Abstract
Background: One of the most common long-term neurological disorders affecting children is epilepsy. Even with effective antiseizure medications, one-third of epileptic patients develop drug-resistant epilepsy (DRE). Numerous treatments have been offered to these DRE patients, though with varying degrees of effectiveness. Objectives: This study aimed to evaluate the effectiveness of probiotics in improving the quality of life (QoL) and lowering the severity and frequency of epileptic episodes in DRE patients. As well as to assess the anti-inflammatory effects of probiotics. Methods: DRE patients were daily supplemented with one probiotic for 4 months. During these 4 months, patients continued their routine anti-epileptics with no change in the doses. Before and following the 4-month trial, patients had their QoL evaluated using the validated Arabic version of QoL in epilepsy-31 inventory (QoLIE-31) questionnaire, an electroencephalogram (EEG) examination, and serum soluble CD14 (sCD14) evaluation by ELISA. Results: Of the 21 DRE patients who completed the study, 42.9% achieved the therapeutic goal, which was a ≥50% reduction in seizures. After probiotic, there was a significant increase in time elapsed since the last seizure (
Introduction
Epilepsy is the most common chronic neurologic disorder affecting children. 1 Over 50 million individuals worldwide have been diagnosed with epilepsy, of whom nearly 80% live in developing countries, and over five million individuals experience epilepsy for the first time each year. 2 Drug therapy using antiseizure drugs is the cornerstone of epilepsy treatment. Nevertheless, around one-third acquire drug-resistant epilepsy (DRE) as a result of having uncontrolled seizures, even with the use of suitable antiseizure drugs. 3 Duration to assess the reaction to an antiepileptic medication varies across patients. If a patient has multiple seizures each week, a few weeks of follow-up will be sufficient to determine medication failure if seizures continue. 4 Despite developing several novel antiseizure medications during the past 30 years, the proportion of individuals with DRE has remained unchanged. 5 These DRE patients have been offered a variety of treatments, all with limited degrees of success, such as surgery, neurostimulator implantation, and ketogenic diet. 6
Most bacterial flora found in the gastrointestinal tract (GIT) make up the gut microbiota. Recent research reveals that the gut flora and the central nervous system (CNS) communicate via various signaling channels and connecting networks, collectively known as the “microbiota-gut-brain axis.”. 7 According to recent research, gut dysbiosis is a likely factor in the etiology and predisposition to epilepsy. Thus, reestablishing the gut flora with a ketogenic diet and probiotic intervention might benefit DRE patients. 8 Probiotics are viable microorganisms that provide the host with health benefits when consumed in sufficient amounts. 9
Recent investigations on human and experimental animal models have also demonstrated a connection between neuro-inflammation and epileptogenesis10,11 and that DRE patients had significantly high percentages of CD14+ monocytes positive for proinflammatory cytokines like interleukin (IL)-1β, IL-6, and tumor necrosis factor-alpha (TNF-α). These percentages were positively correlated with seizure frequency. 12 On the other hand, probiotics were found to prevent inflammation by inhibiting various signaling pathways, including CD14, thereby reducing the production of proinflammatory cytokines. 13
The objective of this study was to assess the efficacy of probiotics in reducing the frequency and severity of epileptic seizures in DRE patients while also enhancing their quality of life (QoL). By comparing the blood levels of soluble CD14 (sCD14) before and after the therapeutic intervention, the current study evaluated the probiotics’ ability to reduce inflammation as well.
Methodology
Ethical considerations
The Ain Shams University Faculty of Medicine’s Ethics Committee approved the study’s protocol (# FMASU MS 690/2022). Prior to enrollment, all participants or their legal guardians provided written informed consent. All participants had the freedom to leave the study at any time without explanation or consequences, and the confidentiality of all data was guaranteed.
