Abstract
The skin serves as the primary defensive barrier of the human body against external stimuli and damage. Keratinocytes, which are the predominant cell type in the human epidermis, undergo a differentiation process that is crucial for the formation of the skin barrier. Myricetin, a dietary flavonoid present in various fruits and vegetables, is known to play a significant role in maintaining intestinal barrier function; however, its impact on the skin barrier remains inadequately understood. Consequently, this study investigates the effects of myricetin on the differentiation of epidermal keratinocytes and the integrity of the skin barrier. Differentiation of primary mouse keratinocytes was induced using 1.8 mM CaCl2. tudy demonstrated that myricetin effectively suppresses cell proliferation and induces both cell cycle arrest and calcium ion (Ca2+) influx, without influencing apoptosis. Concurrently, myricetin enhances the expression of differentiation markers, including K10, TGase1, Filaggrin, and Involucrin, and facilitates the formation of tight junctions. Upon examining the underlying mechanisms, we discovered that myricetin activates the TRPV4 channel, and the promotion of keratinocyte differentiation by myricetin is contingent upon the activation of this channel. In summary, these findings suggested that myricetin could promote keratinocytes differentiation and have well-established skin barrier protective function.
Introduction
Skin is the largest organ of the human body, and consists of the three layers epidermis, dermis, and hypodermis. 1 In particular, the epidermis, composed of keratinocytes and intercellular lipids, is the outermost protective barrier of the skin and protects the body against external environment factors, such as chemicals, microorganisms, antigens, and UV radiation, as well as preventing excessive water loss.2–4 Such a loss of barrier function makes skin more sensitive to various stimuli and allergens, thereby easily giving rise to inflammation and itch, which is also the main cause of skin diseases such as atopic dermatitis and psoriasis.5,6 Therefore, the skin barrier function is essential for maintaining skin homeostasis, and is a key strategy in treatment atopic dermatitis and other skin diseases.
Epidermal keratinocyte (KC) differentiation is essential for the formation and maintenance of the normal skin barrier function and homeostasis. 7 During differentiation, the KC from the basal layer migrate upward through the spinous and granular layers until they finally reach the stratum corneum, accompanied by gene expression related to cell proliferation, differentiation and cycle. Of these, Keratins 5 (K5) and K14 are expressed in the basal layer and substituted by K1 and K10 when the cells move to the spinous layer and finally the cells expressed filaggrin (FLG) and involucrin (IVL) in the stratum corneum.8,9 In the terminal differentiation, the KC lose their nuclei and become corneocytes, which form the bricks in the “bricks and mortar” model, lipids secreted by lamellar body act as mortar, and the tight junctions between KC serve as “steel rebar,” which together constitute the intact epidermal barrier. 7 KC differentiation is a complex and strictly regulated process, and any damage to the structure of the skin results in the dysfunction of the skin barrier. The calcium (Ca2+) in the epidermis, increasing from the basal layer to the granular layer, plays an important regulatory role in the KC differentiation. A large number of studies show that Ca2+ concentration in the epidermis directly affects the balance between KC proliferation and differentiation.10,11 Thus, in this study, in order to mimic keratinocytes differentiation in vitro, the mouse primary keratinocytes were stimulated using 1.8 mM CaCl2.
Transient receptor potential vanilloid 4 (TRPV4), a member of TRPV ion channel family, is mainly expressed in the central nervous system, skin, kidney, and bladder. 12 TRPV4 can be activated by mechanical stimuli, hypoosmolarity, chemical, and physical stimuli, thereby resulting in enhanced Ca2+ influx and regulating many physiological and pathological processes, such as cell growth, signal transduction. 13 In skin, TRPV4 highly expresses in keratinocytes and immune cells, its dysfunction is related to impaired epidermal barrier function, itching and inflammation. 14 Activation of TRPV4 induced by heat and chemical agonists can accelerate skin barrier regeneration. 15 In addition, TRPV4 activation can promote Ca2+ influx and contribute to the tight junction formation, thereby increasing intercellular barrier integrity in vitro.
Myricetin (Myr) is a naturally dietary flavonoid (3, 5, 7, 3’, 4’, 5’-hexahydroxyflavone), which is widely found in a variety of foods, such as fruits, vegetables, berries, tea, and red wine. 16 Myricetin exhibits multiple bioactivities, including anti-inflammatory, immunomodulatory, antibacterial, and analgesic effects.17,18 It has been shown that myricetin significantly reduces the secretion of pro-inflammatory cytokines, such as IL-1β and IL-10, and has inhibitory effect on inflammation induced by LPS. 19 Besides these effects, myricetin also plays an important role in maintaining barrier function. It can reduce permeability of intestinal epithelial cells (IEC-6), and promotes the formation of tight junctions by enhanced the expression of tight junction proteins such as Claudin-1, occluding and Zonula Occludens-1 (ZO-1).20,21 However, it has not been reported whether myricetin has beneficial effects in skin barrier function.
