Abstract
This study aimed to characterize micronutrient deficiencies, including iron, ferritin, folic acid, vitamin D, zinc (Zn), vitamin B12, and copper, in patients with celiac disease, and evaluated the effects of these deficiencies on selected hematological parameters, including hemoglobin and mean corpuscular volume (MCV). Celiac disease (CeD), an immune-mediated disorder affecting the small bowel, is associated with genetic factors and micronutrient deficiencies. This meta-analysis was performed in accordance with the PRISMA guidelines. Literature searches of multiple databases retrieved 4140 studies, of which 45 were selected. Risk of Bias was performed in accordance with the STROBE checklist. Meta-analysis revealed a significant difference in hemoglobin levels between patients with CeD and controls (standardized mean difference (SMD) −0.59 (95% confidence interval (CI) −0.8459 to −0.3382); P = 0.0003). Iron levels were lower in patients with CeD (SMD ≈ −0.4 (95% CI −0.7385 to −0.0407); P = 0.0334), as were ferritin (SMD −0.6358 (95% CI −0.8962 to −0.3755); P = 0.0002), folic acid (SMD −0.5446 (95% CI −0.9749 to −0.1142); P = 0.0187), and vitamin D (SMD −0.4011 (95% CI −0.8020 to −0.0001); P = 0.0499) levels, while Zn levels were significantly reduced (SMD −1.1398 (95% CI −2.0712 to −0.2084); P = 0.0242). No significant differences were found in MCV, or copper or vitamin B12 levels between patients with CeD and controls. This study highlighted significantly higher micronutrient deficiencies in patients diagnosed with CeD than in controls, underscoring the importance of systematic nutritional assessment and multidisciplinary management to address micronutrient deficiencies and minimize negative health impact(s).
Keywords
Introduction
Celiac disease (CeD) is a multifactorial condition influenced by genetic and environmental factors.1,2 More than 90% of patients with CeD carry the human leukocyte antigen (HLA)-DQ2 haplotype, whereas the remainder harbor HLA-DQ8. Although these genetic markers are necessary, they are insufficient for CeD genesis.3,4 Furthermore, the presence of single or double copies of HLA-DQB1*02 has been associated with an increased risk for developing CeD. 5 Gluten consumption is the most important environmental factor in the pathological development of CeD; nevertheless, other factors, including viral infections, gut microbiota, and smoking, have also been implicated in its pathogenesis.3,4 CeD can occur at any age and has a wide range of symptoms. As a result, it is critical to make a diagnosis, not only in individuals experiencing conventional gastrointestinal symptoms, but also in those with extraintestinal clinical features because both forms can have significant and severe implications. 1 The only effective treatment for CeD is strict adherence to gluten-free diet (GFD). Noncompliance with GFD increases the risk for morbidity and mortality due to related illnesses such as infertility, bone ailments, and cancer. According to the 2013 American College of Gastroenterology guidelines, one of the most common complications of CeD at diagnosis is micronutrient deficiency, primarily caused by chronic malabsorption due to villous atrophy (VA) in the small intestine, leading to a significant reduction in the absorptive surface area. 6 Therefore, micronutrient deficiency should be identified and assessed in patients with newly diagnosed CeD. 7 Although a GFD generally leads to mucosal recovery, some patients with CeD undergoing long-term treatment may experience persistent VA on follow-up, with or without ongoing or recurrent symptoms.4,8 This persistent enteropathy appears to be more common in individuals >45 years of age, 9 as indicated by recent findings in which age ≥45 years was established as one of the independent variables predicting the persistence of VA, 10 although it has also been described in 19% of younger patients. 11 Furthermore, the persistence of enteropathy may be exacerbated by poor adherence to a GFD, lack of nutrient fortification in gluten-free products, or generally lower nutritional value of these products, 12 which may further contribute to the persistence of micronutrient deficiencies in patients with CeD. 13 Micronutrient deficiencies in patients with CeD are highly debated. As such, this study aimed to identify anomalies in micronutrient levels, including iron, ferritin, folic acid, vitamin D, zinc (Zn), vitamin B12, and copper, in patients with CeD, and to assess the effects of these deficiencies on specific hematological parameters, such as hemoglobin and mean corpuscular volume (MCV), which are indirectly influenced by deficiencies in iron, folate, and vitamin B12.
Material and methods
Search strategy and study selection
This meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (i.e. “PRISMA”) statement. 14 A comprehensive literature search of the PubMed, Scopus, Google Scholar, and Web of Science databases for relevant studies, published between 1963 and December 2023, was conducted. The search was repeated until March 2024 to identify the most recent studies. The main search terms included “celiac disease,” “celiac,” “coeliac disease,” “micronutrient,” and “deficiency.” Synonyms and alternative spelling methods were used in this study. A full search strategy is presented in Appendix 1. The results from all databases were aggregated using Zotero version 6.0.36 (Zotero.org). Five researchers independently reviewed titles and abstracts to identify potentially eligible studies for full-text review. All reviewers assessed the full text of the articles in detail. Disagreements were resolved by collaborative discussion, when required. In cases of persistent disagreement, the senior author made the final decision. The reference lists of the selected studies were also examined to identify additional, potentially eligible studies.
Inclusion and exclusion criteria
This meta-analysis included all studies reporting raw micronutrient values (mean and standard deviation (SD)) in both the CeD and control groups. Studies were included if they reported raw values for hemoglobin, MCV, iron, ferritin, folic acid, vitamin D, Zn, vitamin B12, and copper in patients diagnosed with CeD according to the American College of Gastroenterology guidelines 15 ) or in the control group. Case reports, case series, commentaries, letters to the editor, and studies that did not report data regarding the research question were excluded. The literature search had no language or data restrictions.
Data extraction
Data were independently extracted by five investigators and entered into spreadsheet software (Excel, Professional Office LTSC Plus, Microsoft Corporation, Redmond, WA, USA). Additionally, we employed various packages from R software to support further data analysis and processing.16–26 Relevant data were extracted independently by five investigators and conflicts were resolved by consensus discussion. The following data were extracted from each study: country; author; year of publication; study design; total number of patients included in the CeD and control groups; mean age in the CeD and control groups; mean and SD values for hemoglobin, MCV, iron, ferritin, folic acid, vitamin D, Zn, vitamin B12, and copper in the CeD and control groups.
Statistical analysis
Meta-analyses were based on a single effect size of the standardized mean. Values were transformed from available statistics (mean and SD) to determine a standardized effect size (Hedges’ g statistic) using a comprehensive meta-analysis software packages.27–33 Hedges’ g is related to Cohen’s d and can be interpreted using the same conventions for effect size, as follows: small (0.2), medium (0.5), and large (0.8).34,35 An added benefit of Hedges’ g is the correction of the biases found in small sample sizes.34,35 The random-effects model was applied in the present meta-analysis, thereby adopting a conservative approach that assumes that the true effect size may vary from study to study, enabling the results to be generalized to populations beyond the study samples.35,36 The Q statistic was used to measure the homogeneity of effect sizes across the studies.35,37 A significant Q statistic indicates dissimilar effect sizes across studies, suggesting that differences in methodology or population samples could introduce variance in the results between studies. 37 To complement the Q test, the I2 statistic was also calculated, which provides an index of the degree of heterogeneity across studies, in which I2 signifies the percentage of the total variability in effect sizes due to the variability between studies and not due to sampling errors within studies. 38 Percentages of approximately 25% (I2 = 25), 50% (I2 = 50), and 75% (I2 = 75) were interpreted as low, medium, and high heterogeneity, respectively.35,39 Egger’s regression test was used to assess publication bias. 40 Rucker’s Limit was used to adjust for suspected publication bias using a random-effects model.35,41 Sensitivity tests (right-skewness and flatness tests) were used to correct for publication bias.35,42 Outliers were addressed by considering studies as outliers if their confidence interval (CIs) did not overlap with those of the pooled effects.35,43
Systematic review registration
This review has been registered on PROSPERO: CRD42024544466. Available from: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42024544466
Results
Study selection
The initial literature search retrieved 4140 studies, of which 145 were assessed by full-text review, and 45 were eligible for inclusion, with perfect agreement between investigators. The study selection process is illustrated in Figure 1.

