Abstract
Introduction
The COVID-19 (the acronym of COronaVIrus Disease 19), or Severe acute respiratory syndrome coronavirus 2, is an infectious disease caused by the SARS-CoV-2 virus, a new member of the human coronaviruses identified for the first time in Wuhan, China. 1
Since the beginning of the COVID-19 pandemic, people have been encouraged in using PPE (Personal Protective Equipment) as masks, eyewear, visors and gloves to protect themselves.
In patients suffering from latex allergy, this massive use of PPE, especially latex gloves, could provoke severe adverse reactions and could negatively affect their quality of life.
Latex hypersensitivity has been increasingly reported worldwide, with a prevalence of 9.7%–12.4% among healthcare workers (HCWs), 7.2%–30.4% among susceptible patients and 4.3%–2.1% in general population. 2 Occupation represents a possible risk factor for natural rubber latex allergy (NRLA); healthcare workers, rubber industry workers, hairdressers and housekeepers are mostly exposed to these sensitisations. Other risk factors include atopic history, exposure to latex-derived products during surgery and sensitizations through skin contact or inhalation.3–6
The clinical manifestations of NRLA include contact dermatitis, urticaria-angioedema, respiratory symptoms and can lead to anaphylactic reactions. 7
The diagnosis of NRL allergy is based on a positive allergological work-up, including allergological tests (skin prick test and specific serum IgE assay-ImmunoCAP System) and provocation challenges (glove-wearing, mucous-oral, nasal, conjunctival and sublingual provocation test) to confirm clinical latex allergy. Sublingual latex immunotherapy provides a 4 days rush protocol, starting with a drop of latex solution (500 μg/ml) diluted 1:1018 until the highest dose of 1 mL of undiluted solution (500 μg of latex), then a maintenance therapy (500 μg of undiluted solution three times a week) is recommended, without discontinuations. 8
Primary prevention remains essential to reduce the risk of reactions in NRLA patients, and it is obtained by avoiding the airborne and contact exposure to natural rubber latex (NRL). 9 During the COVID-19 pandemic primary prevention is very difficult to reach considering NRL is ubiquitous and widely used. Hence, the protection conferred by latex SLIT could be essential for these patients.
Objective
The aim of our study is: (a) to evaluate the incidence of latex allergic reactions in patients during the SARS-CoV-2 pandemic; (b) to evaluate the protective role of continuous latex SLIT during this period; and (c) to evaluate quality of life of NRLA patients during the pandemic.
Material and methods
The single-centre observational study was carried out on 67 adult subjects with a latex allergy diagnosis followed by Allergy Unit of Fondazione Policlinico Universitario A. Gemelli IRCCS of Rome. All data were collected from January 2020 to December 2020.
All patients underwent to a survey focused on their clinical and psychological conditions during COVID-19 pandemic. We analysed the results dividing populations in: never treated with SLIT, treated with continuous SLTI and treated with not-continuous SLIT (Figure 1). Diagram of overall cohort.
For the evaluation of the degree of well-being, we used SF-36 questionnaire (Short-Form 36-Item Health Survey), a validated questionnaire for health perception measurement. 10
The Ethical Committee of our Institute evaluated and approved this study (ID: 3255).
Subjects included in the present study accepted and signed the informed consent, in accordance with the Declaration of Helsinki.
Sample size
For the calculation of the sample size we considered: 1) a population size of 287 latex allergic patients, 2) a margin of error of 10%, 3) a chosen sampling confidence level of 90% and 4) a response rate of 50%; a sample size of 56 subjects was estimated.
Statistical analysis
The sample has been described in its demographic and clinical characteristics applying descriptive statistics techniques. Qualitative variables have been presented as absolute frequencies and percentages. Quantitative variables have been summarized with mean and standard deviations. The normality of data have been verified with the Kolmogorov–Smirnov test. Proportions were compared applying the Chi-square test. A p-value < 0.05 was considered statistically significant. All the statistical analyses have been performed with SPSS 25.
Results
Incidence of latex adverse reactions
Sixty-seven patients (9 males and 58 females, with a mean age of 45.9 ± 11.4 years) suffering from latex allergy were included in the present study.
Our population had a high latex contact risk; 55 patients (82.1%) were healthcare workers (52.2%), hairdressers (17.9%) and housekeepers (11.9%).
Demographic and clinical features of patients.

Data are presented as mean ± SD or %, as indicated. BMI = body mass index; CCS = corticosteroids, anti-H1 = antihistamines, U/A = urticaria-angioedema, RS = respiratory symptoms. All percentages are referred to total of patients
During COVID-19 pandemic, we recorded among our patients 13 cases (34.2%) of urticaria/angioedema (U/A), 9 cases (23.6%) of respiratory symptoms (dyspnoea, shortness of breath and wheezing) and 7 cases (18.4%) of anaphylaxis.
27 patients (40.3%) used antihistamine, 13 patients (19.4%) systemic corticosteroids (CCS) therapy and 5 patients (7.5%) epinephrine auto-injectors, while 2 patient (3%) have been treated in an intensive care Unit. 13 patients used antihistamines or CCS without symptoms.
Role of continuous latex sublingual immunotherapy
Sixty three patients (94%) previously underwent latex SLIT for more than 12 months and, among these patients, 47 subjects (70.1%) regularly followed the SLIT without discontinuations for more than 12 months.
