Abstract
Introduction
The 2007 International Dry Eye Workshop, sponsored by the Tear Film and Ocular Surface Society [TFOS] Committee has defined dry eye disease as follows: “Dry eye is a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability with potential damage to the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface.” 1 After inflammation develops, it gradually increases due to cytokines produced by lymphocytes released from damaged epithelial cells and leaking from dilated conjunctival vessels. 2 In the treatment of dry eye, anti-inflammatory agents are preferred. Cyclosporine A (CsA) is effectively and widely used in drop form due to its inhibitory effect on epithelial apoptosis and cytokine release from activated T cells. 3 Understanding the normal flora of the conjunctiva is essential for tracking changes that might occur in response to various influences and for identifying potential pathological conditions. We know that the normal biological flora of the conjunctiva is formed from birth and that this flora is protective. The microbial flora of the ocular surface is made up of primarily gram-positive microorganisms including staphylococci and diphtheroids. The most frequently isolated causative agent is coagulase-negative staphylococus Epidermidis (CNS). 4 Although rare, the presence of gram-negative rods and fungi in the flora is accepted as temporary ocular microbiota. 5 The predominance of Staphylococcus epidermidis prevents the growth of pathogenic microorganisms and thereby protects the ocular surface. The nasal cavity and ocular surface are connected via the nasolacrimal duct for tear drainage. As a matter of fact, it has been proven that the nasal mucosa is exposed to drops applied to the ocular surface. 6 Studies have shown that chronic usage of certain antibiotic eye drops, antiglaucoma agents, and lubricants containing antiseptic preservatives such as benzalkonium chloride (BAK) alters the flora.7–9 It is thought that when these drops are used, the bacteria that cause endophthalmitis or blebitis, which can develop after intraocular surgery, may develop more resistant strains due to the change in the local flora. 8 A search of major databases including PubMed did not reveal any study to date regarding the changes occurring in the conjunctival and nasal mucosal flora after the use of CsA.
The aim of this study is to analyze the long-term effect on conjunctival and nasal flora and antibiotic resistance in patients on topical cyclosporine for dry eye.
Materials and method
Patients who had been diagnosed with dry eye and had been using CsA eye drops and/or preservative-free artificial tear (PFAT) drops for the last 3 months, seen at Istanbul Medeniyet University’s Göztepe Prof. Dr Süleyman Yalçın City Hospital (Istanbul, Turkey) between January 1st, 2020, and April 30th, 2020, were included in this cross-sectional study. The study was approved by the local university ethics committee. The decision number was 2020/0085. The study was conducted in compliance with the Declaration of Helsinki. Written informed consent with signature was obtained from all participants.
Thirty-eight patients were using CsA 0.05% eye drops (RESTASIS®; Allergan, Inc., Irvine, CA) and PFAT (Refresh Plus (®); Allergan, Inc., Irvine, CA) for the treatment (Group 1), while the 34 patients in the control group were using only PFAT (Group 2). Dry eye diagnoses were established according to the Tear Film and Ocular Surface Society’s Dry Eye Workshop (TFOS DEWS) II criteria. 10 All patients were examined by an ophthalmologist using a slit lamp. The inclusion criteria were as follows: ocular surface disease index (OSDI) ≥13; tear break-up time ≤5 s and Schirmer’s test (without local anesthesia) ≤5 mm. Performing an anesthetized Schirmer test has been shown to be more objective and reliable in patients with dry eye disease than performing the test without anesthesia, and it measures basal tear film secretion. 11 The following inclusion criteria were applied: (1) follow-up with a diagnosis of dry eye at least 3 months; (2) use of PFAT or CsA eye drops for at least 3 months; (3) at least 18 years of age.
Patients with examination findings or history of any of the following were excluded from the study: (1) contact lens wear; (2) ocular surgery; (3) use of topical antibiotics, antiseptics, corticosteroids, or nonsteroidal anti-inflammatory drugs in either eye within the past 3 months; (4) history of any ocular infection within the past 3 months; (5) use of systemic antibiotics within 30 days of enrollment; (6) glaucoma; (7) nasolacrimal duct obstruction; (8) being anormal blood glucose; (9) recent sinonasal infections and diagnosed as allergic rhinitis.
For each patient, one eye and the ipsilateral nasal cavity were included in the study.
The following data were collected: (1) general medical history and demographic information, (2) OSDI score (ocular surface disease score), tear break-up time (TBUT) and schirmer test, (3) conjunctival swab culture. TBUT was evaluated under slit lamp biomicroscopy with cobalt blue filter after application of fluorescein strip. TBUT was measured three consecutive times with a stop watch after each blink. The average of the three measurements was used for analysis.