Study participants
This prospective pilot clinical trial was conducted on 35 children with DRE (age:1-18 years) recruited from the Pediatric Neurology Department, Children’s Hospital, Ain Shams University. DRE diagnosis followed the definition of the International League Against Epilepsy (ILAE), which is the inability to achieve sustained seizure freedom, freedom from all types of seizures, including auras, for 12 months, or three times the inter-seizure interval before treatment started, despite using well-tolerated, carefully selected antiepileptic drugs (whether as monotherapies or in combination).14,15
Which is the failure of adequate trials of two tolerated, appropriately chosen and used antiepileptic drug schedules (whether as mono-therapies or in combination) to achieve sustained seizure freedom, freedom from all types of seizures including auras for 12 months, or 3 times the inter-seizure interval before treatment started.14,15
The selected DRE patients should have no change in their anti-epileptic drug type or dosage during the 4 months of the study, or else the patient was excluded. Other exclusion criteria included patients on a ketogenic diet, patients already on probiotic supplements, patients on chronic antibiotic therapy, or those who had antibiotic therapy within the previous 6 months from the baseline of the study, and patients with GIT problems, especially diarrhea.
Participants were classified into two groups based on their epilepsy type: 1) the global developmental delay (GDD) group, which included participants who had a significant delay in two or more of the following domains: gross/fine motor, speech and language, cognitive, social/personal conduct, and activities of daily living, 16 and 2) the genetic epilepsy group, the idiopathic group, which was characterized by no apparent neuroanatomical or neuropathologic defect and mainly was of genetic or suspected genetic origin. 17 Participants were also subdivided according to the effectiveness of the probiotic in reducing the number of seizures by 50% or more and improving seizure severity (therapeutic goal). 18
Medical history and clinical assessment
All the included DRE patients were subjected to medical history taking, including family history of similar disease, natal and postnatal history, developmental history, dietetic history, and antiepileptic drug history including their side effects and any other supplements taken. Participants were also subjected to clinical examination, including vital signs, anthropometric measurements, and chest, cardiac, abdominal, and neurological examinations. Immediately before and after the 4-months trial, the type, frequency, duration, and time interval of seizures, as well as time elapsed since the last seizure, were assessed by the physician, and the severity of seizures was rated according to the Chalfont Seizure Severity Scale. 19
Therapeutic intervention
The included DRE patients were daily supplemented with one probiotic (spores of antibiotic-resistant
Questionnaire for assessment of QoL
Using the validated Arabic version of QoL in epilepsy-31 inventory (QoLIE-31) scores questionnaire, the QoL of the included DRE patients was assessed before and after therapeutic intervention. QoLIE-31 measures QoL in epileptic patients through seven subscales. Every scale was transformed into a 0–100 scale, with a value distributed equally for every question (the higher the impairment, the lower the score). 20
EEG examination
A 30-min EEG was done for each patient before and after therapeutic intervention using Nicolet REF 515-019000 rev 06, CareFusion, WI 53562, USA. Most patients were recorded during wakefulness. 16 patients were recorded during non-induced sleep, and only five patients used melatonin for sleep induction.
Serum sCD14 evaluation
Three milliliters of whole blood were collected aseptically from each participant before and after therapeutic intervention into a gel vacutainer tube. Sera were separated by centrifugation at 3000 r/min for 20 min after complete blood clotting at room temperature. Sera were used to assess levels of sCD14 using the human ELISA kit (Li StarFish S.r.l., Cernusco Sul Naviglio, Italy, Cat. No. BTB-E0317Hu). The kit detection range is 0.03 mg/L - 9 mg/L and sensitivity is 0.013 mg/L.
Data management and analysis
Version 23 of the Statistical Package for Social Science (IBM SPSS, USA) was used to analyze the data. Whereas the qualitative variables were shown as percentages and numbers, the quantitative non-parametric data were shown as a medians and inter-quartile ranges (IQR). The Wilcoxon Rank test (for quantitative non-parametric data) and the Chi-square test (for qualitative data) were used for the comparisons.
21
When the
Results
Demographic data and characteristics of the studied drug resistant epilepsy patients (

Abbreviations: GDD: Global developmental delay.