Flowchart of study selection process.
Study characteristics
The studies selected were from North America (n = 7), South America (n = 3), Northern Europe (n = 2), Western Europe (n = 19), Southern Europe (n = 4), Eastern Europe (n = 2), Southern Asia (n = 4), Western Asia (n = 3), and Northern Africa (n = 1) (Appendices 2 and 3).
Pooled effect size of hemoglobin in the CeD versus control groups
Twelve studies including 545 patients with CeD and 915 controls were included in this meta-analysis. The pooled results revealed that the SMD of hemoglobin level in CD patients was—0.59 (95% CI −0.8459 to −0.3382]; P = 0.0003) compared with the controls (Table 1; Figure 2). Publication bias was not observed (Table 1.b in Supplemental material). The corrected real effect size estimate was −0.7684 (95% CI −1.2658 to −0.2709) (Table 1.c in Supplemental material). The sensitivity (p-curve test) of the estimated SMD was significant (Table 1.d in Supplemental material). (Detailed data for Tables 1.b to 1.d are provided in the Supplemental material).
Pooled effect size (SMD) results (Hemoglobin).

k = number of studies.

Forest plot of hemoglobin levels.
Pooled effect size of iron in the CeD versus control groups
Eight studies including 519 patients with CeD and 14,566 controls were assessed. The pooled results of the meta-analysis revealed that the SMD of iron level in CD patients was −0.4 (95% CI −0.7385 to −0.0407; P = 0.0334) compared with the controls (Table 2; Figure 3). Publication bias was not observed (Table 2.b in Supplemental material). The estimated corrected true effect size was −0.0837 (95% CI −0.7317 to 0.5643) (Table 2.c in Supplemental material). The sensitivity (p-curve test) of the estimated SMD was significant (Table 2.d in Supplemental material). (Detailed data for Tables 2.b to 2.d are provided in the Supplemental material).
Pooled effect size (SMD) results (Iron).
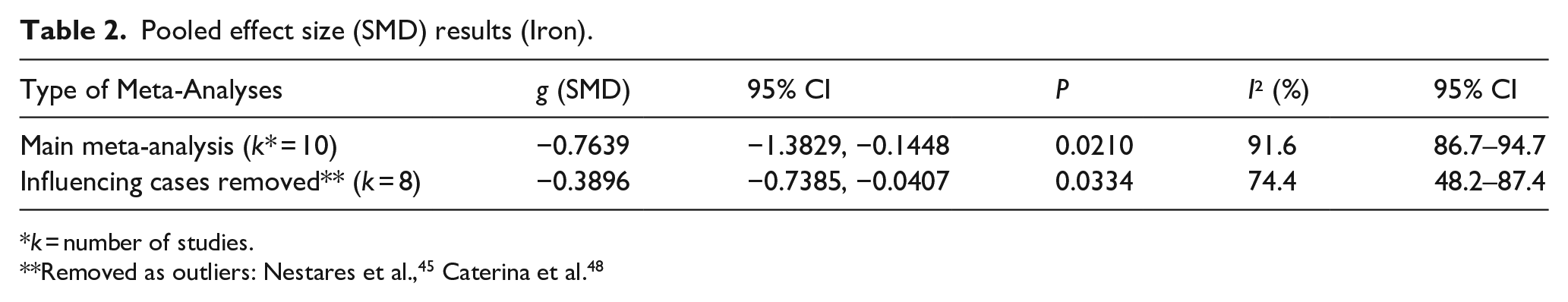
k = number of studies.