Therefore, the aim of this study was to investigate the effects of myricetin on KC differentiation and skin barrier function. To achieve this aim, 1.8 mM CaCl2 was used to induce differentiation of the mouse primary keratinocytes, while intervening with myricetin at different concentrations. Subsequently, the effects of myricetin on differentiation were evaluated by examining the proliferation/differentiation markers and formation of tight junctions, and the mechanism was explored based on TRPV4 channel.
Methods
Drugs and chemicals
Myricetin was purchased from Aladdin (Shanghai, China), and its chemical structure is illustrated in Figure 1(a). Myricetin was dissolved in DMSO and diluted to the desired concentrations. Keratinocyte medium-defined (KM-d) medium was purchased from ScienCell (Carlsbad, California, USA). Ca2+-free DMEM medium was obtained from Yuchun biology (Shanghai, China). Anhydrous calcium chloride (CaCl2) was purchased from Aladdin Co., Ltd (Shanghai, China). Dispase II was obtained from Yuan Ye Biotechnology Co., Ltd (Shanghai, China). CCK8 was obtained from the Dojindo (Tokyo, Japan). Hoechst 33258 and Fluo-4 AM Ca2+ indicator dye were purchased from Biyuntian Biotechnology (Shanghai, China). Biomarker Cell/Tissue Total RNA Isolation kit, BiomarkerScript II 1st Strand cDNA Synthesis Kit and SYBR green dye were obtained from Biomarker Technologies (Beijing, China). Filaggrin antibodies were obtained from Bioss Biotechnology Co. Ltd. (Beijing, China).

The effects of myricetin on the growth of mouse primary keratinocytes. (a) The chemical structure of myricetin. (b) The effect of myricetin on cell morphology during Ca2+ induced differentiation. (c) The cell proliferation was measured on days 0, 2, 4, and 6.
Keratinocyte culture
Mouse primary keratinocytes were isolated from neonatal mouse skin as described previously. 22 Briefly, skins of newborn mice were treated with 0.3% Dispase II for 16–18 h at 4℃. The epidermis was then mechanically separated from the dermis. Isolated epidermis was digested with 0.25% trypsin/EDTA for 10 min and strained through a 200-mesh sieve. The filtrate was centrifuged at 1000 rpm for 5 min to collect keratinocytes. These cells were cultured in keratinocyte medium-defined (KM-d) medium containing 1% (v/v) penicillin/streptomycin solution and Keratinocyte growth supplement. After reaching confluency, the cells were cultured for 6 days in KM-d medium supplemented with 1.8 mM CaCl2 to induce keratinocyte differentiation. At the same time, different concentrations of myricetin (15, 30, 60 μM) were administered during the differentiation induction.
Cell viability assay
Cell viability was determined using cell counting Kit-8 (CCK8). Briefly, the mouse primary keratinocytes were plated onto 96-well plates. They were then switched to KM-d medium containing 1.8 mM CaCl2 and incubated with or without different concentrations of myricetin. Cells in each group cultured for 0, 2, 4, and 6 day respectively and were added 10 μL CCK8 detection solution. After incubation in 5% CO2 for 1 h at 37°C, the cell viability was evaluated by 450 nm absorbance values.
Cell apoptosis assay
Hoechst staining was employed to evaluate cell apoptosis during keratinocyte differentiation. As cells undergo apoptosis, chromatin sequestraters. After Hoechst 33,258 staining, the nucleus of viable cells is normally blue, while the nucleus of apoptotic cells appears abnormally bright white color. The mouse primary keratinocytes were seeded into 6-well plates. The differentiation media was removed at day 6, and the cells were then fixed with paraformaldehyde and stained with Hoechst 33,258 in the dark. After 30 min, the cells were observed under a fluorescence microscope (Olympus, Japan), and five randomly chosen fields were captured from each slide to quantification of dead cells. The apoptosis rate was calculated using the following formula: apoptosis rate = number of apoptotic cells/total number of cells × 100%.
Intracellular Ca2+ concentration determination
Intracellular Ca2+ levels were measured using Fluo-4/AM, a fluorescent Ca2+ indicator. The keratinocytes were grown in 6-well plates and differentiation was induced by 1.8 mM CaCl2 for 6 days in a 5% CO2 incubator at 37°C. Afterward, cells were then incubated with Hank’s balanced salt solution (HBSS) containing 5 µM Fluo-4/AM for 30 min. The cells were washed with HBSS and incubated for another 30 min in HBSS at 37°C to ensure complete conversion of Fluo-4 AM to Fluo 4. Finally, the cells were observed under a fluorescence microscope and five visual fields were randomly chosen to calculate the fluorescence intensity.