After a full ophthalmologic examination including vision measurement, biomicroscopic and fundus examination; a swab sample was taken from the lower conjunctiva using a sterile swab without touching the eyelashes and lids, and without applying local anesthesia to prevent contamination in all cases. In addition, samples were taken from the nasal cavity by gently twisting a cotton-tipped applicator.
The samples were inoculated on blood, chocolate, and EMB (eosin methylene blue) media at the head of the patient. The media were delivered to the laboratory within 1 hour and incubated at 37°C for 24 h.
Laboratory analyses consisted of culture, microorganism identification, and drug sensitivity tests. Drug sensitivity tests were performed by agar diffusion method using the subsequent antibiotics according to clinical and laboratory standards institute (CLSI) guidelines.
The incubation of plaques without growth at 24 h was extended to 48 h. All colonies isolated from the media were identified using a VITEK® MS, MALDI-TOF (bioMérieux, France), and antibiotic susceptibility tests were performed using the VITEK® 2 Compact (bioMérieux, France) system in accordance with the manufacturer’s instructions.
Statistical analysis
In this study, Operational Taxonomic Units (OTU) richness and Shannon index were used to assess alpha diversity. Alpha diversity represents the degree of species richness and equality within a sample, and several metrics are available to assess this diversity. 12 The Shannon index measures the overall diversity of a community by combining species richness and interspecific equality, and is one of the most widely used indices in the literature to measure alpha diversity. 13 For statistical evaluations, analyses were performed using the “vegan” package within the R programming language. Statistical significance was assessed at 0.05 alpha level and all p-values were calculated pairwise. Mann–Whitney U test was used to compare Shannon diversity while chi-square test was used OTU richness values between groups.
The remaining data were analyzed using IBM, SPSS Statistics version 20 (IBM Inc., USA). The normally distributed quantitative data were presented as mean and standard deviation (SD) and compared by t test (tear function tests: TBUT, schirmer test, OSDI score, age). Chi-square test and Fisher’s exact test (gender) were used to compare the antibiotic susceptibility of S. epidermidis between the groups.
Results
Comparison of demographic characteristics of the groups.

CsA: Cyclosporine A, PFAT: Preservative-free artificial tear, m/f: Male/female, OSDI: Ocular surface disease index, TBUT: Tear break-up time.
at test.
bChi-square test.
Bacterial growth in the conjunctiva was observed in 54 (75%) of the 72 patients, while no growth was observed in 25% of patients. While 88.88% of the reproducing microorganisms were S. epidermidis, it was detected in 48 (66.66%) of the total patients. While there was no bacterial growth in the nose in two patients, bacterial growth was detected in the other 70 patients (97.22%). Overgrowth of S. epidermidis was detected in 54 (77.14%) of these 70 patients. This rate was found to be 75% across all patients. In general, reproduction was higher in the nose. S. epidermidis was the dominant pathogen in the eyes and nose.
According to the conjunctival culture findings, there were differences in terms of Shannon diversity and OTU richness in certain bacterial species between Group 1 and Group 2. In the case of S. epidermidis and Strep_viridans bacteria, Group 1 was observed to have higher Shannon diversity and OTU richness values compared to Group 2. However, these differences were not statistically significant. Statistically insignificant, an increase in commensal bacteria has been observed in the group using cyclosporine eye drops. For bacterial species including methicillin sensitive staphylococcus aureus (MSSA), Escherichia coli, Citrobacter_koseri, Klebsiella_pneumoni, Staphilococcus_hominis, and Proteus, Shannon diversity and OTU richness values could not be found in both groups. On the other hand, for methicillin resistant s_Aerus (MRSA) and Corynebacterium bacteria, Shannon diversity and OTU richness values were found only in one group, while these values were zero for the other group.
Comparison of the groups with alpha diversity analysis in terms of bacteria growing in the conjunctiva and nasal cavity.
