Abstract
Different monoclonal antibodies have been used for the treatment of Netherton’s syndrome (NS); secukinumab (anti-IL17A), infliximab (anti-TNF-α), ustekinumab (anti p40 subunit of IL-12 and IL-23), omalizumab (anti-IgE), and dupilumab (anti-IL4 and IL13). We report two sisters with severe NS who were treated with omalizumab in one and with secukinumab in the other. In view of the therapeutic failure, treatment with dupilumab was started in both sisters. The data were analyzed 16 weeks after starting treatment with dupilumab. Treatment response was assessed using the Severity Scoring Atopic Dermatitis (SCORAD); Eczema Area and Severity Index (EASI); Pruritus Numeric Rating Scale (NSR); Netherton Area Severity Assessment (NASA) and Dermatology Life Quality Index Ichthyosis. All scores were reduced after 16 weeks of treatment with dupilumab in both patients. She maintains improvement after 18 months and 12 months of treatment, respectively. No severe adverse events were reported. Treatment with dupilumab in two sisters with NS and atopic diseases produced a marked cutaneous improvement after a failed attempt with omalizumab and secukinumab. Further studies are needed to determine which biologic therapy is the most effective in NS.
Introduction
Netherton syndrome (NS) is a severe autosomal recessive skin disorder caused by mutations in the serine protease inhibitor Kazal-type 5 gene (SPINK5), which encodes the lymphoepithelial Kazal-type-related inhibitor protein (LEKTI). 1 The immunological profile of NS has recently been studied. An IL-17/IL-36 signature has been shown, with predominant complement activation and Th2 allergic response in NS type ichthyosis linearis circumflexa (NS-ILC) and Th9 allergic response in NS type scaly erythroderma (NS-SE). 2 The use of biologics for the treatment of atopic dermatitis and/or psoriasis could be a promising strategy for the treatment of NS. 3
We report two sisters with NS successfully treated with dupilumab (anti-ILR4A) after failed attempts with omalizumab (anti-IgE) in one and secukinumab (anti-IL17) in the other.
Case presentation
Without a history of consanguinity, both sisters were born with ichthyosiform erythroderma and were subsequently diagnosed with NS by genetic testing; the pathogenic variant c.2468dup p. (Lys824Glufs*4) was detected in heterozygosity in the SPINK5 gene in both patients.
Case 1
In 2015, the 28-year-old sister was reevaluated at the Allergy department due to worsening cutaneous and respiratory symptoms. She was diagnosed with severe persistent rhinoconjunctivitis and moderate persistent asthma with sensitization to house dust mites, grass pollen, fish, and shellfish. In vitro tests revealed a total IgE >5000 kU/L.
Skin examination revealed severe, extensive ILC affecting 80% of the body surface, predominantly on the trunk and upper extremities. The eyebrow hairs were sparse, and the scalp hairs were fine and brittle. A cutaneous punch revealed psoriasis form compatible with Netherton syndrome. Despite treatment with high doses of ciclesonide, as she did not tolerate long-acting beta-agonists due to distal tremor, the patient had poor asthma control (ACT 16).
Several topical treatments were tried with low effectiveness, including 0.1% tacrolimus ointment and medium and high-potency corticosteroids.
After signing written informed consent and hospital authorization, compassionate treatment with omalizumab 600 mg every 4 weeks did not improve skin symptoms. However, respiratory symptoms were improved. After 5 years of treatment with omalizumab, the patient presented mild rhinoconjunctivitis and intermittent asthma symptoms (ACT 24).