Quantitative real-time PCR
Briefly, the mouse primary keratinocytes were differentiated by KM-d medium supplemented with 1.8 mM CaCl2 and total RNA was extracted using a Biomarker Cell/Tissue Total RNA Isolation kit following manufacturer’s protocol. The concentration of the total RNA was assessed using the Nanodrop 2000 spectrophotometer (Thermo Fisher Scientific, USA), and its purity was assessed according A260/A280 and A260/A230 ratios. For reverse transcription, BiomarkerScript II 1st Strand cDNA Synthesis Kit was used following the manufacturer’s instructions. Quantitative real-time PCR analysis was conducted using a CFX96 Real-Time PCR detection system (Bio-Rad, USA) with SYBR Green Supermix. The relative expression levels of mRNA of each targeted gene were normalized to the β-actin. The primer sequences for gene-specific amplification were listed in Tab.S1.
Immunofluorescence staining
Immunofluorescence staining was performed as previously described. 23 In brief, the differentiated keratinocytes were fixed with 4% paraformaldehyde for 15 min and then permeabilized with 0.5% triton X-100 for 10 min. After that, the cells were washed with PBS and blocked with immunostaining closure solution for 30 min at room temperature. Subsequently, the cells were incubated at 4℃ overnight with the following antibodies: anti-filaggrin antibody (1:200), anti-involucrin (1:200), and anti-TRPV4 (1:200). They were then washed three times with PBS and incubated with a Cy3-labeled goat anti-rabbit IgG (1:200) for 1 h at room temperature. Finally, the cells were counterstained with DAPI to label cell nuclei, and images were captured by using an inverted fluorescence microscope.
Statistical analysis
All statistical analyses were performed using SPSS statistical software (version 26.0). All experiments were performed independently at least triplicate and the results were expressed as the mean ± standard deviation (SD). Statistical significance differences among groups were analyzed by using one-way ANOVA with Dunnett’s multiple comparison test. p-values < 0.05 were considered to denote statistically significant.
Results
The effects of myricetin on the morphological changes and growth of keratinocytes induced by Ca2+
The effect of myricetin (15, 30, and 60 μM) on the morphology of mouse primary keratinocytes was examined on day 6 post-induction of differentiation. As shown in Figure 1(b), the non-induced control cells maintained an elongated, spindle-shaped morphology. In the intervention group with different concentrations of myricetin, spindle structures disappeared and the cells displayed oval shape, accompanied by tight junction formation between cells. Furthermore, to investigate the effect of myricetin on the growth of cells during differentiation, the proliferation of cells was measured by CCK8 on days 0, 2, 4, and 6, respectively (Figure 1(c)). At the day 0, there was no significant difference of the OD values in each group. Compared with the control group, the OD value of high Ca2+ group remarkably reduced at 2 day, indicating growth inhibition. After treatment with different concentrations of myricetin, the number of cells decreased significantly and the growth was retarded compared with that in the high Ca2+ group, and this inhibitory effect was in concentration- and time-dependent manner. These results indicated that myricetin promoted the growth arrest of keratinocytes in the differentiation induced by Ca2+.
The effects of myricetin on the cell cycle and apoptosis of keratinocytes
The reduced cell proliferation observed in myricetin-treated cells could be due to cycle arrest and increased apoptosis. Therefore, we examined the expression of cell cycle-related genes by qRT-PCR. Cyclin D1 (CCND1) and Cyclin D2 (CCND2), well-known cell cycle regulators, promote cells from G1 to S phase to facilitate cell proliferation. Cyclin-dependent kinase inhibitor 1A (CDKN1A) and CDKN1C inhibit the progression of cells from G1 to S phase and induce G1/S-arrest. At differentiation day 6, the expression of CCND1 and CCND2 was decreased in the high Ca2+ group while CDKN1A and CDKN1C were upregulated. 60 μM myricetin could further downregulate the expression of CCND1 and CCND2 (p < 0.001), and upregulated the mRNA levels of CDKN1A and CDKN1C, indicating that myricetin could promote the cell cycle arrest in the differentiation (Figure 2(a–d)). Immediately after, Hoechst staining was assessed to evaluate cell apoptosis. There was no significant difference in the apoptosis rate among all groups (Figure 2(e and f)). These results indicate that the growth arrest of keratinocytes promoted by myricetin during the induction of differentiation is due to cell cycle arrest rather than apoptosis.
