Abstract
Introduction & Aims:
Optimal neural activity in the central nervous system relies on the myelin sheath formed by oligodendrocytes. During demyelination, changes in the microenvironment can activate oligodendrocyte precursor cells (OPCs) and induce them to generate new oligodendrocytes. However, different demyelination models employ distinct pathways, targeting different cells and cytokines; these, in turn, have varying effects on OPCs. Therefore, it is reasonable to assume that OPCs derived from different pathologic environment may exhibit functional differences. This study aims to investigate the influence of the pre-activation of OPCs, isolated from lipopolysaccharide (LPS)-induced and cuprizone (CPZ)-induced demyelination models, on their regenerative capacity.
Methods:
OPCs were isolated from mice subjected to LPS or CPZ-induced neurodegeneration. Characterization and activation assessment included immunostaining for PDGFRα, OLIG2, and ELISA analysis for IL-1, and SOX10 expression assessment. Then OPCs were intravenously transplanted into LPS-exposed mice. The migration patterns of transplanted OPCs were tracked using DiI labeling. After 7 days, spinal cords were assessed for myelin content and integrity (Luxol fast blue, Transmission electron microscopy, MBP and MOG analysis) and extracellular matrix changes (chondroitin sulfate proteoglycan—CSPG levels).
Results:
Transplantation of LPS-OPCs significantly enhanced their migration to the demyelinated spinal cord, correlating with increased myelin content and integrity and a reduction in CSPG levels compared to the CPZ-pre-activated OPCs and control groups.
Conclusion:
Our findings suggest that the pre-activation environment, determined by the source model, differentially affects the regenerative potential of transplanted OPCs.
Keywords
Introduction
Targeted and regulated communication between different cell types of the central nervous system (CNS) is essential for optimal neural activity. 1 In all complex nervous systems, neurons, along with glial cells, play essential roles in transmitting signals throughout the central nervous system and relaying them to other systems in the body. 2 Neuronal function is supported and modulated by glial cells as they perform many vital functions, such as supplying nutrients, supporting function, disposing of waste, and regulating neurotransmitter metabolism. Glial cells are traditionally defined as astrocytes, microglia, and oligodendrocytes. 2 In addition to the functions mentioned for glial cells, one way that oligodendrocytes support neurons is by generating myelin sheaths through the spiral wrapping and compaction of their plasma membranes around axons, a process essential not only for accelerating electrical signal conduction but also for providing metabolic support.3,4
Myelination can regulate many aspects of information propagation. Also, demyelination often leads to progressive neuronal death, which can cause debilitating neurologic impairments. 5 So, myelination of axons by oligodendrocytes has been considered as a significant function in the body that must be maintained and sought to be compensated in case of disruption.3,6 In the compensatory process of demyelination oligodendrocyte progenitor cells (OPCs), are responsible for generating new oligodendrocytes and play pivotal roles in myelin formation. 7 During demyelination events, changes in the microenvironment can alter the gene expression profiles of OPCs, which can activate them to a regenerative status. 8 In this regard, there are various evidence that suggested OPCs are highly influenced by their surrounding microstructures. By changing the composition of the microstructures around OPCs, researchers have demonstrated that the stability and function of these cells can be affected.9–11 On the other hand, it has been observed that different inflammatory, viral, or toxin-induced models of demyelination employ distinct pathways and target different cells and cytokines, despite the ultimate event being myelin damage. In turn, these models have varying effects on mature oligodendrocytes and OPCs.12–14 So, it is assumed while OPCs are activated by demyelination, OPCs derived from different demyelination models may exhibit functional differences. In this study, we directly assessed the therapeutic efficacy of transplanting OPCs, pre-activated with lipopolysaccharide (LPS) or cuprizone (CPZ) demyelination models, into demyelinated animals. Here we evaluated OPCs survival and migration, myelinogenesis by oligodendrocytes, changes in the nervous system environment, and recovery rate.
Material and method
Study approval and animals
Thirty male C57BL/6 mice, 6 weeks old and weighing 20–25 g, were obtained from the Pasteur Institute in Karaj, Iran. Only animals that exhibited no overt signs of illness prior to the commencement of the study were included, ensuring a healthy and standard baseline for experimental procedures. Animals were excluded from the study if death occurred prior to the study endpoint. In addition, any mice sustaining significant injuries that led to infection or inflammatory responses were also excluded to maintain data integrity and prevent interference with experimental results. Animals were housed at the Tehran University of Medical Sciences Animal Breeding Center under controlled temperature and humidity, with a 12-h light/dark cycle, and both food and water were freely available. All procedures, conducted blindly, were approved by the Institutional Animal Care and Use Committee (IACUC) of the Tehran University of Medical science.