After the patient signed the written informed consent, the ethics committee authorized compassionate subcutaneous dupilumab use. Improvement was evaluated using Severity Scoring Atopic Dermatitis (SCORAD); Eczema Area and Severity Index (EASI); Pruritus Numeric Rating Scale (NSR); Netherton Area Severity Assessment (NASA), and Dermatology Life Quality Index (DLQI).4,5 Before treatment with dupilumab, the patient presented SCORAD 40.2; EASI 28; NSR 8; NASA 44.8, and DLQI 18. Treatment with dupilumab started in 2021 at 600 mg and 300 mg fortnightly. She noticed subjective improvement with the first dose of dupilumab within two to 3 days of its administration. With the successive doses of dupilumab, she experienced fewer skin flare-ups and itching and reduced use of topical corticosteroids. At week 16, all clinical parameters improved (Figure 1(a)). She is receiving dupilumab and maintains improvement 18 months later. (a) Case 1. At baseline vs.16 weeks of treatment with dupilumab (the patient consented to the publication of the photos). (b) Case 2. At baseline vs. 16 weeks of treatment with dupilumab (the patient consented to the publication of the photos).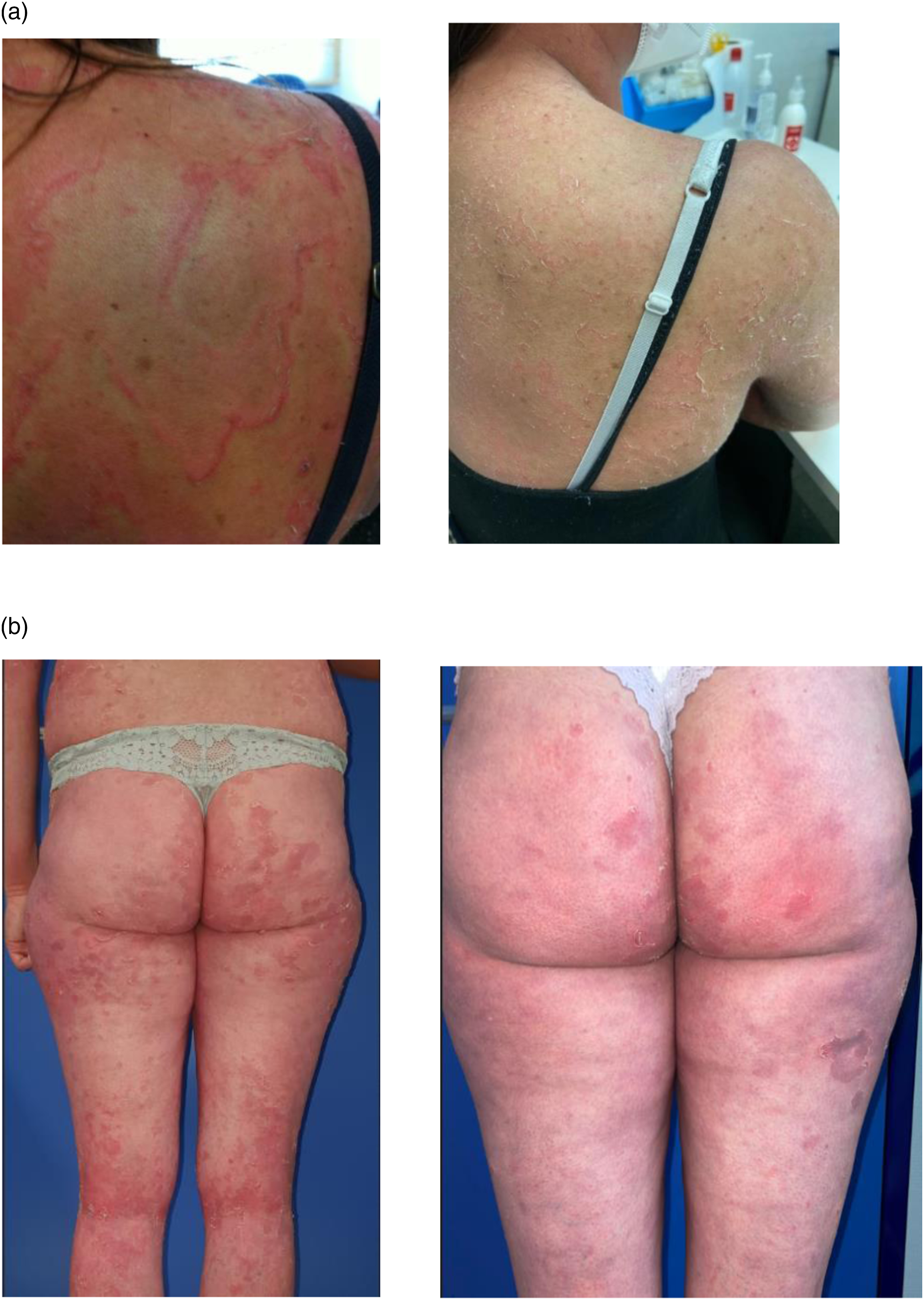
Case 2
In 2019, the younger sister was reevaluated at 27 years at the Dermatology department due to the progressive worsening of her skin symptoms. She was diagnosed with moderate rhinoconjunctivitis, hypersensitivity to fish and egg, and sensitization to house dust mites and grass pollen, egg, fish, and shellfish. In vitro tests revealed a total IgE >5000 kU/L
Skin examination revealed severe, extensive ILC lesions, predominantly on the trunk, arms, and legs, with steroid-induced skin atrophy. Punch biopsy revealed skin lined by an epidermis showing a very irregular thickness with areas of hyperplasia, with hyperkeratosis alternating with areas of parakeratosis.
