Abstract
Objectives
There is a significant decline in the lymphocyte subset counts in the peripheral blood of COVID-19 patients. However, the mitochondrial function of lymphocytes obtained from COVID-19 patients has rarely been studied.
Methods
A case-control study was conducted in 115 COVID-19 patients and 50 healthy controls from December 2022 to February 2023. The extent of lymphocytic mitochondrial damage in these patients using mitochondrial fluorescence staining and flow cytometry. Clinical symptoms were evaluated using the SOFA and APACHE II scores.
Results
The mitochondrial function of lymphocytes was severely impaired in the peripheral blood of COVID-19 patients, compared to healthy controls, and was characterized by an increased single-cell mitochondrial mass (SCMM) and increased percentage of low mitochondrial membrane potential. The increase in the SCMM of T cells was more notable in patients with severe COVID-19 and was positively correlated with the SOFA and APACHE II scores. When the SCMM-CD8 cutoff value was 38.775, the AUC for distinguishing between severe and mild COVID-19 was 0.740, and the sensitivity, specificity, and Youden index were 65.8%, 82.1%, and 0.478, respectively.
Conclusion
SCMM-CD8 could act as a diagnostic biomarker of COVID-19 progression. However, this needs to be verified in other multi-center studies with a larger sample size.
Introduction
Mitochondria play important roles in immune response generation, energy metabolism, and mitochondrial reactive oxygen species (ROS) production.1,2 Increased ROS production stimulates the release of pro-inflammatory mediators and leads to mitochondrial dysfunction. Mitochondrial defects are associated with many pathologies, including diabetes, cardiovascular disease, gastrointestinal disorders, cancer, and aging. 3 The increased population mortality associated with the high transmission and infection of Omicron variants needs to be addressed. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) causes mitochondrial dysfunction (including mitochondrial membrane depolarization, mitochondrial permeability transition pore opening, and increased ROS release) and mitophagy impairment in vitro. 4 SARS-CoV-2 ORF3a induces mitochondrial damage in SARS-CoV-2-infected human cell lines and leads to Mito-ROS production, which helps promote HIF-1α expression and subsequently facilitates SARS-CoV-2 infection and cytokine production. 5 SARS-CoV-2 infection impairs mitochondrial functions and triggers inflammatory responses by activating TLR9 signaling, which leads to endothelial cell dysfunction, potentially contributing to the severity of COVID-19 symptoms. 6 The heightened inflammatory state may result in mitochondrial dysfunction, leading to platelet damage and apoptosis. 7 Studies on mitochondrial dysfunction in COVID-19 mainly focus on platelets, endothelial cells, and infected cell lines, and only a few reports have focused on mitochondrial dysfunction in immune cells to date. Gibellini et al. found that peripheral blood monocytes from patients with COVID-19 pneumonia show signs of altered bioenergetics and mitochondrial dysfunction, had a reduced basal and maximal respiration, reduced spare respiratory capacity, and decreased proton leak. 8
Several well-known Chinese tertiary hospitals have issued treatment guidelines that state that a significant decline in lymphocyte subset counts (especially T cells) in the peripheral blood is an early indicator of severe morbidity. A meta-analysis of 3017 patients with COVID-19 in 20 clinical studies showed that the absolute count of total lymphocyte subsets in critically ill patients was significantly lower than that in mild to moderate patients. Among these, the decrease in T cells is the most notable, and is highly correlated with poor patient prognosis. 9 The mitochondrial function in lymphocyte subsets remain unclear. Current methods for studying mitochondrial function include mitochondrial oxygen consumption, mitochondrial respiratory chain complex activity, mitochondrial membrane potential (MMP), intracellular ATP levels, the open state of mitochondrial membrane ion channels, changes in reactive oxygen species (ROS), immunoprotein blotting, and mitochondrial DNA copy number assays. Isolation of patient PBMC and then magnetic-activated cell sorting of single-cell populations is time-consuming and difficult to accomplish. Flow cytometry is a useful tool for detecting mitochondrial function in a variety of cells in biological samples due to its rapid, high-throughput and multi-parameter analysis.