Syndromes of the included genetic epilepsy patients included generalized tonic-clonic epilepsy (5/8, 62.5%), juvenile absence epilepsy (1/8, 12.5%), childhood absence epilepsy (1/8, 12.5%), and juvenile myoclonic epilepsy (1/8, 12.5%). Figure 1 Disruption of syndromes among the included genetic epilepsy patients (
After probiotic therapy, the number of seizures was reduced by ≥ 50% (the therapeutic goal) in 42.9% of the patients; 6/21 patients (28.6%) had 50%–70% reduction and 3/21 patients (14.3%) had 70%–99% reduction. On the other hand, 7/21 patients (33.3%) showed no improvement at all. And only one patient (4.8%) out of the 21 included had 1%–30% reduction and four patients (19.0%) had 30%–50% reduction. Figure 2 Seizure reduction after the 4-month therapeutic trial (therapeutic goal: ≥50% seizure reduction) (
Comparison of drug resistant epilepsy patients’ (
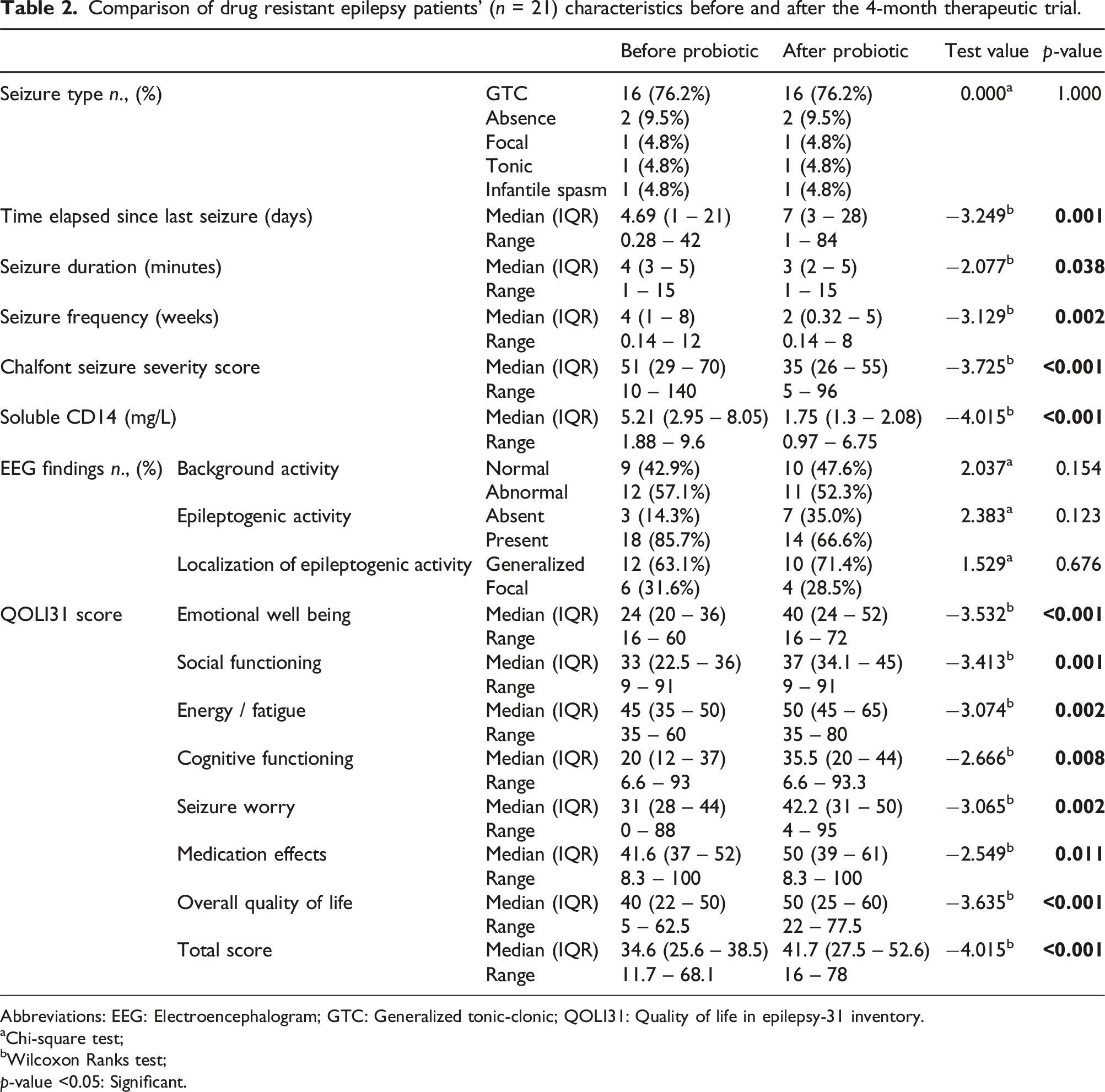
Abbreviations: EEG: Electroencephalogram; GTC: Generalized tonic-clonic; QOLI31: Quality of life in epilepsy-31 inventory.
