Abstract
This study aimed to investigate whether the beneficial effects of PCA on chondrocyte senescence are mediated through the regulation of mitophagy. Chondrocyte senescence plays a significant role in the development and progression of knee osteoarthritis (OA). The compound protocatechuic aldehyde (PCA), which is abundant in the roots of Salvia miltiorrhiza, has been reported to have antioxidant properties and the ability to protect against cellular senescence. To achieve this goal, a destabilization of the medial meniscus (DMM)-induced mouse OA model and a lipopolysaccharide (LPS)-induced chondrocyte senescence model were used, in combination with PINK1 gene knockdown or overexpression. After treatment with PCA, cellular senescence was assessed using Senescence-Associated β-Galactosidase (SA-β-Gal) staining, DNA damage was evaluated using Hosphorylation of the Ser-139 (γH2AX) staining, reactive oxygen species (ROS) levels were measured using Dichlorodihydrofluorescein diacetate (DCFH-DA) staining, mitochondrial membrane potential was determined using a 5,5',6,6'-TETRACHLORO-1,1',3,3'-*. TETRAETHYBENZIMIDA (JC-1) kit, and mitochondrial autophagy was examined using Mitophagy staining. Western blot analysis was also performed to detect changes in senescence-related proteins, PINK1/Parkin pathway proteins, and mitophagy-related proteins. Our results demonstrated that PCA effectively reduced chondrocyte senescence, increased the mitochondrial membrane potential, facilitated mitochondrial autophagy, and upregulated the PINK1/Parkin pathway. Furthermore, silencing PINK1 weakened the protective effects of PCA, whereas PINK1 overexpression enhanced the effects of PCA on LPS-induced chondrocytes. PCA attenuates chondrocyte senescence by regulating PINK1/Parkin-mediated mitochondrial autophagy, ultimately reducing cartilage degeneration.

Keywords
Introduction
Knee osteoarthritis (OA) is a degenerative disease primarily associated with aging. It is characterized by debilitating symptoms, including joint pain, swelling, and stiffness. 1 In the elderly population, knee OA is a leading cause of chronic disability, and its prevalence is expected to double by 2030.2,3 The degeneration of articular cartilage in the knee joint is a hallmark feature of knee OA and serves as a critical indicator of disease progression. 4 Notably, the process of cellular chondrocyte senescence, characterized by irreversible cell cycle arrest, is of utmost importance in initiating and perpetuating cartilage degeneration. 5 Moreover, recent studies have presented compelling evidence revealing that targeted elimination of senescent cells can effectively alleviate the progression of posttraumatic OA. 6 Conversely, intriguing findings have demonstrated that intra-articular injection of senescent cells induces OA in mice. 7 Activation of the p53-P21-RB and p16 INK4A-RB signaling pathways abolishes the cell cycle, causing cells to stop dividing and enter a senescent state. 8 These significant discoveries have sparked substantial interest in revealing the molecular mechanisms underlying chondrocyte senescence and exploring potential therapeutic interventions. 9
Over the years, numerous researchers have devoted efforts to discovering the intricate mechanisms underlying cellular senescence. One widely accepted theory is the mitochondrial damage theory, which was initially proposed by Harman in 1972. 10 Since then, subsequent studies 11–13 have provided compelling evidence that supports the involvement of mitochondrial damage in the accumulation of reactive oxygen species (ROS) and the impairment of ATP synthesis, ultimately leading to cellular senescence. Interestingly, cells have evolved a mechanism known as mitophagy to selectively eliminate damaged mitochondria and maintain mitochondrial quality and stability. 14 However, with aging, the efficiency of mitophagy gradually diminishes, resulting in the build-up of damaged mitochondria. This accumulation further exacerbates mitochondrial instability, increases the levels of reactive oxygen species (ROS), impairs ATP synthesis, and ultimately accelerates cellular senescence. 15 Activation of the PINK1-Parkin-mediated mitochondrial autophagy pathway can cause mitochondrial outer membrane proteins to release “eat me” signals, which are recognized by the autophagy interface protein P62 and initiate mitochondrial autophagy. 16
Protocatechuic aldehyde (PCA), a natural phenolic acid compound, is found at high levels in the roots of the Chinese herbal medicine Salvia miltiorrhiza.
