Abstract
Globally, the fossil record of the co-occurrence of alpheids (mainly claw fingertips) and gobiids (mainly otoliths) is a rarity. Herein, we record a diverse assemblage (comprising a total of nine species) of alpheid shrimps (based on fossilised claw fingertips) assigned to the genus Alpheus (including a new species: Alpheus indicus sp. nov.) from the early Miocene (Burdigalian) Quilon Formation, Kerala Basin, southwest India. These alpheid remains co-occur with fossil remains (mainly otoliths) of numerous gobiid fish genera, including Medoborichthys (represented by M. podolicus) within the Quilon Formation. This fossil evidence supports the idea that the mutualistic relationship(s) within these groups had developed before the Burdigalian. Based on the geographic distribution of alpheids and gobiids fossils, it is evident that these groups quite likely thrived persistently in a mutualistic relationship(s) within the shallow coasts of the Paratethys Sea and the Indian Ocean realms during the Neogene (Aquitanian to Burdigalian). Further, older (Paleogene) records from the subcontinent are certainly warranted to provide support to any argument in favour of the enhancement of a complex snapping claw within alpheids and/or the development of the mutualistic relationship(s) between alpheids and gobiids during the early Paleogene (early Eocene) within India.
INTRODUCTION
Fossils of alpheids (Order: Decapoda; Infraorder: Caridea, Family: Alpheidae), generally referred to as ‘pistol shrimps’ or ‘snapping shrimps,’ mainly consist of strongly calcified pereiopod one claw fingertips of the dactylus (movable finger) and the fixed finger (pollex) (Anker et al., 2006; Hyžný et al., 2017). Globally, the fossils of alpheid shrimps are well-known within the Cenozoic marine deposits (Lima et al., 2020; refer to Figure 1 and supplementary data S1 in Kapur et al., 2022). The world’s oldest fossils of alpheids (claw fingertips) have been reported from the upper Oligocene (~28 Ma) Chickasawhay Limestone deposits, Alabama, southeast USA (Ciampaglio & Weaver, 2008; Feldmann et al., 2019). Early Miocene (Aquitanian) alpheid claw fingertips have been reported from Europe (Cluzaud et al., 2014; Jagt et al., 2015; Müller, 1998) and recently from the Khari Nadi Formation, Kutch Basin, western India (Kapur et al., 2021, 2022). The early-middle Miocene (Burdigalian-Serravallian) records of alpheid claw fingertips commonly occur in the Paratethyian Oceanic realms of Europe (i.e., within Austria, the Czech Republic, the Netherlands, Poland, and Slovakia) (Hyžný et al., 2017, 2018, and references therein). The only occurrence of fossil alpheids within Europe’s upper Miocene (Tortonian) is known from France (Hyžný et al., 2017 and references therein). Alpheid fossils (claw fingertips) are also known from the early and middle Miocene sedimentary deposits of the Pirabas Formation, Brazil, South America (Lima et al., 2020). Pleistocene records of alpheid claw fingertip fossils are known from Japan (Ando et al., 2015, 2016; Karasawa, 2014; Kobayashi et al., 2008) as well as from Egypt (Hyžný et al., 2017). Thus, the early Neogene (mainly Miocene) records of alpheids from Asia (except India) and Africa are absent.
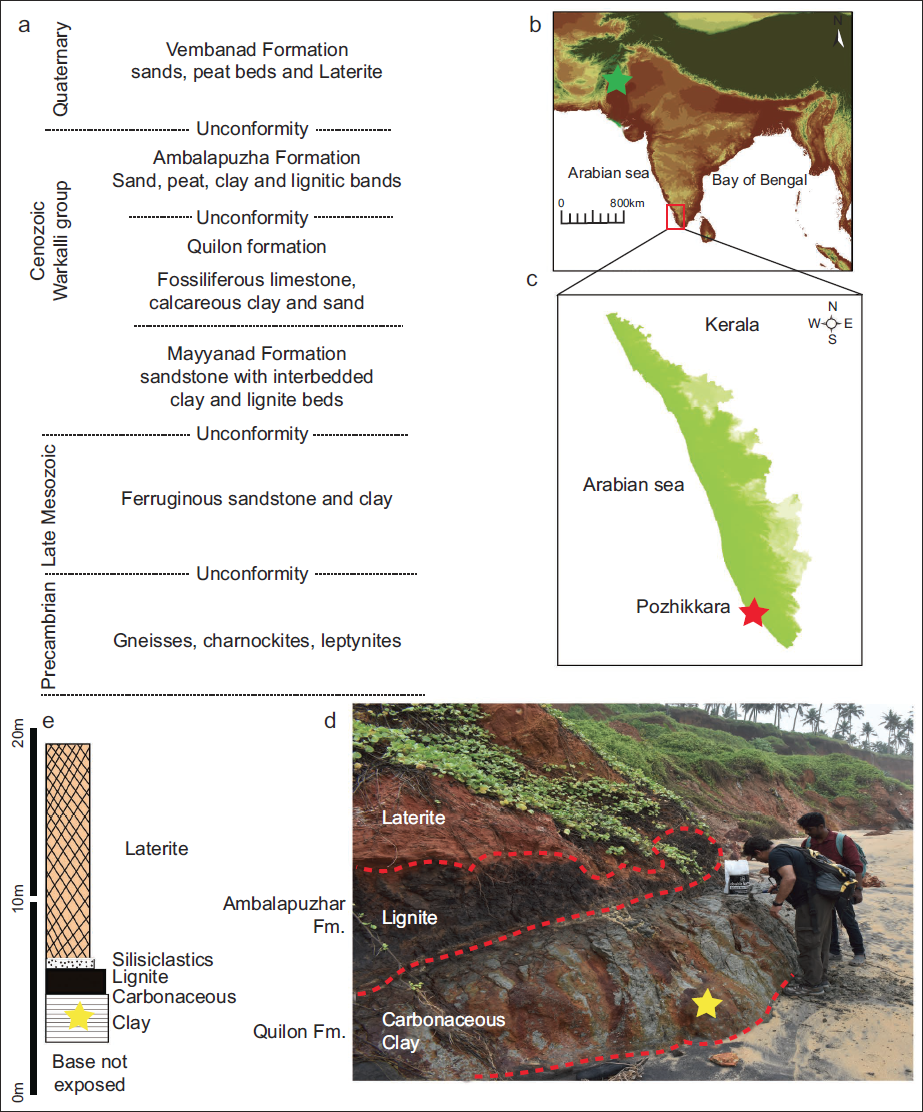
Locality information and geological settings. a) General lithostratigraphic set-up of the onshore Kerala Basin (Raha, 1996; Sajinkumar et al., 2022); b–c) DEM of the Indian subcontinent and the Kerala State (southwest India); d) field photograph showing the sedimentary sequence exposed at the Pozhikkara Cliff Section and the level of the investigated sample within carbonaceous Clay; e) litholog showing the stratigraphic position of the investigated sample. Note: Green Star marks the location of Aquitanian (~22 Ma) Alpheid-Gobiid yielding Khari Nadi Formation location ~1.5 km northeast of Kotada, Kachchh District, Gujarat State, western India) (Kapur et al., 2022), Red Star [marks the location of the Burdigalian (~17 Ma) Alpheid-Gobiid yielding Quilon Formation at Pozhikkara Cliff Section], Yellow Star [marks the location of the Alpheid-Gobiid yielding level (this investigation) within carbonaceous Clay belonging to the Quilon Formation].
Investigations in a phylogenetic framework support that the family Alpheidae is monophyletic and that the true snapping claw in the family Alpheidae evolved only once (Anker et al., 2006). However, the phylogenetic analysis shows that the most diverse genus, Alpheus (represented by more than 400 living species) within Alpheidae, is paraphyletic (Anker et al., 2006). This is owing to many cryptic species within the genus Alpheus and other genera (e.g., Synalpheus) of the family Alpheidae (Anker et al., 2006 and references therein). Nevertheless, the morphology of the claw fingertips in a phylogenetic context is significant in understanding the evolution of the snapping mechanism(s) within alpheids (Anker et al., 2006). Thus, both the field (ex-situ) and the laboratory (in-situ) investigations on extant alpheids have provided valuable contributions towards recognising the acoustic behaviour [i.e., sound patterns produced (underwater) by rapid closing of snapping claws] in terms of the morphological variability in the claw structures [i.e., the snapping apparatus and its functional mechanism] and snapping apparatus’s utilisation in forming the ‘Mutualistic’ relationship(s) with other invertebrate (e.g., crabs) and vertebrate (e.g., goby fish) groups (Au & Banks, 1998; Burns et al., 2019; Kohda et al., 2017; Koukouvinis et al., 2017; Tseng et al., 2019; Werding et al., 2016 and references therein).
