Abstract
The present study investigates seasonal variations in pollen and nectar sources harnessed by honey bees across densely vegetated areas of the Gangetic plains of Uttar Pradesh. The study explores how pollen content in honey reflects the floral diversity, availability of resources, and forage preferences of honey bees. A melissopalynological study in four seasons (spring, summer, rainy, and winter) from ten densely vegetated areas of the Gangetic plain (sub-humid tropics) shows diversity of nineteen pollen types. Pollen of Azadirachta indica A. Juss., Bombax ceiba L., Caesalpinia pulcherima Sw., Syzygium cumini L., and Anacardiaceae were found to be dominant arboreal pollen content in honey samples. The Pollen of Asteroideae, Cichorioideae, Solanaceae, Malvaceae, and Ziziphus sp. Lam., Brassica campestris L., and Coriandrum sativum L. were considerably involved in honey samples. Acacia catechu L., Albizia lebbeck Benth., Cassia fistula L., Eucalyptus citriodora Hook., Lagerstroemia lanceolata Wall., Oleaceae, and Poaceae were also found to be pollen sources in honey. Despite diverse flora in densely vegetated areas, the collected honey samples show less diversity in their pollen content, showing honey bees rarely visit non-preferred species, if their preferred plant species are easily accessible and available during different seasons.
Keywords
INTRODUCTION
Honey bees, the natural pollinators of plants, depend upon diverse floral resources for survival. They glean pollen along with nectar from the flowers of the plant species they forage and proselytise them into honey to satisfy their nutritional needs. Bees collect nectar and consume it either more or less immediately or store it as honey. They collect pollen to feed young larvae or nurse bees, or it is stored in and around the brood nest in the form of ‘bee bread’ for later consumption (Somerville, 2020). Nectar is the chief source of carbohydrate, and pollen grains are rich in proteins, fats, and vitamins. Honey bees gather pollen when available, whether or not it is needed for immediate consumption (Todd & Vansell, 1940). Availability of pollen and nectar for honey bees depends on the type and density of vegetation around the bee hives. Even seasonal variations affect the foraging behaviour of honey bees. Hence, knowledge of local flora helps ascertain bees’ preferences for foraging. This can be done by identifying pollen grains in honey (Melissopalynology). The Melissopalynological data provides botanical and geographical provenance of honey as well as seasonality of nectar flow. Melissopalynology plays a vital role in apiculture and environmental science as it helps to identify the specific floral sources that honey bees visit frequently (Agwu & Akanbi, 1985). Through the microscopic analysis of pollen grains found in honey samples, one can map out the foraging pattern of bees, revealing the type of plants they visit and the extent of their foraging ranges (Chakraborty et al., 2023; Chauhan & Trivedi, 2011).
Understanding these foraging patterns is crucial for several reasons. First, it provides insights into the biodiversity of the regions where the bees are active. By identifying the variety of pollen grains, scientists can infer the presence of different plant species, thereby assessing a particular area’s ecological health and floral diversity (Hernandez & Lopez, 2018; Smith & Brown, 2018). Second, melissopalynology helps determine the seasonal availability of floral resources. Since the flowering periods of plants vary throughout the year, the pollen content in honey changes accordingly, reflecting the seasonal shifts in bee foraging activities (Evans & Roberts, 2019; Singh & Sharma, 2015).
Seasonal variations in pollen availability have a profound impact on honey production and quality. During certain times of the year, the abundance and diversity of flowering plants can lead to higher honey yields and improved honey quality due to the availability of rich and varied nectar sources (Jones & Wang, 2017; Ramirez et al., 2022). Conversely, during seasons when fewer plants are in bloom, honey production can decline, and the quality may be affected if bees are forced to forage from less desirable or less diverse floral sources (Black et al., 2019; Chen et al., 2017). This seasonal dependency underscores the importance of maintaining diverse and healthy ecosystems to support sustainable apiculture (Clark & Wilson, 2017; Thomas & Brown, 2017).
Studies conducted worldwide emphasise the utility of Melissopalynology in various climates and ecosystems. For instance, Louveaux et al. (1978) developed standardised Melissopalynological techniques that continue to guide international pollen identification and quantification research. Research in Europe demonstrates how honey pollen profiles reflect floral diversity and emphasise the role of specific plants, such as Eucalyptus, in Mediterranean regions, which strongly influence honey composition (Floris et al., 2020). In tropical regions, where plant diversity is high, Melissopalynological studies have helped identify key nectar sources and assess floral competition, impacting bee foraging behaviours and honey production (Agwu & Akanbi, 1985).
In recent decades, Melissopalynology has expanded into environmental monitoring, utilising pollen spectra in honey to assess pollution and habitat health (Celli & Maccagnani, 2003). Such studies show how environmental contaminants, such as pesticides and heavy metals, can influence honey composition and reflect broader ecological impacts (Bogdanov et al., 2007). Melissopalynological research has also addressed conservation challenges, encouraging for diverse and pollinator-friendly landscapes to sustain honey bee populations, as evidenced by studies in both urban and agricultural contexts (Persano Oddo et al., 2004).
Continued development in Melissopalynology, including molecular identification methods, enhances the precision of pollen analysis and broadens the field’s applications, underscoring its relevance in ecological research and conservation (Rasmussen & Delgado, 2009). By connecting honey composition with the environment, Melissopalynology remains vital for understanding plant-pollinator interactions, supporting sustainable apiculture, and promoting biodiversity.
In India, Melissopalynology research across various regions has illuminated the diversity of floral resources available to honey bees and their influence on honey composition. A significant contribution has been made in the field of Melissopalynology in India (Basumatary, 2013; Basumatary & Murthy, 2014; Bera et al., 2004; Chauhan & Murthy, 2010; Chauhan & Quamar, 2010; Ponnuchamy et al., 2014; Tripathi et al., 2017). In northern India, including areas like Uttar Pradesh and Punjab, honey samples often feature pollen from Brassica species, prominent during the cooler months, along with other common crops (Singh et al., 2015). In the plains of sub-humid regions, diverse pollen types, including Syzygium sp., Azadirachta, Acacia sp., and cultivated plants, have been identified, reflecting a mix of urban and rural flora that contributes to the botanical richness of the honey (Kumar & Nair, 1994). In semi-arid regions, Melissopalynology has shown that bees rely heavily on drought-resistant flora like Prosopis juliflora and Ziziphus sp., which dominate the pollen spectra of honey produced there, underscoring the adaptability of bees to the local climate and plant availability (Harianja et al., 2023).