17
Numerous studies have demonstrated the diverse biological functions of PCA, including its anti-inflammatory,
18
antioxidant,
19
and antiapoptotic.20,21 effects. Notably, PCA has been shown to alleviate
Methods and materials
Mouse generation and drug treatment
We purchased 30 young male C57BL mice (6 weeks old, 25 ± 2 g) from Jiangsu Huachuang Xinnuo Pharmaceutical Technology Co. (Taizhou, Jiangsu Province, China) for purchase. All experiments (including those used for OA assessment) will be performed using SPSS software to randomly assign 30 mice to control, KOA and PCA treatment groups, 10 mice in each group, five for pathological histological observations and five for molecular biological assays. For drug treatment, mice were orally administered PCA at a dose of 20 mg/kg/d after DMM surgery. All mice were housed under pathogen-free conditions, with a maximum of 5 mice per cage..PCA (≥98%) was purchased from Shanghai Yuanye Biological Co. The structural formula of protocatechuic aldehyde is shown in Figure 1. Structural formula of protocatechuic aldehyde (PCA).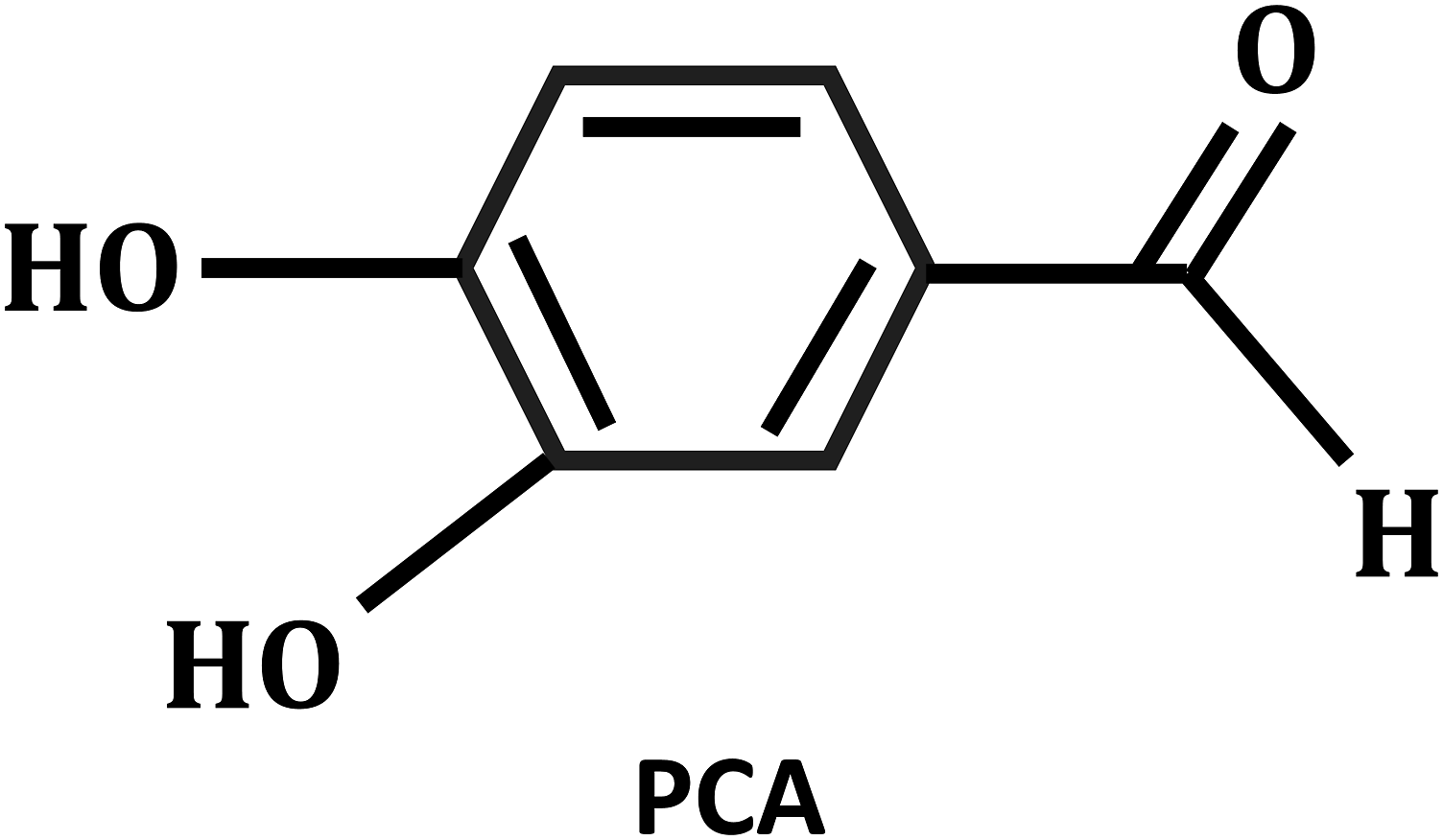
The experimental animals were anesthetized with Nembutal, and all modeling procedures were conducted on the same day by four researchers with a background in orthopedic surgery. Briefly, the DMM model was established by transecting the medial meniscotibial ligament in both knees, as previously described.
25
This animal experiment was reviewed by the Ethics Committee of Nanjing University of Traditional Chinese Medicine (ethics no. 202104A017). The abbreviated flowchart for the animal experiments is shown in Figure 2. Abbreviated flowchart for animal experiments.
Micro-CT scanning
The samples were scanned using a micro-CT system (SKYCAN, Germany) with the following scanning parameters: a scanning voltage of 80 kV, a scanning current of 160 μA, and a scanning layer thickness of 20 μm. The weight-bearing area under the cartilage of the tibial plateau was selected as the region of interest. Subsequently, the system’s software was used to set the parameters for bone tissue detection. Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Histological observation
After the mice were euthanized, cartilage tissues from the knee joint were excised and fixed in 10% neutral formalin. The tissues were then soaked in EDTA, embedded in paraffin, and cut into slices. Subsequently, gross morphologic lesions on the tibial plateau were visualized using HE and Safranin-O staining methods. The OARSI score was used to assess knee pathology. Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Cell isolation and culture
Briefly, the articular cartilage was aseptically dissected and separated from the tibial plateau. The cartilage pieces were then washed with phosphate-buffered saline (PBS) and cut into small fragments. These fragments were subsequently digested in 0.2% collagenase type II for 6 h. After digestion was terminated and the suspension was filtered, the cells were collected by centrifugation at 1500 r/min for 6 min. The chondrocytes were cultured in DMEM supplemented with 10% fetal bovine serum (Gibco, MA, USA) and 1% penicillin/streptomycin (Invitrogen, CA, USA) at 37°C in a 5% CO2 environment.
PINK1 siRNA transfection
To inhibit PINK1 expression in mouse chondrocytes, commercially available PINK1 siRNA and a vehicle control siRNA were used. Chondrocytes were transfected with siRNAs using Lipofectamine 2000 (Invitrogen, CA, USA) following the manufacturer’s instructions. The siRNA was diluted in the transfection reagent and culture medium, and the cells were incubated with 20 pmol of siRNA for 6 h.
PINK1 adenovirus transfection
The MOI was determined through a preliminary experiment to determine the optimal concentration for efficient adenovirus infection. Then, 100 μL of the virus solution was added to each well of a six-well plate and incubated with 900 μL of medium for 72 h. Afterward, the medium was changed to stabilize the cells.