aChi-square test;
bWilcoxon Ranks test;
Comparison of drug resistant epilepsy patients’ (

Abbreviations: EEG: Electroencephalogram; GTC: Generalized tonic-clonic; QOLI31: Quality of life in epilepsy-31 inventory.
aChi-square test;
bWilcoxon Ranks test;
Comparison of characteristics of the included drug resistant epilepsy patients (

Abbreviations: GTC: Generalized tonic-clonic; QOLI31: Quality of life in epilepsy-31 inventory.
aChi-square test;
bWilcoxon Ranks test;
Discussion
Characteristics of the included DRE patients
Only 21 of the 35 DRE children enrolled in the study completed it, and all the findings were based on these 21 DRE children. Their median (IQR) age at the time of the study was 11 (7.5 – 13) years and ranged from 2.5 to 15 years. Their seizure onset age ranged from 6 months to 12 years, with a median (IQR) of 7 (6–9) years. A prospective cohort study by Kwong et al., 2003, compared children with drug-sensitive epilepsy to those with DRE; they discovered that the age of seizure onset was a significant predictor of intractability, meaning that the earlier patients experience seizures, the greater the likelihood that they will become drug-resistant. 22 Similarly, Ramos-Lizana et al.,2012, found that 62% of their DRE cases were younger than 4 years old. 23 In addition, Berg et al., 2001, found that the age of seizure onset of their included participants, which was between 5 and 9 years, was associated with a lower risk for drug resistance. 24
In the current study, most participants were females (71.4%, 15/21), and the rest were males (28.4%, 6/21). Shakeshaft et al., 2022, in a cross-sectional study, investigated the effect of gender on the prognosis of juvenile myoclonic epilepsy and found that the female-to-male ratio was 1.8:1 and that 59% of the females included in the study and 50% of the males reported triggered seizures. 25 Different mechanisms have been postulated to explain gender variations in age-independent seizure susceptibility and epilepsies, but not in DRE. Steroid hormones, cytochrome P450 activity, neurotransmitter systems, and gender-associated neuronal networks in the brain are a few of these variables. 26
Since 2010, the International League Against Epilepsy (ILAE) definition has raised interest and research on DRE. Still, the present guidelines refer to all epilepsies and do not distinguish between types. 15 Our DRE patients were subdivided into two groups based on their diagnosis: 13 patients (61.9%) had GDD, and only eight patients (38.1%) had genetic epilepsy. A similar low prevalence of DRE among genetic epilepsy patients was revealed by Cerulli Irelli et al., 2020, who performed a retrospective study on 199 genetic epilepsy patients and found that only 15% of them had DRE. They also reported that febrile seizures, generalized paroxysmal fast activity EEG patterns, and valproate (VPA) resistance were significantly associated with the risk of DRE among them. Additionally, they found that juvenile myoclonic epilepsy was the sole epileptic syndrome linked to chronic resistance to treatment. 27
Similarly, in a study by Ramos-Lizana et al., 2012, 17% of the included patients had DRE according to the ILAE criteria. Of them, 73% had an associated developmental delay or motor deficit, which agrees with our study in which most patients were of the GDD type. 23
Probiotic effectiveness in seizure reduction
One of the first prospective studies on epileptic children was conducted by Berg et al., 2001; they found that DRE developed more among those with daily seizures compared to those with lower seizure frequency (13.9 vs 6.8%). 24 This result corroborates the current study’s finding that the included DRE patients had a significantly high seizure frequency with a median (IQR) of 4 (1 – 8) weeks before the start of the probiotic supplementation.
Additionally, the current clinical trial demonstrated that probiotics can be a helpful therapeutic adjunct in the management of DRE. A reduction in the number of seizures of ≥50% is the defined therapeutic target. 28 42.9% of the patients in our study fulfilled this goal. Similarly, Gómez-Eguílaz et al., 2018, conducted a pilot, single-center, prospective study on 45 DRE patients who received probiotics as a supplementary agent for 4 months. They found that 28.9% of their participants achieved the therapeutic goal. 18 The gut microbiota can affect brain inflammation, the excitatory-inhibitory balance, and the bioavailability of anti-seizure drugs. This makes the gut microbiome a therapeutic target for the treatment of refractory epilepsy and explains why probiotics are beneficial in reducing seizures. 29
In the current study, the probiotic improved seizure frequency (
In addition, the current study revealed that the QoL differed among DRE patients when they were subdivided according to their diagnosis. QoL was higher in the genetic group than in the GDD group. This finding might be because most of the genetic epilepsy patients had a higher starting point than their counterparts in their developmental history and other aspects.