Experimental design
To obtain pre-activated OPCs isolated from demyelination models, two groups of mice (three mice/group) were used. The first group, specified as the CPZ cells group, mice were fed with 0.2% cuprizone (Sigma-Aldrich- St. Louis, USA) for 12 weeks. The second group, specified as the LPS cells group, received a single intraperitoneal injection of lipopolysaccharide (LPS, serotype 055:B5, Sigma-Aldrich, St. Louis, USA, Product N: L2880) dissolved in 0.9% sterile saline (1 mg/kg).15,16 Subsequently, a separate group of mice(18 weeks old( underwent induction of inflammatory demyelination by a single dose injection of LPS (1 mg/kg; ip). These mice were then randomly divided into three groups (
Isolation of OPCs
Animals from both LPS and CPZ groups (18 weeks old) were sacrificed by carbon dioxide inhalation to obtain primary OPC cultures. At the first step, the brain was removed with forceps and was collected in a glass Petri dish containing 10 cc of ice-cold Hanks’ Balanced Salt Solution (HBSS) (Mg2+ and Ca2+) (Gibco, Cat N: 14025092) and then the tissue was dissociated mechanically, with two needles and small scissors. The pieces of neural tissue were placed in a 50 ml conical tube with 200 µl collagenase1 and 300 µl DNase then were triturated through a pipette several times and incubated at 37°C for 5 min. Then 30 ml of OPC medium including Dulbecco’s modified Eagle’s medium (DMEM; Gibco) containing15% fetal bovine serum (Sigma Aldrich), 100 U/ml streptomycin (Sigma Aldrich), and 100 U/ml penicillin (Sigma Aldrich), were added to stop enzymatic digestion and centrifuged at 900 rpm for 10 min. The supernatant was removed and the pellet was resuspended in 10 ml of OPC medium. The resulting suspension is passed through a 100 μm nylon mesh filter and the cells are transferred into a 75 cm2 culture flask containing a Poly-L-Ornithine coating in a volume of 10 ml per flask and placed in a 37°C, 5% CO2 incubator. After 24 h, the previous culture medium is replaced with OPC culture medium enriched with PDGRF-AA (Gibco PeproTech, USA). After 10 days the supplemented medium was again replaced with an OPC medium until day 25–30 when astrocytes, OPCs, and microglia appeared. To purify OPCs, the flasks were placed in an orbital shaker at 250 rpm at 37°C for 20 h. Then immediately the medium containing the detached OPCs was passed through a 40-μm nylon mesh filter placed over a 50 ml conical tube. Then the suspension was centrifuged at 900 rpm for 10 min, and the pellet was pipetted into 10 cc of the OPC medium and was incubated in an untreated Petri dish for 45 min at 37°C. Centrifugation was performed again for the suspended OPCs at 900 rpm for 5 min and finally, the pellet containing purified OPCs was incubated with the OPC culture medium at 37°C, in 5% CO2. 17
Cell transplantation
To track OPCs, we utilized DiI (1,1-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate), a cross-linkable membrane dye, based on the manufacturer’s protocol. OPCs were placed on 15 ml conical tube centrifuged at 1500 rpm for 5 min. Then, 2 μg DiI/mL DMEM was added to 3 × 106 OPCs and was incubated at 37°C for 30 min. After washing with PBS (D-PBS, Sigma, USA), the Dil-labeled OPCs were ready for injection. Using a 5 μl Hamilton syringe (Hamilton, NV), 2 μl of PBS containing roughly 2 × 10⁵ labeled OPCs were injected into the mice tail vein and a similar volume of PBS was injected into the control group.18,19 To quantify DiI-labeled OPCs in the brain, spinal cord, and other organs, tissue sections were imaged using a high-power fluorescence microscope. For each animal (
Phenotypic analysis by immunostaining