In most cases, flow cytometry was used to measure changes in mitochondrial membrane potential (MMP), mitochondrial mass (MM), and ROS. Damaged mitochondria are usually removed through stress-induced mitochondrial autophagy. Intense stress leads to the misfolding of mitochondrial permeability transition pores, impairing mitochondrial autophagy 10 and increasing MM. 11 Yu et al. found that HIV infection led to an increase in MM in CD4+T and CD8+T cells, but mainly influenced MMP in CD8+T cells and ROS accumulation in CD4+T cells. 12 Therefore, the parameter of mitochondrial mass may reflect mitochondrial disturbance. Lymphocyte subsets were labeled with specific antibodies of CD3, CD4, CD8, CD56, and CD19 for the cell count of each subset. We used a mitochondrial-specific dye (Mito-Tracker Deep Red FM) that labels biologically active mitochondria in cells, and thus median fluorescence intensity (MFI) has been considered an index of MM. The single-cell MM (SCMM) was calculated based on the MFI and the count of cell subsets. Additionally, decreased mitochondrial membrane potential is associated with autophagy, apoptosis, or necrosis, etc. This study used single-cell of MM (SCMM) and the percent of MMP-low to indicate mitochondrial function.
Therefore, this study aimed to determine the SCMM and MMP-low of lymphocytes in COVID-19 patients and healthy individuals using flow cytometric analysis to understand mitochondrial dysfunction in COVID-19 and quantify the value of the mitochondrial function of lymphocytes in the severity assessment of COVID-19 pneumonia.
Materials and methods
Subjects
In this case-control study, 115 COVID-19 patients and 50 healthy controls were admitted to Zhongda Hospital, Southeast University, from December 2022 to February 2023. COVID-19 was diagnosed if the results of the analysis of a pharyngeal swab were positive in a SARS-CoV-2-real-time reverse transcription–polymerase chain reaction assay. Clinical data for COVID-19 patients, such as the Sequential Organ Failure Assessment (SOFA) score and Acute Physiology and Chronic Health Evaluation II (APACHE II) score, 13 were obtained from their medical records. COVID-19 patients were classified into three clinical subtypes: mild, moderate, and severe based on the Guidelines of the COVID-19 Diagnosis and Treatment. Exclusion criteria: (1) severe immune diseases; (2) previous history of organ transplantation; (3) combined with malignant tumors; (4) other systemic infectious diseases caused by other reasons; (5) recently taken immunosuppressants; (6) with bronchial asthma, pulmonary interstitial fibrosis; (7) pregnant and lactating women. The controls were matched by sex and age, and were selected during simultaneously conducted routine health examinations. All controls were unrelated ethnic Han Chinese individuals. The clinical data and laboratory parameters (routine blood test, cytokine results, D-Dimer, Total protein, Albumin, and blood gas analysis) were collected from the information-management system at the hospital.
Ethical approval for this study was obtained from the Institutional review board of Zhongda Hospital, Southeast University (IRB: 2021ZDSYLL331-P01). Written informed consent was obtained from all participants before admission.
Flow cytometry
We collected 2 mL of peripheral blood at the time of admission. Then, 100 µl of whole blood was placed into one absolute counting tube and two normal flow tubes (labeled MM and MMP). 20 μL of commercial antibody mixture (CD3-FITC, CD16/56-PE, CD45-PerCP-Cy5.5, CD4-PE-Cy7, CD19-APC, and CD8-APC-Cy7) was added to the absolute counting tubes, followed by incubation for 15 min and addition of 450 μL of diluted hemolysin. The antibody combinations for MM tubes were CD4-FITC, CD19-PE, CD45-percp, CD8-PE-Cy7, CD56-BV421, CD3-BV510; while for MMP tubes the antibody combinations were CD19-BB700, CD45-APC, CD8-PE-Cy7, CD4-APC-Cy7, CD56-BV421, CD3-BV510 (all BD bioscience). The corresponding volumes of CD4, CD19, CD45, CD8, CD56, and CD3 antibodies were added to the flow tube and incubated at 37°C for 15 min and hemolyzed. The tubes were washed twice and the supernatant was discarded. The MM tube was incubated with 1 μL of Mito-Tracker Deep Red probe (Beyotime, Shanghai, China) for 20 min at 37°C. The MMP tube was incubated with 500 μL of pre-warmed JC-1 working solution for 20 min at 37°C. The cells were then washed twice with 1 mL of pre-chilled JC-1 buffer. Both tubes were assayed using a FACSCanto II flow cytometer (BD bioscience, California, USA) (Supplementary Figures 1 and 2). MM was expressed as the median fluorescence intensity of mitochondria. The SCMM of lymphocytes was determined by calculating the MM values and absolute counts of lymphocyte subsets.