In southern India, Melissopalynological studies have identified Eucalyptus sp., Coconut, and Cashew pollen as dominant components in honey, reflecting the regional flora. These plant species contribute to the specific taste and quality of honey in this subcontinent. Meanwhile, in the Western Ghats, known for its biodiversity, studies have shown a unique pollen profile in honey that includes endemic plants, highlighting the ecological importance of conserving native flora for sustainable apiculture (Ramanujam & Kalpana, 1992). This regional variation in pollen sources underscores the importance of localised studies, as Melissopalynology not only aids in authenticating honey but also provides insights for managing floral resources and supporting bee populations throughout India (Richard et al., 2014). The studies collectively highlight the importance of Melissopalynology in supporting apiculture and enhancing our understanding of bee ecology.
By providing detailed insights into honey bees’ foraging patterns and the floral resources they depend on, Melissopalynology not only helps improve honey production practices but also contributes to the conservation of biodiversity and the sustainability of agricultural ecosystems (Baker & Hill, 2018; Fernandez & Rodriguez, 2015; Jackson & Green, 2016).
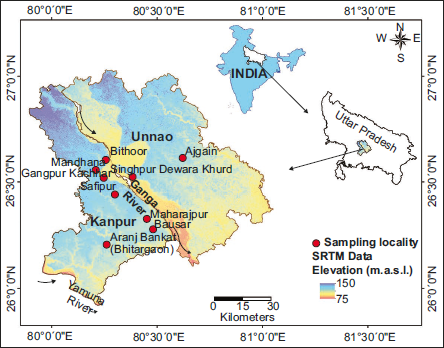
The present work incorporates Melissopalynological studies from densely vegetated areas (forest patches), namely Aranj Bankat (Bhitargaon), Bausar, Maharajpur, Safipur, Singhpur, Gangpur Kachhar, Mandhana, Bithoor, Dewara Khurd, and Ajgain in the Gangetic plains of Kanpur and Unnao districts of Uttar Pradesh, India, during four different seasons, namely spring, summer, rainy, and winter, in order to understand foraging habits and seasonal preferences in areas of rich floral diversity. In addition, the representation of pollen taxa has been observed with the earlier published work on modern pollen and vegetation relationships in and around the study area (Tripathi et al., 2016).
Study Area, Climate, and Vegetation
The honey samples for the present study were collected from the densely vegetated regions of Kanpur and Unnao districts in Uttar Pradesh, Central India. These districts lie along the fertile Gangetic Plain, with the Ganga River forming a major geographical hallmark. Kanpur Nagar spans urban and peri-urban regions as a significant industrial and commercial hub. Unnao, on the other hand, is predominantly rural, with agriculture and small-scale industries forming the backbone of its economy. The districts are situated between approximately 26°N and 27°N latitude and 80°E and 81°E longitude. Kanpur Nagar covers an area of 3,155 km², while Unnao encompasses around 4,558 km². The region experiences a subtropical monsoon climate, characterised by hot summer, humid monsoon, and cool winter (Singh, 1966). There are four distinct seasons namely spring (middle of February - middle of March), summer (middle of March - June) which is hot and dry, followed by monsoon (July - September) representing humid, rainy and winter (November - middle of February) varying from mild cool to very cold. The summer peak temperature often exceeds 40°C (104°F), leading many deciduous trees to shed leaves as a water conservation strategy. In contrast, during winters, the average temperature ranges from 5°C to 15°C (41°F to 59°F) (Indhu et al., 2023). The low temperature below 10°C supports the flowering of late-season plants like Brassica sp., Coriandrum sp., Triticum sp., etc. The monsoon brings moderate to heavy rainfall, rejuvenating the forests and stimulating flowering in species like Acacia sp., Syzygium sp., etc., vital for honeybee foraging. The annual rainfall averages between 800 and 1,000 mm, with the southwest monsoon winds contributing most of the precipitation. Occasional light showers occur during winter, influenced by western disturbances and pre-monsoon periods. These showers provide crucial support for winter crops such as wheat (Triticum aestivum) and mustard (Brassica campestris) (Mall et al., 2006). These seasonal changes ensure year-round availability of floral resources for honeybees, though with significant temporal variation.
The humidity level in the region is generally low, ranging from 20% to 40%. However, it increases slightly in June as the moisture level rises in anticipation of the monsoon. During the monsoon season, humidity peaks at 70%–90%, creating a warm and sticky environment (IMD, 2024). These districts, located in the upper Gangetic Plains, are characterised by diverse ecosystems influenced by the mighty Ganga River, agricultural landscapes, and pockets of dry deciduous forests. These regions support a mix of natural and human-modified habitats, hosting a variety of plant and animal species (Kanaujia & Kumar, 2023).
The fertile alluvial soil, deposited by the Ganga River and its tributaries, makes these districts highly suitable for agriculture. Typical features of the Indo-Gangetic Alluvial Plain dominate the geomorphology of the districts. Key geomorphological units include floodplains, Older Alluvial Plains (OAPs), ravines and gullies, and aeolian deposits (Singh, 1996). Overall, the region has a flat to gently undulating topography, with elevation ranging between 100 and 150 meters above sea level (Singh, 2002). Oxbow lakes and abandoned river channels near the Ganga River indicate active fluvial processes shaping the landscape over time.
The flora of Kanpur and Unnao reflects the region’s subtropical monsoon climate and fertile alluvial soils. Vegetation can be broadly categorised into natural forests, riparian vegetation, crops, and planted species in urban areas (Champion & Seth, 1968). Among the moist/dry deciduous forests, dominant tree species include Acacia catechu L., Acacia nilotica L., Aegle marmelos L., Ailanthus excelsa Roxb., Albizia lebbeck Benth., Alstonia scholaris R. Br., Anthocephalus Kadamba Roxb., Azadirachta indica A. Juss., Bauhinia sp. L., Bombax ceiba L., Butea monosperma (Lam.) taub., Ceiba pentendra L., Dalbergia sissoo Roxb., Feronia limonia L., Ficus religiosa L., Ficus benghalensis L., Grewia asiatica L., Holoptelia integrifolia Planch., Madhuca indica J.F. Gmel, Melia azedarach L., Millettia ovalifolia (Wight & Arn.) Kurz., Moringa oleifera Lam., Pithecelobium dulce Benth., Polyalthia longifolia Thw., Pongamia pinnata L., Prosopis spicegera, Syzygium cumini L., Tamarindus indica L., Tectona grandis L.f., Terminalia arjuna (Roxb.) Wight & Arn., Terminalia bellerica (Gaertn.) Roxb. and Ziziphus mauritiana Lam. Amidst the shrubs, the common species include Prosopis juliflora (Sw.) DC., Adhatoda vasica Medic., Calotropis gigantea, Cassia sp. L., Ipomoea sp. L., Hyptis suaveolens, Murraya sp. L., Nyctanthes arbortistis L., Ziziphus nummularia (Burm.f.) Wight & Arn., and various grasses such as Cynodon dactylon (L.) Pers., Dactyloctenium aegyptium (L.) Willd., Echinochloa sp. L., Eleusine indica Gaertn., Panicum sp. Retz., Saccharum munja Roxb., Setaria sp. Beauv. and Sonchus sp. L. The crops dominate the landscape, with wheat, rice, sugarcane, mustard, pulses, and vegetables being widely grown. Fruit trees such as mango (Mangifera indica L.), guava (Psidium guajava L.), lemon (Citrus sp. L.), and banana (Musa sp. Colla.) are also common in orchards. In urban and semi-urban areas, planted trees/shrubs like Callistemon lanceolatus, Cassia fistula, Caesalpinia pulcherrima, Delonix regia (Bojer ex Hook.) Raf., Eucalyptus sp., Lagerstroemia lanceolata Wall., Murraya koenigii L., Plumeria sp. L., Thevetia peruviana (Pers.) Merrill is prominent in parks, roadsides, and residential areas (Kanaujia & Kumar, 2023).