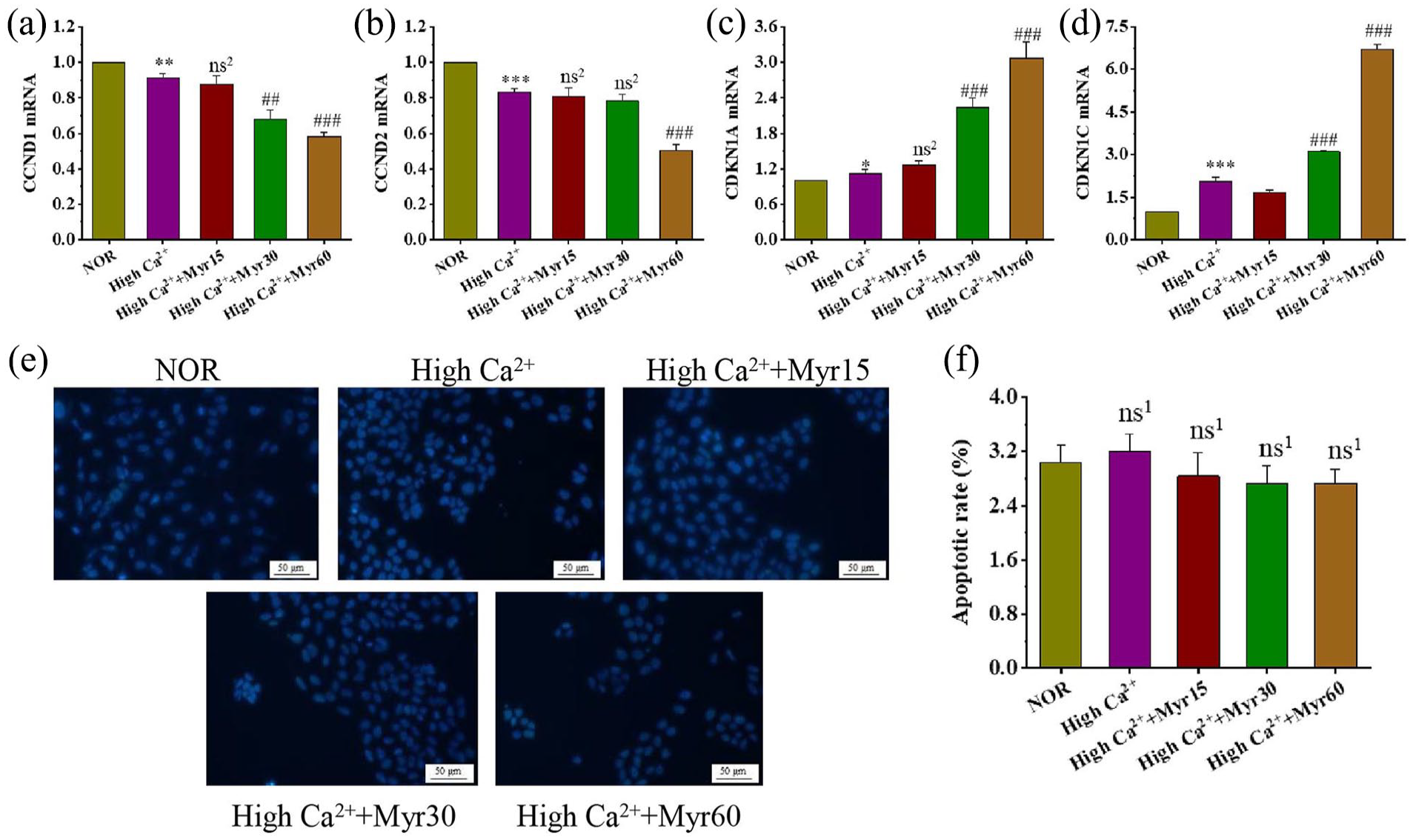
The effects of myricetin on the cell cycle and apoptosis. The mRNA levels of cell cycle genes CCND1 (a), CCND2 (b), CDKN1A (c), and CDKN1C (d). (e) Apoptotic cells were detected by Hoechst staining (200 magnification, scale bars = 50 μm). (f) Quantification of apoptosis rates, which was counted by the number of apoptotic cells to the total number of cells in three randomly visual fields in per section.
The effect of myricetin on intracellular Ca2+ level
The intracellular concentrations of Ca2+ in mouse primary keratinocytes were detected by Fluo-4 AM. The results were shown in Figure 3(a and b), Fluo-4 fluorescence intensity in the untreated control group was very low, whereas it was significantly increased in the high Ca2+ group indicating an increase in nuclear calcium. Three different concentrations of myricetin intervention groups showed significantly increased green fluorescence intensities compared to the high Ca2+ group, suggesting that myricetin could promote Ca2+ influx.