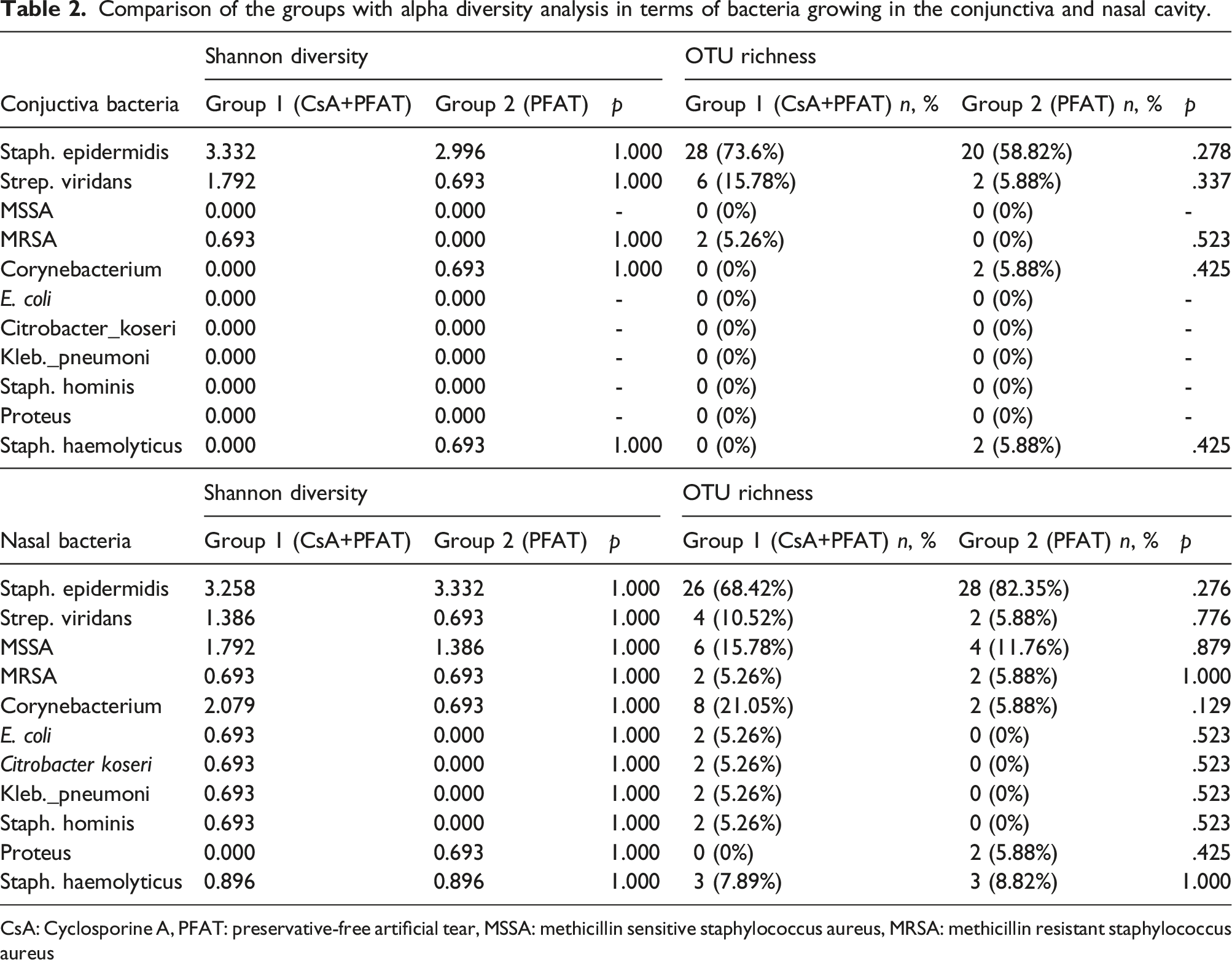
CsA: Cyclosporine A, PFAT: preservative-free artificial tear, MSSA: methicillin sensitive staphylococcus aureus, MRSA: methicillin resistant staphylococcus aureus
Comparison of the groups in terms of antibiotic susceptibility of S. Epidermidis.

TMP-SMX: trimethoprim sulfamethoxazole, CsA: Cyclosporine A, PFAT: preservative-free artificial tear.
Discussion
Various studies have determined that the conjunctival flora can change in response to the age of a person, contact lens use, drug use, dry eye, immunosuppression, and environmental factors. 4
Endophthalmitis is the most feared complication following intraocular surgery. Although rare, endophthalmitis can lead to severe vision loss. After cataract surgery, the reported rate of endophthalmitis ranges between 0.04 and 0.2%. 14 In endophthalmitis, the main source of infection is the patient’s endogenous bacterial flora, which is usually determined by postoperative conjunctival cultures. 15 Therefore, especially in intraocular surgeries, the factors that change the conjunctival flora need to be well known and studied in order to be protected from postoperative endophthalmitis in the preoperative period.
Conjunctival culture positivity ranges between 30.0% and 93.0%.4,16 S. epidermidis is known to be the most densely colonized bacterium in the conjunctival flora. 17 Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus spp., Moraxella spp., Neisseria spp., Bacillus spp., and gram-negative rods have been isolated, but this is usually temporary colonization. 18 In our study, the conjunctival growth rate was found to be 75%. In addition, in accordance with the literature, the bacteria that reproduced in the conjunctiva was predominantly S. epidermidis (88.88%).
The type of medium used also plays an important role in the growth of microorganisms. Coşkun et al. used three types of media (blood agar, chocolate agar, EMB agar) and found a conjunctival growth rate of 93.6%. 4 In this present study, all three types of media were used to achieve the highest growth rate. In a study showing that local anesthetic drops affect the flora during sample collection, the CNS growth rate was found to be 52.8% in samples taken before anesthesia, while this rate was found to be 33.3% in samples taken after local anesthetic drops were applied. 19 In our study, anesthetic drops were avoided with the aim of increasing the reproduction rate.