Cytotoxicity assay
CCK-8 assays (APExBIO, USA) were used to evaluate the cytotoxicity of PCA toward cells. Mouse chondrocytes were seeded in a 96-well plate in the presence of PCA. CCK-8 reagent (10 μL) was added to the supernatant of each well, which was then incubated for 1 h. The absorbance was then measured at 452 nm using a PerkinElmer EnVision microplate reader. The ratio of the absorbance in the experimental group to that in the control group served as an indicator of cell viability. Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Measurement of mitochondrial membrane potential
Changes in the mitochondrial membrane potential were analyzed using a mitochondrial membrane potential assay kit with JC-1 (Beyotime, China). Cells were seeded at a density of 1 × 106 cells per well in 6-well plates and incubated with medium containing JC-1 working solution at 37°C for 20 min. Fluorescence images were captured using a fluorescence microscope (Leica DMI8, Germany). Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Flow cytometry
The cells were incubated in an incubator at 37°C for 20 min with 1 mL of diluted DCFH-DA (Beyotime, China), with mixing and inverting every 5 min. The cells were washed 3 times with PBS to fully remove any DCFH-DA that had not entered the cells. PBS (500 μL) was used to resuspend the cells, and then flow cytometry was used for detection. Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Immunohistochemistry staining
For immunohistochemical staining, the endogenous peroxidase activity of the sections was quenched using 2.5% (v/v) hydrogen peroxide in methanol, and the sections were then incubated at 3°C with 0.25% (w/v) hyaluronidase. Primary antibodies against P21 (1:200; Affinity, AF6290) and PINK1 (1:200; Affinity, DF7742) were used and diluted in 4% BSA dissolved in PBST (2.5% Triton X-100 in PBS). Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Immunofluorescence staining
Briefly, prefixed tissue sections and adherent cell sections were subjected to osmotic treatment followed by blocking with 4% BSA solution to block nonspecific binding. Finally, the primary antibody was added, and the sections were incubated at 4°C overnight. Fluorescent-labeled secondary antibodies were added at room temperature and incubated for 2 h the next day. After that, DAPI staining solution was added for nuclear staining. After washing, the slides were subjected to fluorescence microscopy to observe and record the images. Primary antibodies against COL2 (1:200; Affinity, AF0135) and phospho-histone H2A.X (1:200; Affinity, AF8482) were used and diluted in 4% BSA dissolved in PBST (2.5% Triton X-100 in PBS). Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Senescence-associated β-galactosidase (SA-β-gal) staining
Chondrocyte senescence was evaluated using senescence-associated β-galactosidase (SA-β-gal) staining with a cellular senescence assay kit (product code C0602; Beyotime). The procedure was conducted according to the manufacturer’s instructions. Briefly, the cells were washed twice with PBS and fixed with 4% paraformaldehyde for 15 min at room temperature. Subsequently, the cells were incubated with the staining solution overnight at 37°C. The number of SA-β-gal-positive cells was quantified relative to the total number of cells. Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Protein extraction and western blotting
Western blotting was performed as previously described. 24 The blots were incubated with primary antibodies against beta actin (1:3000; Affinity, AF7018), ACAN (1:1000; Affinity, DF7561), COL2 (1:1000; Affinity, AF0135), MMP-3 (1:1000; Affinity, AF0217), MMP-13 (1:1000; Affinity, AF5355), P53 (1:1000; Affinity, AF0879), P21 (1:1000; Affinity, AF6290), PINK1 (1:1000; Affinity, DF7742), Parkin (1:1000; Affinity, AF0235), P62 (1:1000; CST, 23,214), and LC3A/B (1:1000; CST, 4108). Statistical results were first assessed using one-way ANOVA to assess all between-group differences, and a t test was used to compare differences between groups on a two-by-two basis.
Statistical analysis
The data are expressed as the mean ± standard deviation (SD) for continuous variables. All experiments tested sample sizes greater than 3 (n ≥ 3). Statistical comparisons between groups were conducted using either one-way analysis of variance (ANOVA) or paired t tests. A p value less than 0.05 (two-tailed) was considered to indicate statistical significance. All the statistical analyses and data visualizations were performed using GraphPad Prism 5.
Results
PCA treatment reduces the development of posttraumatic OA
