Abstract
Introduction
Type I hypersensitivity is a representative T helper 2 (Th2) cell immune response that reacts excessively to antigens introduced from the outside and includes various allergic diseases such as allergic asthma, dermatitis, rhinitis, anaphylaxis, food allergy, and drug allergy. 1 In these diseases, mast cells, derived from hematopoietic stem cells, play a crucial role in inducing allergic symptoms. Mast cell progenitors migrate through the blood to peripheral tissues, such as the skin and mucosa, and complete the process of maturation. 2 Mast cells are distributed throughout the body and are abundant at sites where pathogens or harmful substances from the external environment meet the host, such as the skin, gastrointestinal tract, and respiratory mucosa, which are located near nerves, smooth muscle cells, blood vessels, mucus-producing glands, and hair follicles in the adjacent connective tissue.3,4 When mast cells are activated by antigens, they release mediators, such as histamine, prostaglandins, leukotrienes, and platelet-activating factor, and cytokines, such as IL-3, IL-4, IL-5, IL-13, and tumor necrosis factor (TNF)-α. These allergic mediators increase vascular permeability, peripheral vasodilation, and smooth muscle contraction, and facilitate the migration of various immune cells, including Th2 cells, thereby increasing allergic symptoms.5,6
IgE generated by antigens from the exterior binds to the α-subunit of FcεRI in mast cells, leading to mast cell activation when the same antigen is re-exposed. 7 Crosslinking of IgE on the surface of mast cells by antigen activates the Src family kinase Lyn. Activated Lyn phosphorylates the immunoreceptor tyrosine-based activating motifs (ITAMs) of FcεRI β and γ chains. The phosphorylated ITAMs of FcεRⅠ β and γ chains serve as docking sites for the ZAP70-related tyrosine kinase Syk. Syk can increase its catalytic activity through trans- and auto-phosphorylation, in addition to its phosphorylation by Lyn.8,9 Activated Syk phosphorylates the essential adaptor protein linker for the activation of T cells (LAT), which plays a crucial role in activating downstream signaling for the release of various pro-inflammatory mediators. Phosphorylated LAT recruits various proteins, including cytosolic adaptor molecules such as GRB2, SHC, and SLP76, and signaling enzymes such as PLCγ1. 10 PLCγ1 hydrolyzes phosphatidylinositol bisphosphate in the plasma membrane to inositol triphosphate and diacylglycerol. Inositol triphosphate induces the mobilization of cytosolic calcium, whereas diacylglycerol induces the activation of PKC, leading to mast cell degranulation. Other signaling pathways are also activated by PI3K, Akt, and mitogen-activated protein kinases (MAPKs) such as JNK, p38, and Erk1/2. MAPK activation activates transcription factors, including Fos, Jun, nuclear factor of activated T cells, and nuclear factor-κB, increasing the generation of inflammatory cytokines such as IL-4 and IL-13. 11
Nifuratel, 5-[(methylthio)methyl]-3-[(5-nitrofurfurylidene) amino]-2-oxozolidinone, is a nitrofuran derivative with anti-protozoal, anti-fungal, and anti-bacterial activities. Nifuratel is used to remove microorganisms associated with infections of the genitourinary tract, such as Trichomonas vaginalis, Gardnerella vaginalis, aerobic and anaerobic bacteria, Candida spp., Chlamydia trachomatis, and Mycoplasma spp.12,13 Nifuratel is a drug with relatively few side effects and has been demonstrated as non-toxic after oral or intravaginal administration in mice and rats. 14 Nifuratel is used alone or in combination with nystatin for the treatment of vulvovaginal infections. 15 However, there are currently no reports on the efficacy of nifuratel for the treatment of type I allergic diseases. Therefore, the present study aimed to investigate the potential of nifuratel to treat mast cell–associated allergic disorders.
Materials and methods
Antibodies and reagents
The chemical compound nifuratel (structurally represented as 5-[(methylthio)methyl]-3-[(5-nitrofurfurylidene) amino]-2-oxozolidinone, Figure 1) was procured from MedChemExpress (Monmouth Junction, NJ, USA). The chemical 4-amino-5-(4-chlorophenyl)-7-(dimethylethyl) pyrazolo [3,4-d] pyrimidine (PP2) was obtained from Calbiochem (La Jolla, CA, USA). Monoclonal dinitrophenol-specific IgE, DNP-human serum albumin (HSA), Evans blue, toluidine blue O, cetirizine, ionomycin, and thapsigargin were purchased from Sigma-Aldrich (St. Louis, MO, USA). Antibodies against the phosphorylated forms of Syk (Tyr352, 1:1000), LAT (Tyr191, 1:1000), Akt (Ser473, 1:1000), ERK1/2 (Thr202/Tyr204, 1:1000), JNK (Thr183/Tyr185, 1:1000), p38 (Thr180/Tyr182, 1:1000), and β-actin:HRP-conjugated (1:5000) were purchased from Cell Signaling Technology (Danvers, MA, USA). Antibodies against Syk (1:200), Akt (1:200), ERK1/2 (1:200), JNK (1:200), p38 (1:200), FcεRⅠγ (1:200), and Protein A/G PLUS-Agarose were obtained from Santa Cruz Biotechnology (Dallas, TX, USA). The antibody against LAT (1:1000) was obtained from EMD Millipore (Billerica, MA, USA). The medium used for cell culture was acquired from Welgene (Gyeongsangbuk-do, Korea). Molecular structure of nifuratel.