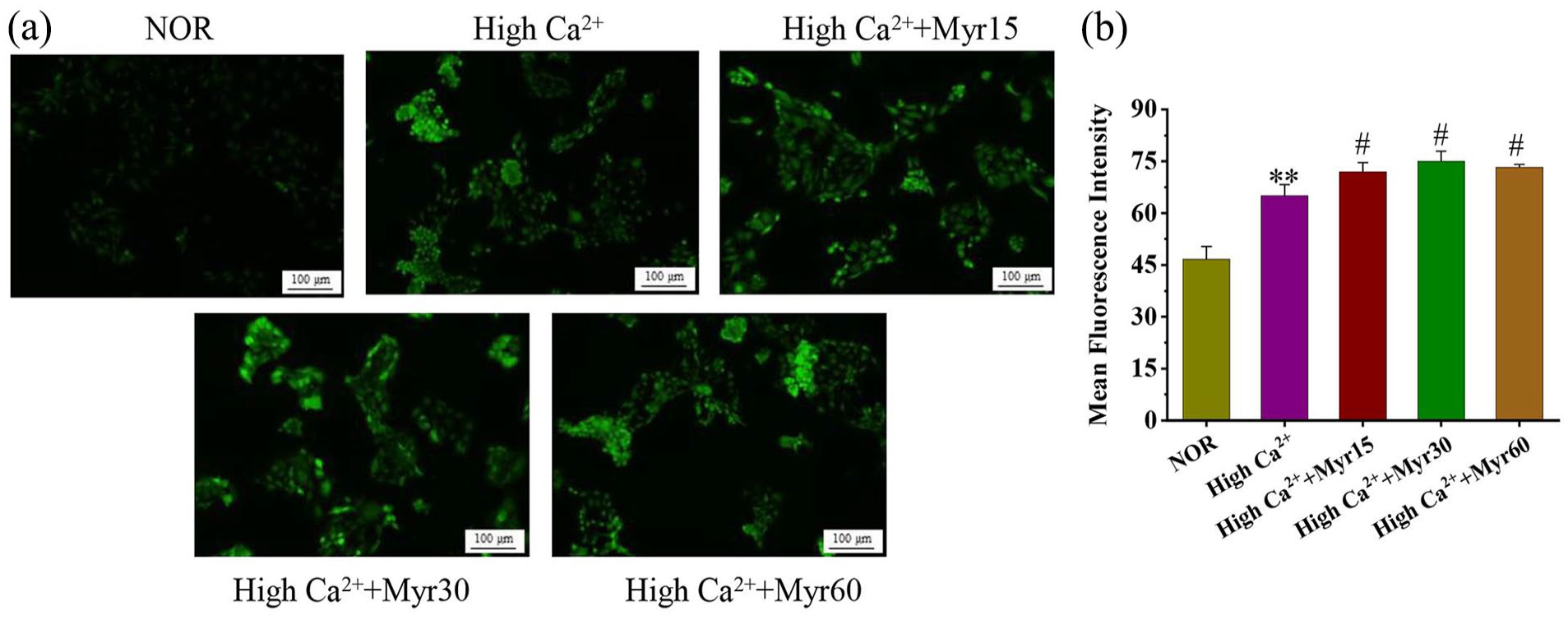
The effect of myricetin on intracellular Ca2+ levels during differentiation. (a) The concentrations of Ca2+ in keratinocytes were detected by Fluo-4/AM fluorescent probe (100 magnification, scale bars = 100 μm). (b) Quantitative analysis of intracellular Ca2+ levels, fluorescence intensity was measured by Image J software.
The effects of myricetin on differentiation and tight junctions of keratinocytes
In epidermis, basal layer keratinocytes are in a proliferating state and mainly expresse Keratins 5 (K5) and K14. When they leave the basal layer and gradually migrate upwards, TGase l is expressed in the spinous layer, K1 and K10 expression is evident mainly in the granular layer, and terminal differentiation markers FLG and IVL are expressed in the stratum corneum. 24 To investigate the regulatory effects of myricetin on differentiation, the expression of proliferation/differentiation markers were detected by qRT-PCR in the keratinocyte differentiation. The expression of K5 and K14 in the high Ca2+ group was significantly reduced compared to the normal control group, while differentiation markers TGase l, K10, FLG, and IVL were significantly increased (Figure 4(a–f)). Compared with the high Ca2+ group, the expression levels of K5 and K14 were further downregulated in the 60 μM myricetin group, and differentiation markers were up-regulated, indicating that high concentrations of Ca2+ could induce keratinocytes differentiation, and myricetin could promote this process (Figure 4(a–f)). In addition, we also examined expression of the tight junction markers during differentiation. Intervention with myricetin could further upregulate the expression of E-cadherin and Claudin-1 in the high Ca2+ group (Figure 4(g–h)), and the results showed significant differences. All the results indicated that myricetin could promote the keratinocyte differentiation induced by Ca2+.

The effect of myricetin on the balance of proliferation and differentiation. (a) Keratin5 mRNA. (b) Keratin 14 mRNA. (c) TGasel mRNA. (d) Keratin10 mRNA. (e) Filaggrin mRNA. (f) Involucrin mRNA. (g) Ecadherin mRNA. (h) Claudin-1 mRNA.