To investigate the potential beneficial effects of PCA administration on pathological changes in knee OA, we constructed a DMM surgery model, which is known to induce inflammation and articular cartilage defects. Micro-CT scanning revealed that mice in the DMM model group exhibited an abnormally narrow joint space, increased cartilage surface density, and noticeable osteophyte formation compared to those in the normal group. Osteoarthritic processes were accompanied by a reduction in the bone density area (Tb.BS/TV), elevated trabecular separation (Tb.Sp) and SMI structural model coefficients. However, these effects were significantly attenuated by treatment with PCA (Figure 3(a)). Histological analysis of cartilage tissue using HE and safranin O staining further validated these findings (Figure 3(b)). Additionally, western blotting analysis demonstrated that the expression of ACAN and COL2 decreased, while MMP-3 and MMP-13 expression increased in association with cartilage damage in the DMM model. Importantly, PCA treatment reversed these changes (Figure 3(c)). PCA treatment reduces the development of posttraumatic OA. (a) Micro-CT scanning image of the knee joint on Day 35 after DMM surgery. (b) Representative images of baicalin O and H&E staining and OARSI scores of different groups showing changes in cartilage structure and proteoglycan content. (c) The protein expression levels of ACAN, COL-2, MMP-3 and MMP-13 in the different groups of mice were determined by western blotting. The results are expressed as the mean ± SD. The statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; DMM, destabilization of the medial meniscus; OA, osteoarthritis; ACAN, aggrecan core protein; COL, collagen; MMP, matrix metalloproteinase.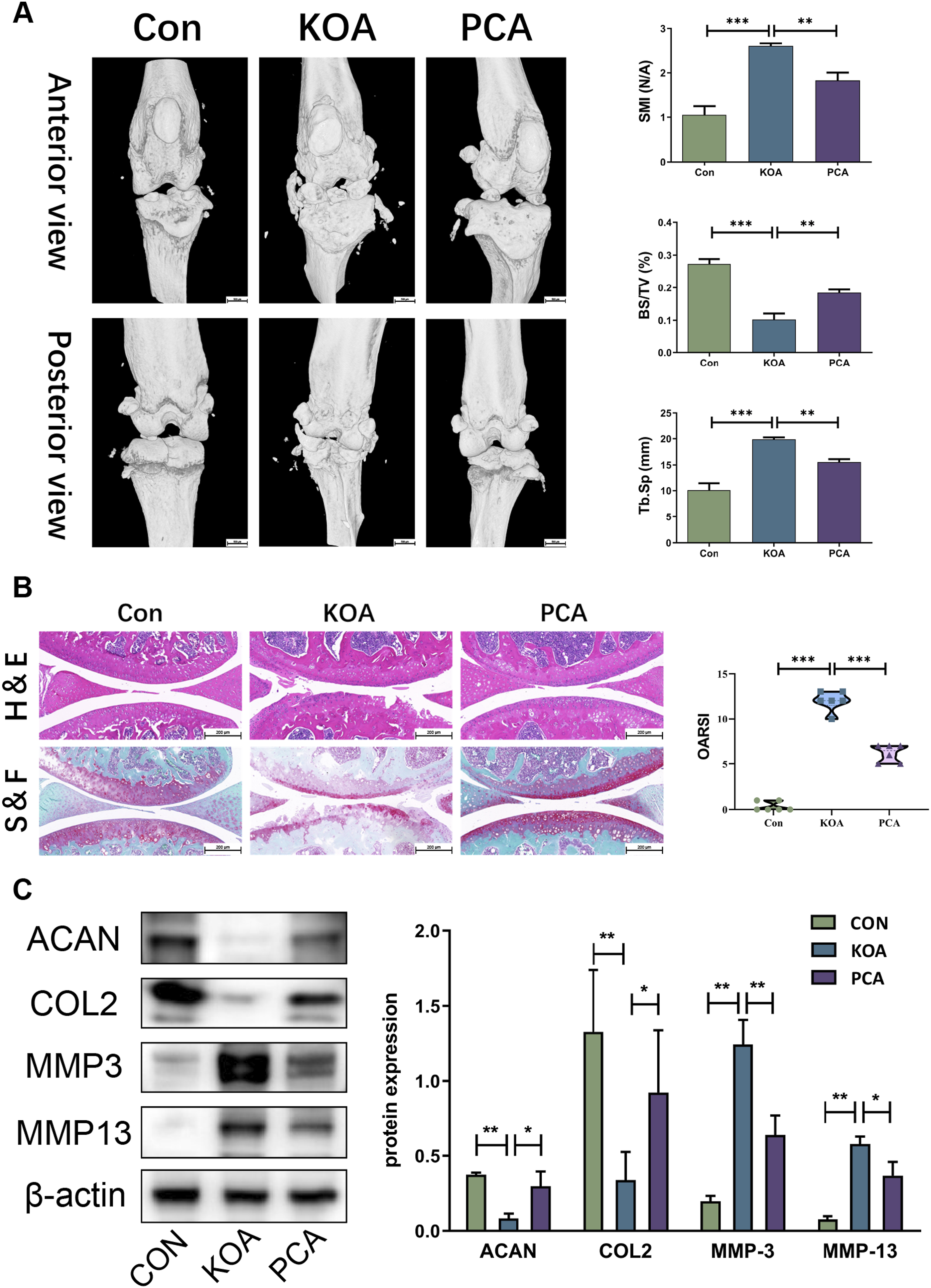
PCA treatment improves cartilage senescence in mice with posttraumatic OA
The expression levels of the senescence-associated proteins P21 and p53 were examined to investigate the effects of PCA on the DMM model mice. Notably, treatment with PCA significantly improved the abnormal expression of these proteins in the joint cartilage (Figure 4(a)). Mitochondrial autophagy is a well-recognized cause of cellular senescence in chondrocytes during OA development. In this study, western blot analysis revealed that PCA treatment markedly attenuated the levels of mitophagy-related proteins, including PINK1, Parkin, P62, and LC3 (Figure 4(a)). Immunohistochemical staining analysis further confirmed these results by revealing a decreased number of P21- and PINK1-positive cells in response to PCA treatment (Figure 4(b)). PCA treatment improves cartilage senescence in mice with posttraumatic OA. (a) The protein expression levels of the senescence-associated proteins p53, P21, PINK1, Parkin, P62, and LC3 in the different groups of mice were determined by western blotting. (b) Representative images of P21 and PINK1 immunostaining in articular cartilage from different groups of mice are shown. The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; OA, osteoarthritis; PINK1, PTEN-induced putative kinase 1.
PCA treatment attenuates LPS-stimulated chondrocyte senescence
To investigate the potential protective effects of PCA on chondrocytes, we treated mouse chondrocytes with various concentrations and durations of PCA and assessed cell viability using the CCK-8 assay. Treatment with 600 μmol PCA for 24 h did not induce significant cytotoxicity in the chondrocytes (Figure 5(a)). Next, we assessed the anti-aging effect of PCA by measuring the cellular senescence index using SA-β-gal staining. Interestingly, our findings demonstrated that the percentage of SA-β-gal-positive cells significantly increased following LPS treatment, but this effect was markedly attenuated by treatment with PCA (Figure 5(b)). Furthermore, we examined the effect of PCA on DNA damage by performing a γH2AX assay. The results demonstrated that PCA treatment reduced DNA damage in LPS-treated chondrocytes, as indicated by decreased levels of γH2AX (Figure 5(c)). In addition, the senescence-associated secretory phenotype (SASP) plays a crucial role in the aging microenvironment. We examined the expression levels of SASP-related molecules, including MMP-3, MMP-13, ACAN, and COL2, in mouse chondrocytes. Our results revealed that PCA treatment reduced the LPS-induced increase in the expression of these SASP-related molecules (Figure 5(d)). PCA treatment attenuates LPS-stimulated chondrocyte senescence. (a) The cytotoxicity of PCA was evaluated using the CCK-8 assay. Mouse chondrocytes were treated with various concentrations of PCA (1–1000 μmol/ml) for 24 h and with a concentration of 600 μmol/ml for different durations (0–72 h). Cell viability was then measured. (b) Chondrocyte senescence was assessed using SA-β-Gal staining, a widely used marker for cellular senescence. The number of SA-β-Gal-positive cells was quantified and is presented in a bar chart. (c) Immunofluorescence staining was performed to visualize γH2AX, a marker for DNA damage, in LPS-treated chondrocytes. Representative images were captured using a fluorescence microscope. (d) Protein expression levels of ACAN, COL2, MMP-3 and MMP-13 were examined using western blotting in chondrocytes stimulated with different conditions. The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; LPS, lipopolysaccharide; ACAN, aggrecan core protein; COL, collagen; MMP, matrix metalloproteinase.