Statistical analysis
The data were processed using SPSS 22.0 statistical software (IBM, New York, USA). The Shapiro-Wilk test was used to illustrate whether the data were normally distributed. Normal distribution statistics are expressed as mean ± SD values, and an independent t-test or one-way ANOVA was used for comparing between groups. Otherwise, the Mann–Whitney U-test was applied for skewed distribution statistics. Count data (n,%) of the two groups were compared using the χ2 test. The correlation between the SCMM of lymphocyte subsets and laboratory parameters was assessed using the Spearman test. The receiver operating characteristic curve was used to analyze the efficacy of SCMM of lymphocyte subsets for predicting the occurrence and severity of COVID-19 based on the area under the curve (AUC), specificity, sensitivity, and Youden index. 14 Values were considered statistically significant if p < .05.
Results
Demographic and clinical characteristics
The demographic and clinical characteristics of COVID-19 patients and controls.

Continuous variables were expressed as mean ± SD or median [Q1, Q3].
Abbreviations: SOFA, Sequential Organ Failure Assessment; IL-6, interleukin-6; IL-8, interleukin-8; PaO2, partial pressure of oxygen; SPO2, arterial blood oxygen saturation; APACHE II score, Acute Physiology, and Chronic Health Evaluation II score.
Mitochondrial function analysis in lymphocytes
Comparison of mitochondrial function in lymphocyte subsets between three groups.
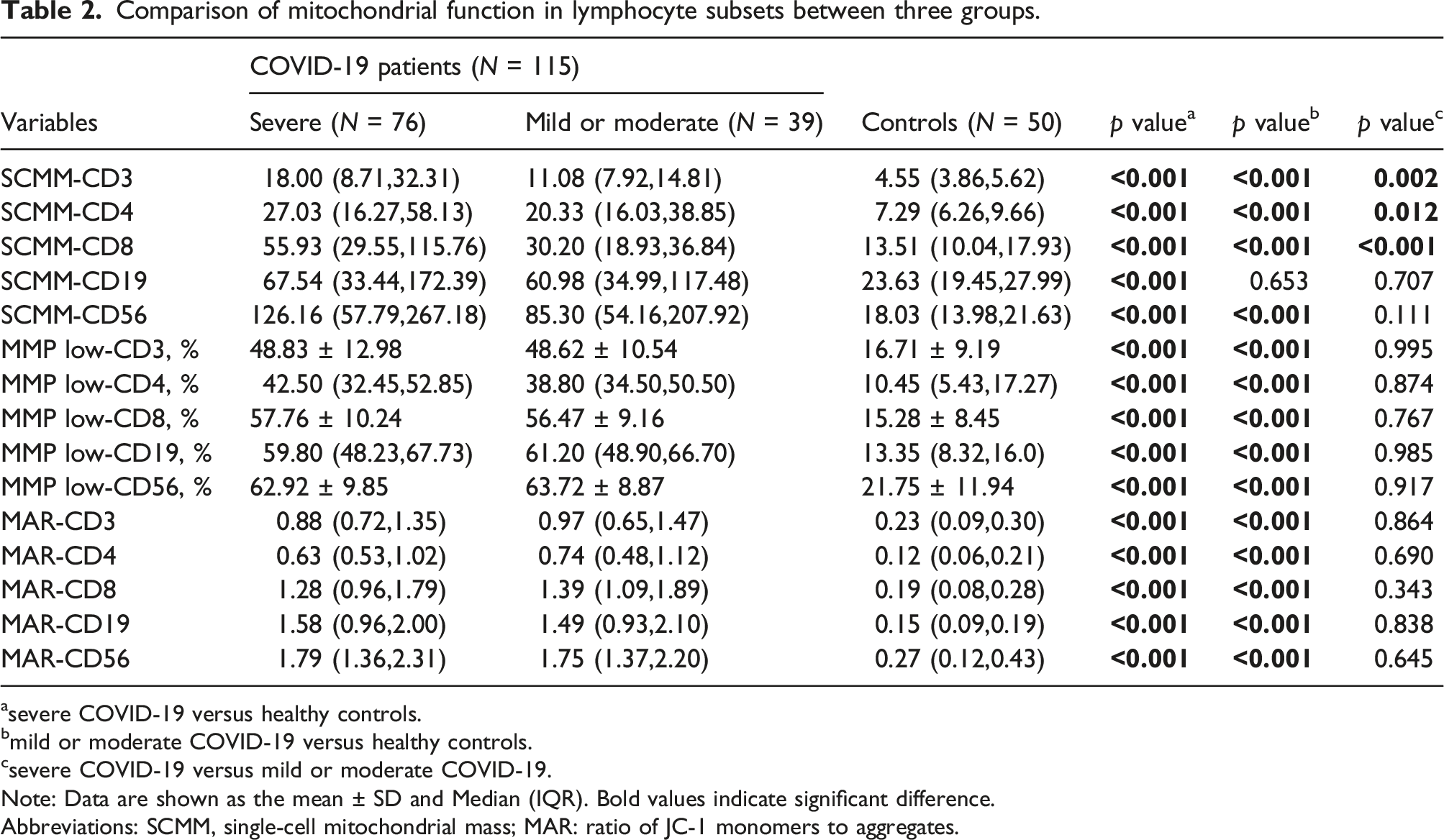
asevere COVID-19 versus healthy controls.