In a study, the sensitivity rate of S. epidermidis to penicillin G was found to be 14% in normal conjunctival flora. 4 In a recent large series study, CNSs in normal conjunctival flora were shown to be 90.8% resistant to penicillin, 84.4% resistant to erythromycin, and 6.9% resistant to gentamicin. 20 In our samples, S. epidermidis had similar rates of resistance.
The ocular surface and nasal cavity are connected via the nasolacrimal canal. Ocular drops are known to reach the nasal mucosa and be absorbed from there. 21 Forty percent of the standard 50-μL eye drops reach the nasal mucosa through the nasolacrimal duct where they are then absorbed. 6 The nasal mucosa acts as a reservoir of bacteria for the ocular surface. In one study worth noting, it was found that bacteria isolated in an endophthalmitis case series were genetically similar to bacteria taken from the nose. 22 In our study, no difference was observed between the eyes and noses of the patients in terms of bacterial growth and antibiotic susceptibility.
Bacteria that make up the normal flora of the conjunctiva protect the eye from diseases by preventing the colonization of resistant and pathogenic species. 23 In this phenomenon known as “competitive exclusion,” S. epidermidis, which makes up the majority of the normal flora of the conjunctiva, functions as a probiotic and provides protection against a more pathogenic species, S. aureus. 24 Despite the protective effect of S. epidermis, it is the most frequently isolated opportunistic bacteria in ocular surface and intraocular infections. In addition, these resistant strains can develop resistance to antibiotics rapidly by making changes in their biofilms. 25 In “The Antibiotic Resistance of Conjunctiva and Nasopharynx Evaluation” study (ARCANE), which is the most comprehensive study to have examined antibiotic resistance developing in the conjunctiva and nasal mucosa after repeated use of topical antibiotics, colonization of resistant strains was observed. 21 In another study, it was found that resistant strains of S. aureus colonized rapidly after repeated use of macrolide and fluoroquinolone antibiotic eye drops. 9 Many studies have shown that BAK, which has antiseptic properties and is used as a preservative in eye drops, causes changes in the conjunctival flora.21,26
These studies show the conditions caused by drugs that suppress the flora. However, as far as we know, no studies have investigated the effect of cyclosporine—an immunosuppressive drug—on flora.
It has been reported that at a dose of 0.05%, cyclosporine A is effective at suppressing ocular inflammation by blocking TH2 lymphocyte proliferation and interleukin 2 (IL-2) production. 27 Cyclosporine A is effectively and widely used because of its inhibitory effect on epithelial apoptosis and cytokine release from activated T cells. 28 In a study that observed changes to flora when cyclosporine is applied to human skin grafts, after 6 days of application S. aureus and coagulase-negative streptococci were replaced by the more pathogenic organisms E. coli, E. faecium, Micrococcus, and Pseudomonas. 29
Pathogenic microorganisms kept under control by the immune system may become dominant in the case of immunosuppression. 29 This study; we noticed that the use of cyclosporine drops significantly increased the sensitivity of epidermidis to penicillin in the conjunctiva compared to the group that did not use it. The immunomodulatory effect of cyclosporine may decrease antibiotic resistance in the conjunctiva.
In this study, conducted with the hypothesis that the suppression of inflammatory cells and mediators on the conjunctival surface might increase the number of pathogenic species or contribute to antibiotic resistance development, such an effect was not observed. This situation can be explained by the fact that cyclosporine inhibits an excessive inflammatory response, does not affect the normal inflammatory response, and plays a stabilizing role in the protection of the normal flora.
In the nasal cavity, although the presence of different and more pathogenic microorganisms in Group 1 compared to Group 2 was worth noting, no significant difference was found. This can be interpreted as indicating that the use of cyclosporine does not create a tendency to infection. This result is especially important in terms of assessing whether patients who use cyclosporine for a long time and are set to undergo intraocular surgery are at increased risk of infection. However, studies with a larger patient series will provide more precise information on this subject and will form the basis for studies on the potential to reduce the prevalence of postoperative endophthalmitis.
One of the limitations of our study is that the data for minimum inhibitory concentrations were not collected, and instead, bacteria were classified as “resistant” or “susceptible.” The second limitation is that our study was not prospective. It would have been much more informative to perform antibiotic sensitivity prior to initiation of dry eye therapy in both groups and then after 3 months of therapy. The lack of a sample size calculation is another limitation of this study.
Conclusion
According to the findings obtained in this study, cyclosporine drops with anti-inflammatory properties do not have any effect on conjunctival and nasal flora and do not affect antibiotic resistance. For this reason, we think that patients using cyclosporine before intraocular surgery are safe in terms of postoperative endophthalmitis. Larger studies are needed for more precise results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