PCA treatment improves mitochondrial autophagy in LPS-treated chondrocytes
Mitochondria serve as central hubs for cellular energy regulation. Hence, we used flow cytometry and fluorescence microscopy to assess ROS levels and mitochondrial membrane potential. Our results showed that PCA effectively reduced ROS levels (Figure 6(a)and (b)) and even restored the mitochondrial membrane potential in LPS-treated chondrocytes (Figure 6(c)). Subsequently, we performed MitoTracker O (MITO) staining to assess mitochondrial autophagy. Remarkably, the results revealed a significant decrease in the number of senescent chondrocytes following LPS treatment, which was notably restored upon treatment with PCA (Figure 6(d)). Furthermore, western blot analysis demonstrated that PCA treatment markedly attenuated the expression of mitophagy-related proteins, including PINK, Parkin, P62, and LC3 (Figure 6(e)). PCA treatment improves mitochondrial autophagy in LPS-treated chondrocytes. (a) Chondrocytes incubated with the DCFH-DA probe were detected using flow cytometry to characterize ROS levels in chondrocytes treated with LPS and PCA. (b) Immunofluorescence staining images depicting the levels of ROS in chondrocytes treated with LPS were captured using DCFH-DA staining. (c) Mitochondrial membrane potential was assessed using fluorescence imaging, and the red/green immunofluorescence intensity ratio was quantified and is displayed in a bar chart. (d) Immunofluorescence staining images illustrating mitochondrial autophagy in LPS-treated chondrocytes were obtained using MITO staining. (e) Western blotting was performed to analyze the expression levels of mitophagy-associated proteins, namely, PINK1, Parkin, P62, and LC3, in chondrocytes subjected to different treatments. The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; LPS, lipopolysaccharide; ROS, reactive oxygen species; PINK1, PTEN-induced putative kinase 1.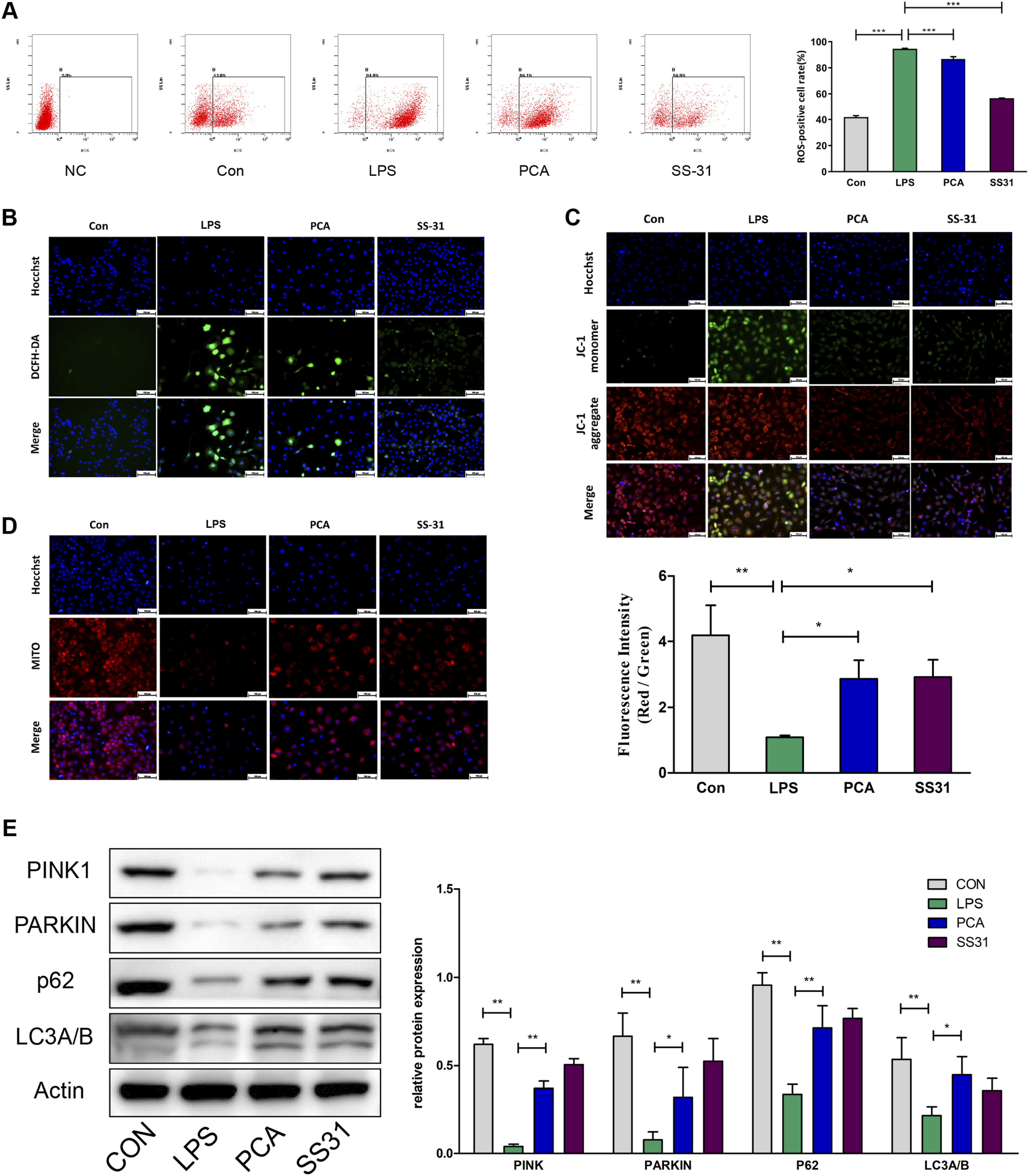
The protective effect of PINK1 on LPS-stimulated chondrocyte senescence
To examine the role of PINK1/Parkin signaling in LPS-stimulated chondrocytes, we performed gene silencing and overexpression experiments targeting PINK1. Figure 7(a) and (b) shows the protein expression levels of PINK1 after siRNA or adenovirus transfection, confirming the successful manipulation of PINK1 expression. Remarkably, our results demonstrated that PINK1 gene silencing significantly increased chondrocyte senescence induced by LPS stimulation (Figure 7(c)), which was dramatically attenuated by PINK1 overexpression (Figure 7(d)). Similar trends were observed in the expression levels of the senescence-associated proteins P21 and P53, as assessed by western blot analysis. Consistent with these findings, western blot analysis revealed that PINK1 gene silencing led to reduced expression of mitophagy-related proteins, including Parkin, P62, and LC3 (Figure 7(e)), whereas PINK1 overexpression promoted mitophagy (Figure 7(f)). The protective effect of PINK1 on LPS-stimulated chondrocyte senescence. To assess the impact of (a) PINK1 siRNA knockdown and (b) PINK1 adenovirus-induced overexpression, western blotting analysis was performed to measure the protein expression levels. Chondrocyte senescence was evaluated using SA-β-Gal staining, and the quantification of SA-β-Gal-positive cells is represented by a bar chart following (c) PINK1 gene silencing and (d) PINK1 overexpression. Moreover, western blotting was employed to examine the expression levels of senescence-associated proteins, including P53, P21, Parkin, P62, and LC3, in chondrocytes subjected to different stimuli. The results are presented in Figure 5(e) and 5(f). The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; LPS, lipopolysaccharide; PINK1, PTEN-induced putative kinase 1.