Longley and Hildebrand (1941) were the first to recognise the mutual relationship between the alpheid shrimps (family Alpheidae) and the gobiid fishes (family Gobiidae) in the coastal marine environment of Florida, USA. Subsequently, many workers recognised this symbiotic behaviour between different extant species of alpheids and gobiid fishes across the coastal environments of the world; however, the partner choices in terms of their behavioural biology in varied environments is yet to be fully understood (Banner & Banner, 1980; Burns et al., 2019; Farrow, 1971; Jaafar & Hou, 2012; Karplus, 1987; Karplus & Thompson, 2011; Luther, 1958; Thomson, 2004; Wirtz et al., 2008 and references therein). Generally, the symbiotic ‘mutualistic’ relationship between these animal groups is due to the poor vision and the burrowing behaviour of the alpheid shrimps. These burrows serve as a shelter for gobiids that have good eyesight and guard the burrows constructed by the partner alpheid(s) (Werding et al., 2016 and references therein). Additionally, the opportunistic omnivorous alpheid shrimps benefit from the vital food sources provided by the gobiids, including the excreta of the gobiids within the burrow (Jaafar & Hou, 2012; Kohda et al., 2017 and references therein).
Globally, the fossil records on the co-occurrence of alpheids (mainly claw fingertips) and gobiids (mainly otoliths) are a rarity and restricted to the Miocene (Kapur et al., 2021, 2022; Radwańska, 2018). These studies support the mutualistic relationship between alpheid shrimps and gobiids developed during the Neogene (Aquitanian). However, our understanding of the development of this mutualistic relationship is limited due to the paucity of fossil data on alpheids, particularly within the Paleogene and Neogene intervals across the globe. As already mentioned, in India, prior to the present investigation, the only published Neogene record on the co-occurrence of alpheid shrimps and the gobiid fish comes from the early Miocene (Aquitanian) Khari Nadi Formation, Kutch Basin, western India (Kapur et al., 2021, 2022). In this palaeontological investigation, we record the most diverse assemblage of fossil alpheids (based on fingertips) within the Neogene of south Asia, that is, from the Quilon Formation, Kerala Basin, southwest India. Additionally, we discuss the significance of the fossil evidence on the co-occurrence of alpheids (fingertips) and gobiids (based on otoliths) in the context of the timing(s) and development of mutualistic relationship(s) within these groups.
Geological Setting
The peri-cratonic Kerala Basin is situated on the south-western margin of India (Figure 1). The Cenozoic sedimentary deposits (Warkalli Group) of the Kerala Basin overly the Archean crystalline basement and underlies the Quaternary sediments belonging to the Vembanad Formation (Figure 1). The Cenozoic Warkalli Group has been divided into three litho-units, that is, the Mayanad Formation, the Quilon Formation, and the Ambalapuzha Formation, in ascending stratigraphic order (Raha et al., 1983, 1996). The Quilon Formation (thickness up to 130 m) consists of limestones, carbonaceous clays, marls, and sand in varying proportions (Kumar & Pachaimuthu, 1933; Menon, 1967; Narayanan et al., 2007; Poulose & Narayanaswami, 1968). In the present micropalaeontological study, carbonaceous clays belonging to the Quilon Formation exposed at the Pozhikkara Cliff Section (Co-ordinates: 08°48’14.8” N; 76°39’16.4” E), Kollam District (Kerala State, southwest India) were investigated (Figure 1). Micropalaeontological data in a biostratigraphic framework have provided an early Miocene (Burdigalian) age for the Quilon Formation deposits exposed at Channakodi Type Section (~30 km north of the Pozhikkara Cliff Section), Kerala State, southwest India (Reuters et al., 2011 and references therein). Considering the above, published literature also argues in favour of an early Miocene (Burdigalian) age for the Quilon Formation exposed at the Pozhikkara Cliff Section (Carolin, 2012; Prasanna et al., 2021; Prasanna & Kapur, 2022; Carolin et al., 2022 and references therein). In a recent study, an age range from 17.08 Ma to 17.53 Ma has been assigned to the Quilon Formation exposed at the Pozhikkara Cliff Section (Kerala State, southwest India) based on the strontium isotopic composition of the molluscan shells (refer to Table 2 in Carolin et al., 2022).
MATERIALS AND METHODS
A bulk sample was collected from a ~1 m thick carbonaceous clay unit from the early Miocene (Burdigalian) Quilon Formation at the Pozhikkara Cliff section, Kerala State, southwest India (Figure 1). The sample was dried in an oven at 50°C for about 48 hours and later macerated without chemical treatment. The sample slurry was wet and dry sieved using standard ASTM (American Standards for Testing of Materials) sieves of varying sizes (ASTM - 10, 35, 60, 120, and 230). The dried, well-sorted sample was examined for microfossils (recorded herein) under a stereomicroscope (Model: Leica S8APO). The fossil specimens reported in this investigation were photographed utilising the Field Emission Scanning Electron Microscope (FESEM Model: JEOL-JSM7610F). The Energy Dispersive Spectroscopy (EDS) utilised for accessing the elemental composition of the alpheid claw fingertips (recorded herein) involved only the multiple spot technique using the EDAX (Model: Octane Plus with TEAM software version V4.2.1) instrument. The referred and/or illustrated microfossil specimens (this article) are housed in the Birbal Sahni Institute of Palaeosciences, Lucknow, India (BSIP Museum locality no. 10526; BSIP Museum micropalaeontology slide nos. 17610-17611) under the Acronym(s): VPPL/QLN/L1/ALP and VPPL/QLN/L1/OTH; Abbreviations: VPPL, Vertebrate Palaeontology and Preparation Laboratory; QLN, Quilon Formation; L1, yielding sample number within carbonaceous clay unit; ALP, alpheid; OTH, otolith.
RESULTS
In our micropalaeontological investigation, we recovered twenty isolated major (snapping) claw fingertips (comprising both dactyli and pollex) of alpheid shrimps, herein assigned to the genus Alpheus (Order: Decapoda; Family: Alpheidae) from the Quilon Formation exposed at the Pozhikkara Cliff Section, Kerala State, southwest India. We herein followed the classification scheme of De Grave et al. (2009). Generally, a highly calcium-rich composition of these claw fingertips is known to assist in their preservation as fossils in the global geological records (Hyžný et al., 2017 and references therein). The multispot EDS analyses of the isolated alpheid claw fingertips (recorded herein) from the Quilon Formation confirm a high calcium-rich composition of these fossils (refer to Supplementary Data S1; Plates 1–3). The investigated diverse assemblage of alpheids from the Quilon Formation (Kerala Basin, southwest India) comprises one new species and another eight unidentified species (refer to section 5 ‘Discussion’). Thus, it becomes the most diverse record of fossil alpheids from India, which is a rarity in the Neogene fossil record of the subcontinent (see Kapur et al., 2022). This is to mention that one specimen for each recorded taxon of alpheid is illustrated in the present investigation (refer to Plates 1–3).
Alpheid shrimps
Alpheus indicus sp. nov.