MATERIALS AND METHODS
Field work
The sampling locations namely Aranj Bankat (26.2123°N, 80.2732°E), Bausar (26.2842°N, 80.4886°E), Maharajpur (26.3328°N, 80.4602°E), Safipur (26.4450°N, 80.3103°E), Gangpur Kachhar (26.5212°N, 80.2590°E), Singhpur (26.5232°N, 80.2586°E), Mandhana (26.5560°N, 80.2233°E), Bithoor (26.6057°N, 80.2698°E), Dewara Khurd (26.5252°N, 80.3953°E), and Ajgain (26.6151°N, 80.6258°E) (Figure 1) were strategically selected to understand diverse range of floral resources crucial for honeybee foraging in vegetated area.
Laboratory work
The standard acetolysis method (Erdtman, 1952) was used for the pollen analysis from the honey samples. To create the cluster bar graph of pollen taxa, 500–600 pollen spores were counted. Identification and characterisation of pollen spores were performed using published literature (Basumatary, 2013; Basumatary & Murthy, 2014; Bera et al., 2004; Chauhan & Murthy, 2010; Chauhan & Quamar, 2010; Ponnuchamy et al., 2014; Tripathi et al., 2017). Microscopic observation and photo-documentation of pollen spores were made using an Olympus BX-61 microscope with a DP-25 digital camera under 40X magnification.
RESULTS
Localities and pollen composition
The composition of honey samples collected from different localities during four seasons is discussed below:
Aranj Bankat (Bhitargaon)
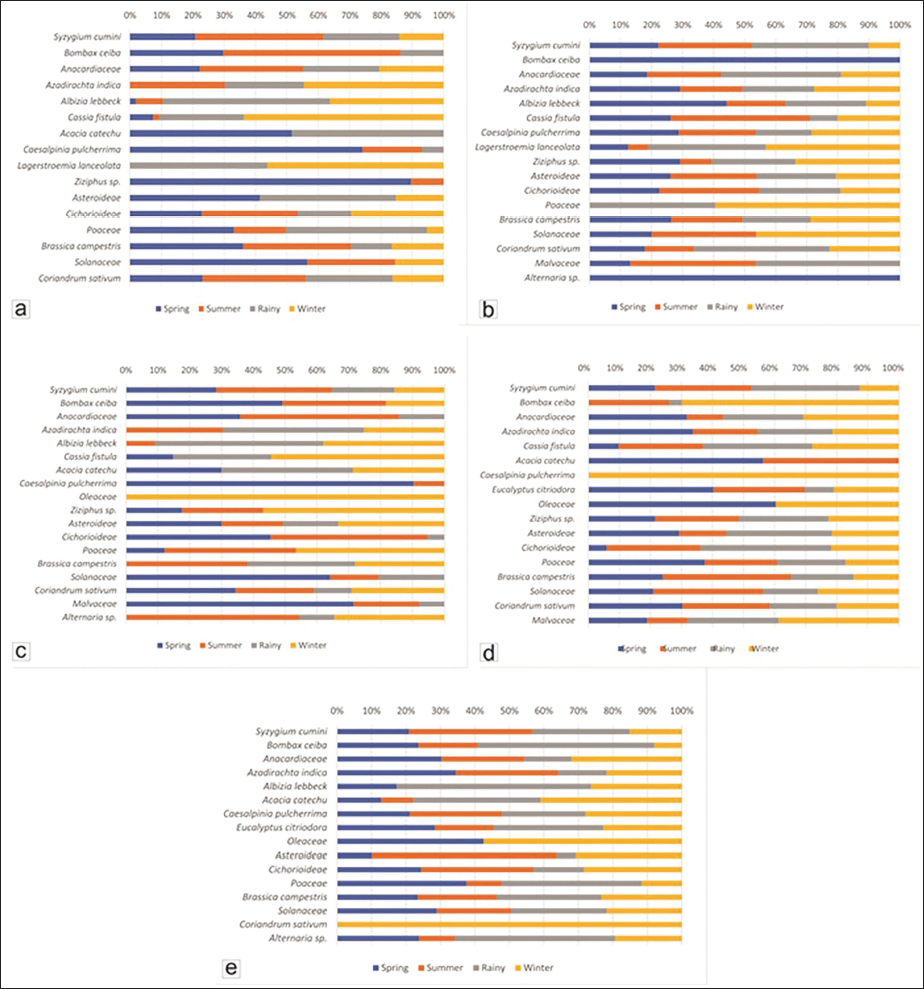
The honey sample collected from densely vegetated area of Aranj Bankat is found to be multi-floral with dominance of Brassica campestris pollen (23.80%) and arboreal pollen comprising of Caesalpinia pulcherrima (13.31%), Acacia catechu (10.76%), Syzygium sp. (8.22%), Bombax ceiba (3.4%), Cassia fistula (2.27%), Albizia lebbeck (0.57%) along with pollen of Ziziphus sp. (4.82%), Coriandrum sativum (3.97%), Anacardiaceae (12.18%), Asteroideae (4.25%), Cichorioideae (1.70%), Solanaceae (4.53%), Malvaceae (0.28%) and Poaceae (3.97%) with Alternaria sp. Sorauer, spores (1.98%) in the spring season.
Summer honey also shows more or less similar pollen assemblage with dominance of Brassica (22.82%), Anacardiaceae (18.03%), Syzygium sp. (16.06%), Azadirachta indica (14.65%) along with Bombax ceiba (6.48%), Coriandrum sativum (5.63%), Caesalpinia pucherrima (3.38%), Albizia lebbeck (2.54%), Solanaceae (2.25%), Cichorioideae (2.25%), Poaceae (1.97%), Malvaceae (1.13%) Ziziphus sp. (0.56%), Cassia fistula (0.56%) and Alternaria spores (1.69%). Brassica campestris and Coriandrum sativum pollen show the cultivation area near the densely vegetated region.