The effect of PCA on the PINK1/Parkin signaling pathway in chondrocytes
Furthermore, we investigated the effect of PCA on the PINK1/Parkin signaling pathway. Interestingly, PINK1 gene silencing slightly increased the expression of the senescence-related proteins P21 and P53 but decreased the expression of the mitophagy-associated proteins PINK1, Parkin, P62, and LC3 (Figure 8(a)). However, treatment with PCA significantly attenuated the effects of PINK1 gene silencing on these proteins (Figure 8(a)). Conversely, PINK1 overexpression had a protective effect on mitophagy, which was further enhanced by PCA treatment (Figure 8(b)). These findings were confirmed by MITO staining, which demonstrated the induction of mitochondrial autophagy (Figure 8(c) and (d)). The effect of PCA on the PINK1/Parkin signaling pathway in chondrocytes. Chondrocytes were prestimulated with PINK siRNA (20 μmol/L) or virus solution (100 μL), followed by coculture with LPS (1 μM) and PCA (600 mmol/L) for 24 h. The expression levels of senescence-associated proteins, including P21 and P53, as well as mitophagy-associated proteins, such as PINK1, Parkin, P62, and LC3, were assessed using western blotting in cells subjected to (a) PINK1 gene silencing and (b) PINK1 overexpression. Furthermore, immunofluorescence staining was used to capture and visualize MITO images, which revealed mitochondrial autophagy in cells subjected to (c) PINK1 gene silencing or (d) PINK1 overexpression. The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; LPS, lipopolysaccharide; PINK1, PTEN-induced putative kinase 1.
The effect of PINK1 on the protective effect of PCA on LPS-stimulated chondrocyte senescence
To determine whether the protective effects of PCA described above might be mediated through increasing the activity of PINK1, we performed PINK1 inhibition and overexpression experiments (Figure 9(a)). As a result, silencing of PINK1 abolished the protective effects of PCA, as determined via the measurement of SA-β-Gal activity (Figure 9(b)). However, PINK1 overexpression enhanced these effects, confirming the involvement of PINK1 in mediating the chondroprotective effects of PCA. Similar results were also obtained by western blotting, indicating that PINK gene silencing markedly inhibited the effects of PCA on senescence-related molecules, including P21, P53, ACAN, COL2, MMP-3, and MMP-13. In contrast, PINK1 overexpression showed collaborative enhancement with PCA (Figure 9(c)). The effect of PINK1 on the protective effect of PCA on LPS-stimulated chondrocyte senescence. (a) Western blotting analysis was performed to examine PINK1 protein expression following PINK1 gene silencing and PINK1 overexpression. (b) To assess the impact of PINK1 on the protective effect of PCA on LPS-stimulated chondrocytes, SA-β-Gal staining was performed to identify senescent cells, and the quantification of SA-β-Gal-positive cells is represented by a bar chart. (c) Moreover, western blotting was used to examine the expression levels of senescence-associated proteins, including P53, P21, ACAN, COL-2, MMP-3 and MMP-13, in chondrocytes subjected to different stimuli. The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PINK1, PTEN-induced putative kinase 1; PCA, protocatechuic aldehyde; LPS, lipopolysaccharide.