The effect of myricetin on the TRPV4 expression
To explore the potential mechanism of myricetin promoting keratinocyte differentiation, the TRPV4 expression was detected using qRT-PCR and cellular immunofluorescence staining on the 6th day of differentiation. TRPV4 mRNA level in the high Ca2+ group showed no significant change relative to normal control group, but its expression was markedly increased in the myricetin intervention groups (Figure 5(a)). Meanwhile, immunofluorescence staining showed weak TRPV4 staining in control group and high Ca2+ group, whereas the red fluorescence intensity was significantly enhanced in the myricetin intervention group compared with that in the high Ca2+ group (Figure 5(b and c)). These results showed that myricetin promoted keratinocyte differentiation, which might be related to the TRPV4-mediated Ca2+ influx.
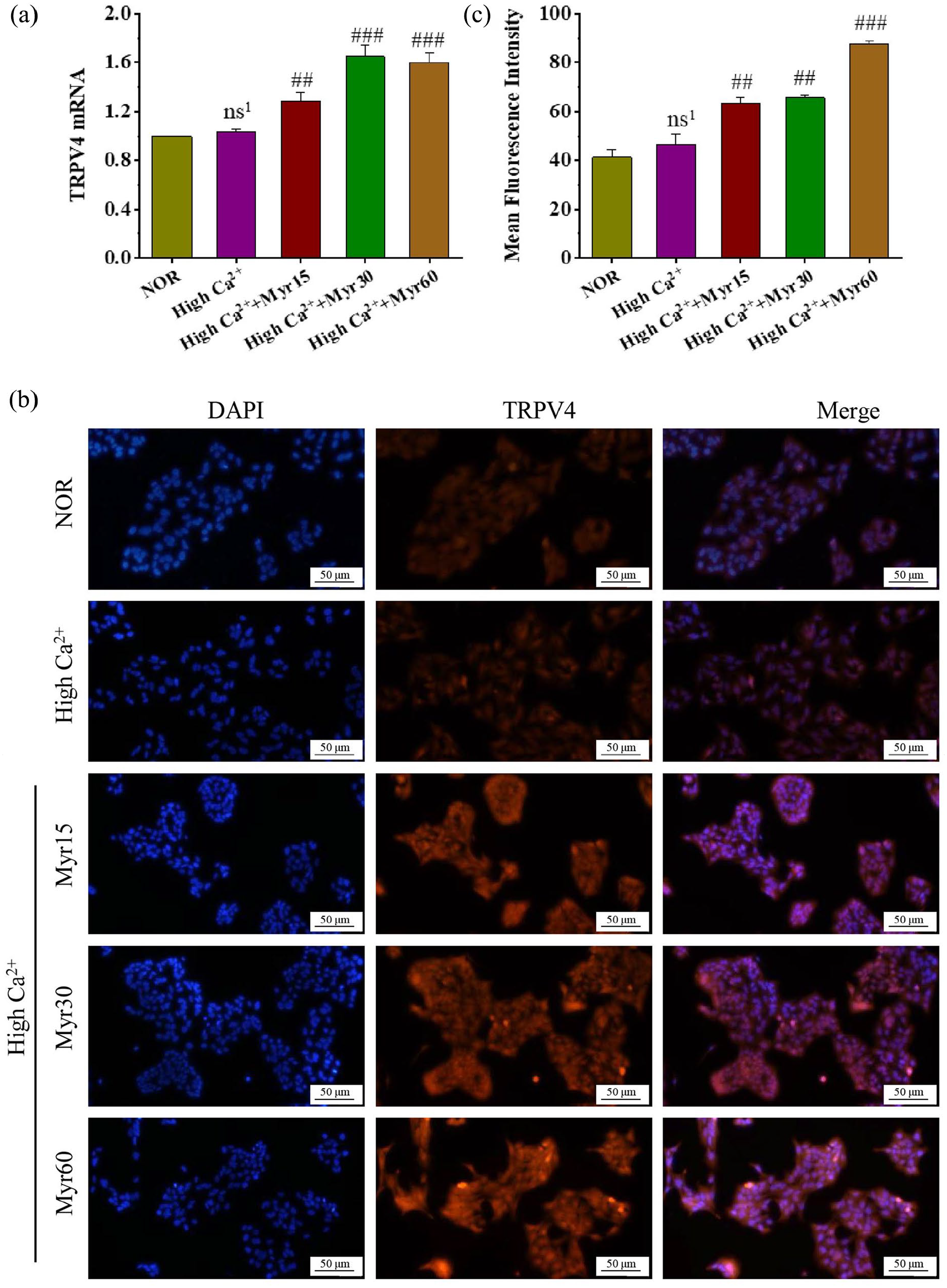
The effect of myricetin on the TRPV4 expression during the differentiation. (a) TRPV4 mRNA. (b) Immunofluorescence staining of TRPV4, 200 magnification, scale bars = 50 μm. (c) Mean fluorescence intensity of TRPV4, which was calculated with three random visual fields of each section by Image J.