(Plate 1, Figure 1a–c)
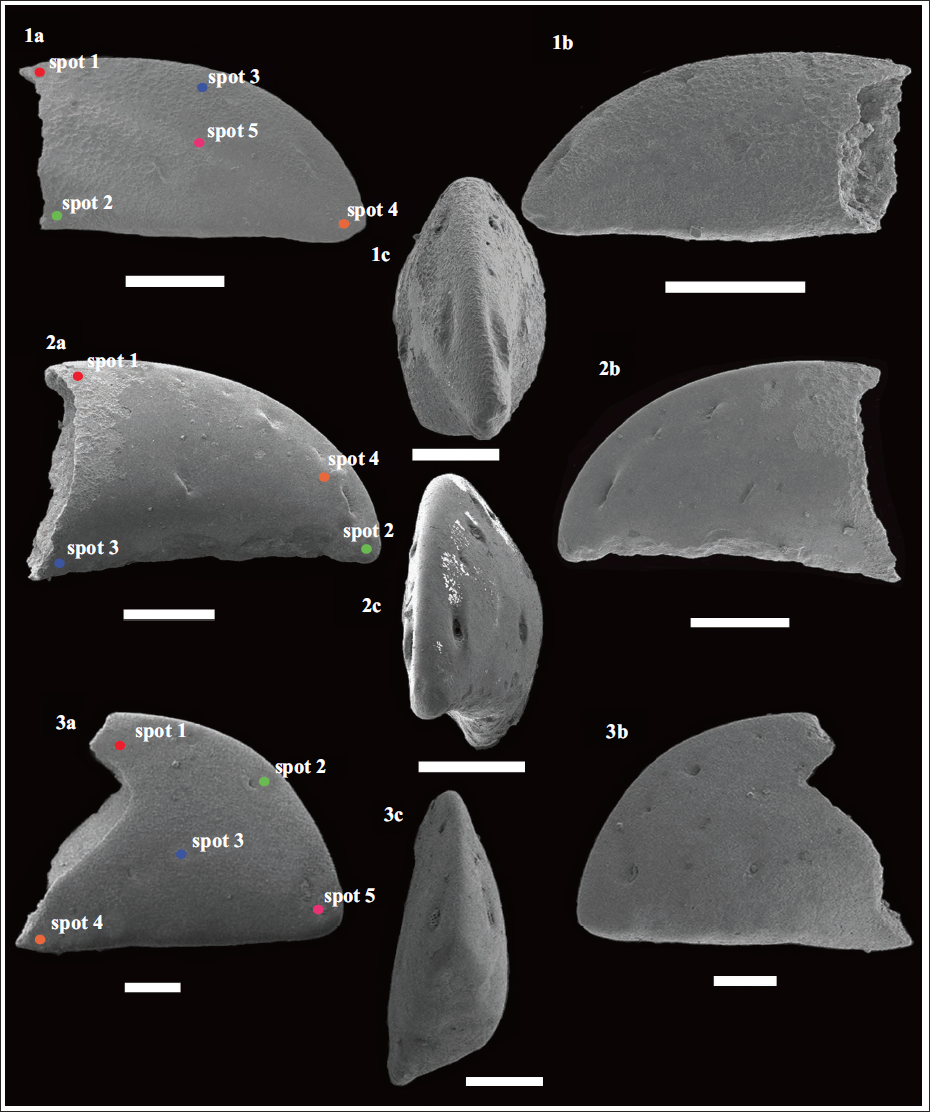
Scanning electron microphotographs of the alpheid claw fingertips recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation, Kerala Basin, southwest India. (1a–c) Dactylus of Alpheus indicus sp. nov. (Specimen no. VPPL/QLN/L1/ALP1); (2a–c) Dactylus of Alpheus sp. 1 (Specimen no. VPPL/QLN/L1/ALP11); (3a-c) Dactylus of Alpheus sp. 2 (Specimen no. VPPL/QLN/L1/ALP12). Scale bar equals 300 µm for all. The position of the individual spots (in each specimen) analysed using Electron Dispersive Spectroscopy (EDS) has been marked. Profiles of the EDS analysis of the fossil alpheid claw fingertips recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation (Kerala Basin, southwest India) have been provided as Supplementary Data S1.
LSID. urn:lsid:zoobank.org:pub:A15BDBF0-AAFE-46D4-9521-B3AEB442024C
Derivation of name: The species name is after the country, India.
Holotype: VPPL/QLN/L1/ALP1.
Referred specimens: Nine isolated dactyli specimens (VPPL/QLN/L1/ALP2; VPPL/QLN/L1/ALP3; VPPL/QLN/L1/ALP4; VPPL/QLN/L1/ALP5; VPPL/QLN/L1/ALP6; VPPL/QLN/L1/ALP7; VPPL/QLN/L1/ALP8; VPPL/QLN/L1/ALP9; VPPL/QLN/L1/ALP10).
Locality, Horizon and Age: Pozhikkara Cliff Section, Kerala State, southwest India; Carbonaceous shales belonging to Quilon Formation; early Miocene (Burdigalian; ~17 Ma).
Diagnosis: The dactyli showcase an almost rectangular structure with a short tip, a straight to subtly concave ventral margin, and an elongated convex dorsal margin. A row of five setal pores positioned in a depression is observed along the convex dorsal margin, with a large setal pore positioned in the central part of the fingertip; the first setal pore is conspicuously larger compared to the others; the tip is weakly blunt.
Remarks: The dactyli assigned herein to Alpheus indicus sp. nov. differ morphologically from the other investigated alpheid fingertips (dactyli) as part of the present study, and from the previously recorded alpheid fingertips reported from the early Miocene (Aquitanian; ~22 Ma) Khari Nadi Formation (Kachchh District, Gujarat State, western India) (see Kapur et al., 2022). However, Alpheus indicus sp. nov. is similar to ‘Morphotype IV’ recorded by Hyžný et al. (2017) in being rectangular; however, it differs in the absence of a strong, blunt tip (refer to Figure 3j–l in Hyžný et al., 2017).
Alpheus sp. 1
(Plate 1, Figures 2a–c)
Description: The dactylus (specimen no.: VPPL/QLN/L1/ALP11) has a pointed claw fingertip representing a triangular structure having a row of six setal pores positioned in a depression along the convex dorsal margin. In the specimen no. VPPL/QLN/L1/ALP11, the ventral margin is slightly eroded but may have showcased a slightly concave margin. The occlusal margin of the dactylus forms a sharp tip followed by a concavity.
Remarks: This species is represented by a single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP11); however, it is morphologically similar to ‘Alpheus sp. 1’ (specimen no. VVK/KOT4050) recorded by Kapur et al. (2022) from the early Miocene (Aquitanian) Khari Nadi Formation (Kachchh (=Kutch) District, Gujarat State, western India), ‘Morphotype I’ sensu Hyžný et al., 2017 (refer to Figure 4a–c in Kapur et al., 2022 and Figure 3a–c in Hyžný et al., 2017) and middle Miocene dactyli recorded from Poland (refer to ‘Text-fig. 3(5)’ in Radwańska (2018).
Alpheus sp. 2
(Plate 1, Figures 3a–c)
Description: The single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP12) displays a triangular structure with a strong, blunt tip; having a broadly convex dorsal margin, and a straight ventral margin. No depression is observed along the dorsal margin, and four setal pores can be observed along the dorsal margin, and another two setal pores are located centrally.
Remarks: This species is represented by a single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP12) and differs morphologically from previous known records of Neogene fossil alpheid fingertips, including the ones reported from the early Miocene (Aquitanian; ~22 Ma) Khari Nadi Formation (Kachchh District, Gujarat State, western India) (see Kapur et al., 2022).
Alpheus sp. 3
(Plate 2, Figures 1a–c, 2a–c)
Scanning electron microphotographs of the alpheid claw fingertips recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation, Kerala Basin, southwest India. (1a–c) Dactylus of Alpheus sp. 3 (Specimen no. VPPL/QLN/L1/ALP13); (2a–c) Pollex of Alpheus sp. 3 (Specimen no. VPPL/QLN/L1/ALP14); (3a-c) Dactylus of Alpheus sp. 4 (Specimen no. VPPL/QLN/L1/ALP15). Scale bar equals 300 µm for all. The position of the individual spots (in each specimen) analysed using Electron Dispersive Spectroscopy (EDS) has been marked. Profiles of the EDS analysis of the fossil alpheid claw fingertips recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation (Kerala Basin, southwest India) have been provided as Supplementary Data S1.