In rainy season the honey is composed of pollen of Albizia lebbeck (15.51%), Anacardiaceae (13.29%), Azadirachta indica (12.03%), Acacia catechu (10.13%), Syzygium cumini (9.49%), Brassica campestris (8.54%), Cassia fistula (8.23%), Poaceae (5.38%), Coriandrum sativum (4.75%), Asteroideae (4.43%), Lagerstroemia lanceolata (4.11%), Bombax ceiba (1.58%), Caesalpinia pulcherrima (1.27%) and Cichorioideae (1.27%).
Winter honey is dominated by pollen of Albizia lebbeck (21.50%) and Cassia fistula (19.31%) with co-dominance of Azadirachta indica (11.21%), Brassica campestris (10.90%) and Anacardiaceae (10.59%) along with Malvaceae (5.92%), Syzygium cumini (5.61%), Lagerstroemia lanceolata (5.30%), Coriandrum sativum (2.80%), Cichorioideae (2.18%), Asteroideae (1.56%), Solanaceae (1.25%), Poaceae (0.62%) pollen and spores of Alternaria sp. (1.25%) an endophytic fungi with highly bioactive metabolites (Karwehl & Stadler, 2016, Figure 2a).
Bausar
Spring honey has dominance of Azadirachta indica pollen (18.01%) after Brassica campestris (19.11%) along with pollen of Caesalpinia pulcherrima (10.25%), Syzygium sp. (9.70%), Asteroideae (8.31%), Anacardiaceae (8.03%), Solanaceae (6.65%), Cichorioideae (5.82%), Albizia lebbeck (3.32%), Ziziphus sp. (3.32%), Cassia fistula (2.49%), Coriandrum sativum (1.94%), Lagerstroemia lanceolata (0.55%), Malvaceae (0.55%) and Bombax ceiba (0.28%).
Summer honey comprises of pollen of Brassica campestris (16.57%), Syzygium sp. (13.14%), Azadirachta indica (12.29%), Solanaceae (11.14%), Anacardiaceae (10.29%), Caesalpinia pulcherrima (8.86%), Asteroideae (8.86%), Cichoroideae (8.29%), Cassia fistula (4.29%), Coriandrum sativum (1.71%), Malvaceae (1.71%), Albizia lebbeck (1.43%), Ziziphus sp. (1.14%) and Lagerstroemia lanceolata (0.29%).
In the rainy season, the pollen assemblage is dominated by Anacardiaceae (16.76%), Syzygium sp. (16.48%), Azadirachta indica (14.25%), and Brassica campestris (15.92%) pollen. Pollen of Asteroideae (8.10%), Cichoroideae (6.70%), Coriandrum sativum (4.75%), Ziziphus sp. (3.07%), Malvaceae (1.96%), Albizia lebbeck (1.96%), Lagerstroemia lanceolata (1.68%), Poaceae (1.12%) and Cassia fistula (0.84%) are also present.
Winter season honey is dominated by pollen of Brassica campestris (20.82%), Azadirachta indica (16.99%), Solanaceae (15.34%) and Caesalpinia pulcherrima (10.14%). Pollen of Anacardiaceae (8.22%), Asteroideae (6.58%), Cichoroideae (4.93%), Syzygium sp. (4.38%), Ziziphus sp. (3.84%), Coriandrum sativum (2.47%), Lagerstroemia lanceolata (1.92%), Cassia fistula (1.92%), Poaceae (1.64%) and Albizia lebbeck (0.82%) are also present (Figure 2b).
In the Bausar region, honey is also multi-floral, with Brassica campestris pollen’s dominance in all four seasons, suggesting agricultural land near a forested area and less anthropogenic activity.
Maharajpur
Spring honey comprises of pollen of Caesalpinia pulcherrima (19.81%), Solanaceae (15.17%), Syzygium sp. (11.46%), Anacardiaceae (9.29%), Malvaceae (8.36%), Coriandrum sativum (7.12%), Bombax ceiba (6.81%), Cassia fistula (5.26%), Cichorioideae (5.26%), Acacia catechu (4.64%), Asteroideae (3.72%), Ziziphus sp. (1.86%), Poaceae (1.24%).
While summer honey has pollen of Brassica campestris (18.98%), Azadirachta indica (15.36%), Syzygium sp. (14.76%), Anacardiaceae (12.95%), Cichorioideae (5.72%), Coriandrum sativum (5.12%), Bombax ceiba (4.52%), Solanaceae (3.61%), Poaceae (4.21%), Albizia lebbeck (3.61%), Ziziphus sp. (2.71%), Malvaceae (2.41%), Asteroideae (2.41%), Caesalpinia pulcherrima (2.11%), Alternaria sp. (1.51%).
In rainy season Azadirachta indica (22.26%), Albizia lebbeck (20.73%), Brassica campestris (16.77%), Cassia fistula (10.98%), Syzygium sp. (7.93%), Acacia catechu (6.40%), Solanaceae (4.88%), Anacardiaceae (3.66%), Coriandrum sativum (2.44%), Asteroideae (2.13%), Malvaceae (0.91%), Cichorioideae (0.61%) pollen are present along with Alternaria sp. (0.30%).
Winter honey shows presence of Pollen of Cassia fistula (19.37%), Albizia lebbeck (14.92%), Brassica campestris (13.97%), Azadirachta indica (12.70%), Syzygium sp. (6.35%), Coriandrum sativum (6.03%), Ziziphus sp. (6.03%), Acacia catechu (4.44%), Asteroideae (4.13%), Oleaceae (3.81%), Cereals (2.86%), Bombax ceiba (2.54%), Poaceae (1.90%) and Alternaria sp. spores (0.95%) (Figure 2c).
Beautiful gardens, public parks, echo parks, and amusement parks have been developed nearby in the densely vegetated area of Maharajpur. This is why the pollen of ornamental plants, dry/moist deciduous forest trees, and cultivated plants is present.
Safipur
Multi-floral honey of spring season in Safipur comprises of pollen of Anacardiaceae (17.81%), Coriandrum sativum (15.07%), Azadirachta indica (12.33%), Syzygium sp. (8.22%), Ziziphus sp. (7.40%), Solanaceae (6.03%), Brassica campestris (5.75%), Acacia catechu (5.48%), Eucalyptus citriodora (4.66%), Asteroideae (4.38%), Poaceae (4.11%), Malvaceae (3.29%), Cassia fistula (2.47%), Oleaceae (1.64%) and Cichorioideae (1.37%).
Summer honey dominates herbaceous pollen of Coriandrum sativum (13.92%), Brassica campestris (9.94%), Solanaceae (10.23%), Cichorioideae (7.10%), Poaceae (2.56%), Asteroideae (2.27%), Malvaceae (2.27%) along with arboreal pollen of Syzygium sp. (11.93%), Ziziphus sp. (9.38%), Azadirachta indica (7.67%), Cassia fistula (6.82%), Anacardiaceae (6.53%), Acacia catechu (4.26%), Eucalyptus citriodora (3.41%), and Bombax ceiba (1.70%).