Forest plot of iron levels.
Pooled effect size of ferritin in the CeD versus control groups
Twelve studies, including 799 patients with CeD and 1442 controls, were included. The pooled results of meta-analysis revealed that the SMD of ferritin levels in CeD patients was −0.6358 (95% CI −0.8962 to −0.3755; P = 0.0002) compared with the controls (Table 3; Figure 4). No publication bias was observed (Table 3.b in Supplemental material). The estimated corrected true effect size was −0.3885 (95% CI −0.8173 to 0.0403) (Table 3.c in Supplemental material). The sensitivity (p-curve test) of the estimated SMD was significant (Table 3.d in Supplemental material). (Detailed data for Tables 3.b to 3.d are provided in the Supplemental material).
Pooled effect size (SMD) results (Ferritin).

k = number of studies.
Removed as outliers: Caterina et al. 48

Forest plot of ferritin levels.
Pooled effect size of folic acid in the CeD versus control groups
Ten studies, including 834 patients with CeD and 16,378 controls, were included in this meta-analysis. The pooled results revealed that the SMD of folic acid in patients with CeD was −0.5446 (95% CI −0.9749 to −0.1142; P = 0.0187) compared with the controls (Table 4; Figure 5). No publication bias was observed (Table 4.b in Supplemental material). The estimate of the corrected true effect size was −0.2540 (95% CI −0.7134 to 0.2055) (Table 4.c in Supplemental material). The sensitivity (p-curve test) of the estimated SMD was significant (Table 4.d in Supplemental material). (Detailed data for Tables 4.b to 4.d are provided in the Supplemental material).
Pooled effect size (SMD) results (Folic acid).

k = number of studies.
Removed as outliers: Ballestero-Fernández et al. 44

Forest plot of folic acid levels.
Pooled effect size of vitamin D in the CeD versus control groups
Fifteen studies were analyzed, including 655 patients with CeD and 14,717 controls. The pooled results revealed that the SMD of vitamin D in patients with CeD was −0.4011 (95% CI −0.8020 to −0.0001; P = 0.0499) compared with the controls (Table 5; Figure 6). Publication bias was not observed (Table 5.b in Supplemental material). The sensitivity test (right-skewness) for the estimated SMD was significant (Table 5.c in Supplemental material). (Detailed data for Tables 5.b to 5.c are provided in the Supplemental material).
Pooled effect size (SMD) results (Vitamin D).

k = number of studies.

Forest plot of vitamin D levels.
Pooled effect size of Zn in the CeD versus control groups
Eight studies were analyzed, including 343 patients with CeD and 14,250 controls. The pooled results of the meta-analysis revealed that the SMD of Zn in patients with CeD was −1.1398 (95% CI −2.0712 to −0.2084; P = 0.0242) compared with the controls (Table 6; Figure 7). There was publication bias (Table 6.b in Supplemental material). However, the sensitivity test (right-skewness) for the estimated SMD was significant (Table 6.c in Supplemental material). (Detailed data for Tables 6.b to 6.c are provided in the Supplemental material).
Pooled effect size (SMD) results (Zn).

k = number of studies.
Removed as outlier: Idris et al. 54

Forest plot of zinc levels.
Pooled effect size of MCV in the CeD versus control groups
Six studies comprising 134 patients with CeD and 806 controls were included in this meta-analysis. The pooled results of the meta-analysis revealed that the SMD of MCV in patients with CeD was −0.16 (95% CI−0.8 to 0.47; P >0.05) compared with the controls (Table 7; Figure 8). Publication bias was not observed (Table 7.b in Supplemental material). The sensitivity test (right skewness) for the estimated SMD was significant (Table 7.d in Supplemental material). (Detailed data for Tables 7.b to 7.d are provided in the Supplemental material).
Pooled effect size (SMD) results (MCV).

k = number of studies.

Forest plot of MCV levels.
Pooled effect size of copper in the CeD versus control groups
Six studies comprising 189 patients with CeD and 3396 controls. The pooled results revealed that the SMD of copper in patients with CeD was −0.6429 (95% CI −1.5264 to 0.2407; P > 0.05) compared with controls (Table 8; Figure 9). No publication biases were observed.
Pooled effect size (SMD) results (Copper).

k = number of studies.
Removed as outlier: Guerrieri et al. 55

Forest plot of copper levels.
Pooled effect size of vitamin B12 in the CeD versus control groups
Ten studies were conducted to assess the vitamin B12 levels, including 838 patients with CeD and 16,437 controls. The pooled results of meta-analysis revealed that the SMD of vitamin B12 in patients with CeD was 0.01 (95% CI −0.0121 to 0.15; P > 0.05) compared with controls (Table 9; Figure 10). No publication bias was observed (Table 9.b in Supplemental material). The estimated corrected true effect size was 0.1563 (95% CI −0.0205 to 0.3331; P = 0.0831) (Table 9.c in Supplemental material). (Detailed data for Tables 9.b to 9.c are provided in the Supplemental material).
Pooled effect size (SMD) results (Vitamin B12).