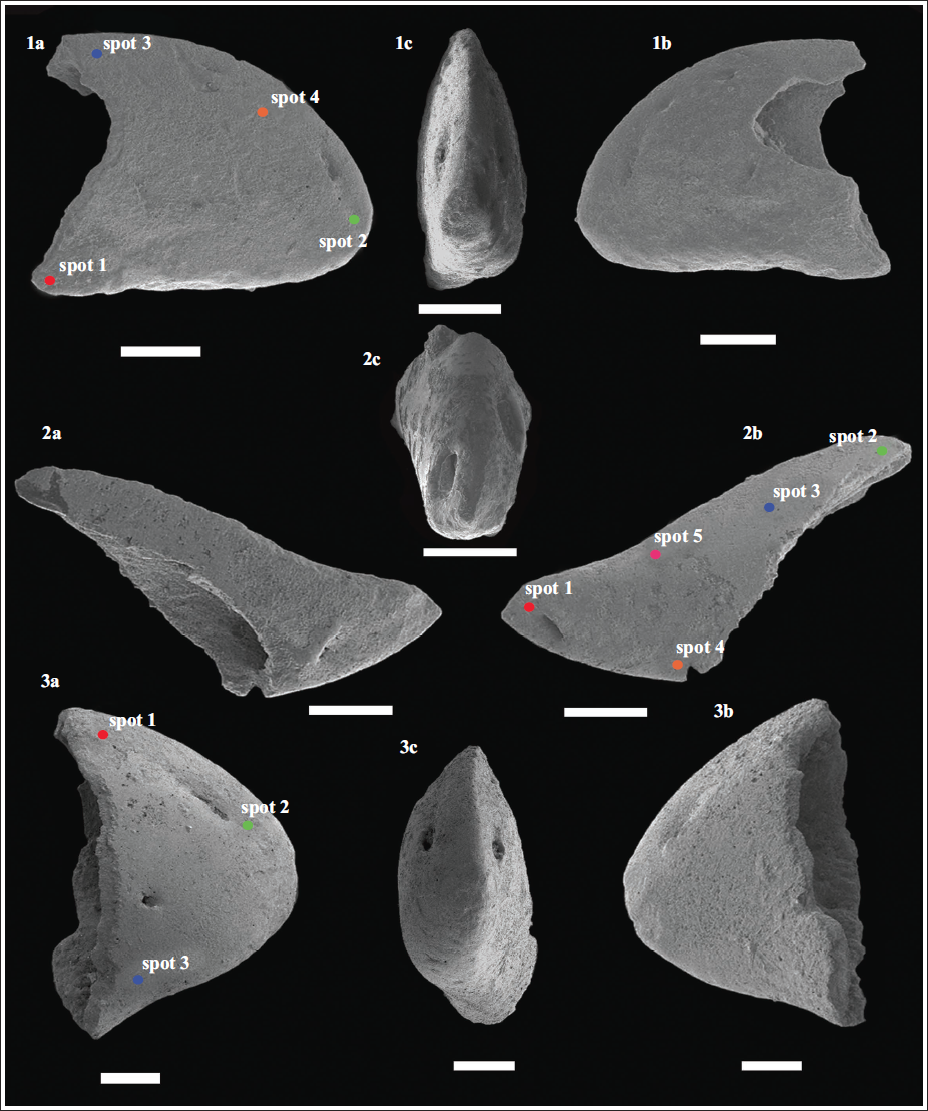
Description: A single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP13) showcasing an overall triangular structure with a strong but blunt tip; having a broadly convex dorsal margin; a straight ventral margin that slightly concaves proximally. Three large setal pores are observed within a depression along the dorsal margin, and one large setal pore is present in the central position. The isolated pollex specimen (specimen no.: VPPL/QLN/L1/ALP14) exhibits a strong, rounded, blunt, dorsally curved spine. One large setal pore is observed on the proximal portion (on both sides).
Remarks: A single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP13) is morphologically similar to ‘Dac-1’ recorded by Lima et al. (2020) from the middle Miocene Pirabas Formation, south America (refer to Figure 4A, C in Lima et al., 2020). However, ‘Dac-I’ sensu Lima et al. (2020) hosts a setal pore on the ventral margin, with a prominent dorsal hook, unlike specimen no. VPPL/QLN/L1/ALP13. The isolated pollex specimen from Quilon (specimen no.: VPPL/QLN/L1/ALP14) is morphologically similar to the pollex morphotype ‘Pol-4’ (Specimen no. MCT7035-I) sensu Lima et al. (2020), also recorded from the middle Miocene Pirabas Formation, South America (see Figure 4F in Lima et al., 2020). However, ‘Pol-4’ sensu Lima et al. (2020) have numerous small setal pores on the outer and inner surfaces, unlike specimen no. VPPL/QLN/L1/ALP14. Taking into consideration the co-occurrence of dactyli ‘Dac-I’ and pollex ‘Pol-4’ recorded from the middle Miocene Pirabas Formation, South America (Lima et al., 2020) and the co-occurrence of dactylus and pollex specimens (specimen no.: VPPL/QLN/L1/ALP13 and VPPL/QLN/L1/ALP14) within the Quilon Formation (herein assigned to Alpheus sp. 3) along with the above-mentioned morphological comparisons, it is quite plausible that the specimen nos. VPPL/QLN/L1/ALP13 and VPPL/QLN/L1/ALP14 may belong to the same species.
Alpheus sp. 4
(Plate 2, Figures 3a–c)
Description: A single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP15) showcasing an overall triangular structure; an obscured fingertip; dorsal margin broadly convex; ventral margin is slightly concave proximally. One large setal pore is positioned in the depression along the dorsal margin, while another smaller setal pore is positioned centrally.
Remarks: The specimen (specimen no.: VPPL/QLN/L1/ALP15) morphologically differs from the recorded dactyli from the Quilon Formation (Kerala Basin, southwest India) as part of the present investigation and the ones recorded previously from the early Miocene (Aquitanian; ~22 Ma) Khari Nadi Formation (Kachchh District, Gujarat State, western India) (see Kapur et al., 2022). The specimen (specimen no.: VPPL/QLN/L1/ALP15) also differs morphologically from the alpheid shrimp fingertips (dactyli) recorded from the Neogene sedimentary successions (Hyžný et al., 2017, 2018; Lima et al., 2020; Pasini & Garassino, 2019 and references therein) from various locations across the globe.
Alpheus sp. 5
(Plate 3, Figures 1a–c)
Scanning electron microphotographs of the alpheid claw fingertips recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation, Kerala Basin, southwest India. (1a–c) Dactylus of Alpheus sp. 5 (Specimen no. VPPL/QLN/L1/ALP16); (2a–c) Dactylus of Alpheus sp. 6 (Specimen no. VPPL/QLN/L1/ALP17); (3a-c) Dactylus of Alpheus sp. 7 (Specimen no. VPPL/QLN/L1/ALP19); Dactylus of Alpheus sp. 8 (Specimen no. VPPL/QLN/L1/ALP20). Scale bar equals 300 µm for all. The position of the individual spots (in each specimen) analysed using Electron Dispersive Spectroscopy (EDS) has been marked. Profiles of the EDS analysis of the fossil alpheid claw fingertips recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation (Kerala Basin, southwest India) have been provided as Supplementary Data S1.