Cells were rinsed in PBS, fixed with 4% paraformaldehyde (PFA, Merck, Germany) at room temperature (15 min), and then rinsed three times (10 min each). In the next step, cells were incubated with 10% normal goat serum (NGS) in PBS (D-PBS, Sigma, USA), with 0.3% Triton X-100; RT; 1 h. Immunostaining using primary antibody in blocking solution containing OLG2 (a marker for OPCs; recognizes a cell surface chondroitin sulfate proteoglycan) and PDGFRα (the earliest OPC markers; recognize the receptor tyrosine kinase) was conducted using standard protocols. The dilution ratio of antibodies is as follows anti-PDGFRα (ab203491) 1:500 and anti-OLG2 (Santa Cruz, Cat N: sc-518069), 1:500 at overnight. On the second day, the cultures incubated with the secondary antibody Goat Anti-Rabbit IgG (Elabscience, Cat N: E-AB-1014) for anti-PDGFRα and Goat Anti-Mouse IgG (Elabscience, Cat N: E-AB-1011) for anti-OLG2 in blocking solution (1:100; RT; 1 h). Then Samples were rinsed with PBS and mounted with DAPI (1:1000). The slides were observed by fluorescent microscope (Olympus, BX50). 20
ELISA analyses for inflammatory assay in OPCs culture
Before cell purification, the culture supernatants from OPCs were collected and examined for IL1, using enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, USA). Flat-bottom 24-well plates were coated overnight with coating IL-1 mAbs at 4°C. After discarding the primary mAbs, the plates were blocked with blocking buffer for an hour at room temperature. After three times washing with buffer (0.05% Tween 20 in PBS) the plates were blotted on a paper towel. Then, after adding dilute standards and samples, the plates were incubated overnight at room temperature. The supernatant was discarded and the wells were washed twice with buffer. A second antibody capable of detecting IL-1 was added and incubated for 30 min at room temperature. The plates were washed and tetramethyl benzidine substrate solution was added. In the end, the results were read at 450 nm and the sample concentrations were calculated based on a standard curve. 21
Gene expression assay
The real-time-PCR assay was performed to determine expression patterns of SOX10 in OPCs and the expression of MOG and MBP were evaluated in spinal cord tissue after 7 days. Total RNA was isolated from frozen cells using an RNA-Sinaclon extraction kit (SinaPure) according to the manufacturer’s instructions (Sinaclon, Iran) and cDNA was synthesized using Easy cDNA synthesis kit- Parstous (Parstous, Iran). Quantitative real-time PCR (qRT-PCR) was run by SYBER Green Master mix- VERNER (Iran) besides Real Time PCR system (Applied Biosystems, USA) using corresponding primers: SOX10,F:GACGZTGACAAGTTCCCCGT,R:TCCTCAATGAAGGGGCGCTT;MOG:F:CAGATGAAGGAGGCTACACC,R:GCACGAAGTTTTCCTCTCAGTC;MBP:F:GTCCCTGAGCAGATTTAGCTT,R:GAATCCCTTGTGAGCCGATT;
Sacrifice and tissue preparation
At the end of the experimental course (end of the first week), mice from all three experimental groups (19 weeks old) were anesthetized using administration of 10:1 Ketamine (Sigma, St. Louis, USA): Xylazine (Heidelberg, Germany) (ip). For histological assay, transcardially perfusion with phosphate-buffered saline (PBS) followed by 4% Paraformaldehyde (PFA, Merck, Germany) was done. The back skin was trimmed off and the spinal cord (SC) were carefully exited then SC was post-fixed in a jar of 4% PFA for 24 h. After paraffin (Merck, Germany) embedding, 5 μm transverse sections were prepared from the collected SC samples for immunofluorescence and hematoxylin and eosin (H&E) staining. To analyze gene expression, mice were transcardially perfused with ice-cold Dulbecco’s PBS (D-PBS, Sigma, USA). Subsequently, SC sections were frozen in liquid nitrogen and stored at −80°C until further use. 23
Immunofluorescence staining