The modulation of mitochondrial autophagy by PCA is related to the PINK1/Parkin signaling pathway
Finally, we investigated whether the activation of PINK1 by PCA is necessary for mitophagy induction and explored the underlying mechanisms involved. Our findings demonstrated that the overexpression of PINK1 enhanced the restorative effect of PCA on the mitochondrial membrane potential and induced mitochondrial autophagy, as evidenced by MITO staining in chondrocytes treated with LPS. In contrast, silencing of the PINK1 gene had the opposite effect (Figure 10(a) and (b)). Furthermore, western blot analysis revealed similar results, showing altered expression levels of mitophagy-related proteins, including Parkin, P62, and LC3 (Figure 10(c)). These findings suggest that the mechanism underlying the induction of mitophagy by PCA involves the PINK1-Parkin pathway. The modulation of mitochondrial autophagy by PCA is related to the PINK1/Parkin signaling pathway. (a) Mitochondrial membrane potential was assessed using fluorescence imaging, and the red/green immunofluorescence intensity ratio was quantified and is displayed in a bar chart. (b) Immunofluorescence staining images illustrating mitochondrial autophagy in LPS-treated chondrocytes were obtained using MITO staining. (c) Western blotting was performed to analyze the expression levels of mitophagy-associated proteins, namely, Parkin, P62, and LC3, in chondrocytes subjected to different treatments. The results are expressed as the mean ± SD. Statistical significance is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001. PCA, protocatechuic aldehyde; PINK1, PTEN-induced putative kinase 1; LPS, lipopolysaccharide.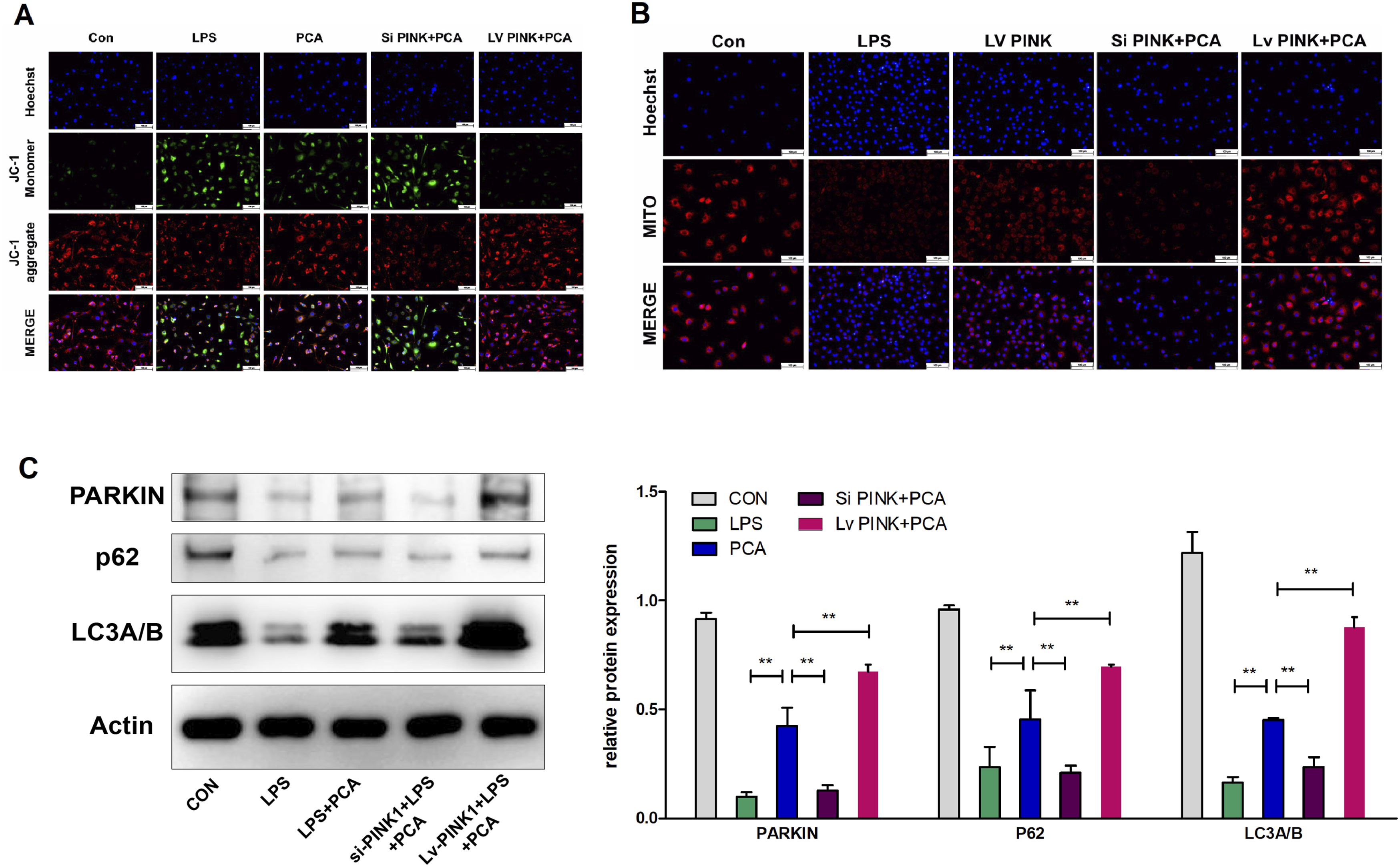
Discussion
Knee OA is a prevalent degenerative joint disease that imposes significant costs on society. 3 The use of traditional Chinese medicine for the treatment of OA has attracted considerable interest from researchers. One such naturally occurring phenolic compound, protocatechuic aldehyde, derived from Salvia miltiorrhiza, has shown various pharmacological activities, including anti-inflammatory, antioxidant, and antiapoptotic effects.18–22 However, the specific role of PCA in OA remains unclear. In a previous study, 24 we discovered that PCA exhibits potential protective effects on cartilage degeneration and chondrocyte senescence. Importantly, these protective effects may be mediated through the promotion of mitochondrial autophagy via the upregulation of the PINK1/Parkin pathway.
