korean, Russian, and Danish.
Dr Baethge and Dr Bschor had the idea for the study and its design. Dr Henssler conducted the literature search and screened the articles. All authors reviewed all full texts for inclusion and collected the data independently. Dr Baethge and Dr Henssler analyzed the data. All authors drafted and revised the paper, and approved the final version.
Both Dr Henssler and Dr Bschor contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and (or) publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and (or) publication of this article: Until June 2014, Dr Bschor has received honoraria for speaking from Lilly, AstraZeneca, Esparma, Bristol-Myers Squibb, Sanofi-Aventis, Servier, and Lundbeck, and has accepted reimbursements of travelling expenses to congresses from AstraZeneca and Lilly. Since then, Dr Bschor has discontinued all financial relations with pharmaceutical companies..