Forest plot of vitamin B12 levels.
Risk of bias
Risk of bias was calculated using STROBE. 56 Using this tool, the studies were assessed using a 22-point checklist and grouped into low, moderate, and high risks of bias. Studies with a score <50 were considered to be poor, 50–70 as fair, 70–85 as good, and ≥85 as excellent. Studies with a high risk of bias were excluded (Supplemental Table 1).
Subgroup analysis
A subgroup analysis was used to investigate the sources of heterogeneity in the meta-analyses. The included studies were separated into ≥2 subgroups and the pooled effect sizes observed in these subgroups were examined to determine whether they differed significantly from one subgroup to another. The results of this subgroup analysis revealed significant unexplained heterogeneity within each subgroup as well as smaller and/or unequal data points. Consequently, the validity of the effect estimate for each subgroup is questionable, implying that the subgroup analysis is unlikely to yield valuable results (results not shown).
Discussion
Results of the present meta-analysis revealed that the pooled global effect of hemoglobin, ferritin, iron, and MCV was reduced in patients with CeD compared with the control group (−0.6 (95% CI −0.8459 to −0.3382), P = 0.0003; −0.6358 (95% CI −0.8962 to −0.3755), P = 0.0002; −0.4 (95% CI −0.7385 to −0.0407), P = 0.0334; and −0.16 (95% CI −0.8 to 0.4), P > 0.05, respectively). Furthermore, iron deficiency anemia (IDA) is not unusual because it is one of the main manifestations of CeD and is the predominant abnormality in approximately 45% of individuals with subclinical forms. 57 Notably, Simon et al. 58 reported that IDA could be the sole presenting feature in 39% of patients with CeD, underscoring its significance in the clinical assessment of this disorder. Furthermore, patients with CeD presenting with anemia at diagnosis have more advanced disease and a slower dietary response than those without anemia. This observation was reported in an excellent review of the extraintestinal manifestations of CeD, highlighting that when anemia is the primary reason for presentation of the disease, patients exhibit higher anti-transglutaminase levels, lower serum cholesterol, and higher degrees of VA than those presenting with diarrhea alone. 59 Therefore, CeD should be considered as a possible cause of IDA in all patients. The underlying reason why some patients with CeD develop IDA while others do not remains poorly understood. However, it may be associated with deficiencies in specific regulatory proteins that are crucial for iron absorption at the enterocyte level, reflecting an imbalance between iron loss and absorption.59,60 Several disorders can affect the upper gastrointestinal tract, which is crucial for the absorption of dietary iron. 61 GFD is recognized as the primary intervention for managing mild cases of IDA in CeD patients. 62 However, the recovery of iron levels through GFD alone can be slow, particularly in severe cases. 63 To accelerate the restoration of iron stores, oral iron supplementation may be implemented, which is especially beneficial for patients with mild enteropathy or those with inconsistent adherence to the GFD. 59 In cases of advanced enteropathy, oral iron supplementation may lead to adverse effects, necessitating alternative strategies such as intravenous iron administration or methods to improve tolerability. 59 Therefore, while dietary measures, such as a high-iron diet, can complement therapy, they are insufficient as standalone treatments and cannot replace the essential role of iron supplementation in managing anemia in CeD. 59
CeD is a well-known cause of duodenal intraepithelial lymphocytosis, inflammation, and VA. It is mostly observed in the duodenum and upper jejunum. This may explain why folate deficiency has been reported in 8–85% of adult patients with CeD. 64 The disparity in prevalence may be explained, in part, by the technical problems of measuring “folate” and “folic acid” because the bioavailability of folic acid is twice that of folate. 65 Furthermore, patients with CeD exhibit megaloblastic anemia and neurological symptoms, and their chance of acquiring this deficiency is >5 times higher than that of healthy individuals. This was most likely caused by loss of villi in the proximal small intestine. As a result, the greater the degree of VA, the greater the folate insufficiency. 66 Furthermore, a GFD appears to improve or even normalize folic acid levels in those affected by CeD. 6 Our results are consistent with those of previous reports, given that the global pooled effect of folic acid in our study was −0.5446 (95% CI −0.9749 to −0.1142; P = 0.0187) in the CeD group compared with that in the control group.