bmild or moderate COVID-19 versus healthy controls.
csevere COVID-19 versus mild or moderate COVID-19.
Note: Data are shown as the mean ± SD and Median (IQR). Bold values indicate significant difference.
Abbreviations: SCMM, single-cell mitochondrial mass; MAR: ratio of JC-1 monomers to aggregates.
Associations between clinical parameters and SCMM values of T cells
The correlations between laboratory parameters and mitochondrial damage in T lymphocytes.
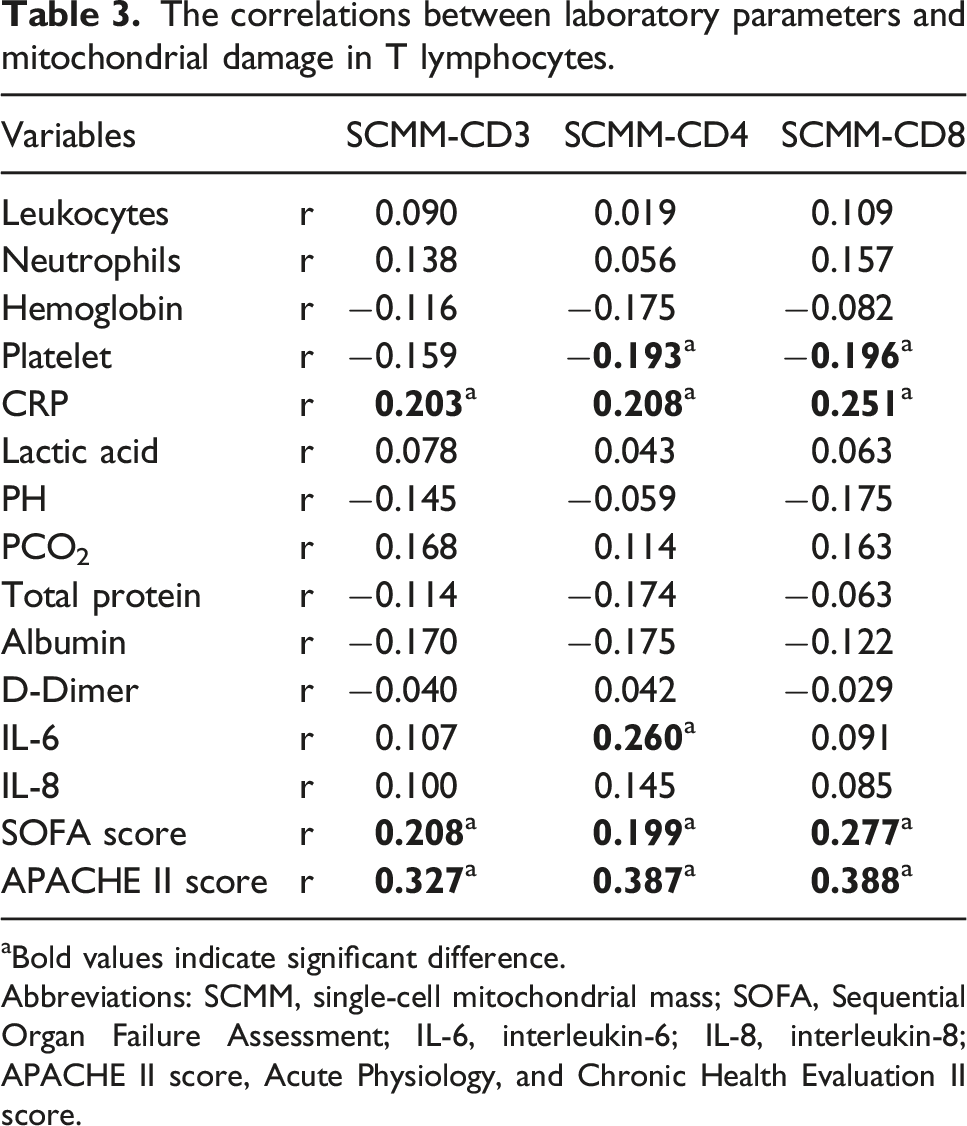
aBold values indicate significant difference.
Abbreviations: SCMM, single-cell mitochondrial mass; SOFA, Sequential Organ Failure Assessment; IL-6, interleukin-6; IL-8, interleukin-8; APACHE II score, Acute Physiology, and Chronic Health Evaluation II score.
Predictive efficacy of SCMM-CD3, SCMM-CD4, and SCMM-CD8
The diagnostic efficacy of various indicators for the severity of COVID-19 patients.

aBold values indicate significant difference.
Abbreviations: AUC, area under the curve; CRP, SCMM, single-cell mitochondrial mass.

The diagnostic performance of SCMM-CD3, SCMM-CD4, and SCMM-CD8 in predicting COVID-19 progression.
Discussion
This study provides evidence of mitochondrial damage in lymphocytes obtained from COVID-19 patients, including data showing elevated SCMM and MMP-low values. In contrast to the MMP-low level, the SCMM of T cells was significantly higher in severe COVID-19 patients, compared to mild or moderate COVID-19 patients, and was significantly correlated with the SOFA and APACHE II scores. Additionally, the SCMM of T cells exhibits improved performance in assessing COVID-19 patient severity.