Following deparaffinization and rehydration, transverse sections of SC were incubated in antigen retrieval buffer (10 mM sodium citrate buffer, 20% tween for 30 min; RT). Non-specific labeling was blocked using 0.1% BSA (BSA, sigma, USA) in 0.1% Triton X-100 (1 h). Then SC sections were incubated overnight with primary anti-chondroitin sulfate (1:500, Santa Cruz, Cat N: sc-166251). Then we added anti-mouse IgG as a secondary antibody (ab6785) on the sections for 1 h (RT) and visualized the cell nucleus was done using DAPI staining (1/1500). A fluorescence Microscope (Olympus BX50) set with a digital camera (Olympus DP72) was used for Immunostained sections assessment. 24
Transmission electron microscopy
The mice were perfused with a solution containing 2% PFA and 2.5% glutaraldehyde fixative. The collected SC was fixed with 3% glutaraldehyde for 2 h and then washed three times with 0.1 PBS. Afterward, it was fixed with 1% osmic acid and washed three times with 0.1 PBS. After dehydration, the tissues were embedded in epoxy resin. Ultrathin sections were then prepared by a Reichert ultramicrotome with a diamond-tipped knife, contrasted with uranyl acetate and lead citrate. The sections were then viewed under an electron microscope (EM) (LEO 906; Carl-Zeiss) at 80 kV. Five fields of view were selected randomly in each section. G-ratio (the ratio of the axonal diameter to the total fiber diameter) was evaluated according to the protocols mentioned in previous reports.22,25
Luxol fast blue (LFB) staining
To evaluate myelin, SC slides underwent Luxol fast blue (LFB) staining (Sigma-Aldrich, USA) following a previously established protocol. 24 After deparaffinization and rehydration, SC sections were incubated overnight in LFB solution (1% in 95% ethanol with 0.05% acetic acid; 60°C). For quantification blue color intensity as the myelinated regions were measured using a light microscope (Olympus CX310, Japan) set with a digital camera (Olympus, Japan) and Image J software. The myelination index (%) was measured as the size of myelinated regions in SC slides to the total area and multiplied by 100.
Statistical analysis
The sample size was determined based on extensive previous research on the mouse spinal cord. Aligning with methodologies from similar studies, we selected eight mice per group as optimal for the final evaluation, ensuring compliance with biological testing standards.15,26 The obtained results of the current study were expressed as mean values ± standard error (Mean ± SD) and were analyzed using Graph Pad Prism (Version 6.01, CA, and the USA) and SPSS software version 20 (IBM, Armonk, NY). Comparison between the three studied groups was carried out by Kruskal-Wallis H test and one-way analysis of variance (ANOVA) test followed by Tukey post hoc tests. The significance is indicated by *
Results
Characterization of the isolated OPCs exhibits immunoreactivity to certain markers and proving the status of cells
After cell isolation, the mixed neural stem cells were cultured to perform the OPC purification process (Figure 1(a)). Then, purified OPCs showed specific morphological characteristics of this cell lineage as follows: the cell bodies were round or spindle-shaped, and their protrusions were short or bead-like (Figure 1(b)). After examining OPCs inflammatory status, the results showed that there was a significant difference (
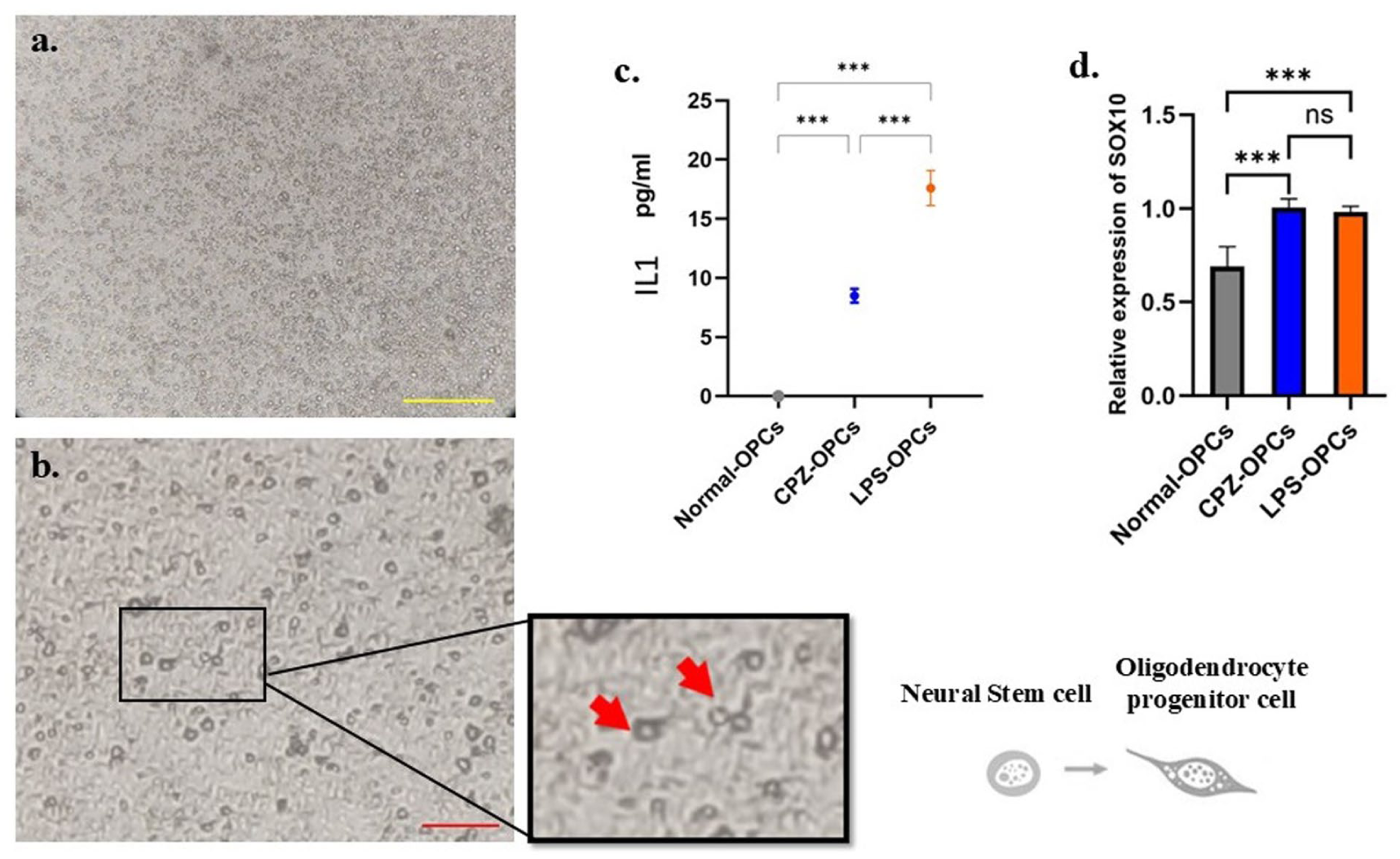
The observation of cell culture and the activation and inflammatory status of OPCs. (a) The cell culture of mixed neural stem cells after isolation (scale bar = 300 μm). (b) Purified OPCs showed specific morphological characteristics: cell bodies were round or spindle-shaped, and their protrusions were short or bead-like. Arrows indicate OPCs (scale bar = 100 μm). (c) The effects of two demyelination models, lipopolysaccharide (LPS) and cuprizone(CPZ), on the production of interleukin-1
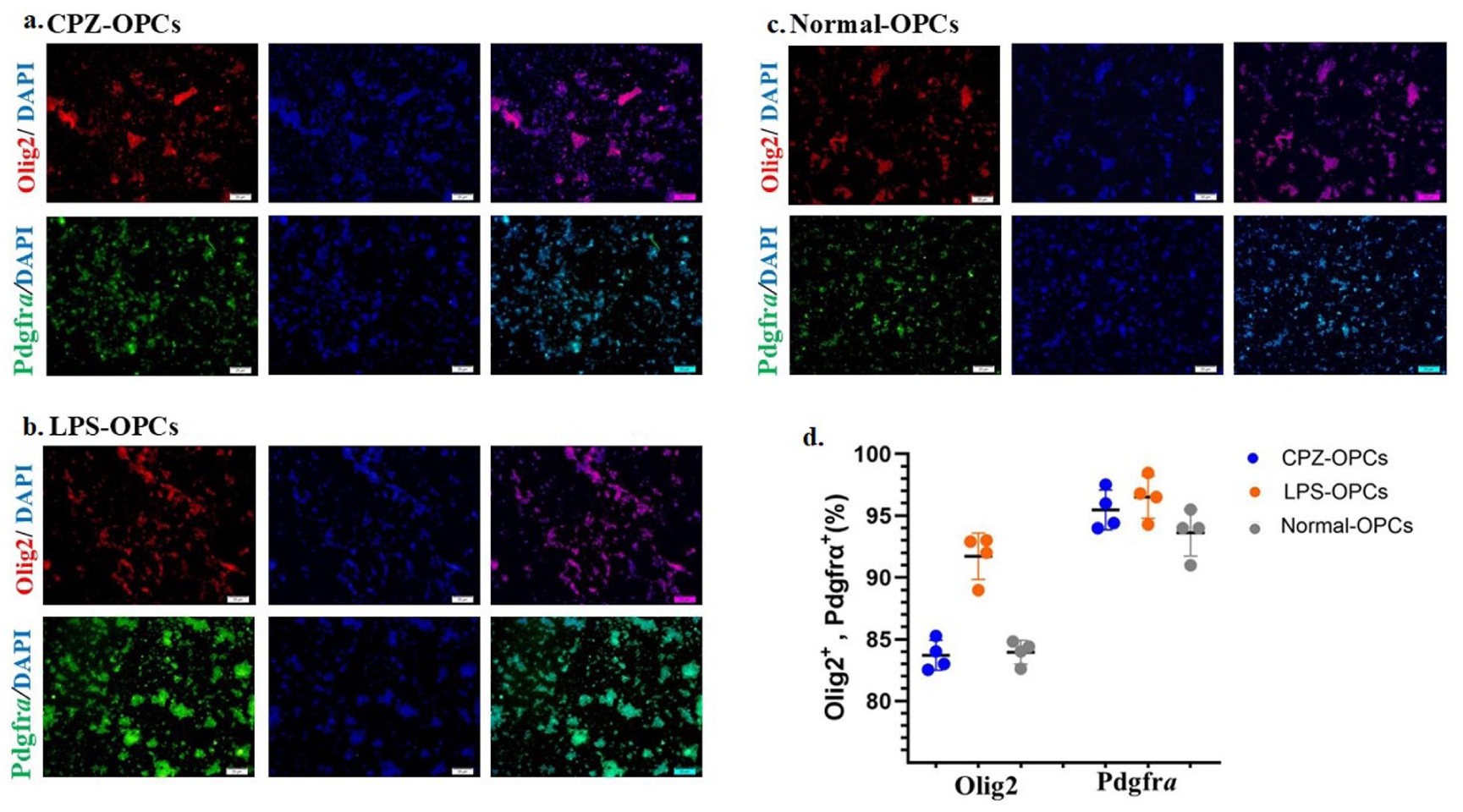
Characterization of the isolated OPCs. (a–d) The graphs describe the percentage of Olig2 (red) and Pdgfr-α (green) positive cells as OPC-specific antigens. Nuclei were stained with 4,6-diamidino-2-phenylindole (DAPI, blue); (scale bar = 20 μm).