The small intestine plays a critical role in Zn homeostasis. Zn deficiency in patients with CeD can be caused by an increased endogenous loss of this mineral rather than by abnormal Zn absorption. 67 This cumulative loss can occur through several mechanisms, including the formation of insoluble Zn complexes with fat and phosphate, exudation of Zn protein complexes into the intestinal lumen, massive loss of intestinal secretions, and impaired Zn absorption resulting from damage to the intestinal epithelial cell membrane. 68 Some CeD symptoms (e.g. anorexia and slow growth rate) may be linked to Zn deficiency. In recent years, Zn has emerged as a critical micronutrient for maintaining the integrity of the intestinal mucosa, immunity, and growth. Moreover, patients with CeD have been shown to have lower plasma Zn concentrations.68–73 Similar results were found in our meta-analysis, in which the pooled effect of Zn was −1.1398 (95% CI −2.0712 to −0.2084; P = 0.0242) in the CeD group compared with that in the control group.
CeD is linked to a wide range of endocrine concerns,74,75 the most prevalent of which are low bone mineral density (BMD), osteopenia, and osteoporosis, 76 resulting in a high risk for bone fracture(s). Therefore, BMD measurements in adult patients are recommended. 77 Although BMD was not considered in this meta-analysis, we found that the pooled effect of vitamin D in the CeD group was −0.4011 (95% CI −0.8020 to −0.0001; P = 0.0499) compared with that in the control group. In light of these results, the P-value analysis and p-curve results (P-Full and P-half ≤ 0.05), revealed that the pooled effect is not completely spurious; it is not merely a “mirage” produced by selective reporting. 35 Our results suggest that vitamin D levels are low in patients with CeD. In addition to repairing and protecting the skeletal system during calcium metabolism, other roles of vitamin D have recently been reported. Vitamin D plays an important modulatory role in inflammation, immunological processes, and mucosal barrier control. In this context, vitamin D can cause immunological disorders and the role of vitamin D in immune regulation may be a major element in the initiation of CeD. 78 Nonetheless, the results of studies investigating vitamin D levels and screening for vitamin D deficiency in patients are conflicting. 79 Most vitamin D investigations on adult CeD have demonstrated that 25(OH) D insufficiency improves with a GFD, regardless of supplementation. 80 The active form of 1,25 (OH) vitamin D was within normal range at the time of CeD diagnosis. It has been suggested that a GFD can boost vitamin D levels without the need for supplementation. 81 However, our results provide evidence that patients with CeD should undergo nutritional assessment and receive nutritional counseling, as well as a strict GFD, and that dietary supplements should be recommended for those with severe deficiencies.
True deficits are difficult to demonstrate due to the complicated interplay between the elements. For example, folate requires vitamin B12 activation; therefore, low intracellular folate levels may result from vitamin B12 deficiency. 82 Vitamin B12 deficiency appears to be rare in patients with CeD because it binds to intrinsic factors in the duodenum and the complex is absorbed in the terminal ileum, which is supposed to be protected from harm in CeD. Although the precise etiology of vitamin B12 deficiency in CeD remains unclear, potential contributing factors, such as reduced gastric acid production, small intestinal bacterial overgrowth (SIBO), autoimmune gastritis, and subtle dysfunction of the distal small intestine, have been suggested. 67 Moreover, Dahele and Gosh, 83 reported that 41% of adults with untreated CeD exhibited vitamin B12 deficiency despite the absence of intrinsic factor antibodies in all patients, with only one-third experiencing concurrent folate deficiency. We found no evidence of compromised vitamin B12 status (the pooled effect of vitamin B12 in the CD group was 0.01 (95% CI −0.12 to 0.15); P > 0.05). This could be due to higher dietary intake. Some patients with CeD have been reported to use vitamin and mineral supplements (vitamin B-complex) before being diagnosed with CeD. 84
Primary dietary copper deficit is uncommon and is mostly caused by malabsorption syndrome. In our study, the pooled effect of copper in the CeD group was −0.6429 (95% CI −1.5264 to 0.2407)]. Although our results were not statistically significant, the trend was toward copper deficiency, which is consistent with many previous studies.54,71,85,86 An Iranian study reported in 2013 that the mean levels of Zn in patients with CeD were significantly lower than those in control group (75.97 ± 12 vs 92.83 ± 18, P < 0.0001). 68 Similarly, Singhal et al. noted that serum Zn levels in patients with newly diagnosed CeD were significantly reduced (0.64 ± 0.34 mg/mL vs 0.94 ± 0.14 mg/mL in controls (95% CI −0.44 to −1.4)). 73 Similarly, a recent study by Adam et al. showed that Zn levels were decreased in 59.4% of patients with CeD compared with 33.2% in controls. 70