In 2007, Petrovas et al. demonstrated that a deficiency in HIV-specific CD8 (+) T cell survival was accompanied by increased MM levels. 15 Subsequently, several studies have reported the association of MM of T cells with disease progression. Higher levels of SCMM in peripheral blood T lymphocytes were observed during the acute relapse of schizophrenia. The SCMM exhibited a good performance in predicting the response to treatment for schizophrenia. 16 In addition, lymphocyte dysfunction occurs in T cells during the early stages of sepsis. SCMM-CD4 is more effective for diagnosing sepsis at an early stage than the traditional biomarker PCT, and the diagnostic value of PCT, when combined with SCMM-CD4, is superior to that observed for SCMM-CD4 alone during early sepsis. 17 To our knowledge, this study is the first to examine the association between the mitochondrial function of lymphocyte subsets and COVID-19 risk and might provide guidance to researchers conducting future studies in this field. Our results revealed that the mitochondrial dysfunction of lymphocyte subsets, especially T cells, occurred in COVID-19 patients. An elevated MM level was associated with impaired mitochondrial autophagy, while the MMP level decreased before the early pathological changes associated with apoptosis. Therefore, we hypothesized that lymphocytes from COVID-19 patients exhibited impaired mitochondrial autophagy and increased early apoptosis, leading to a significant decrease in the absolute number of lymphocyte subsets.