The detection of transplanted pre-activated OPCs in various organs and homing to the spinal cord
To evaluate the migratory capacity of OPCs, we determined the distribution of DiI-labeled OPCs across various organs. Red fluorescence, indicating the presence of DiI-labeled CPZ-OPCs and DiI-labeled LPS-OPCs, was observed in sections of the specified organs under a fluorescence microscope. Figures 3 and 4 give us a view of how the transplanted cells are distributed in different tissues. The presence of labeled cells among the neuronal and glial cells of the host brain tissue was proven in both groups of transplanted cells and, LPS-OPCs group showed a stronger tendency for cell migration toward brain tissue (78 ± 3.16 (LPS) vs 32.75 ± 8 (CPZ),

Homing and the detection of transplanted OPCs in the nervous system: brain and spinal cord. (A, B) Red fluorescence, indicating the labeled OPCs localized to the brain and spinal cord region in both groups. Nuclei were stained with DAPI (blue) (4,6-diamidino-2-phenylindole). Arrows indicate DiI-labeled cells overlap with nuclei;

The detection of transplanted OPCs in the various organs: lung, liver, and spleen. (A–C) Representative immunofluorescent images of the lung, liver, and spleen sections showing labeled OPCs (DiI, red), and nuclei (DAPI, blue). Arrows indicate DiI-labeled cells overlap with nuclei;
Figure 4(A) and (a) aimed at determining the impact of the activation model on OPCs migration to the lung, revealed a striking difference (
Effect of transplanted pre-activated OPCs on the myelogenesis and integrity of myelin content in the spinal cord
The effect of transplantation of pre-activated OPCs was investigated on the myelogenesis of oligodendrocytes (MBP) and integrity of myelin content (MOG) among the groups (Figure 5). Our results revealed a significant (

Assessment of MBP and MOG expression by RT-qPCR. Myelogenesis and integrity of myelin content were examined by assessment of MBP and MOG expression, respectively. (a) Relative MBP expression in Control, LPS-OPCs transplanted, and CPZ-OPCs transplanted groups. OPCs transplantation increased MBP mRNA expressions. (b) LPS-OPCs transplantation demonstrated high levels of MOG expression compared to control mice and CPZ-OPCs transplanted mice.
In addition, compared to the control (0.99 ± 0.1), MOG expression was significantly elevated in both LPS-OPCs (88.87 ± 6.5,
Effect of pre-activated OPCs transplantation on the neural tissue matrix in the spinal cord of mice
Our results demonstrated that during the 7 days after cell transplantation, a change occurred in the neural tissue matrix in both OPCs groups. Therefore, evaluation on the amount of chondroitin sulfate as an important glycoprotein showed that a significant (
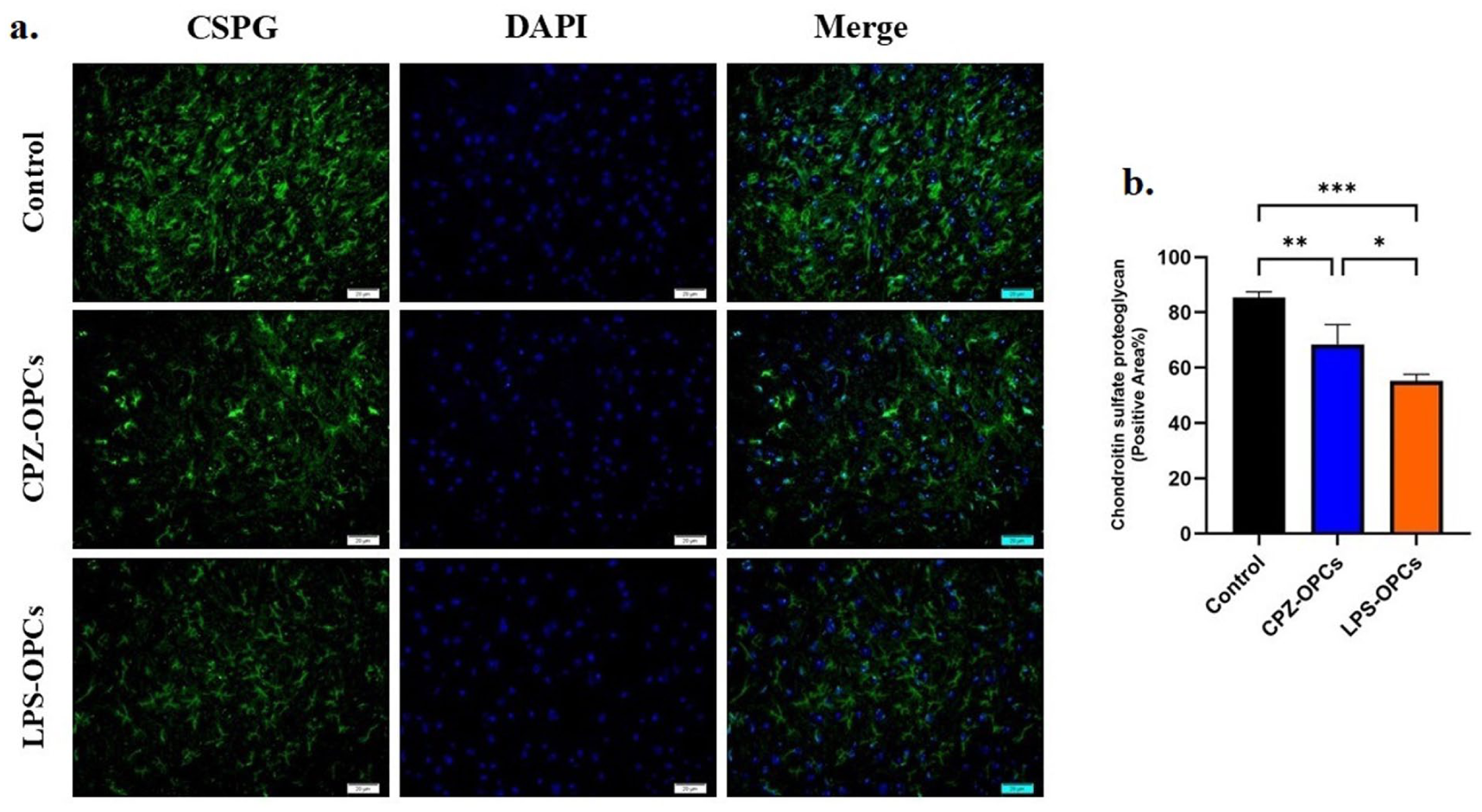
Immunofluorescence staining for assessment of CSPG in the spinal cord tissue. (a) Representative images of the spinal cord sections showing CSPG (green), and nuclei (DAPI, blue). (b) The graph depicts the quantitative analysis for the percentage of the CSPG.