Micronutrient deficiencies observed in patients with CeD can be attributed to several factors related to disease pathophysiology. The CeD pathway is characterized by alterations in the small intestine, including intraepithelial lymphocytosis, crypt hyperplasia, and VA, which reduce nutrient absorption. 87 Moreover, inflammation and small intestinal mucosal damage lead to loss of absorptive surfaces and nutrient malabsorption. 88 Refined flours used in GFDs often lack fortification, potentially contributing to nutritional deficiencies in this population.89,90 In addition, GFDs commonly followed by patients with CeD are characterized by reduced intake of cereals, fruits, and vegetables, along with increased consumption of meat and meat-derived products. 91 Whole-grain barley, rye, and wheat products are typically replaced by specialized gluten-free alternatives, which have been shown to possess lower nutritional value compared with their gluten-containing counterparts. 91 These gluten-free products are often associated with higher levels of fats, particularly saturated and trans-fats, as well as refined sugars, phosphorus, and salt, which can reduce the intake of fibers, complex carbohydrates, and proteins. 91 Furthermore, the inadequacy of dietary habits specific to this group may exacerbate the issue.92,93 In addition, the low demand for nutritional counseling from registered dietitians may foster insufficient food intake, particularly in rural areas. Despite being straightforward, GFD implementation poses significant challenges for patients and their families, 94 one of which is the risk for cross-contamination, often leading to unintentional gluten transgression. These inadvertent exposures can perpetuate VA and contribute to ongoing nutritional deficiencies in individuals with CeD even when they adhere to a strict GFD. 95 Registered dietitians play a critical role in guiding patients with CeD by adopting a GFD that is not only healthy but also interesting and practical, helping to mitigate these challenges. 96 While it is acknowledged that a GFD entails dietary restrictions, patients who receive nutritional counseling from a registered dietitian can achieve a well-balanced and healthy diet. The dietary recommendations for a healthy GFD should align closely with those of a regular healthy diet, emphasizing nutritious and safe alternatives to cereal-based foods while avoiding excessive consumption of highly processed products. Such a diet should prioritize the intake of fresh, unprocessed, and naturally gluten-free foods, including a variety of fruits, vegetables, and proteins, preferably from plant sources such as legumes, whole grains, pseudocereals, tubers, and nuts. 91 As such, a tailored diet could be beneficial in restoring a balanced gut microbiota. 67 Our study has the merit of using a standardized meta-analytical methodology (with random-effects analyses) to assess the impact of CeD on different micronutrient categories. However, there were several limitations, including the lack of sample size calculation, the high level of heterogeneity observed among the included studies, and the disproportionate number of studies addressing the nutrients analyzed.
Conclusion
The present analysis revealed substantial differences in micronutrient levels between patients with CeD and controls. Decreases in hemoglobin, ferritin, iron, folic acid, Zn, and vitamin D levels highlight the multidimensional characteristics of nutritional deficits in CeD. These findings highlight the crucial role of a thorough nutritional evaluation and intervention techniques in CeD care to address a wide range of micronutrient deficits. Thus, it is critical to use a multidisciplinary strategy that includes registered dietitian counseling, supplementation when needed, and continued monitoring to reduce the negative health effects of micronutrient deficiencies in patients with CeD. Furthermore, additional studies should focus on identifying the underlying processes that contribute to micronutrient deficits in patients with CeD, as well as investigating novel techniques to improve nutrient absorption and overall nutritional status in this susceptible group.
Supplemental Material
sj-docx-1-iji-10.1177_03946320241313426 – Supplemental material for Micronutrient deficiencies in patients with celiac disease: A systematic review and meta-analysis
Supplemental material, sj-docx-1-iji-10.1177_03946320241313426 for Micronutrient deficiencies in patients with celiac disease: A systematic review and meta-analysis by Saad Lamjadli, Ider Oujamaa, Ikram Souli, Fatima ezzohra Eddehbi, Nadia Lakhouaja, Bouchra M’raouni, Abdelmouine Salami, Morad Guennouni, Moulay Yassine Belghali, Raja Hazime and Brahim Admou in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-html-2-iji-10.1177_03946320241313426 – Supplemental material for Micronutrient deficiencies in patients with celiac disease: A systematic review and meta-analysis
Supplemental material, sj-html-2-iji-10.1177_03946320241313426 for Micronutrient deficiencies in patients with celiac disease: A systematic review and meta-analysis by Saad Lamjadli, Ider Oujamaa, Ikram Souli, Fatima ezzohra Eddehbi, Nadia Lakhouaja, Bouchra M’raouni, Abdelmouine Salami, Morad Guennouni, Moulay Yassine Belghali, Raja Hazime and Brahim Admou in International Journal of Immunopathology and Pharmacology
Footnotes
Appendix
Articles involving Zinc included in the meta-analysis.