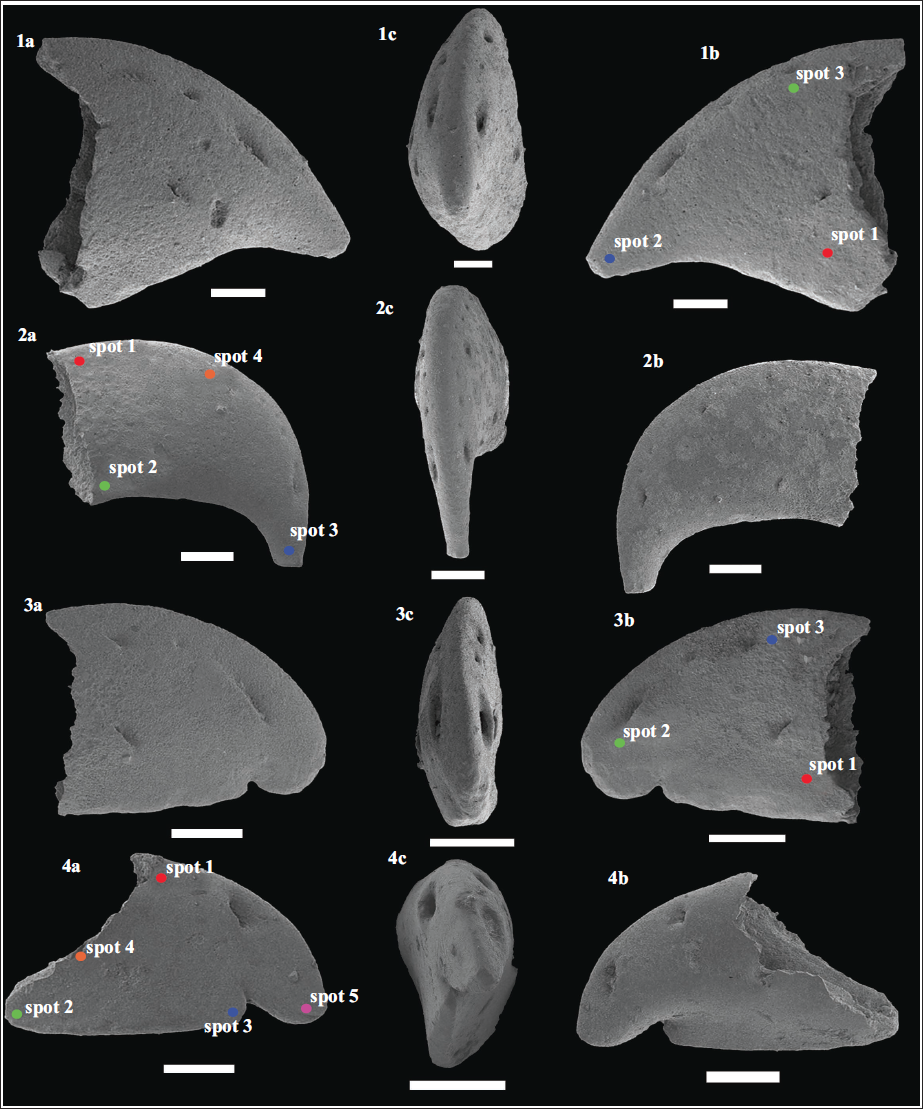
Description: A single isolated dactylus specimen (specimen no.: VPPL/QLN/L1/ALP16) displaying a triangular structure; having a prominent but short fingertip; dorsal margin broadly convex; ventral margin is concave. Three setal pores are positioned in the depression along the dorsal margin, with the anterior-most setal pore larger than the other two. Another comparatively smaller setal pore is positioned close to the proximal margin.
Remarks: The specimen (specimen no.: VPPL/QLN/L1/ALP16) morphologically differs from the recorded dactyli from the Quilon Formation, Kerala Basin, southwest India (this investigation) and the previously recorded dactyli from the early Miocene (Aquitanian; ~22 Ma) Khari Nadi Formation, Kutch Basin, western India (Kapur et al., 2022). Specimen no.: VPPL/QLN/L1/ALP16 also differs morphologically from the Neogene dactyli reported globally (see Hyžný et al., 2017, 2018; Lima et al., 2020; Pasini & Garassino, 2019 and references therein).
Alpheus sp. 6
(Plate 3, Figures 2a–c)
Description: The specimen (specimen no.: VPPL/QLN/L1/ALP17) presents a rectangular structure, having a prominent blunt tip; the dorsal margin is broadly convex; the ventral margin is slightly concave. Numerous small setal pores are positioned in the depression along the dorsal margin.
Remarks: Only two isolated dactyli specimens (specimen nos.: VPPL/QLN/L1/ALP17 and VPPL/QLN/L1/ALP18) were recovered in the present investigation. Only the specimen no. VPPL/QLN/L1/ALP17 is illustrated herein. These isolated dactyli specimens (specimen nos.: VPPL/QLN/L1/ALP17 and VPPL/QLN/L1/ALP18) are quite similar in morphology to the ‘Morphotype IV’ sensu Hyžný et al., 2017 (refer to Figure 3j–l in Hyžný et al., 2017). However, the ventral margin is straight in the dactyli ‘Morphotype IV’ sensu Hyžný et al. (2017), unlike Alpheus sp. 6 recorded in the study.
Alpheus sp. 7
(Plate 3, Figures 3a–c)
Description: The specimen (specimen no.: VPPL/QLN/L1/ALP19) exhibits a broadly triangular structure; having a small blunt tip; the dorsal margin is broadly convex; the ventral margin is straight having a prominent shallow notch towards the anterior portion; three setal pores are positioned in the depression along the dorsal margin and an another setal pore is positioned proximally.
Remarks: The specimen (specimen no.: VPPL/QLN/L1/ALP19) morphologically differs from the previously recorded dactyli from the early Miocene (Aquitanian; ~22 Ma) Khari Nadi Formation, Kutch Basin, western India (Kapur et al., 2022) and other Neogene dactyli reported globally (see Hyžný et al., 2017, 2018; Lima et al., 2020; Pasini & Garassino, 2019 and references therein). The specimen (specimen no.: VPPL/QLN/L1/ALP19) also differs from the other recorded dactyli from the Quilon Formation (Kerala Basin, southwest India), in this investigation.
Alpheus sp. 8
(Plate 3, Figures 4a–c)
Description: The specimen (specimen no.: VPPL/QLN/L1/ALP20) showcases a prominently triangular structure; having a large blunt rounded tip; the dorsal margin is broadly convex; the ventral margin is straight having a prominently deep notch towards the anterior portion; two large setal pores are positioned along the dorsal margin.
Remarks: The specimen (specimen no.: VPPL/QLN/L1/ALP20) morphologically differs from the previously recorded dactyli from the early Miocene (Aquitanian; ~22 Ma) Khari Nadi Formation, Kutch Basin, western India (Kapur et al., 2022) and previously known Neogene dactyli across the globe (see Hyžný et al., 2017, 2018; Lima et al., 2020; Pasini & Garassino, 2019 and references therein). The specimen (specimen no.: VPPL/QLN/L1/ALP20) also differs from the recorded dactyli from the Quilon Formation, Kerala Basin, southwest India (this investigation); however, both Alpheus sp. 7 and Alpheus sp. 8 display a unique presence of a notch on the ventral margin.