The effect of transplantation of pre-activated OPCs on myelin recovery in demyelinated spinal cord
We performed LFB staining and TEM assessment on SC sections from each group to evaluate demyelination and remyelination rates. Myelin content was also quantified using ImageJ software. Normal SC displayed typical myelin content with no disruption, which was evident by the blue color in LFB staining (Figure 8(a)) and confirmed by the intact myelin structures observed in TEM images (Figure 7(a)). In contrast, demyelination (appearing as white color in LFB staining) was observed in the LPS-exposed SC (Figure 8(b)). This demyelination was further characterized in TEM images by elevated myelin loss, gaps between myelin layers, and disrupted myelin sheaths (Figure 7(b)).

Ultrastructural analysis of myelin in the spinal cord following OPC transplantation. In normal spinal cord (a), TEM images revealed intact myelin, whereas LPS-exposed spinal cord (b) showed demyelination, a loss of myelin, and gaps in myelin layers. Scale bar = 1 μm, 500 nm. (c) The effect of OPCs transplantation on myelin damage recovery. Myelin sheaths and structures of axons are shown in the spinal cord in TEM sections. Scale bar = 1 μm. (d) Quantitative data showed the G-ratio in the spinal cord.
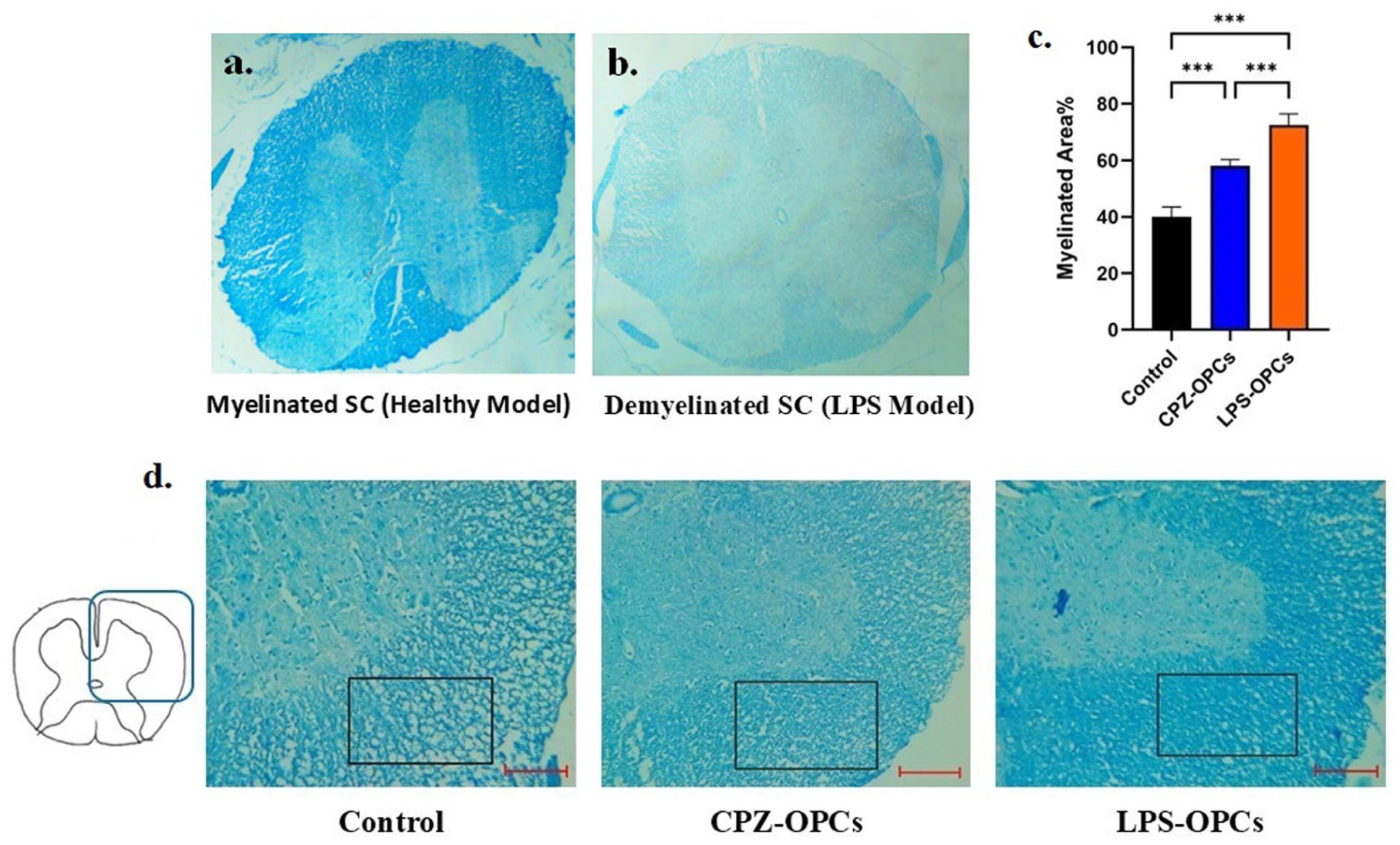
The Effect of transplantation of OPCs on myelin damage recovery. (a, b) Myelin sheaths stained with LFB showed demyelination in the spinal cord section of LPS model. Scale bar = 400 μm. (c) The graph depicts the quantitative analysis for the percentage of myelinated areas. (d) Transplantation of OPCs facilitated remyelination of demyelinated spinal cord in LPS-OPCs and CPZ-OPCs.