Animals
A cohort of male Balb/c mice, aged 5 weeks, was obtained from Orient Bio (Gyeonggi-do, Korea). The study was approved by the Institutional Animal Care and Use Committee (IACUC) of Konkuk University (permit number KU22156). All animal experiments were performed in accordance with the institutional guidelines.
Cultures of rat basophilic leukemia (RBL)-2H3 cells and bone marrow mast cells (BMMCs)
RBL-2H3 cells were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultivated in complete minimal essential medium (MEM) supplemented with L-glutamine (100 Units/ml), penicillin (100 μg/mL), streptomycin (100 μg/mL), and 15% fetal bovine serum at 37°C in a CO2-enriched environment. Bone marrow cells were harvested from the tibias and femurs of 5-week-old mice for BMMC culture and were cultured in complete RPMI 1640 medium containing 4 mM L-glutamine, 100 Units/ml penicillin, 100 μg/mL streptomycin, 1 mM sodium pyruvate, 0.1 mM non-essential amino acids, 25 mM HEPES, 50 mM 2-mercaptoethanol, 10% FBS, and 10 ng/mL IL-3. After 4-5 weeks, the purity of BMMCs was evaluated using FcεRI and c-Kit expression as mast cell maturation markers. Only cells with purity greater than 95% were used in subsequent experiments.
β-Hexosaminidase assay
RBL-2H3 cells (2.0 × 105 cells/well) were sensitized overnight with 20 ng/mL DNP-specific IgE in the culture medium. BMMCs (3.0 × 105 cells/well) were sensitized with 100 ng/mL DNP-specific IgE for 4 h in the culture medium. The cells were then transferred to Siraganian buffer (pH 7.2, containing 119 mM NaCl, 5 mM KCl, 40 mM NaOH, 25 mM PIPES, 5.6 mM glucose, 4 mM MgCl2, 1 mM CaCl2, and 0.5% BSA) for RBL-2H3 cells or Tyrode buffer (pH 7.4, containing 135 mM NaCl, 5 mM KCl, 20 mM HEPES, 5.6 mM glucose, 1.8 mM CaCl2, 1 mM MgCl2, and 0.5% BSA) for BMMCs. Nifuratel was prepared in each buffer solution. The cells were incubated with or without nifuratel for 30 min and then stimulated with 25 ng/mL (RBL-2H3 cells) or 50 ng/mL (BMMCs) of DNP-HSA for 15 min at 37°C. For mast cell stimulation using ionomycin or thapsigargin, BMMCs (3.0 × 105 cells/well) were incubated with nifuratel for 30 min without IgE sensitization and then stimulated with 1 μM ionomycin or 300 nM thapsigargin for 15 min. Thirty microliters of supernatant and 30 μL of p-nitrophenyl N-acetyl-β-
Cell viability assay
RBL-2H3 cells (2 × 104 cells/well) were cultured in 96-well plates for 12 h, followed by treatment with nifuratel for 4 h. BMMCs (2 × 104 cells/well) were incubated in 96-well plates with nifuratel for 4 h. Cell viability was evaluated using the Cell Counting Kit-8 (CCK-8) (Dojindo Laboratories, Kumamoto, Japan) in accordance with the manufacturer’s protocol. A viability assay was conducted and the absorbance was measured at 450 nm to determine cell viability.
Assessment of histamine release from MCs
BMMCs (1.0 × 106 cells/well) were sensitized with 100 ng/mL of DNP-specific IgE for 4 h. Subsequently, the cells were thoroughly washed with fresh culture medium, and then treated with nifuratel or PP2 for 30 min at 37°C. The cells were then stimulated with 50 ng/mL DNP-HSA for 15 min in culture media. The amount of histamine in supernatant were measured using Abcam Histamine ELISA kit (Cambridge, MA, UK).
Enzyme-linked immunosorbent assay (ELISA)
RBL-2H3 cells (8 × 105 cells/well) were sensitized overnight with 20 ng/mL DNP-IgE. BMMCs (1.0 × 106 cells/well) were similarly sensitized with 100 ng/mL of DNP-specific IgE for 4 h. Subsequently, the cells were thoroughly washed with fresh culture medium, and then treated with nifuratel or PP2 for 30 min. The cells were then stimulated with 25 ng/mL (for RBL-2H3 cells) or 50 ng/mL (for BMMCs) DNP-HSA for 4 h at 37°C. The concentrations of TNF-α and IL-4 in the resulting culture medium were measured using Rat OptEIA ELISA kits from BD Biosciences (San Jose, CA, USA) or TNF alpha Mouse ELISA Kit from Invitrogen (Waltham, MA, USA) according to the manufacturer’s protocol.