Gobiid fishes
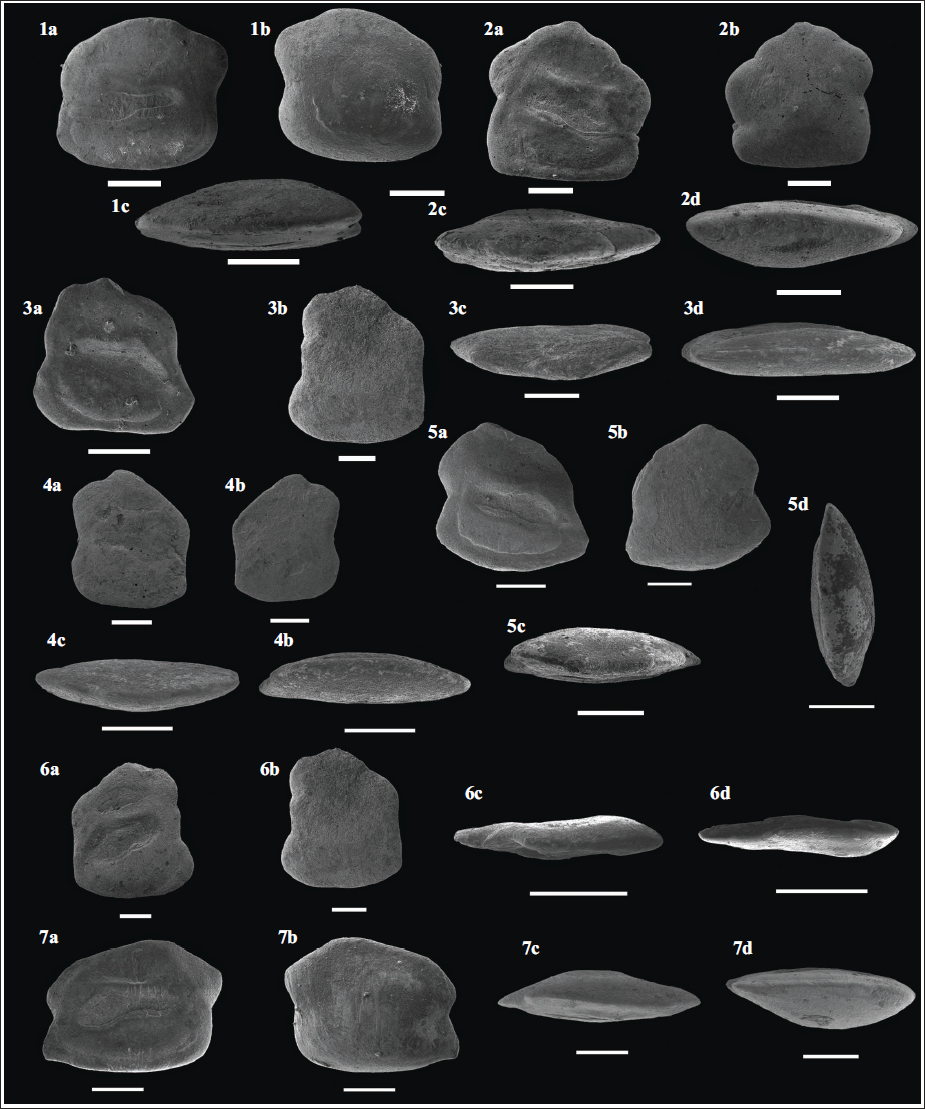
In co-occurrence with the above-mentioned alpheids remains, we recovered >200 otolith specimens belonging to the Family: Gobiidae (Subfamily: Gobiinae) from the early Miocene (Burdigalian) Quilon Formation at the Pozhikkara Cliff Section, Kerala State, southwest India. Of these, only a few select specimens, that is, one specimen for each species, have been illustrated in this investigation (Plate 4, this article). These six illustrated taxa (Ancistrogobius indicus, Acentrogobius matsya, Amblyeleotris kireedam, Amblyeleotris cf. radwanskaae, Drombus thackerae, Gobiodon burdigalicus), and a few other gobiid taxa were also recorded previously from the Quilon Formation at the Pozhikkara Cliff Section, Kerala Basin, southwest India (Figures 5–8 in Carolin et al., 2023) except for Medoborichthys podolicus (refer to Plate 4, this study). Earlier, Prasanna et al. (2021) also recorded otoliths of ‘genus Ambassidarum’ sp. and ‘genus Gobiidarum’ sp. from the Quilon Formation at Pozhikkara Cliff Section (Kerala State, southwest India); however, these authors focused on the palaeoenvironmental reconstruction based on stable and clumped isotopic analysis of the otoliths.
Published record(s) on otoliths from the early Miocene (Burdigalian) Quilon Formation at the Pozhikkara Cliff Section (Kerala Basin, southwest India) suggest that the gobiid genus Ancistrogobius is represented by two species, namely Ancistrogobius indicus and A. yanoi (Carolin et al., 2023). Otoliths belonging to the genus Ancistrogobius are also known from the early Miocene (Aquitanian) Pirabas Formation, Brazil (refer to Figure 12: 5-6 in Aguilera et al., 2014). Of these, a few unidentified otolith specimens recorded by Aguilera et al. (2014) are morphologically very similar to Ancistrogobius indicus (refer to Figure 12: 5a–c in Aguilera et al., 2014; Figure 6a–g in Carolin et al., 2023; Plate 4, Figure 1a–c, this article). The genus Acentrogobius is represented by one named (Acentrogobius matsya) and another unidentified species from the early Miocene (Burdigalian) Quilon Formation at the Pozhikkara Cliff Section, Kerala Basin, southwest India (Carolin et al., 2023). We also observe the presence of Acentrogobius matsya within the Quilon Formation (Plate 4, Figure 2a–d). It is noteworthy that a few unidentified otolith specimens (specimen no. MPEG-1820-V; Figure 12: 6 in Aguilera et al., 2014) recorded from the early Miocene (Aquitanian) Pirabas Formation (Brazil) showcase a similar morphology (e.g., a conspicuously broad concavity on the posterior margin at the level of the cauda) to Acentrogobius sp. recorded previously from the Quilon Formation at the Pozhikkara Cliff Section, Kerala Basin, southwest India (refer to Figure 5g–h in Carolin et al., 2023). The goby genus Amblyeleotris [Asterropteryx-lineage (sensu Agorreta et al. 2013)/reef shrimp gobies (sensu Thacker & Roje, 2011)] from the Quilon Formation at the Pozhikkara Cliff Section is represented by two named species, that is, Amblyeleotris kireedam and Amblyeleotris cf. radwanskaae (Carolin et al., 2023). Both these species were observed in the present investigation (Plate 4, Figures 3a–d, 4a–d) in co-occurrence with alpheids remains (also refer to section ‘Discussion’, this article). The genus Drombus, represented by a single species D. thackerae, was also observed in our investigation (Plate 4: 5a–d). This species was initially recorded by Carolin et al. (2023) from the Quilon Formation at the Pozhikkara Cliff Section, Kerala Basin, southwest India. The goby genus Gobiodon [Gobiodon-lineage (sensu Agorreta et al., 2013)/coral gobies (sensu Thacker & Roje, 2011)] is represented by a single taxon Gobiodon burdigalicus, from the Quilon Formation at the Pozhikkara Cliff Section, Kerala Basin, southwest India (Carolin et al., 2023; also refer to Plate 4: 6a–d, this article). For the first time, we observe the presence of Medoborichthys podolicus in our collection (based on well-preserved otoliths) recovered from the Quilon Formation at the Pozhikkara Cliff Section, Kerala Basin, southwest India (Plate 4: 7a–d). Otoliths belonging to the genus Medoborichthys podolicus have been recorded previously from the middle Miocene deposits of Ukraine (Schwarzhans et al., 2022).
Scanning electron microphotographs of the isolated gobiid (family Gobiidae) otoliths recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation, Kerala Basin, southwest India. (1a–c) Ancistrogobius indicus, right otolith (Specimen no. VPPL/QLN/L1/OTH1); (2a–d) Acentrogobius matsya, left otolith (Specimen no. VPPL/QLN/L1/OTH2); (3a–d) Amblyeleotris kireedam, left otolith (Specimen no. VPPL/QLN/L1/OTH3); (4a–d) Amblyeleotris cf. radwanskaae, left otolith (Specimen no. VPPL/QLN/L1/OTH4); (5a–d) Drombus thackerae, left otolith (Specimen no. VPPL/QLN/L1/OTH5); (6a–d) Gobiodon burdigalicus, right otolith (Specimen no. VPPL/QLN/L1/OTH6); (7a–d) Medoborichthys podolicus, right otolith (Specimen no. VPPL/QLN/L1/OTH7). Scale bar equals 300 µm for all. Note: suffix (a) = inner side view; suffix (b) = outer side view; suffix (c) = dorsal view; suffix (d) = ventral view.
DISCUSSION
The alpheid shrimps belonging to the genus Alpheus (represented by >400 species) are strictly benthic and dominantly thrive within the modern inter-tidal environments, apart from deep marine, estuarine, to freshwater environments, across the globe (Anker et al., 2006 and references therein). Alpheid shrimps (including the ones belonging to the genus Alpheus) are known to exhibit diverse social behaviours such as monogamy, and symbiotic relationships with other diverse faunal groups (e.g., fishes, molluscs, crabs, echinoderms, sponges, and other invertebrates) (Alencar Rodrigues et al., 2009; Anker et al., 2006; Oliveira et al., 2015 and references therein). Alpheid shrimps are generally territorial, that is, have a high inter- and intraspecific competition for shelters; however, symbiotic associations with other alpheids have been observed in ex-situ investigations (Alencar Rodrigues et al., 2009; Costa-Souza et al., 2018, Oliveira et al., 2015, and references therein; Alonso-Domínguez et al., 2022 and references therein). For instance, the burrows constructed by Alpheus estuariensis have been observed to be inhabited by other alpheid shrimps within the modern intertidal environments (Costa-Souza et al., 2018 and references therein). Additionally, a common occurrence of different genera of alpheid shrimps as well as numerous species of the genus Alpheus have been recorded at the same sampling site in ex-situ investigations (Oliveira et al., 2015 and references therein; Hyžný et al., 2017 and references therein; Costa-Souza et al., 2018 and references therein). A recent investigation by Nascimento et al. (2025) on static allometry considering the modulation of shape of the snapping and cutting claws in Alpheus (A. angulosus, A. carlae, and A. nuttingi) suggests that sexual selection tends to favour larger, more robust claws in males that augment their effectiveness during agonistic behaviours. Published literature on fossil alpheids generally considers that the <5 mm-sized strongly calcified triangular and hook-shaped structures are the claw fingertips of the major chelae (Pasini & Garassino, 2019, and references therein). Likewise, we consider that the <5 mm-sized strongly calcified, variously shaped claw fingertips recovered in the present investigation as belonging to the major chelae. Overall, the morphologies of the recovered fingertips do not rule out the presence of both dactyli (movable fingers) and pollex (fixed fingers) pairs (refer to Plate 2, Figure 1a–c, 2a–c).