| ID | YEAR | COUNTRY | StudyType | AGE | Nc | Nd | ZINCc | ZINCd | SdZINCc | SdZINCd | AdherenceScore |
|---|---|---|---|---|---|---|---|---|---|---|---|
| BoteroLopez et al. 99 | 2020 | Chile | Cross-sectional | Adulte | 36 | 73 | 90 | 82.5 | 16.25 | 20 | Excellent |
| UnalpArida et al. 64 | 2022 | USA | Cross-sectional | Adulte | 14000 | 26 | 82.2 | 75.1 | 34.33 | 10.97 | Excellent |
| Rawal et al. 122 | 2010 | India | Prospective | Children | 48 | 48 | 74.9 | 71.9 | 29.2 | 19.3 | Good |
| Ince et al. 86 | 2007 | Turkey | Cross-sectional | Adulte | 35 | 32 | 101 | 70 | 20 | 14 | Good |
| Naveh et al. 123 | 1983 | Israel | Prospective | Children | 31 | 34 | 100 | 62 | 15 | 11 | Fair |
| Fathi et al. 68 | 2013 | Tehran | Case-control | Adulte | 30 | 30 | 92.83 | 75.97 | 18 | 12 | Good |
| Karnani et al. 85 | 2022 | India | Case-control | Children | 30 | 60 | 102.13 | 16.52 | 80.63 | 21.18 | Good |
| Idris et al. 54 | 2019 | Sudan | Case-control | Adulte | 40 | 40 | 1 | 0.285 | 0.245 | 0.1776 | Excellent |
Acknowledgements
We gratefully acknowledge Professor Daniel S. Quintana for his significant contributions to this study. His insightful feedback and dedication to rigorous scientific inquiry greatly enhanced the quality of this research.
Author contributions
Conceptualization: Saad Lamjadli and Ikram Souli; Methodology: Ider Oujamaa, Morad Guennouni, Moulay Yassine Belghali, Raja Hazime and Brahim Admou; Software: Saad Lamjadli and Morad Guennouni; Data curation: Ikram Souli, Fatima ezzohra Eddehbi, Nadia Lakhouaja, Bouchra M’raouni and Abdelmouine Salami; Writing—Original Draft, Saad Lamjadli, Ider Oujamaa, Ikram Souli; Validation: Ider Oujamaa, Moulay Yassine Belghali, Raja Hazime and Brahim Admou; Writing—Review and Editing, all authors; Supervision: Brahim Admou.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Not applicable.
Informed consent
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