Western blot and immunoprecipitation
RBL-2H3 cells (8.0 × 105 cells/well) were sensitized with 20 ng/mL of DNP-specific IgE in a culture medium and incubated overnight. BMMCs (2.0 × 106 cells/well) were similarly sensitized with 100 ng/mL of DNP-specific IgE for 4 h. Subsequently, the cells were thoroughly washed with fresh culture medium, and then treated with nifuratel or PP2 for 30 min. The cells were then stimulated with 25 ng/mL (for RBL-2H3 cells) or 50 ng/mL (for BMMCs) DNP-HSA for 15 min at 37°C. Upon completion of the reaction, it was promptly terminated by placing the sample on ice. RBL-2H3 cells were lysed using a carefully formulated lysis buffer consisting of 70 μL of solution (pH 7.5), comprising 20 mM HEPES, 150 mM NaCl, 1% Nonidet P–40, 10% glycerol, 60 mM octyl β-glucoside, 10 mM NaF, 1 mM Na3VO4, 1 mM phenylmethylsulfonyl fluoride, 2.5 mM nitrophenyl phosphate, 0.7 mg/mL pepstatin, and a protease-inhibitor cocktail tablet. The BMMCs were lysed using RIPA buffer (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 1 mM PMSF, 0.7 μg/mL pepstatin, and a protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, USA). The resulting cell lysates were subjected to high-speed centrifugation at 13,000 rpm/min for 10 min, after which the supernatant was mixed with 4× Nu-PAGETM sample buffer (Thermo Fisher Scientific, Waltham, MA, USA) and denatured by heating for 5 min in a 96°C heating block. For immunoprecipitation, BMMCs (1.0 × 107 cells/well) were sensitized with 100 ng/mL of DNP-specific IgE for 4 h. Subsequently, the cells were treated with or without nifuratel for 30 min and then stimulated with 50 ng/mL DNP-HSA for 5 min at 37°C. After the reaction, the cells were washed with Dulbecco’s phosphate buffered saline (DPBS) and lysed using IP Lysis Buffer (Thermo Fisher Scientific, Waltham, MA, USA). Lysates were centrifugated at 13,000 rpm/min for 10 min, and then the supernatants were treated with the antibody against FcεRⅠγ for 3 h at 4°C. Protein-Ab complexes were added into 40 μL of protein A/G agarose and incubated at 4°C for 4 h. The agarose complexes were washed twice with DPBS, mixed 2× Nu-PAGETM sample buffer, and denatured at 100°C for 10 min. Proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred to a polyvinylidene fluoride membrane using transfer cassettes (Bio-Rad, Hercules, CA, USA). The membrane was then incubated with specific primary antibodies in a Tris-buffered saline containing 0.1% Tween 20 containing 5% BSA or skim milk. Subsequently, the membrane was incubated with a horseradish peroxidase-labeled secondary antibody. Protein bands were detected using an enhanced Luminata Crescendo Western HRP substrate (EMD Millipore Corp., Billerica, MA, USA). The protein bands were extracted horizontally, and a molecular weight marker was used as a reference to determine their positions.
Passive cutaneous anaphylaxis (PCA)
Male Balb/c mice, approximately 6 weeks of age, were subjected to intradermal injections of 50 ng of DNP-specific IgE suspended in 10 μL of PBS into the ear skin. Twelve hours later, the mice were administered oral doses of nifuratel, in doses of 10, 30, or 100 mg/kg, suspended in 5% Arabic gum (Sigma-Aldrich, St. Louis, MO, USA). One hour after oral administration, mice were intravenously injected with 100 μg of DNP-HSA suspended in 250 μL of a solution containing 5 mg/mL Evans blue. One hour after antigen challenge, the mice were humanely euthanized, and the ear tissue was removed. The extracted dye from the ear tissue was analyzed by measuring its absorbance at 620 nm after incubation in 1 mL of formamide at 63°C for 24 h.
Histologic analysis
After the PCA reaction, the ear skin of the mice was fixed with a solution of 4% paraformaldehyde in PBS. Fixed skin was dehydrated with ethanol and embedded in paraffin. Serial sections (5 μm in thickness) were cut from paraffin-embedded tissues and stained with toluidine blue. To determine the percentage of degranulated mast cells in ear tissues, representative images used for evaluation of degranulation were obtained at a total magnification of ×400. The total number of mast cells and their degranulation were counted on 5-7 sites randomly chosen among 5-7 sections per ear tissue (n = 5 mice). The data were expressed as the ratio of the number of degranulated mast cells to the total number of mast cells present in the tissue.