Chondrocytes, the exclusive inhabitants of articular cartilage, play a crucial role in maintaining the metabolic balance within cartilage tissue.26,27 However, their ability to maintain a youthful state diminishes over time, eventually leading to an irreversible state of cellular senescence. 28 Chondrocyte senescence is characterized by a reduction in metabolic capacity and an increased susceptibility to stress, compromising the structural integrity of cartilage tissue and ultimately leading to cartilage degradation.29,30 In our study, we also observed chondrocytes undergoing accelerated senescence in LPS-stimulated cells and in DMM mice, which is consistent with the observed cartilage degradation. Furthermore, we found that treatment with PCA both in vitro and in vivo alleviated these effects. The senescence-associated secretory phenotype (SASP) is an umbrella term for a range of cytokines, such as proinflammatory factors, chemokines and proteases, which are key features of senescent cells. Our results showed that PCA treatment improved the expression levels of SASP protein, such as MMP-3 and MMP-13, in chondrocytes to exert anti-inflammatory effects for alleviating KOA. These findings suggest that PCA may have therapeutic potential in preventing or slowing the progression of cartilage degradation by attenuating chondrocyte senescence.
Mitochondria have long been recognized as important contributors to the establishment and maintenance of cellular senescence. 31 Numerous studies have demonstrated that mitochondrial damage can lead to the accumulation of ROS within cells, disrupt ATP synthesis, and induce phenotypic changes associated with cellular aging.32–34 In this study, we aimed to investigate the effects of PCA on ROS levels and mitochondrial function in chondrocytes. To assess ROS levels, we utilized DCFH-DA staining, a commonly used method for measuring intracellular ROS production. Additionally, we measured the mitochondrial membrane potential, a crucial indicator of mitochondrial function. Our results revealed that treatment with PCA effectively inhibited ROS production and improved the mitochondrial membrane potential in chondrocytes stimulated with LPS. These findings suggest that PCA possesses potent antioxidant activity and may have therapeutic potential in mitigating oxidative stress and mitochondrial dysfunction in chondrocytes.
In this study, we aimed to elucidate the precise molecular mechanisms underlying the antioxidative effects of PCA. To achieve this goal, we investigated the effect of PCA on mitophagy in both LPS-treated cells and the cartilage of mice with DMM. Insufficient mitophagy is known to contribute to the accumulation of dysfunctional mitochondria and the production of ROS, which are closely associated with the development of cellular senescence. Therefore, it is imperative to not only protect against mitochondrial dysfunction but also trigger mitophagy to eliminate damaged mitochondria in senescent cells. Previous studies have reported that PCA can promote autophagy in various cell types.22,23,35 Our results also demonstrated that PCA significantly enhances mitophagy in both LPS-treated cells and in the cartilage of mice with DMM. These findings suggest that PCA may have a potential therapeutic role in promoting mitophagy and reducing cellular senescence-related damage.
Previous studies have classified PINK1 as a serine-threonine protein kinase,36,37 and it is known to regulate the PINK1/Parkin pathway, which is involved in mitophagy.38,39 In this pathway, PINK1 accumulates on the outer membrane of damaged mitochondria and phosphorylates Parkin. This leads to the ubiquitination of proteins on the mitochondrial outer membrane, which are recognized by P62 and subsequently facilitate the engulfment of damaged mitochondria by autophagosomes. 40 However, the specific role of PINK1 in the context of OA has not been thoroughly investigated. Thus, in our recent study, we used genetic editing methods to validate the involvement of PINK1 in chondrocyte senescence. The results indicated that downregulation of the PINK1 protein in chondrocytes markedly decreased the expression of mitophagy-related proteins, elevated ROS levels, aggravated the decrease in the mitochondrial membrane potential, and intensified the progression of chondrocyte senescence. On the other hand, by upregulating PINK1 protein expression using a lentivirus, we found that the overexpression of PINK1 alleviated chondrocyte senescence by promoting mitophagy.
Next, OA chondrocytes were treated with PCA to investigate the role of PCA in the of PINK1/Parkin pathway. Treatment with PCA reversed the changes induced by PINK1 knockdown. Specifically, PCA treatment reversed the alterations in mitochondrial morphology and function and decreased the expression of SASP-related proteins in OA chondrocytes. Moreover, silencing PINK1 abolished the protective effects of PCA against LPS-induced cellular senescence. These findings collectively suggested that PCA reduced cellular senescence through the regulation of PINK1/Parkin-mediated mitochondrial autophagy. Further investigations are warranted to fully understand the underlying mechanisms responsible for these protective effects.
However, there were some limitations in our study. One limitation is that we did not investigate the effects of different concentrations of PCA on cartilage in osteoarthritis, either in vivo or in vitro experiments. Therefore, we cannot fully understand the range of its effects on cartilage. Moreover, further exploration of the molecular mechanism of PCA is needed.
Conclusion
In this study, we evaluated the anti-aging effect of PCA on chondrocytes by in vivo and in vitro experiments and found that PCA protected the mitochondrial membrane potential and reduced the intracellular ROS level, which may be related to the maintenance of mitochondrial autophagy homeostasis in chondrocytes. In future studies, we will explore the relationship between PCA and mitochondrial autophagy in more depth. In conclusion, PCA can target chondrocyte senescence, which provides an effective foundation for its development as a treatment for KOA to improve quality of life.
Footnotes
Acknowledgements
The authors wish to express their gratitude to all staffs in the medical research center of first college of clinical medicine, the Nanjing University of Chinese Medicine, Nanjing, China.
Authors’ contributions
Lishi Jie: Writing – original draft. Xiaoqing Shi: Writing – original draft. Junfeng Kang: Data curation. Houyu Fu: Resources, Data curation. Likai Yu: Methodology, Investigation. Di Tian: Methodology. Wei Mei: Writing – review & editing, Project administration. Songjiang Yin: Writing – review & editing, Funding acquisition, Project administration.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82104893), the Natural Science Foundation of Jiangsu Province (BK20200842), Research Project at Jiangsu Provincial Hospital of Traditional Chinese Medicine (Y21029), the Peak Academic Talent Project of Jiangsu Province Hospital of Chinese Medicine (y2021rc48).
Ethical statement
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Consent for publication
Data was de-identified, after collection, and therefore there was no consent required from individual patients, according to the ethical approval.
Data availability statement
All results and data are kept in the section for Departments of orthopedics, the Affiliated Hospital of Nanjing University of Chinese Medicine, Nanjing, China. These will be made available from the corresponding author on reasonable request.