Probiotics and inflammation
Previous studies provided evidence that inflammation plays a role in the pathophysiology of epilepsy, and the frequency and intensity of seizures may be correlated with the levels of proinflammatory cytokines. 18
We employed serum sCD14, a marker of monocyte activation, as an inflammatory marker to investigate the possible reason for the reduction in epileptic seizures following probiotic therapy. We discovered that sCD14 levels drastically decreased (
Furthermore, Matei et al., 2021, studied the effect of probiotics on serum endotoxin and sCD14 in dogs with intestinal dysbiosis. They assessed their levels before initiating probiotic treatment and after 30 days of therapy and found that they significantly dropped after therapy. 32 This discovery supports the anti-inflammatory properties of probiotics. It could be explained by the several, mostly contact-dependent ways that probiotics affect the expression of immune-related genes, the activity of inflammatory pathways, and the levels of immunological markers. 9
Probiotics and EEG findings
The current study demonstrated no statistically significant improvement in the EEG epileptogenic activity nor background activity among patients pre- and post-probiotic intake. On the other hand, Aygun et al., 2022, conducted a study to evaluate the effect of probiotics on pentylenetetrazole (PTZ)-induced seizures in Wistar albino rats. They followed them up with EEG and other parameters and found that probiotic treatment significantly reduced the spike frequency in EEG. 33 The difference in the initial conditions between our study and theirs may account for the discrepancy, as their research involved rats rather than humans, even though they confirmed our findings that probiotics can lessen the severity of seizures and have neuroprotective effects by lowering oxidative stress and neuroinflammation.
Probiotics and QoL
The current study also discovered that probiotic use considerably enhanced the QoL of the included subjects in every subscale of the QOLIE31 questionnaire. This improvement can be explained by the fact that probiotics reduced the functional and social burden, improved seizure intensity and frequency, and enabled the study group to experience events they might not have been able to before using the probiotic. Similarly, Gómez-Eguílaz et al., 2018, found that the QoL of their DRE patients, according to the QOLIE-10 questionnaire, improved after receiving probiotics. 18 Also, Wang et al., 2022, found that temporal lobe epilepsy patients who received probiotics experienced an improvement in their QoL according to the QOLIE-89 and in their cognitive function as measured by the Wechsler Adult Intelligence Scale-Fourth Edition (WALS-IV). 31
Study limitations
Even though our patients came from various cities around Egypt, the current study was a single-center design. The tiny participant pool of the present study is seen as a weakness because it restricts the generalization of the data collected. Furthermore, the absence of a control group of children and adolescents who did not receive the probiotic is a study limitation. Moreover, the questionnaire used for QoL assessment had some questions limited to the study’s duration of the last 4 weeks. These conditions can change over time and do not necessarily reflect the effect of the probiotic per se. The questionnaire also does not consider the variable perspectives of patients and their guardians regarding their QoL in general, irrespective of probiotics. Additional multicenter prospective studies that continuously monitor seizures throughout the study, not just at the beginning and the end, are needed to determine when the claimed probiotic effect on seizures should occur. It is also advised to screen stool samples for assessment of the microbiome before starting the treatment and the changes caused by the treatment.
Conclusion
The present study revealed that probiotics could be a potential adjuvant treatment for DRE. They can decrease the frequency and severity of seizures, reduce the associated inflammation, and improve the QoL of DRE patients. Since this is a pilot study and there are deficient data in this area, more studies are needed to evaluate probiotics’ applicability, safety, and mechanism of action in DRE patients.
Footnotes
Authors contribution
The study design was done by all authors. O.S.E shared in clinical evaluation and sample collection and wrote the manuscript. O.F.E did the investigations and reviewed the manuscript. I.A.A.E. shared in clinical evaluation and reviewed the manuscript. B.A.N shared in clinical evaluation and reviewed the manuscript. S. I. T. did the investigations and wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Informed consent
Written informed consent was obtained from legally authorized representatives before the study.
Data availability statement
All the data needed to support the findings will be presented upon request.