In vitro kinase activity assay
Lyn and Fyn kinase assay based on radiometry was performed at Eurofins (Celle-Lévescault, France) using KinaseProfilerTM. Eurofins kinase enzymatic radiometric assays were carried out at the Km = ATP in dose response for each kinase. Briefly, Lyn (mouse) is incubated with 50 mM Tris pH 7.5, 0.1 mM EGTA, 0.1 mM Na3VO4, 0.1% β-Mercaptoethanol, 0.1 mg/mL Poly (Glu, Tyr) 4:1, 10 mM Magnesium acetate and [γ33P]-ATP. Fyn (human) is incubated with 50 mM Tris pH 7.5, 0.1 mM EGTA, 0.1 mM Na3VO4, 250 μM KVEKIGEGTYGVVYK (Cdc2 peptide), 10 mM Magnesium acetate and [γ33P]-ATP. To check the activity of Lyn or Fyn, the reaction was initiated by the addition of the Mg/ATP mix. After incubation for 40 min at room temperature, the reaction was stopped by the addition of 0.5% phosphoric acid. Ten μL of the reaction mixture wass then spotted onto a Filtermat A and washed four times for 4 min in 0.42% phosphoric acid and once in methanol prior to drying and scintillation counting.
Statistical analysis
Data are presented as mean ± standard error of the mean obtained from three independent experiments. In each animal experiment, a minimum of five mice were used per experimental group. The in vitro experiments were conducted in triplicates for each trial. Statistical analysis was performed using a one-way analysis of variance and Student’s t-test for unpaired data. Statistical significance was set at P < 0.05 or P < 0.01 and all statistical computations were performed using GraphPad Prism 7 software (GraphPad Software Inc., San Diego, CA). The IC50 values were calculated using the AAT bioquest IC50 calculator (https://www.aatbio.com/tools/ic50-calculator).
Results
Effect of nifuratel on mast cell degranulation stimulated by antigen
Mast cells contain many granules, including histamine, serotonin, platelet-activating factors, and other proinflammatory mediators. To determine the effect of nifuratel on the degranulation of mast cells induced by antigen stimulation, we measured the release of granule marker β-hexosaminidase from mast cells after antigen stimulation with or without nifuratel. Nifuratel inhibited the degranulation induced by antigen in a concentration-dependent manner in both RBL-2H3 cells and BMMCs (Figure 2b, IC50, approximately 0.3 μM for RBL-2H3 cells; approximately 0.9 μM for BMMCs). In addition, we investigated the impact of nifuratel on the release of histamine, a type I allergy-inducing mediator, in mast cells. Similar to the finding illustrated in Figure 2b, nifuratel suppressed histamine secretion in mast cells by antigen in a dose-dependent manner (Figure 2c). To check reversibility of the inhibitory effect of nifuratel on mast cell degranulation, RBL-2H3 cells were incubated with nifuratel (3 μM) for 30 min and then subjected to three washing steps with Siraganian buffer before antigen stimulation. After washing, the inhibitory effect of nifuratel disappeared, and the degranulation induced by antigen stimulation was recovered (Figure 2d). Nifuratel showed no cytotoxicity at the highest concentrations used in this experiment (Figure 2a). These results suggest that nifuratel, currently used as an anti-microbial drug for genitourinary tract infections, may have new efficacy as a mast cell–dependent allergy therapy. Nifuratel suppresses antigen-induced degranulation in mast cells. (a) The effect of nifuratel on cell viability of RBL-2H3 cells and BMMCs was evaluated by incubating the cells with nifuratel for 4 h and measuring the absorbance at 450 nm using the CCK-8 kit. (b) RBL-2H3 cells and BMMCs, which were sensitized with DNP-specific IgE, were treated with nifuratel or PP2 for 30 min and then stimulated with DNP-HSA for 15 min. (c) DNP-specific IgE primed BMMCs (1.0 × 106 cells/well) were treated with nifuratel for 30 min and then stimulated with 50 ng/mL DNP-HSA for 15 min. The measurement of histamine secretion was performed as described in “Materials and methods”. (d) RBL-2H3 cells were incubated with or without nifuratel for 30 min, washed three times, and then stimulated with antigen for 15 min. (b, d) The extent of degranulation from the mast cells was determined by the ratio of β-hexosaminidase activity released outside the cells to the total β-hexosaminidase activity. The results are the mean ± SEM from three independent experiments with triplicate measurements. Statistical significance is indicated by asterisks; *p < 0.05, **p < 0.01. PP2 is a general Src-family kinase inhibitor.
Effect of nifuratel on the secretion of inflammatory cytokines in mast cells
In addition to degranulation, release of inflammatory cytokines is another response that occurs in mast cells during IgE-mediated allergic reactions. Cytokines produced by antigen stimulation in mast cells include TNF-α and IL-4. TNF-α plays a major role in inducing late-phase allergic reactions by recruiting leukocytes, such as neutrophils and eosinophils, to the site of inflammation.
16
IL-4, a typical Th2 cytokine, is essential for allergic reactions because it induces leukotriene synthesis and promotes mast cell proliferation and survival.