Pollens present in rainy season are of Anacardiaceae (14.48%), Syzygium sp. (13.40%), Coriandrum sativum (10.72%), Ziziphus sp. (9.92%), Cichorioideae (9.92%), Azadirachta indica (8.85%), Cassia fistula (8.85%), Solanaceae (5.09%), Asteroideae (5.09%), Malvaceae (5.09%), Brassica campestris (4.83%), Poaceae (2.41%), Eucalyptus citriodora (1.07%) and Bombax ceiba (0.27%).
Winter honey has Anacardiaceae (17.25%), Coriandrum sativum (9.97%), Caesalpinia pulcherrima (9.16%), Ziziphus sp. (7.82%), Azadirachta indica (7.82%), Solanaceae (7.55%), Cassia fistula (7.01%), Malvaceae (6.74%), Cichorioideae (5.12%), Syzygium sp. (4.85%), Bombax ceiba (4.58%), Brassica campestris (3.50%), Asteroideae (3.23%), Eucalyptus citriodora (2.43%), Poaceae (1.89%) and Oleaceae (1.08%) pollen (Figure 2d).
Anacardiaceae pollen dominates the honey samples of Safipur.
Gangpur Kachhar
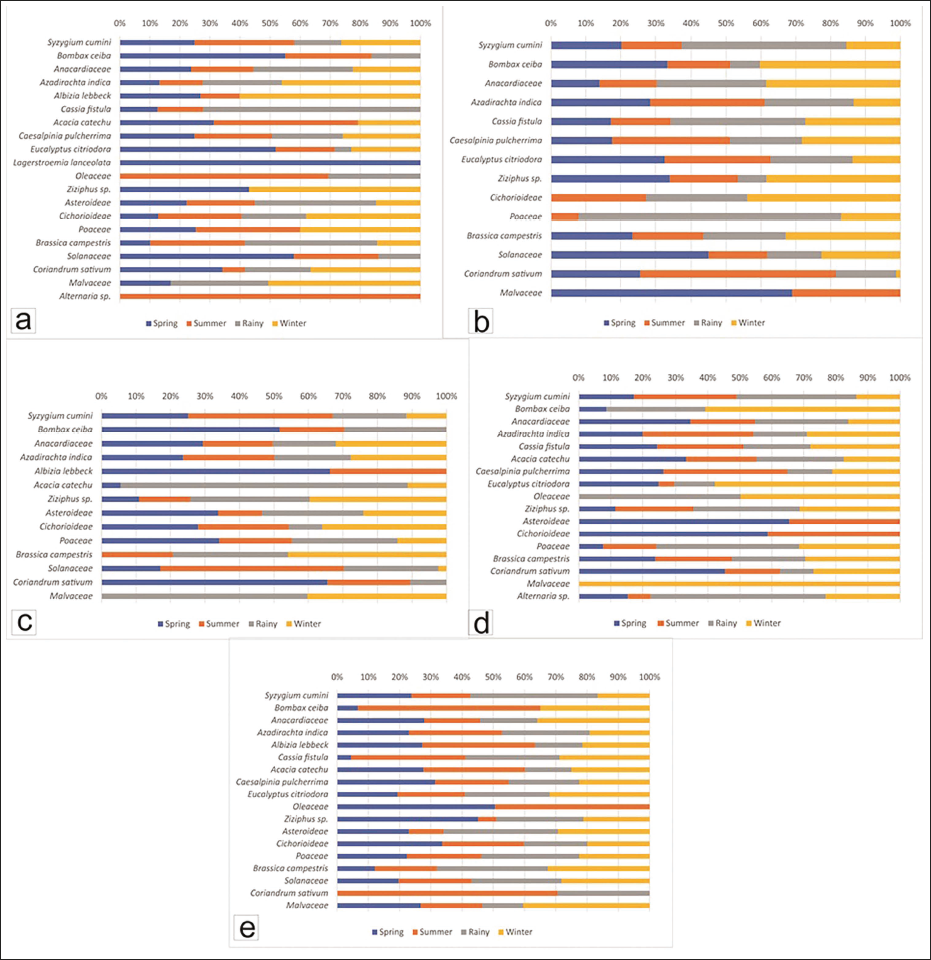
Spring honey is found to have pollen of Anacardiaceae (17.36%), Brassica campestris (15.80%), Caesalpinia pulcherrima (12.69%), Poaceae (9.33%), Solanaceae (9.07%), Eucalyptus citriodora (7.51%), Syzygium sp. (7.25%), Cichorioideae (6.99%), Azadirachta indicia (6.22%), Asteroideae (2.07%), Bombax ceiba (1.55%), Acacia catechu (1.55%), Alternaria sp. (1.30%), Oleaceae (0.78%) and Albizia lebbeck (0.52%).
Summer honey has Caesalpinia pulcherrima (16.06%), Brassica campestris (15.49%), Anacardiaceae (13.80%), Syzygium sp. (12.39%), Asteroideae (10.99%), Cichorioideae (9.30%), Solanaceae (6.76%), Azadirachta indica (5.35%), Eucalyptus citriodora (4.51%), Poaceae (2.54%), Bombax ceiba (1.13%) and Acacia catechu (1.13%) pollen.
Brassica campestris (20.51%), Caesalpinia pulcherrima (14.61%), Poaceae (10.11%), Syzygium sp. (9.83%), Solanaceae (8.71%), Eucalyptus citriodora (8.43%), Anacardiaceae (7.87%), Acacia catechu (4.49%), Cichorioideae (4.21%), Bombax ceiba (3.37%), Azadirachta indica (2.53%), Alternaria sp. (2.53%), Albizia lebbeck (1.69%), Asteroideae (1.12%) pollen are obtained from rainy season honey.
Winter honey takes in pollen of Anacardiaceae (18.37%), Caesalpinia pulcherrima (16.80%), Brassica campestris (15.75%), Cichorioideae (8.14%), Solanaceae (6.82%), Asteroideae (6.30%), Eucalyptus citriodora (6.04%), Syzygium sp. (5.25%), Acacia catechu (4.99%), Azadirachta indica (3.94%), Poaceae (2.88%), Coriandrum sativum (1.31%), Oleaceae (1.05%), Albizia lebbeck (0.79%) and Bombax ceiba (0.52%) (Figure 2e).
Singhpur
Spring honey contains Caesalpinia pulcherrima (18.87%), Solanaceae (13.52%), Anacardiaceae (11.27%), Syzygium sp. (9.01%), Coriandrum sativum (7.61%), Eucalyptus citriodora (5.07%), Asteroideae (5.07%), Poaceae (4.79%), Azadirachta indica (4.51%), Brassica campestris (3.94%), Bombax ceiba (3.66%), Acacia catechu (3.38%), Cichorioideae (3.38%), Lagerstroemia lanIceolata (2.25%), Albizia lebbeck (1.13%), Cassia fistula (1.13%), Ziziphus sp. (0.85%) and Malvaceae (0.56%) pollen.