IL-6 levels were significantly higher in the severe and dead groups of COVID-19 patients and negatively correlated with lymphocyte subsets counts. 18 IL-6 is an adequate predictor of severe disease in patients infected with the COVID-19. 19 There is a positive correlation between SCMM-CD4 and IL-6. This may be attributed to a decrease in the absolute number of CD4+T cells, which leads to an increased risk of opportunistic infections and a tendency to evolve into severe disease. Additionally, SCMM-CD8 was significantly elevated in the severe COVID-19 group and outperformed SCMM-CD3 and SCMM-CD4 for distinguishing between COVID-19 patients with mild and severe disease. Petrovas et al. found that there was remarkably higher MM in HIV-specific CD8+T cell from HIV patients and MM (High) phenotype was sensitive to spontaneous and CD95/Fas –induced apoptosis. 15 Therefore, mitochondrial dysfunction resulted in the decreased survival of CD8+T cells. There are many similarities between COVID-19 and sepsis. Tang et al. demonstrated that a low-CD8+ T cell count was a risk factor for sepsis progression. 20 The mitochondrial dysfunction of CD8+T cells may be an early indicator of severe COVID-19. When the cutoff value is 38.775, the AUC, sensitivity, and specificity are 0.731, 65.8%, and 82.1%, respectively. There was no significant difference between mild and severe patients with regard to SCMM-CD19 and SCMM-CD56. Lymphocytes upregulate glycolysis and mitochondrial oxidative phosphorylation (OxPhos) during their activation. 21 The process of metabolic reprogramming occurs differently between B cells and T cells, and also within regulatory T cell subsets. 21 For instance, naïve murine splenic CD4+ T cells upregulate glycolysis and glucose uptake and reveal a strongly enhanced ratio of glycolysis versus OxPhos upon stimulation with anti-CD3/CD28. In contrast, B cells upregulate both glycolysis and OxPhos upon stimulation with LPS or anti-B-cell receptor, and maintain the glycolysis/OxPhos ratio in resting B cells. 22
Díaz-Resendiz et al. compare the MMP in PBMC isolated from healthy control (HC) subjects, patients with COVID-19 (C-19), recovered subjects at 40 ± 13 (R1) and 335 ± 20 (R2) days after infection. They found that MMP decreased in Human PBMC of subjects with C-19, R1, and R2 compared with HC. 23 In this study, the MMP-low values of lymphocyte subsets were significantly higher in the case group than in the control group (p < .001). Possible explanations for the decrease in MMP in COVID-19 patients are as follows. Individuals recovering from moderate- and critical illness also exhibit higher plasma medium-chain acylcarnitines and lower levels of circulating TCA cycle intermediates such as succinate. 24 A decrease in TCA cycle intermediates in neocoronary patients may lead to an obstacle in the electron transfer process in the mitochondrial respiratory chain, which affects the formation of a transmembrane gradient of protons (H+) within the matrix, and this leads to a decrease in the outer positive and inner negative mitochondrial membrane potential, that is, depolarization. Reduced intermediates of the TCA cycle may directly reflect mitochondrial damage.
Several limitations are associated with this study. First, the sample size/power analysis was not performed for this study, thus the relatively small sample size might undermine significant relationships. Therefore, the positive results of this study are preliminary and need to be verified in a multi-center study with large samples. Second, our study focused on the mitochondrial function at admission and did not dynamically monitor the mitochondrial damage in lymphocytes. Third, other confounding factors influencing mitochondrial function might have led to certain inaccuracies. Fourth, the mitochondrial damage to the lymphocytes in peripheral blood might only be an indirect indicator of the pathological changes in the lung. Fifth, we cannot evaluate whether high MM corresponds to larger organelles, which would lead to higher levels of staining.
Conclusion
In conclusion, the level of mitochondrial damage in T lymphocytes in the peripheral blood was significantly more severe in COVID-19 patients, as compared to healthy controls, and was positively associated with the SOFA and APACHE II scores. An increase in the circulating levels of SCMM-CD8 can help us distinguish between patients with mild and severe COVID-19 infections. Therefore, an assessment of the mitochondrial function may indicate disease progression and help optimize treatment strategies.
Supplemental Material
Supplemental Material - Analysis of mitochondrial function in lymphocytes obtained from COVID-19 patients
Supplemental Material for Analysis of mitochondrial function in lymphocytes obtained from COVID-19 patients by Zhi He, Jing-Jing Liu, and Shao-Lei Ma in International Journal of Immunopathology and Pharmacology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