Interestingly, the oldest record of alpheid fingertips from the Oligocene of Alabama, USA, was inadvertently identified as coleoid guard-like sheaths of spirulid Cephalopods (Ciampaglio & Weaver, 2008). These authors recognised two new genera and species, that is, Oligorostra alabami, and Oligosella longi. However, both Oligorostra alabami and Oligosella longi were synonymised and assigned to the genus Alpheus, considering sexual dimorphism in extant alpheids, for example, in Alpheus lottini (refer to Feldmann et al., 2019; Hyžný et al., 2017, 2018). Subsequent investigations on fossil alpheids (based on claw fingertips) restricted taxonomic assignments of the alpheid fingertips to the generic level, that is, confined to the genus Alpheus (Feldmann et al., 2019; Hyžný et al., 2017, 2018; Kapur et al., 2022; Lima et al., 2020 and references therein). However, palaeontological investigations do acknowledge the presence of several species of Alpheus, considering a large morphological diversity at individual sampling sites (see Hyžný et al., 2017; Lima et al., 2020 and references therein). It is important to mention here that both allopatric and sympatric speciation are known in extant alpheids (Alencar Rodrigues et al., 2009; Oliveira et al., 2015). Morphological investigations in a phylogenetic framework considering the size of the major chelae suggest that sexual dimorphism may have evolved multiple times within Alpheus (Anker et al., 2006 and references therein). However, investigations also propose a lack of size dimorphism within Alpheus species complexes (Costa-Souza et al., 2018). Owing to the ecologically cryptic behaviour of alpheid shrimps, challenges in taxonomy (i.e., the genus Alpheus being paraphyletic with the presence of species complexes, see Anker et al., 2006), the population biology of numerous species within the genus Alpheus is sparsely understood (Costa-Souza et al., 2018 and references therein; Rouzé et al., 2024 and references therein). Thus, the interpretation that the large morphological diversity of isolated claw fingertips in the fossil record solely represents sexual dimorphism is possibly untenable (see Hyžný et al., 2018) and hints at the presence of >1 species (Lima et al., 2020 and references therein). Likewise, the morphological variations within the alpheid fingertips (this study) possibly reflect the taxonomic diversity within the genus Alpheus during the early Miocene (Burdigalian), and the presence of new species cannot be ruled out within the early Miocene (Burdigalian) on the western coast of India.
As already mentioned, the genus Alpheus is paraphyletic and is the most speciose genus of the family Alpheidae, with the presence of numerous species complexes; consequently, their taxonomic classification is very challenging (Anker et al., 2006 and references therein). Palaeontological data on fossil alpheids generally yield variously shaped strongly calcified fingertips of the major chelae that are commonly assigned to the genus Alpheus (see Feldmann et al., 2019; Hyžný et al., 2018 and references therein). Published phylogenetic analysis incorporates morphological characters of the pereiopod 1 ‘P1’ claw of the alpheid shrimps (Anker et al., 2006). These characters include but not limited to P1 dactylus shape; absence or presence of minute, flattened, and rounded granules on the chelae of both minor and major P1 claw; absence or presence of one or several tubercles on the ventral margin of the palm of one (major) or both P1 claw; presence or absence of stout, blunt or subacute projections on the mesial portion of the PI ischium; and the appearance or composition of the PI fingertips in terms of being calcareous, corneous, or amber coloured (refer to Appendix 1 in Anker et al., 2006). Additionally, owing to the ecologically cryptic behaviour of alpheid shrimps, the presence of similar and highly variable species occurring in sympatry, with the common taxonomic characters appearing to form mosaic-like groupings in different geographic regions (e.g., within Alpheus edwardsi species complex), the development and understanding of alpheid systematics has been a difficult task for the biologists. Likewise, taphonomic biases leading to a general absence of soft tissues and the common presence of variously shaped strongly calcified alpheid fingertips in the fossil record are a challenge for palaeontologists. Nevertheless, biologists do consider the presence of additional species of Alpheus in the modern intertidal environments across the globe. The recovery of variously shaped fingertip morphotypes in the fossil record also hints at the presence of new species within the Neogene sedimentary deposits across the globe (Lima et al., 2020 and references therein). Given the above, integrated investigations on both modern and fossil alpheids, especially considering morphological variations in the calcified fingertips of the major chelae of the pereiopod 1 ‘P1’ claw in a morphological phylogenetic framework, should be encouraged in the future.
The highly diverse assemblage of alpheid shrimps (based on fingertips) comprising of one new taxon Alpheus indicus sp. nov. and eight unnamed species belonging to the genus Alpheus, recovered from the early Miocene (Burdigalian; ~17 Ma) Quilon Formation (Kerala Basin, southwest India) is a rarity considering the fossil record(s) of this group from Asia. Considering the fossil evidence from the Quilon Formation (Kerala Basin, southwest India), it is noteworthy that apart from a diverse assemblage of alpheids (this investigation), numerous genera/species of extant gobiid fishes have been reported previously from the Quilon Formation at the Pozikkara Cliff Section (Kerala State, southwestern India). These include Ancistrogobius indicus, Acentrogobius, Amblyeleotris kireedam, Amblyeleotris cf. radwanskaae, Drombus thackerae, Gobiodon burdigalicus) to name a few (see Carolin et al., 2023). However, we herein also record the presence of gobiid fish Medoborichthys podolicus based on isolated otoliths within the Quilon Formation, Kerala Basin, southwestern India (refer to Plate 4, this article).
Numerous living species of the extant genus Alpheus (snapping shrimps or pistol shrimps) are known to form mutualistic relationships with goby fish in both natural (in situ) and controlled (ex situ) environments (Karplus & Thompson, 2011 and references therein). As mentioned, this mutualistic behaviour generally involves the construction and maintenance of the burrow by the shrimp while the goby fish utilises the burrow as a shelter. In return, the goby guards the burrow and acts as an early warning system towards predators, so that both the shrimp and the goby can retreat into the burrow. However, the behavioural interdependence within these groups in varied environments is not entirely comprehended (Karplus & Thompson, 2011 and references therein). For instance, many Alpheus species live in pairs, often sharing their burrow with one or two goby fishes (Wirtz, 2008 and references therein). The living Alpheus bellulus and its partner goby Amblyeleotris japonica are known to form symbiotic relationships wherein the goby provides vital nutrition (in the form of faecal matter) to the alpheid. In contrast, the alpheid shelters the goby fish (see Kohda et al, 2017). Mutualistic relationship(s) between a few species of living goby fish genus Acentrogobius and the alpheid Alpheus brevicristatus are known to affect the morphology of the burrow constructed by the alpheid (Henmi et al., 2020). The living Drombus triangularis is also known to shelter within the burrows constructed by alpheids belonging to the genus Alpheus (Bray, 2018). The gobiid fish otoliths belonging to the genus Medoborichthys resemble otoliths of the Priolepis lineage, particularly of the genus Priolepis (see Schwarzhans et al., 2020, 2022). The extant Banded reef goby, Priolepis cincta, is known to exhibit a mutualistic relationship with pistol shrimps (family Alpheidae), where the shrimp maintains the burrow, and the goby acts as a lookout to warn the shrimp of predators. Shrimp species Alpheus lobidens has been in association (possible mutualistic relationship) with an unidentified goby in the mixed mudflats (intertidal zone) of the north coast of the Gulf of Kutch, western India (Figure 10 in Kapur et al., 2022).