Summer honey is composed of Caesalpinia pulcherrima (19.67%), Brassica campestris (12.30%), Syzygium sp. (12.02%), Anacardiaceae (9.84%), Cichorioideae (7.38%), Solanaceae (6.56%), Poaceae (6.56%), Asteroideae (5.19%), Acacia catechu (5.19%), Azadirachta indica (4.92%), Oleaceae (2.46%), Eucalyptus citriodora (1.91%), Bombax ceiba (1.91%), Coriandrum sativum (1.64%), Cassia fistula (1.37) and Albizia lebbeck (0.55%) pollen.
Rainy season honey incorporates pollen of Caesalpinia pulcherrima (17.84%), Brassica campestris (17.30%), Anacardiaceae (15.68%), Asteroideae (9.19%), Azadirachta indica (8.92%), Cassia fistula (6.49%), Syzygium sp. (5.68%), Cichorioideae (5.68%), Coriandrum sativum (4.86%), Solanaceae (3.24%), Poaceae (1.35%), Oleaceae (1.08%), Bombax ceiba (1.08%), Malvaceae (1.08%) and Eucalyptus citriodora (0.54%).
Winter honey includes Caesalpinia pulcherrima (19.61%), Azadirachta indica (15.69%), Anacardiaceae (10.64%), Cichorioideae (10.08%), Syzygium sp. (9.52%), Coriandrum sativum (8.12%), Poaceae (7.56%), Brassica campestris (5.60%), Asteroideae (3.36%), Albizia lebbeck (2.52%), Eucalyptus citriodora (2.24%), Acacia catechu (2.24%), Malvaceae (1.68%) and Ziziphus sp. (1.12%) pollen (Figure 3a).
Mandhana
Azadirachta indica (15.34%), Solanaceae (12.39%), Ziziphus sp. (11.50%), Brassica campestris (11.50%), Syzygium sp. (9.44%), Caesalpinia pulcherrima (8.55%), Anacardiaceae (7.67%), Coriandrum sativum (6.78%), Bombax ceiba (5.90%), Eucalyptus citriodora (5.90%), Malvaceae (3.24%) and Cassia fistula (1.77%) account for pollen in spring honey.
Summer honey embraces pollen of Azadirachta indica (17.44%), Caesalpinia pulcherrima (16.28%), Coriandrum sativum (14.83%), Brassica campestris (9.88%), Anacardiaceae (9.01%), Syzygium sp. (7.85%), Ziziphus sp. (6.69%), Eucalyptus citriodora (5.52%), Solanaceae (4.65%), Bombax ceiba (3.20%), Cassia fistula (1.74%), Malvaceae (1.45%), Cichorioideae (1.16%) and Poaceae (0.29%).
In rainy season honey is composed of Syzygium sp. (22.02%), Anacardiaceae (17.13%), Azadirachta indica (13.76%), Brassica campestris (11.62%), Caesalpinia pulcherrima (10.09%), Coriandrum sativum (4.59%), Eucalyptus citriodora (4.28%), Solanaceae (4.28%), Cassia fistula (3.98%), Ziziphus sp. (2.75%), Poaceae (2.75%), Bombax ceiba (1.53%) and Cichorioideae (1.22%) pollen.
In winter Anacardiaceae (21.18%), Brassica campestris (16.20%), Caesalpinia pulcherrima (13.71%), Ziziphus sp. (13.08%), Syzygium sp. (7.17%), Azadirachta indica (7.17%), Bombax ceiba (7.17), Solanaceae (6.23%), Cassia fistula (2.80%), Eucalyptus citriodora (2.49%), Cichorioideae (1.87%), Poaceae (0.62%) and Coriandrum sativum (0.31%) pollen are found (Figure 3b).
Bithoor
Azadirachta indica (21.01%), Anacardiaceae (19.05%), Poaceae (15.68%), Syzygium sp. (9.24%), Asteroideae (8.40%), Cichorioideae (7.28%), Coriandrum sativum (7.00%), Solanaceae (4.20%), Bombax ceiba (3.92%), Ziziphus sp. (3.36%), Albizia lebbeck (0.56%) and Acacia catechu (0.28%) constitute pollen component of spring honey.
Summer honey is made up of Azadirachta indica (23.65%), Syzygium sp. (15.38%), Anacardiaceae (13.11%), Solanaceae (13.11%), Poaceae (9.69%), Cichorioideae (6.84%), Brassica campestris (6.27%), Ziziphus sp. (4.56%), Asteroideae (3.13%), Coriandrum sativum (2.56%), Bombax ceiba (1.42%) and Albizia lebbeck (0.28%) pollen.
Azadirachta indica (19.66%), Poaceae (14.05%), Anacardiaceae (11.80%), Ziziphus sp. (10.67%), Brassica campestris (10.11%), Syzygium sp. (7.87%), Asteroideae (7.30%), Solanaceae (6.74%), Acacia catechu (4.21%), Cichorioideae (2.53%), Bombax ceiba (2.25%), Malvaceae (1.69%) and Coriandrum sativum (1.12%) pollen are included in honey sample of rainy season.
Winter honey shows pollen content of Azadirachta indica (24.72%), Anacardiaceae (20.74%), Brassica campestris (13.92%), Ziziphus sp. (12.22%), Cichorioideae (9.38%), Asteroideae (5.97%), Syzygium sp. (4.26%), Poaceae (6.53%), Malvaceae (1.14%), Solanaceae (0.57%) and Acacia catechu (0.57%) (Figure 3c).
Dewara Khurd
Spring honey embraces pollen of Anacardiaceae (15.04%), Brassica campestris (14.48%), Cassia fistula (12.26%), Acacia catechu (11.70%), Azadirachta indica (9.47%), Syzygium sp. (7.80%), Cichorioideae (6.96%), Coriandrum sativum (6.13%), Caesalpinia pulcherrima (4.18%), Asteroideae (3.90%), Eucalyptus citriodora (2.79%), Poaceae (2.22%), Ziziphus sp. (1.95%), and Bombax ceiba (0.56%).
Summer honey contains Azadirachta indica (16.20%), Brassica campestris (14.40%), Syzygium sp. (14.40%), Cassia fistula (13.37%), Anacardiaceae (8.74%), Acacia catechu (7.71%), Caesalpinia pulcherrima (6.17%), Cichorioideae (4.88%), Poaceae (4.88%), Ziziphus sp. (4.11%), Coriandrum sativum (2.31%), Asteroideae (2.06%) and Eucalyptus citriodora (0.51%).