Our quantitative analysis of OPCs transplantation demonstrated greater recovery of myelin sheaths. Specifically, the G-ratio quantitative analysis of TEM data showed that myelin recovery significantly increased after OPCs transplantation. This was evident in the CPZ-OPCs group (0.71 ± 0.02;
Discussion
In the current study, we investigated how transplanted OPCs pre-activated with LPS and CPZ models affect the therapeutic process of the changed neural microenvironment. OPCs isolated from either demyelination model underwent a cascade of activation and further differentiation, evidenced by simultaneous SOX10, Olig2, and PDGFR-a expression. Our study indicated that the LPS model activated OPCs also induces more secretory activity for IL-1 secretion in the neural stem cells population isolated from this model in cell culture. We found that transplantation of OPCs pre-activated could markedly enhance remyelination capacity in the demyelinated spinal cord of the mouse model while reducing the CSPG and increasing the density and amount of myelin. Our results support the hypothesis that activation of OPCs that occurs during LPS demyelination and neuroinflammation model can promote remyelination by positively influencing OPCs function, including cells migration, differentiation, and ECM modulation.
Cuprizone, a copper chelator, targets oligodendrocytes and leads to their destruction, primarily through multiple intracellular mechanisms and then through environmental changes. These are characterized by mitochondrial dysfunction and ferroptosis, an adjusted cell death process driven by iron-dependent lipid peroxidation.12,13
The LPS model is widely used for in vivo and in vitro studies, contributing to our understanding of neuroinflammation and neurodegeneration when simulates ongoing inflammation in neurodegenerative diseases.27,28 LPS administration triggers microglial activation which subsequently leads to enhancement of pro-inflammatory cytokines: tumor necrosis factor-
Our initial cultures contained a mixture of neural stem cells and glial cells, derived from two distinct models that differentially engage the immune and nervous systems, ultimately leading to neurodegeneration. These differences extend beyond short-term phenotypic states such as M1/M2 polarization in microglia/macrophages or A1/A2 activation in astrocytes, which typically persist for only a few days.32,33 Increasing evidence suggests that distinct models of neural injury can induce longer-lasting effects, imprinting memories and lasting responses in glial cells.34,35 Moreover, neurodegenerative conditions are known to trigger epigenetic modifications and persistent changes within neural progenitor cells. 36 In line with this, the gene profile of OPCs undergoes substantial changes when exposed to microenvironmental stresses following demyelination, shifting toward compensatory patterns—a stable and enduring process. 37 Thus, it is confirmed that different models induce lasting alterations within NSCs, while interactions with glial cells in the initial culture conditions further reinforce these differences, even under same culture conditions. In response to demyelination, OPCs are activated and induced to reprogram to a more immature gene expression profile similar to developmental OPCs, leading to increased migratory capacity and production of cytokines and chemokines.8,37–39 Researchers have observed that the Wnt pathway, Shh signaling, and developmental signaling pathways such as PDGF, FGF, and EGF are activated in the damaged nervous system following demyelination. These changes within OPCs contribute to an increase in OPC numbers and subsequently enhanced oligodendrogenesis in demyelinated lesions.40–42 In addition, research findings indicate that SOX10 expression levels increase during periods of neurological stress when there is an enhancement in the differentiation of neural stem cells into oligodendrocytes. In fact, Sox10 plays a pivotal role in determining the fate of activated OPCs that have been stimulated to proliferate and differentiate into oligodendrocytes.43,44 Here we report, in both demyelination models, isolated OPCs showed higher expression of SOX10 compared to the normal condition, following the induction of a degenerative wave in the CNS and stimulation of neural stem cells.