17
Using ELISA, we measured the release of inflammatory cytokines from mast cells following antigen stimulation. Nifuratel inhibited the release of the inflammatory cytokines TNF-α and IL-4 in antigen-stimulated RBL-2H3 cells (Figure 3a–b, IC50, approximately 0.48 and 0.75 μM for TNF-α and IL-4, respectively). Furthermore, we observed that nifuratel suppressed the secretion of TNF-α in BMMCs in response to antigen stimulation (Figure 3c, IC50, approximately 3.01 μM). These results indicate that nifuratel may have the ability to reduce late-phase allergy symptoms by blocking the secretion of inflammatory cytokines from mast cells. Nifuratel suppresses the release of inflammatory cytokines in RBL-2H3 cells and BMMCs by antigen. (a, b) RBL-2H3 cells (8.0 × 105 cells/well) sensitized with DNP-specific IgE were treated with nifuratel or PP2 for 30 min and then stimulated with 25 ng/mL DNP-HSA for 3 h. (c) BMMCs (1.0 × 106 cells/well) sensitized with DNP-specific IgE were treated with nifuratel or PP2 for 30 min and then stimulated with 50 ng/mL DNP-HSA for 3 h. After 3 h of incubation, the amount of IL-4 and TNF-α released into the culture media was measured by ELISA. The values obtained from three independent experiments are shown as mean ± SEM. Statistical significance is indicated by asterisks; *p < 0.05, **p < 0.01. PP2 is a general Src-family kinase inhibitor.
Mechanism of nifuratel for inhibition of mast cells
Next, we investigated the mechanism by which nifuratel inhibits antigen stimulation-induced mast cell activation. Syk is a well-known protein that plays a crucial role in the IgE-mediated signaling pathway of mast cells. When mast cells are activated by an antigen, Syk is phosphorylated by binding to the ITAMs of FcεRI γ-subunit.
18
In RBL-2H3 cells, nifuratel inhibited the activation of Syk and its downstream signaling molecule, LAT, upon antigen stimulation in a concentration-dependent manner (Figure 4a–b). Akt and MAPKs (Erk1/2, JNK, and p38) play important roles in mast cell survival and the synthesis and release of inflammatory cytokines.
19
Nifuratel suppressed the phosphorylation of Akt, ERK1/2, JNK, and p38 in response to antigen stimulation in RBL-2H3 cells in a concentration-dependent manner (Figure 4a–b). Compared to the group treated with antigen only, nifuratel almost completely inhibited the Syk and Syk-mediated signaling pathways at a concentration of 3 μM. Additionally, nifuratel also inhibited the Syk and LAT signaling pathways in BMMCs (Figure 4c–d). These results demonstrate that nifuratel inhibits mast cell activation by suppressing Syk activaiton in FcεRI-mediated signaling in response to antigen stimulation in mast cells. Nifuratel inhibits the activation of IgE-mediated signaling proteins in mast cells stimulated by antigens. DNP-specific IgE primed RBL-2H3 cells (a and b, 8.0 × 105 cells/well) or BMMCs (c and d, 2.0 × 106 cells/well) were treated with nifuratel for 30 min and then stimulated with 25 ng/mL and 50 ng/mL DNP-HSA, respectively, for 15 min. The reaction was stopped by placing on ice, and then, the cells were washed with PBS and lysed. Western blotting was performed as described in the “Materials and methods” section. The representative images (a and c) and band densities (b and d) of phosphorylated proteins were obtained from three independent experiments. Densitometric analysis of phosphorylated proteins was performed using Multi Gauge Ver 3.0 (FUJIFILM, Valhalla, NY, USA). The values indicate the mean ± SEM from three independent experiments. The asterisks indicate significant differences from antigen-stimulated groups without inhibitors; *p < 0.05, **p < 0.01. PP2 is a general Src-family kinase inhibitor.