Since the patient was using topical corticosteroids in a limited way due to her Addison’s disease and low efficiency, it was decided to start treatment with secukinumab. The patient signed the written informed consent, and the hospital’s ethics committee authorized secukinumab as a drug for compassionate use. In 2020, she started treatment with secukinumab 300 mg, with a favorable initial response and an apparent decrease in topical corticosteroid use. After 9 months of treatment, the patient presented a progressive loss of efficacy, with flare-ups prior to secukinumab administration, and needed more frequent treatment with topical corticosteroids.
Given the lack of efficacy with secukinumab and the improvement of her sister with NS in treatment with dupilumab, the patient signed the written informed consent, and dupilumab was authorized by our hospital’s ethics committee for compassionate use. Severity parameters were SCORAD: 47.5, EASI: 33.21, NASA: 49, NSR: 9, and DLQI: 16. In September 2021, treatment with dupilumab 600 mg followed by 300 mg every 2 weeks was started. One month after administration, the patient noticed less cutaneous itching and did not report using topical corticosteroids. At week 16, the patient improved all severity parameters. (Figure 1(b)). The improvement is maintained after 12 months of treatment.
Discussion
There are some case reports of treatment of severe NS with biologics targeting proinflammatory cytokines or specific immunoglobulins, such as anti-IL-17 ixekizumab, 6 and secukinumab,7,8 anti-TNF-α infliximab, 9 anti-IL-12/IL-2 ustekinumab, 10 and anti-IgE omalizumab, 11 as well as anti-IL-4/IL-13 dupilumab.12–19
There is two case reports of NS treated with omalizumab.11,16 In one patient, allergic skin symptoms and mucosal symptoms decreased. 11 In the other patient, the treatment with omalizumab was not effective. 16
There are two reports on NS treated with secukinumab.7,8 Blanchard et al. 8 reported a case of NS successfully treated with secukinumab after a prior unsuccessful attempt with omalizumab. Luchsinger et al. 7 published four cases, all successfully treated with secukinumab.
List of published case reports of NS patients treated with dupilumab.

Abbreviations: CS: corticosteroid; IVIg: intravenous immunoglobulin; M: months.
Conclusions
The success of dupilumab treatment is possibly related to the phenotype of NS-ILC in both patients. Dupilumab reduces T2 inflammation. In addition, blocking IL-13 reduces epithelial permeability, and blocking IL-4 reduces IgE synthesis, thus further reducing inflammation. 20
Dupilumab could be effective and safe treatment in patients with NS, probably those with NS-ILC phenotype.
Footnotes
Acknowledgment
We would like to thank the two patients for their consent to the publication of the photos.
Authors’ contributions
Martin- García C, Godoy E, Cañueto J and Muñoz-Bellido FJ and Davila I: designed the study and wrote the manuscript. Perez-Pazos J: performed the genetic study of patients. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