Myricetin promoted primary keratinocyte differentiation through activation of TRPV4 channel
To investigate the role of TRPV4 in the process of keratinocyte differentiation, the agonist GSK1016790A and antagonist HC-067047 were used to upregulate or inhibit TRPV4 expression. TRPV4 mRNA expression was detected by qRT-PCR during Ca2+ induced differentiation. As shown in Figure 6(a and b), the TRPV4 expression was significantly upregulated upon 40 nM GSK1016790A treatment and further increased with the combined treatment of GSK1016790A and myricetin. In contrast, the TRPV4 expression in the inhibitor HC-067047 group was significantly reduced to the high Ca2+ group, and the combination of myricetin and HC-067047 largely restored its mRNA level, suggesting that myricetin could promote TRPV4 expression. Next, to investigate whether myricetin promoted Ca2+ induced differentiation dependent on TRPV4 channel, GSK1016790A and HC-067047 were administered to upregulate or inhibit TRPV4 expression and then detected the expression of proliferation/differentiation markers. GSK1016790A could enhance the expression of differentiation genes (TGase l, K10, FLG, and IVL), and suppressed proliferation genes expression. Combination of myricetin and GSK1016790A further promoted differentiation and inhibited proliferation in keratinocytes (Figure 7(a–f)). In contrast, inhibitor HC-067047 increased the levels of basal K5 and K14, and decreased the expression of TGase l, K10, FLG and IVL, whereas this trend was reversed by myricetin treatment (Figure 8(a–f)). Additionally, immunofluorescence staining was used to detect terminal differentiation genes FLG and IVL, and similar trends were found (Figure S1 and S2). In summary, these experiments indicated that myricetin promoted mouse primary keratinocyte differentiation through activation of TRPV4 channel.

Effect of myricetin on TRPV4 mRNA expression in activation and inhibition experiments. (a) TRPV4 mRNA level in TRPV4 activation experiments. (b) TRPV4 mRNA level in TRPV4 inhibition experiments.

The effect of myricetin on proliferation/differentiation balance in TRPV4 activation experiment. (a) Keratin 5 mRNA. (b) Keratin 14 mRNA. (c) TGase 1 mRNA. (d) Keratin 10 mRNA. (e) Filaggrin mRNA. (f) Involucrin mRNA.

The effect of myricetin on proliferation/differentiation balance in TRPV4 inhibition experiment. (a) Keratin 5 mRNA. (b) Keratin 14 mRNA. (c) TGase 1 mRNA. (d) Keratin 10 mRNA. (e) Filaggrin mRNA. (f) Involucrin mRNA.
The effects of TRPV4 on the biological functions of keratinocyte cells
To investigate the TRPV4 channel on cellular biological function during the differentiation of keratinocytes, cell proliferation, cycle, apoptosis and intracellular Ca2+ levels were detected on day 6 following the administration of GSK1016790A and HC-067047. CCK8 assay showed cell proliferation was significantly inhibited by agonist GSK1016790A, and further inhibition was observed by the combination treatment of GSK1016790A and myricetin. In contrast, antagonist HC-067047 could promote keratinocyte cell proliferation and treatment with myricetin attenuated this phenomenon (Figure S3). Immediately after, we further analyzed cell cycle distribution. GSK1016790A significantly upregulated the expression of CDKN1A and CDKN1C, and the effect was further enhanced by myricetin co-administration. HC-067047 promoted the expression of CCND1 and CCND2, while CDKN1A and CDKN1C were downregulated. This trend, however, was reversed in the HC-067047+myricetin group (Figure S4), suggesting that myricetin induced cell cycle arrest and growth arrest depending on the TRPV4 activation. Then, apoptosis analysis was conducted to assess cell apoptosis. Myricetin, GSK1016790A and HC-067047 did not increase apoptosis in keratinocyte cells compared with the High Ca2+ group (Figure S5). To test the effect of TRPV4 channel on myricetin-promoted Ca2+ influx, we employed the Fluo-4/AM to detect Ca2+ influx. Compared with the high Ca2+ group, fluo-4 fluorescence intensities were significantly increased by GSK1016790A and myricetin, and further enhanced by a combined treatment. In contrast, HC-067047 was able to block Ca2+ influx mediated by TRPV4, this effect was notably ablated in the combined treatment of myricetin and HC-067047 (Figure S6). The above results suggested that myricetin promoted keratinocyte differentiation, which may be depend on the TRPV4-mediated Ca2+ influx.
Discussion
The epidermis is located on the outermost layer of the skin, and is the first barrier against invading cutaneous pathogens and environmental stimuli. The epidermis is in a process of constant renewal, KC produced in the basal layer move upward through the stratum spinosum and stratum granulosum, and finally culminate in the stratum corneum. The proliferation, migration and differentiation of KC is a precise regulation, problems in any links may lead to skin barrier dysfunction, causing various cutaneous diseases including AD and psoriasis.25,26 Therefore, finding compounds that regulate the differentiation of keratinocytes has important scientific implications.