Inhibitory effect of nifuratel on antigen-induced PCA in mice
We previously observed that nifuratel curtails the release of allergy-inducing substances from mast cells in response to antigen stimulation. To confirm its inhibitory effect on in vivo allergic reactions caused by mast cell activation, we employed a widely recognized IgE-dependent PCA mouse model, a type 1 hypersensitivity immune response. IgE (50 ng) was intradermally injected into each ear of the mouse, followed by oral administration of nifuratel (10, 30, or 100 mg/kg) and cetirizine (50 mg/kg) the following day. One hour after oral administration, the mice received an intravenous injection of 100 μg antigen. The PCA response in the ears of mice treated with nifuratel was visibly reduced compared to that in mice that received only the antigen (Figure 5a). Furthermore, a dose-dependent reduction in Evans blue dye extracted from the ears of nifuratel-treated mice was observed compared to the mice that received only the antigen (Figure 5b, ED50, approximately 22 mg/kg). Finally, histological analysis of ear tissue from PCA-induced mice and measurement of mast cell degranulation using toluidine blue O staining showed a dose-dependent reduction in the proportion of mast cell degranulation in nifuratel-treated ear tissue (Figure 5c). Collectively, our results indicate that nifuratel has an inhibitory effect on IgE-dependent PCA reactions by antigen in mice. Nifuratel mitigates IgE-mediated passive cutaneous anaphylaxis (PCA) reaction. Mice were subjected to intradermal injection of IgE (50 ng) in their ear. Subsequently, they were administered orally with nifuratel (10, 30, or 100 mg/kg) and cetirizine (CZ, 50 mg/kg) and, after 1 h, underwent antigen challenge containing Evans blue. The images in (a) depict representative ear images for each group. The quantity of Evans blue leaked from the ears, as measured via formamide extraction, is presented at 620 nm in (b). (c) The mice ears from the experiment depicted in (a) underwent histological analysis, with ear sections being stained with toluidine blue and total and degranulated mast cells being counted. The scale bar in histological images represents 10 μm. The red arrows indicate degranulated mast cells. The representative images were obtained at a total magnification of ×400. The representative images (a, c) and values (b, c) were generated from three independent experiments. The values (b and c) are presented as mean ± SEM. Significance was indicated by asterisks, with *p < 0.05 and **p < 0.01.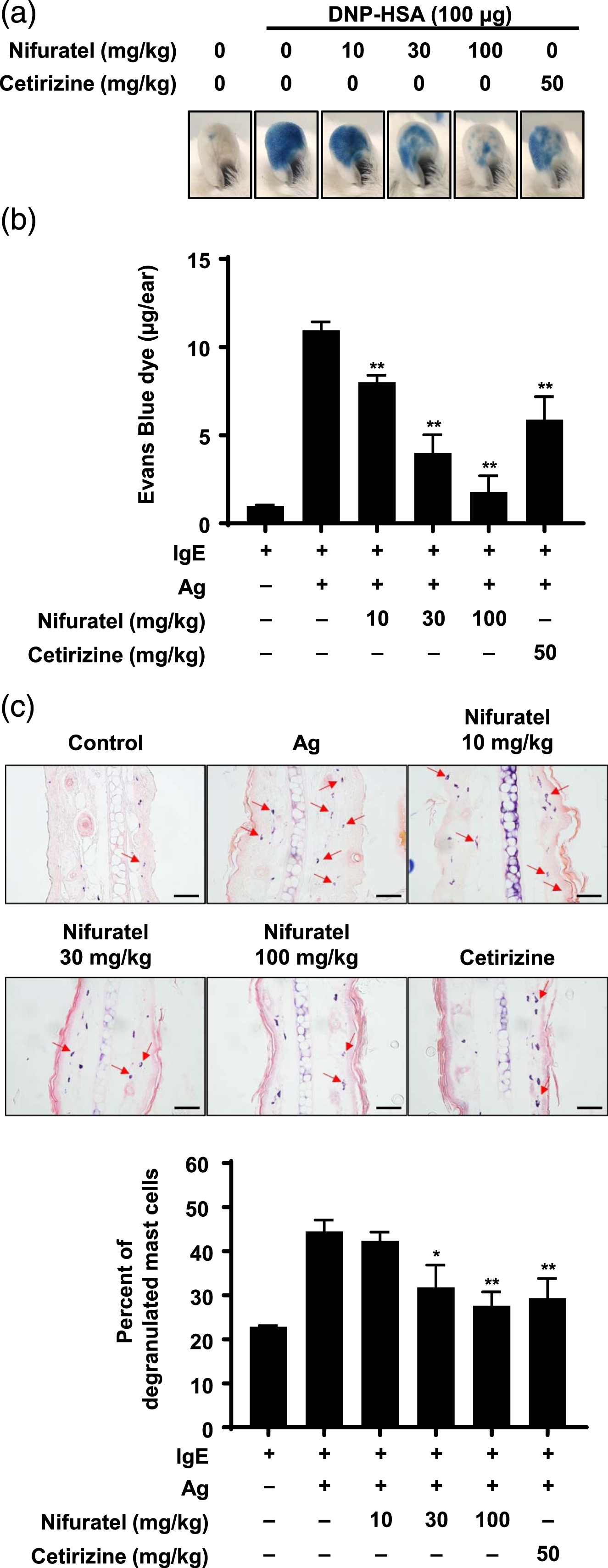
Suppression of the Syk and FcεRⅠγ association by nifuratel during antigen stimulation
Nifuratel inhibited Syk and its downstream signaling pathways in FcεRⅠ signaling pathways activated by antigen (Figure 4). To investigate the further detail mechanism by which nifuratel inhibits FcεRⅠ signaling pathways, we initially conducted the in vitro kinase activity analysis with Lyn and Fyn, which play essential roles in early signaling events of FcεRⅠ. As a result of the dose-response assay, nifuratel did not affect Fyn and Lyn kinase activity (Figure 6a), suggesting that nifuratel doesn’t regulate the upstream kinase of Syk, at least, in our experimental condition. The PLCɣ/calcium signal axis constitutes the fundamental downstream signal of Syk in mast cell degranulation.