Pollen of Syzygium sp. (16.95%), Brassica campestris (13.84%), Poaceae (12.99%), Anacardiaceae (12.71%), Cassia fistula (10.45%), Acacia catechu (9.60%), Azadirachta indica (7.91%), Ziziphus sp. (5.65%), Caesalpinia pulcherrima (2.26%), Bombax ceiba (1.98%), Coriandrum sativum (1.41%), Eucalyptus citriodora (1.41%) and Oleaceae (0.85%), are found in honey of rainy season.
Winter honey incorporates Brassica campestris (17.93%), Cassia fistula (14.01%), Azadirachta indica (13.73%), Poaceae (9.24%), Anacardiaceae (7.00%), Eucalyptus citriodora (6.44%), Syzygium sp. (6.16%), Acacia catechu (6.16%), Ziziphus sp. (5.32%), Bombax ceiba (3.92%), Coriandrum sativum (3.64%), Caesalpinia pulcherrima (3.36%), Malvaceae (1.40%) and Oleaceae (0.84%) pollen (Figure 3d).
Ajgain
Honey sample collected from Ajgain in spring season is dominated by Caesalpinia pulcherrima (20.94%), Anacardiaceae (11.26%), Syzygium sp. (9.42%) pollen along with pollen of Cichorioideae (8.90%), Ziziphus sp. (7.85%), Azadirachta indica (6.28%), Poaceae (6.02%), Asteroideae (5.76%), Acacia catechu (5.24%), Malvaceae (4.19%), Albizia lebbeck (3.66%), Eucalyptus citriodora (3.66%), Solanaceae (3.66%), Bombax ceiba (1.05%), Oleaceae (0.79%), Brassica campestris (0.79%) and Cassia fistula (0.52%).
Summer honey comprises of Caesalpinia pulcherrima (15.64%), Bombax ceiba (9.23%), Azadirachta indica (8.21%), Syzygium sp. (7.44%), Anacardiaceae (7.18%), Cichorioideae (6.92%), Acacia catechu (6.15%), Coriandrum sativum (6.15%), Albizia lebbeck (4.87%), Poaceae (6.41%), Solanaceae (4.36%), Cassia fistula (4.36%), Eucalyptus citriodora (4.10%), Malvaceae (3.08%), Asteroideae (2.82%), Brassica campestris (1.28%), Ziziphus sp. (1.03%) and Oleaceae (0.77%) pollen.
Multi-floral honey of rainy season involves pollen of Syzygium sp. (16.11%), Caesalpinia pulcherrima (15.09%), Asteroideae (9.21%), Poaceae (8.44%), Azadirachta indica (7.67%), Anacardiaceae (7.42%), Cichorioideae (5.37%), Solanaceae (5.37%), Eucalyptus citriodora (5.12%), Ziziphus sp. (4.86%), Cassia fistula (3.58%), Acacia catechu (2.81%), Coriandrum sativum (2.56%), Brassica campestris (2.30%), Albizia lebbeck (2.05%) and Malvaceae (2.05%).
Winter honey takes in pollen of Caesalpinia pulcherrima (15.00%), Anacardiaceae (14.47%), Asteroideae (7.37%), Syzygium sp. (6.58%), Malvaceae (6.32%), Eucalyptus citriodora (6.05%), Poaceae (6.05%), Bombax ceiba (5.53%), Azadirachta indica (5.26%), Cichorioideae (5.26%), Solanaceae (5.26%), Acacia catechu (4.74%), Ziziphus sp. (3.68%), Cassia fistula (3.42%), Albizia lebbeck (2.89) and Brassica campestris (2.11%).
Honey sample and its potential for pollen-vegetation relationship
Ancient pollen deposits can be better understood with the use of modern pollen spectra. When pollen from modern habitats is studied, it is easier to connect particular pollen types to their parent plants and surrounding vegetation. This makes it simpler to identify the kinds of flora that were present in the past, such as grasslands or forests. Understanding historical habitats is enhanced by how modern pollen travels and settles in various locations.
Another application for these spectra is studying historical climates. Pollen samples from the past and present can be compared to detect changes in the climate throughout time. This is particularly helpful for comprehending times of rapid climate change. Pollen spectra from today are a reliable resource for learning about prehistoric vegetation and climates.
It is observed that the vegetation composition based on the pollen taxa with the different areas studied from honey samples indicates a mixed deciduous vegetated area, along with associated cropland and open land in and around the study area. After analysing the earlier published work on modern pollen and vegetation relationship (Ghosh & Srivastava, 2004; Gupta, 2013; Singh & Pandey, 2007; Trivedi & Chauhan, 2011) it is revealed that our generated pollen data is close similar about the representation of the arboreal taxa such as Syzygium sp., Bombax ceiba, Azadirachta indica, Anacardiaceae, Ziziphus sp., Cassia fistula, Acacia catechu and Eucalyptus citriodora. Similarly, the pollen assemblage also represents non-arboreal taxa such as Solanaceae, Asteroideae, Malvaceae, Brassica campestris, and Poaceae. So, our generated data could be a complementary dataset for the surface soil samples in palaeopalynological studies. Accordingly, this data could be a baseline and reliable for the palaeovegetation and climate reconstruction in the regional and vicinity area (Figure 3e).
DISCUSSION AND CONCLUSIONS
The honey samples collected from densely vegetated areas of Kanpur and Unnao districts are multi-floral, an amalgamation of arboreal and non-arboreal pollen (Plate 1). In the spring and summer seasons, Brassica campestris pollen dominates in honey samples along with arboreal pollen. Brassica campestris, an oil-yielding crop, blooms in winter. Bees collect pollen and nectar and transform them into honey in about 45 days. So, it is evident that Brassica pollen is present in these honey samples in spring and summer. The presence of Brassica campestris pollen shows the cultivation area near the densely vegetated region and the foraging preference of honeybees in densely vegetated land, too. Honey bees commonly forage within a circular distance of approximately 1–2 km from their hives (The Keepers Collective, 2021).