Patients with a higher risk of latex contact had more adherence to SLIT (p = 0.017) and did not show a significant increase rate of latex allergic reactions (U/A p = 0.79; respiratory symptoms p = 0.19; anaphylaxis p = 0.43).
Continuous latex immunotherapy effectiveness.

Natural rubber latex allergy patients quality of life in continuous/not continuous sublingual immunotherapy
Results of the SF-36 questionnaire in C-SLIT and NC-SLIT.
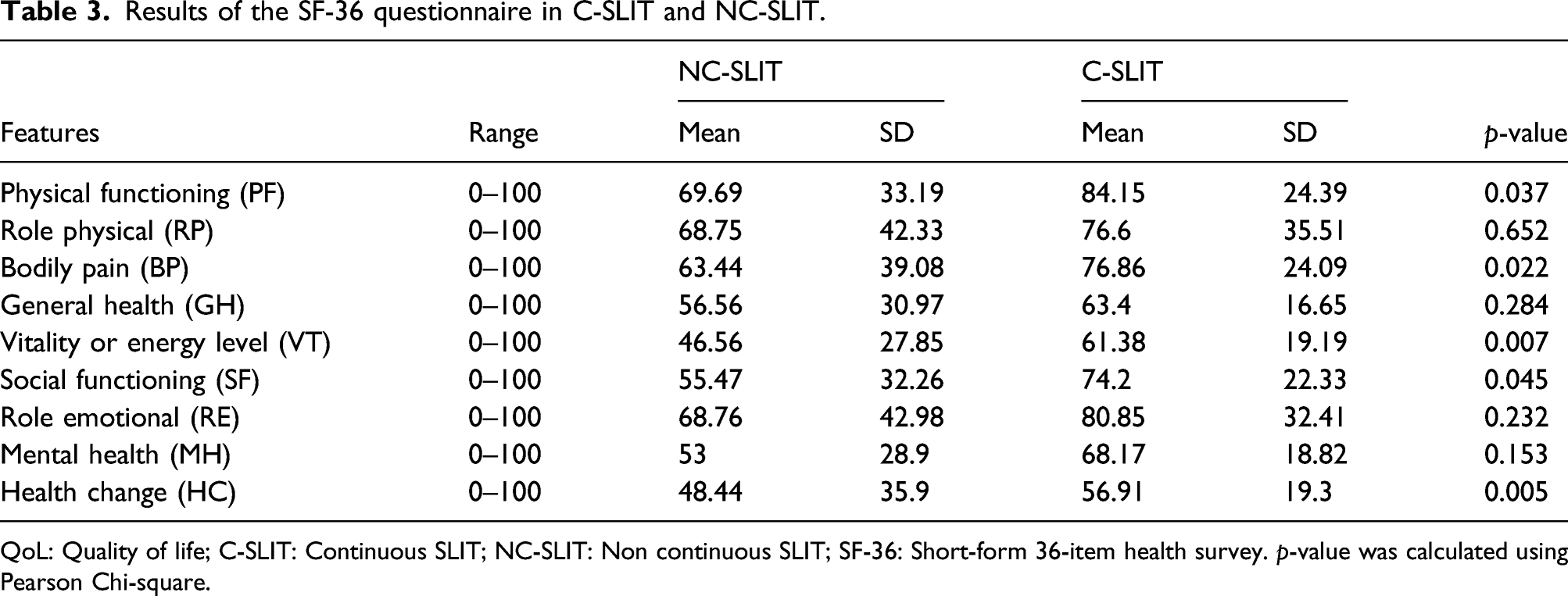
QoL: Quality of life; C-SLIT: Continuous SLIT; NC-SLIT: Non continuous SLIT; SF-36: Short-form 36-item health survey. p-value was calculated using Pearson Chi-square.
Discussion
Previous studies focused on risk of latex and contacts dermatitis in HCWs,11,12 but, to our knowledge, this is the first study that investigates the clinical status and quality of life of NRLA patients during COVID-19 pandemic.
The COVID-19 pandemic and the Lockdown measures have worsened the quality of life of our patients as well as of general population. 13
The pandemic challenged the capacity of healthcare systems to provide adequate management of NRLA patients, 14 and in this context, we performed a survey to monitor their health status.
During the pandemic, the risk of latex exposure has had a significant increase considering the possibility of direct skin contact, airborne exposure, contamination of food and medical evaluations.
We recorded a higher use of antihistamines therapy in relation to the adverse reactions rate; this finding was probably due to fear of NRLA patients in front of a wide spread of latex NRL items.
As already has been demonstrated,15,16 also in our populations the continuous SLIT proved effective in preventing latex severe reactions and farther in increasing the quality of life of patients.
Our paper presents several limitations. First all, data collected are self-reported by the patients without any medical records evaluation. Moreover, a largest number of patients should need to establish the exact impact of immunotherapy in the prevention of adverse reactions.
Conclusion
Our study is the first that investigated the clinical and quality of life effects of COVID-19 pandemia in NRLA patients demonstrating the importance of SLIT adherence also in this complex period. Further studies are needed to investigate the protective role of immunotherapy also in the complex pandemic period.
Footnotes
Author contributions
The authors are responsible for the content and the writing of this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