7
Therefore, we conducted experiments to ascertain the potential inhibitory effects of nifuratel on degranulation induced by calcium inducers. Our experimental findings revealed that nifuratel did not exhibit any inhibitory activity on mast cell degranulation by ionomycin or thapsigargin (Figure 6b). These results strongly imply that nifuratel exerts its inhibitory action on the upstream signal of the PLCɣ/calcium signaling pathway. Next, for activation of Syk in MCs stimulated by antigen, the recruitment of cytosolic Syk to FcεRⅠγ is a crucial event. Therefore, we investigated the effect of nifuratel on the recruitment of Syk to FcεRⅠγ by antigen using immunoprecipitation. The results showed that the recruitment Syk to FcεRⅠγ was remarkably inhibited by nifuratel (Figure 6c). Our results suggest that nifuratel inhibits IgE-mediated MC activation by suppressing the recruitment of Syk to FcεRⅠγ. Nifuratel suppresses the recruitment of Syk to FcεRⅠγ in mast cells. (a) KinaseProfiler radiometric protein kinase assays were performed with Lyn or Fyn kinase. Kinase assay protocols were described in the “Materials and methods” section. (b) The BMMCs (3.0 × 105 cells/well) were pre-incubated with or without nifuratel (0.3, 1, 3 μM) for 30 min and then stimulated with 1 μM ionomycin or 300 nM thapsigargin for 15 min. (C) DNP-specific IgE-primed BMMCs (1.0 × 107 cells/well) were treated with nifuratel for 30 min and then stimulated with 50 ng/mL DNP-HSA for 5 min. The reaction was stopped by placing on ice, and then, the cells were washed with PBS and lysed. Immunoprecipitation was performed as described in the “Materials and methods” section. (a, b) The values are expressed as the mean ± S.E.M. from two independent experiments. (c) The representative images were generated from three independent experiments.
Discussion
Allergy is a serious disease that is increasing globally, with 20%-30% of the global population suffering from allergic diseases. 20 The prevalence of allergic diseases in the European population is >25%. 21 Allergens that cause allergies include various foreign materials, such as pollen, house dust mites, animal dander, foods, drugs, and insect venoms. Allergic diseases are largely associated with an IgE-dependent immune response, which is primarily manifested in the mucosa (gastrointestinal tract, airway, and nose) or skin surface, with various symptoms appearing depending on the site of contact with the antigen. These include food allergy, allergic dermatitis, allergic rhinitis, and asthma. 22 Allergic anaphylaxis, which is closely linked to mast cell activation, is a life-threatening acute and systemic reaction that can cause death within minutes. 23 This has led many researchers worldwide to focus on developing allergy treatments that inhibit mast cells.24,25
Researchers are working continuously to reduce the discomfort and threat of allergic responses.26,27 Antihistamine drugs and glucocorticoids are commonly used to control the symptoms of allergic conditions. Epinephrine, bronchodilators, antihistamines, and glucocorticoids are used to relieve anaphylactic symptoms, and inhaled beta-agonists are primarily used for the treatment of allergic asthma. 1 Despite the efficacy of many drugs used to treat allergic diseases, side effects persist, and the drugs can lose effectiveness over time with repetitive use.28,29 Hence, it is crucial to continue searching for drugs that can alleviate allergic responses while minimizing the side effects. 30
Mast cells are important cells involved in both innate and adaptive immunity, possessing various receptors such as TLRs, NOD-like receptors, FcεRI, and MRGPRX2. 31 Mast cells can respond directly to pathogens and PAMPs by binding to or recognizing them. For example, TLR4 responds to LPS from Gram-negative bacteria by inducing the release of proinflammatory cytokines such as IL-1 and IL-6 without degranulation. 32 Immediate IgE-mediated type I hypersensitivity is an acute reaction that occurs within minutes of allergen exposure. IgE binds to FcεRI on mast cells, and basophils crosslink upon re-exposure to allergens, releasing various preformed and newly synthesized inflammatory mediators. 16 Histamine is one of the best-known preformed mediators present in mast cell granules. Histamine released from mast cells elicits bronchoconstriction, smooth muscle contraction, and vasodilation, thereby contributing to the progression of allergic inflammation from the early to the late phase.4,33 In the present study, the objective was to identify drugs with the ability to regulate mast cell activity by inhibiting activation by allergens, by exploring drugs already utilized in the treatment of other diseases, and to determine the underlying mechanisms. Nifuratel was effective in ameliorating MC activation in both in vitro and in vivo models (Figures 2 and 5). Histological analysis revealed that nifuratel inhibited the degranulation of mast cells in mouse ear tissue in a dose-dependent manner (Figure 5c). These findings indicate that nifuratel may have potential for the treatment of symptoms associated with mast cell–dependent type 1 hypersensitivity disorders, in addition to its commonly employed antimicrobial activities in the female population.
Drug repositioning refers to a strategy that seeks to uncover new uses for existing drugs. 34 The primary targets for drug repositioning efforts are drugs that have already been successfully marketed, or new drug candidates that exhibit no safety concerns in clinical trials. These candidates have already demonstrated sufficient safety in preclinical trials by reducing the risk of toxicity and shortening the drug development process. The main objective of drug repositioning is to identify the relationship between drugs and various diseases. 35 Our research focused on exploring the potential of various compounds for drug repositioning in the treatment of mast cell–dependent allergic diseases.36–38 5-Nitrofuran has been used in clinical settings for over 60 years. The furan ring system is a widely utilized skeleton for various compounds and has been observed to impart biological activities, including antibacterial, anti-inflammatory, and antifungal effects.39,40 Nifuratel belongs to the nitrofuran drug class and has been clinically demonstrated to be effective and safe for the treatment of bacterial vaginosis, Candida, and Trichomonas vaginalis.12,13 The mechanism underlying nifuratel activity is not fully understood, but it is known to have few side effects, with limited reports of gastrointestinal disturbance, peripheral neuropathy, and thrombocytopenic purpura. In addition, drug resistance has not been reported in nifuratel treatment. 14 In other studies, nifuratel was shown to be an effective new anti-leishmanial agent, reducing the infection of L. donovani and L. major parasites in vivo. 41 Nifuratel has also been shown to be an effective treatment for acute uncomplicated cystitis in patients after oral administration and is a good candidate for recurrent cystitis treatment. 42 Recent studies have suggested that nifuratel may have anticancer activity in gastric cancer by inhibiting STAT3 signaling. 43 However, to date, there has been no research on the inhibitory effect or mechanism of action of nifuratel on mast cell degranulation.