Under normal conditions, there is a concentration gradient of Ca2+ in the epidermis, which gradually increases from the basal layer to the granular layer. 27 The maintenance of this concentration gradient is essential for the differentiation of KC. Low concentrations (0.05–0.15 mM) of Ca2+ promotes KC proliferation and inhibits differentiation, but high concentrations (>1 mM) induces differentiation and causes cell growth arrest. 28 As a differentiation inducer of KC, Ca2+ can also induce KC differentiation in vitro. After the Ca2+ concentration was adjusted to 1.8 mM, the morphology of the cells changed significantly on day 6, which was composed of spindle to ovoid shaped. In addition, high concentration Ca2+ suppresses cell proliferation, accompanied with the cycle arrest, and keratinocytes remained in G2/M phase to continue DNA replication. 29 Martinez et al. 30 found that the protein expression levels of Cyclin D1 and Cyclin D2 were significantly decreased in the keratinocyte differentiation induced by Ca2+, a similar trend was observed in our results. Concomitantly, we observed an increase in the expression of cycle arrest genes CDKN1A and CDKN1C, which was consistent with the inhibition of cell proliferation. Treatment with myricetin further inhibited cell proliferation and the cell cycle, but no increase in cell apoptosis was detected, suggesting that myricetin might promote keratinocyte differentiation.
Gene expression is a highly coordinated process during the keratinocyte differentiation. K1 and K10 are mainly expressed in the granular layer and are the markers of differentiation, while terminal differentiation proteins such as FLG and IVL are mainly expressed in the corneous layer. 31 In vitro, expression of these differentiation markers is tightly regulated by extracellular Ca2+ concentration. We found that the expression of differentiation markers TGase1, K10, LOR and IVL increased on the day 6 of Ca2+ induction, while the expression of basal layer proliferation markers K5 and K14 was reduced. In addition, Claudin-1 and E-adherin mRNA level was significantly upregulated during differentiation, which was compatible with the study of Sayedyahossein et al. 32 These results suggested that myricetin promoted the keratinocytes differentiation induced by Ca2+.
Transient receptor potential vanilloid 4 (TRPV4) belongs to the family of transient receptor potential channels (TRP), which is nonselective cation channels. 33 TRPV4, which is mainly expressed in keratinocytes of the skin, is one of the main Ca2+ inflow channels and plays an important role in skin barrier function. 34 Some studies have demonstrated that selective activation of TRPV4 in mice was able to accelerate skin barrier regeneration, while knockout of TRPV4 resulted in impaired skin barrier and abnormal differentiation in AD.35,36 In addition, some studies showed that TRPV4 activation by heating or chemical stimuli could induce extracellular Ca2+ influx, and promote the expression of differentiation markers K1 and K10, which were helpful in enhancing skin barrier function. 37 On the other hand, TRPV4 activation could up-regulate the expression of occluding and claudin-4, thereby strengthening the tight junction barrier of keratinocytes. 36 In this study, we found that myricetin could activate TRPV4 channels and promote extracellular Ca2+ flow, thereby accelerating keratinocyte differentiation and tight junction formation. The combined intervention of myricetin and TRPV4 agonists further promoted keratinocyte differentiation induced by Ca2+, while combination with inhibitors could restore the differentiation process to a certain extent, suggesting that myricetin promoted the keratinocytes differentiation of through the activation of TRPV4 channels.
Conclusions
In summary, myricetin could promote the differentiation of primary keratinocytes induced by Ca2+. During the differentiation of keratinocytes induced by 1.8 mM Ca2+, myricetin could inhibit keratinocyte growth, and promote cell cycle arrest. In addition, myricetin inhibited the expression of proliferation markers K5 and K14, while the differentiation markers TGasel, K10, FLG, and IVL were up-regulated to promote keratinocyte differentiation. Moreover, the expression of Claudin-1 and E-adherin in keratinocytes were significantly up-regulated, suggesting that myricetin could promote the tight junction formation during differentiation. Exploring its mechanism, we found that it might be dependent on the activation of TRPV4 channel and promotion of Ca2+ influx.
Supplemental Material
sj-docx-1-iji-10.1177_03946320251317287 – Supplemental material for Myricetin enhances keratinocytes differentiation via TRPV4 channel activation in mouse primary keratinocytes
Supplemental material, sj-docx-1-iji-10.1177_03946320251317287 for Myricetin enhances keratinocytes differentiation via TRPV4 channel activation in mouse primary keratinocytes by Jie-Fang Gao, Tong-Xuan Li and Guo-Qiang Zhang in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
None
Author contributions
Jiefang Gao was responsible for the whole experiment implementation and wrote the paper, Tongxuan Li checked all the statistical analyses, Guoqiang Zhang did final proof reading and approved the final manuscript. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hebei Province (H2024206180).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