Considering the above symbiotic relationship(s) demonstrated by living alpheids and gobiids, the co-occurrence of alpheid and gobiid fossils (within the same geological sample, that is, at the same level within the same litho-unit) are suggestive of prevalence of mutualistic behaviour within these groups in the geological past (Kapur et al., 2022; Radwańska, 2018). It is noteworthy that Carolin et al. (2023) mentioned the presence of alpheid shrimp remains (quite possibly fingertips) in co-occurrence with otoliths belonging to the family Gobiidae. However, to the best of authors’ knowledge, prior to our investigation, neither illustration(s) nor morphological description(s) are available for alpheids in the published literature from the Quilon Formation exposed at the Pozhikkara Cliff Section, Kerala State, southwestern India (see Carolin et al., 2023). Thus, the detailed morphological comparisons on alpheids’ fingertips (dactyli and pollex) (this study) are limited to published literature on this group from across the globe, including India (refer to supplementary data S1 in Kapur et al., 2022). Based on the co-occurrence of extant goby Amblyeleotris kireedam (otolith remains) and unidentified alpheid elements, Carolin et al. (2023) envisaged the possibility of a mutualistic relationship between these groups during the Burdigalian. Previously, Radwańska (2018) provided middle Miocene evidence from the Paratethys, while Kapur et al. (2022) provided early Miocene (Aquitanian) evidence from the Indian Ocean in support of the development of the mutualistic relationship(s) between these groups during the early Neogene. Additionally, considering the known geographic distribution of Paleogene-Neogene alpheids (Hyžný et al., 2017, 2018; Kapur et al., 2022; Radwańska, 2018; this study), it is evident that alpheids and gobiids thrived in mutualistic relationship(s) persistently within the shallow coasts of the Paratethys Sea and the Indian Ocean region during the Neogene, that is, from the Aquitanian to Burdigalian.
We herein also note the common presence of alpheid ‘Alpheus sp. 1’ within the Aquitanian (~22 Ma) Khari Nadi Formation (Kutch Basin, western India) (Kapur et al., 2022) and the Burdigalian (~17 Ma) Quilon Formation (Kerala Basin, southwest India). Generally, the common presence of fauna showcasing similar morphologies in geographically distant locations during different geological time intervals is argued to favour shared ancestry. For instance, the historical distribution of faunal groups recorded within the Paratethys, Mediterranean and the Indian Ocean realms is suggestive of shared ancestry in the geological past (see Harzhauser et al., 2007 and references therein). Thus, the presence of the gobiid genus Ancistrogobius within the Quilon Formation (southwest India) and the middle Miocene Pirabas Formation, Brazil, south America (see Carolin et al., 2023) and the common presence of Medoborichthys within the late Badenian (Serravallian; ~13-12 Ma) of Ukraine (Schwarzhans et al., 2022) and Burdigalian (~17 Ma) Quilon Formation (this investigation) implies shared ancestry and hints at biotic connectivity between these shallow marine Oceanic realms during the Miocene (see Kapur et al., 2022). However, difficulties exist in interpreting the historical distribution of fauna across the Tethyan Seaway owing to sporadic occurrence of fossils, stratigraphic incompleteness of the fossil record, and heterochrony considered in various faunal groups (Harzhauser et al., 2007 and references therein). Thus, any new palaeontological findings on gobiids and alpheids become crucial to assist in developing palaeobiogeographic scenario(s) within the Paratethys, Mediterranean and the Indian Ocean realms during the early Miocene interval (see Figure 12 and Table 1 in Kapur et al., 2022).
Hyžný et al. (2017) discussed some valuable information on the origin of the family Alpheidae. Molecular data in a phylogenetic framework provide an estimate of 150 Ma for the origin of this family (Bracken et al., 2010). However, the development of a complex snapping claw within alpheids occurred prior to the Miocene (i.e., prior to 23 Ma), and most likely before the late Oligocene (Hyžný et al., 2017) considering that the oldest known fossil alpheids (based on fingertips) have been recorded within the late Oligocene (~28 Ma) Chickasawhay Limestone deposits, Alabama, southeast USA (Ciampaglio & Weaver, 2008; Feldmann et al., 2019). Alpheid records from India, though rare, are the oldest Neogene records for this group in Asia (Kapur et al., 2022a; this study). The Indian subcontinent has been considered a diversity hotspot for numerous marine (e.g., cetaceans, sirenians) and terrestrial (e.g., perissodactyls) fauna (Kapur, 2024 and references therein). Interestingly, the globally oldest record on gobiid fishes (based on otoliths) has been recorded from the early Eocene (~54.5 Ma) Cambay Shale deposits, western India (Bajpai & Kapur, 2004). Thus, it can be postulated that the enhancement of a complex snapping claw within alpheids and/or the mutualistic relationship(s) between alpheids and gobiids developed during the early Paleogene in India. However, further palaeontological investigations are warranted within the older sedimentary deposits (Paleogene) of the subcontinent to support this hypothesis.
CONCLUSIONS
In this investigation, we record a diverse assemblage (comprising a total of nine species) of alpheid shrimps (based on fossilised claw fingertips) assigned to the genus Alpheus (including a new species: Alpheus indicus sp. nov.) from the early Miocene (Burdigalian) Quilon Formation, Kerala Basin, southwest India. Published literature acknowledges the challenges in developing systematics for the paraphyletic genus Alpheus owing to cryptic ecological behaviour of alpheid shrimps, and the presence of similar and highly variable species occurring in sympatry in different geographic regions. Likewise, taphonomic biases resulting in the general absence of complete specimens (soft and hard tissues) and the common presence of variously shaped, strongly calcified claw fingertips in the fossil record are a challenge for palaeontologists. However, numerous investigations do not rule out the presence of new species belonging to the genus Alpheus in modern intertidal environments and the Neogene fossil record. In this regard, integrated biological and palaeontological investigations taking into account the morphological variations in the calcified fingertips of the major chelae of the pereiopod 1 ‘P1’ claw in a morphological phylogenetic framework are certainly warranted.
The alpheid remains (this investigation) co-occur with fossil remains (mainly otoliths) of numerous gobiid fish genera, including Medoborichthys (represented by M. podolicus) within the Quilon Formation. This fossil evidence supports the idea (see Hyžný et al., 2017) that the mutualistic relationship(s) within these groups had developed prior to the Burdigalian. Based on the available Paleogene-Neogene geographic distribution of alpheids and gobiids, it is evident that these groups quite likely thrived in mutualistic relationship(s) persistently within the shallow coasts of the Paratethys Sea and the Indian Ocean region during the Neogene (Aquitanian to Burdigalian). Further, any argument in favour of the enhancement of a complex snapping claw within alpheids and/or the development of the mutualistic relationship(s) between alpheids and gobiids during the early Paleogene in India warrants older records from the subcontinent.
Footnotes
Acknowledgements
The authors are grateful to Professor Mahesh G Thakkar (Director, BSIP) for providing the necessary infrastructure, support, and permissions to carry out this investigation (Permission no. BSIP/RDCC/Publication no. 05; 2025-26). Funding support in the form of Anusandhan National Research Foundation (ANRF) (formerly SERB) Core Research Grant (CRG/2022/003341) is thankfully acknowledged. AMS acknowledges funding support from ANRF (formerly SERB) CRG in the form of a Junior Research Fellowship. The authors thank the Chief Editor, (Professor Mukund Sharma) for the invitation. The authors also thank Professor Carrie E. Schweitzer (USA) and anonymous reviewers for providing constructive commentaries.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding support from the Anusandhan National Research Foundation (ANRF) (formerly SERB) under the Core Research Grant (CRG), Grant No. CRG/2022/003341, which included both a research grant and a Junior Research Fellowship.
Repository
The referred and/or illustrated microfossil specimens (this article) are housed in the Birbal Sahni Institute of Palaeosciences, Lucknow, India (BSIP Museum locality no. 10526; BSIP Museum micropalaeontology slide nos. 17610-17611) under the Acronym(s): VPPL/QLN/L1/ALP and VPPL/QLN/L1/OTH; Abbreviations: VPPL, Vertebrate Palaeontology and Preparation Laboratory; QLN, Quilon Formation; L1, yielding sample number within carbonaceous clay unit; ALP, alpheid; OTH, otolith.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