Mast cells contain heterogeneous granules, which release preformed mediators, such as histamine, β-hexosaminidase, and chymase, as well as newly synthesized prostaglandin D2 and E2, leukotrienes B4 and C4, and platelet-activating factors. 6 We found that nifuratel inhibited mast cell degranulation and histamine release caused by antigen at very low concentrations (Figure 2b–c). In addition to the degranulation triggered by antigen stimulation, mast cells also synthesize and release proinflammatory cytokines, especially IL-4, TNF-α, and IL-6, which play an important role in the late-phase allergic response. TNF-α released by mast cells in response to antigen stimulation contributes to the worsening of the allergic response by upregulating leukocyte adhesion molecules, such as vascular cell adhesion molecule 1 (VCAM-1) and E-selectin, in vascular endothelial cells. 33 IL-4 signaling has been shown to increase FcεRI expression and granule content, and to promote the growth of both mature mast cells and mast cell progenitors.44,45 Our experiments showed that the release of IL-4 and TNF-α from mast cells in response to antigens was inhibited by nifuratel (Figure 3). These results suggest that nifuratel can alleviate allergy symptoms not only in the acute phase but also in the late-phase allergic response.
To verify the mechanism of the inhibitory effect of nifuratel on mast cell activation, we explored which pathway is inhibited in the FcεRI-mediated signaling pathway. Nifuratel decreased the activation of Syk and its downstream signaling pathways after antigen stimulation in mast cells (Figure 4). Syk is a member of the non-receptor tyrosine kinase family and is an initial signaling protein activated in the FcεRI-mediated signaling pathway in mast cells. Research is ongoing on drugs that inhibit Syk or its upstream signaling molecules, Fyn and Lyn, which play a crucial role in the signaling pathway for mast cell activation, as a treatment for allergic diseases.46–48 In our study, contrary to what was expected, nifuratel inhibits neither Lyn and Fyn (Figure 6a), nor does it inhibit mast cell degranulation induced by ionomycin or thapsigargin (Figure 6b). On the other hand, it is known that cytosolic Syk in mast cells is recruited to the phosphorylated ITAM motif of the gamma subunit of FcεRI upon antigen stimulation, followed by phosphorylation and activation by Lyn or Fyn. Therefore, we investigated whether nifuratel inhibits the recruitment of Syk to the ɣ subunit of FcεRI. The experimental results showed an increased recruitment of cytosolic Syk to the FcεRI gamma subunit upon antigen stimulation. Under these conditions, nifuratel inhibited this recruitment of Syk in a concentration-dependent manner (Figure 6c). Taking these results into consideration, we discovered that the inhibition mechanism of nifuratel for antigen-induced anaphylaxis is not a direct inhibition of the phosphorylation of Syk by Lyn and Fyn in mast cells but the inhibition of the recruitment to the gamma subunit of FcεRI. Considering that most existing mast cell inhibitors have mechanisms based on protein kinase inhibition, which may have a high risk of side effects, the fact that nifuratel has a different mechanism raises further interest in its potential as a new drug with a novel mechanism. However, further research is needed to elucidate how nifuratel specifically inhibits the recruitment of Syk to the gamma subunit of FcεRI.
Conclusions
Nifuratel is used to treat microbial infections in humans, but its effect on mast cells has not been reported. This study demonstrates that nifuratel suppresses mast cell–mediated allergic responses to antigens in both in vitro and in vivo models. Our results indicate that nifuratel suppresses Syk in activated mast cells, leading to alleviation of the allergic response (Figure 7). Therefore, we propose nifuratel as a potential new drug candidate for the treatment of various mast cell–dependent allergic diseases. Proposed diagram illustrating the mechanism of nifuratel in mast cells.
Footnotes
Authors’ contributions
All authors have contributed significantly to the completion of this paper as follows. Ji Eon Lee: Investigation, Formal analysis, Writing - original draft. Min Yeong Choi: Investigation, Validation, Writing – original and final draft. Keun Young Min: Methodology, Formal analysis. Min Geun Jo: Investigation, Visualization, Formal analysis. Young Mi Kim: Validation, Formal analysis, Writing – review and editing. Wahn Soo Choi and Hyuk Soon Kim: Conceptualization, Data curation, Funding, Supervision, Writing – review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (NRF-2021R1A2B5B03002157).