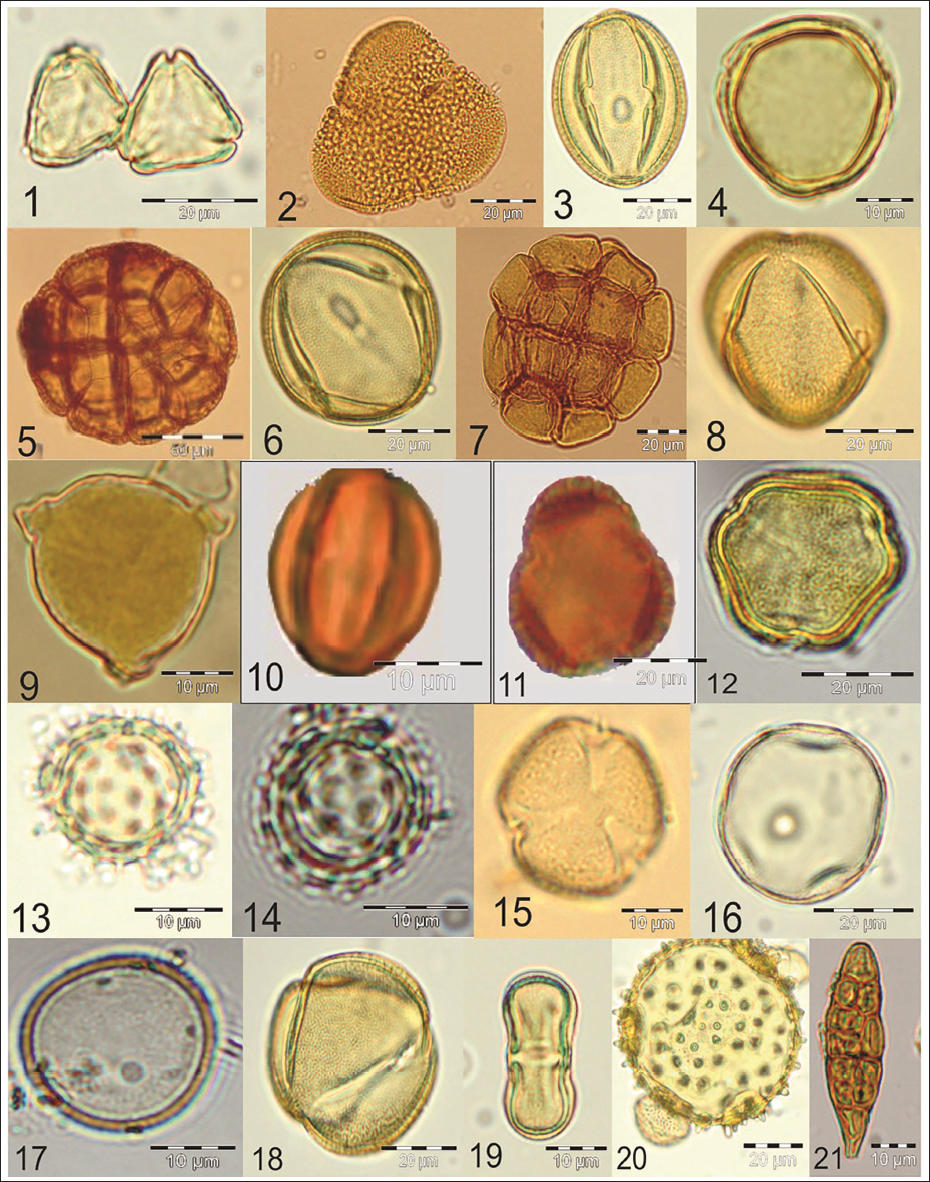
Pollen plate illustrating the morphological diversity of pollen taxa identified in Honey Samples: 1. Syzygium sp.; 2. Bombax ceiba; 3. Anacardiaceae; 4. Azadirachta indica; 5. Albizia lebbeck; 6. Cassia fistula; 7. Acacia catechu; 8. Caesalpinia pulcherrima; 9. Eucalyptus citriodora; 10. Lagerstroemia lanceolata; 11. Oleaceae; 12. Ziziphus sp.; 13. Asteroideae; 14. Cichorioideae; 15. Brassica campestris; 16&17. Poaceae; 18. Solanaceae; 19. Coriandrum sativum; 20. Malvaceae; 21. Alternaria sp.
Pollen of Acacia catechu, Albizzia lebbeck, Azadirachta indica, Caesalpinia pulcherrima, Cassia fistula, Syzygium sp., with members of Anacardiaceae, are present in significant proportions in honey samples of all the seasons, highlighting their role as reliable floral resources. Dominance of arboreal pollen, mainly in lean seasons, shows a shift in the foraging pattern of honey bees from non-arboreal to arboreal taxa, which may be due to the non-availability of annual flowering plants (Roubik, 1995). The seasonal dependency on various plant communities emphasises their critical role in sustaining pollinator populations, as emphasised by Gupta (2014) in India and by Klein et al. (2007) in Germany.
Regional variations in pollen spectra further reflect the influence of habitat characteristics. Locations with dense vegetation, such as Maharajpur, Mandhana, Singhpur, Safipur, Gangpur Kachhar, Bithoor and Ajgain exhibited a dominance of tree pollen, in parallel to findings from native forests in Brazil (Behling & Negrelle, 2006) and temperate zones in Europe (Banaszak-Cibicka et al., 2018), where forested habitats provide year-round resources while in areas like Aranj Bankat (Bhitargaon), Bausar and Dewara Khurd closer to agricultural zones, were dominated by crop species, consistent with studies in the agricultural landscapes of Punjab and Haryana, India (Tripathi et al., 2014). Such spatial differentiation underscores the importance of preserving diverse habitats, as each provides unique floral resources essential to the foraging needs of honey bees (Ollerton et al., 2011). The interplay between agricultural and wild flora creates a dynamic foraging environment, observed in India and internationally (Chauhan & Singh, 2021; Klein et al., 2007). Conservation strategies integrating afforestation and urban greening with agricultural and apicultural practices are vital to ensure the sustainability of both pollinator populations and ecosystem services, as emphasised by Gupta (2014) and Ollerton et al. (2011).
The presence of Eucalyptus citriodora pollen in honey samples of Safipur, Gangpur Kachhar, Singhpur, Mandhana, Dewara Khurd, and Ajgain shows that this tree is cultivated near these areas, promoting afforestation strategies. The study aligns with global and national efforts to promote pollinator-friendly practices and habitat conservation.
Studies in South India (Tripathi et al., 2014) confirm the importance of native flora, such as Syzygium cumini, in maintaining pollinator health. Internationally, similar roles are played by species like Quercus in Europe (Banaszak-Cibicka et al., 2018) and Dalbergia in tropical forests (Behling & Negrelle, 2006). This highlights the ecological importance of native and wild flora, which Tripathi et al. (2014) also stressed in India. Conservation efforts should prioritise such species to enhance ecosystem stability, echoing global recommendations for pollinator conservation (Ollerton et al., 2011; Roubik, 1995).
Footnotes
Acknowledgements
We express our sincere gratitude to the Principal, Dayanand Girls’ PG College, Kanpur, and the Director, Birbal Sahni Institute of Palaeosciences, Lucknow, for providing the necessary facilities and support to successfully complete this work. Sneh Trivedi is deeply thankful to the University Grants Commission (UGC) for their financial assistance under the Savitribai Jyotirao Phule Single Girl Child Fellowship scheme. We are also thankful to the chief editor Prof. Mukund Sharma for inviting us to contribute research work for the special issue of the Journal of Palaeontological Society of India and anonymous reviewers for their valuable suggestions that helped to improve the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research is financially supported by the University Grants Commission (UGC) under the Savitribai Jyotirao Phule Single Girl Child Fellowship scheme.