In line with previous reports, our findings provide additional insight into the biodistribution and migratory behavior of transplanted OPCs. Previous studies have shown that intravenous cell delivery is associated with widespread dissemination and entrapment of cells in various peripheral organs. 45 It has also been suggested that the migratory dynamics of systemically administered cells are influenced by various factors, including age, systemic health status, and the condition of the nervous system. 46 Consistent with these findings, we observed that under identical transplantation conditions, LPS-isolated OPCs exhibited greater pulmonary entrapment, while CPZ-isolated OPCs showed preferential accumulation in the liver and spleen. These results suggest that the in vivo fate of OPCs is influenced by the origin and inflammatory status of the donor model. Such differences may affect both the efficiency of CNS targeting and the cells’ ability to localize to areas of demyelination. We found that transplantation of OPCs derived from the LPS model led to enhanced myelogenesis of oligodendrocytes and improved recovery. Previous studies have shown that endogenous OPCs, in response to demyelination, are guided by chemokines (CCL2, CCL5, CCL12) and axonal damaged cues to migrate toward the injury site. This migration facilitates myelin repair through subsequent differentiation into mature oligodendrocytes.11,37 In addition, the neuroinflammation associated with demyelination triggers a molecular cascade, including MCP-1 (Monocyte Chemoattractant Protein-1), MIP-1α (Macrophage Inflammatory Protein-1α), and SDF (Stromal Cell-Derived Factor), that recruits neural stem cells to the demyelinated area.47,48
Our results suggest that transplanted OPCs may migrate toward areas of inflammatory demyelinated within the spinal cord, where they can effectively contribute to remyelination events. Our findings are supported by Soo-Kyung et al., who demonstrated that intravenous injection of adipose tissue-derived stromal cells (ATSCs)-OPCs ameliorated neurological conditions of SCI. This was achieved by the migration of transplanted cells to the injured spinal cord and their interaction with endogenous spinal cord stem cells. 49 In this regard, Wang et al. 50 reported that transplanted CG-4 cells improved the remyelination process in a zymosan-induced focal demyelination model. Another study also reported that transplantation of OPCs into an experimental autoimmune encephalomyelitis (EAE) model caused improvement in neurological outcomes. Notably, the researchers observed that inflammation within the EAE model enhanced the migration and survival of the transplanted oligodendrocyte progenitors within the central nervous system. 51 Based on previously mentioned researches, the LPS demyelinated model induces neuroinflammation and we demonstrated that activation in LPS inflammatory demyelinated model can positively impact OPC myelination ability and their influence on other oligodendrocytes. The observations of some studies align with our results demonstrating a strong association between active inflammatory cells in demyelinated lesions and facilitating remyelination processes.52,53 In this line, Arnett et al. addressed the issue of the potential benefits of MHC-mediated antigen presentation in inflammation. Their findings showed that MHC-null mice exhibited delayed remyelination. 54 Moreover, the research in an animal model study that examined NF-kB signaling and TLR4 activation, revealed that the innate immune system has crucial neuroprotective properties. This includes the clearance of cellular debris and the facilitation of OPC mobilization to demyelinated area. 55 In this regard, another study stated that pro-inflammatory signaling not only does not inhibit recovery but actively promotes remyelination. This process involves the induction of oligodendrogenesis. 56
Oligodendrogenesis and subsequent remyelination are subject to regulation by numerous molecules, particularly extracellular matrix (ECM) proteins. The Chondroitin sulfate proteoglycans (CSPGs) localized in ECM modulate the CNS microenvironment and cellular function in neural tissues. 57 In the CNS, CSPGs are known to have a highly inhibitory effect on remyelination, axonal elongation, as well as the migration and differentiation of OPCs into mature oligodendrocytes.58,59 Researchers examined the effects of the enzyme chondroitinase ABC (cABC) on the migration of endogenous OPCs within spinal cord contusion lesions. They stated that following the removal of the glycosaminoglycan chains of CSPGs, the CSPG-related suppression on OPCs are reversed and regenerative axonal sprouting is enhanced. 60 In this line, researchers also investigated repair in a model of hypoxic-ischemic brain injury following modification of CSPGs. Their results suggest that CSPGs inhibit both the maturation of OLs and the remyelination process. 61 Researcher also showed that the Aggrecan, neurocan, and NG2, CSPG family, significantly inhibited oligodendrocyte projections and myelination. They found that the downregulation of PTPσ (protein tyrosine phosphatase sigma), a receptor for CSPGs, and the involvement of Rho-associated kinase (ROCK) downstream can improve white matter myelination. 58 OPCs, macrophages/microglia, and astrocytes are key cellular players in the CNS that can modify the surrounding environment. However, not only OPCs are affected by their surroundings, some evidence also shows that they can mediate a wide range of effects on the ECM.62,63 Many of the factors that influence the ECM during neuroinflammation are downstream of the effects of OPCs on cytokine and chemokine production, as well as its effect on other cellular activities.64,65 Furthermore, data from studies suggest that NG2, a chondroitin sulfate proteoglycan expressed by OPCs, is a major species whose deposition at the lesion site in the CNS directly inhibits regeneration.66,67 Recognizing CSPGs as known inhibitors of remyelination in the CNS, we investigated whether preactivated-OPC transplantation could modulate their expression at the spinal cord. Using cell isolation from two different models, we found that, compared to these two groups, after CPZ-OPCs transplantation, the ECM was associated with greater CSPG deposition. Moreover, in the absence of cell transplantation, the highest amount of CSPG was observed. Consistent with findings from the aforementioned studies, we also observed that neuronal remyelination occurred to a lesser extent in the group with high CSPG deposition.
Conclusion
In conclusion, our study demonstrated that transplantation of OPCs pre-activated with the LPS model significantly improves demyelination compared to CPZ-OPCs. In sum, our study has shown that transplanted OPCs survive and migrate in demyelinated mice, and their behavior depends on the demyelination model with which the cells were activated. These findings emphasize the impact of the demyelinated activation environment on OPC behavior in neurodegenerative diseases. Further research is needed to comprehensively investigate molecular changes in OPCs during activation in response to neurological events.
Footnotes
Acknowledgements
The authors extend their gratitude to the professors of the Anatomy Department, Tehran University of Medical Sciences, and the Department of Postgraduate Studies at Tehran University of Medical Sciences for their collaborative efforts and assistance in conducting this research.
Author contributions
Professor Iraj Ragerdi Kashani: Conceived and designed the study, and served as the primary supervisor. Farzaneh Rezaei Yazdi: Performed the experimental part of the research and conducted the statistical analysis and drafted the manuscript. Hoda Akbari: Contributed to the experimental work and the statistical analysis. Professor Parihar Pasbakhsh: Provided consultation on the study design and served as the secondary supervisor.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was supported by a Grant (Number: 71252) from the Tehran University of Medical Sciences.
Ethical approval
This study was approved by the Ethics Committee (
Animal welfare
The present study followed Institutional Animal Care and Use Committee (IACUC) of the Tehran University of Medical science guidelines for animal treatment and complied with relevant legislation.
