Abstract
Background
Bisphenol (BPA) and ionizing radiation exposure (IR) are potent oxidants that cause free radical induction, leading to signaling pathway activation that alters cell growth. Due to the insufficient knowledge of the impact of BPA and IR on the lungs, the current study determined the impact of BPA and IR on the lung tissue of adult female Wistar rats.
Methods
Forty Wister female rats were used in this study and were randomly divided into four groups. The rats received BPA (150 mg/kg body weight/day for 6 weeks) and were exposed to IR at 2 Gy/week up to 12 Gy for 6 weeks.
Results
It was found that BPA and IR possess a harmful effect on the lungs via induction of oxidative stress, confirmed by increasing levels of malondialdehyde (MDA), nitric oxide, myeloperoxidase (MPO), and lactate dehydrogenase (LDH). Exposure to BPA and IR activates inflammatory cytokines TNF-α, IL-6, IL-1β, growth factors such as TGF-β, and gastrin-releasing peptides. BPA/IR exposures induced phosphorylated expression p-ERK1/2 and p-MEK1/2 associated with triggering of the GPER/EGFR/KRAS signaling factors, resulting in matrix metalloproteinase-2 and 9 overexpression and the development of lung tumors. Our findings support the causal role of two deleterious environmental pollutants BPA and IR, via the cytotoxicity in the respiratory system in the form of severe lung damage resulting in cancerous cells.
Introduction
Bisphenol A is one of the ubiquitous major environmental contaminants where the presence of two phenolic functional groups helps BPA achieve properties beneficial in manufacturing processes. The main use of BPA is in the manufacture of polycarbonate plastics. BPA is a common ingredient in food-related consumer products such as baby bottles, food storage containers, cling film, water bottles, and the liners of metal cans. 1 Due to the widespread use of BPA-containing products, people worldwide are highly exposed to food containers. 2
Bisphenol A has been the subject of great concern because it interacts with endocrine signaling pathways even at very low concentrations. BPA is a diphenyl molecule with two hydroxyl groups in the “para” position, which makes it very similar to diethylstilbestrol, a synthetic estrogen. BPA has been proven in several studies to activate estrogen receptors (ERs) a and b. BPA has estrogen-like action and developmental harm in the reproductive organs, according to many lines of evidence. 3 BPA’s estrogen-mimicking causes a variety of health concerns, including ovarian cancer and breast cancer, as well as reproductive abnormalities, even at extremely low concentrations. 4 In addition, BPA exposure has been linked to lung damage. BPA also has additional effects that are not related to estrogenic action, such as inflammatory cytokine dysregulation and increased oxidative stress. 5 BPA may interfere with and disturb the immune system through a number of cytokine signals. 6 As a result, cytokine signaling dysfunction can lead to allergies, autoimmune disorders, inflammation, and cancer, among other illnesses. BPA also stimulates MAPK/ERK and p-JNK signaling pathway members' transcription and translocation. 7 Moreover, living organisms are exposed to ionizing radiation (IR) in everyday life, which is a phenomenon that has the potential to cause harmful human health impacts. Staff in the sectors of radiation and nuclear-powered engineering, patients having diagnostic procedures and/or treatment activities, and medical workers accidentally exposed to some degree of radiation are among the people and humanities who are exposed to IR from natural and manufactured sources. 8 Increased endogenous signaling and the culmination of free radicals in a direct sequence of activities that are damaging to numerous cellular macromolecules such as DNA, lipids, and proteins resulting in an IR-induced cellular damage. 9
Direct DNA damage, which leads to the death of epithelial cells owing to the overproduction of reactive oxygen and nitrogen species ROS and RNS, is the postulated mechanism by which both BPA and IR cause lung tissue damage. The injured area is infiltrated by cytokines, lymphocytes, and leucocytes. Pneumonitis and fibrosis were induced by a severe inflammatory accumulation. Intracellular signaling has been triggered by DNA or cytoplasmic organelles damage, resulting in altered gene expression and the immediate release of growth factors such as TGF-ß, platelet-derived growth factor (PDGF), and interleukin 1ß (IL-1ß), as well as aggravating the Smad2/3/EMT signaling pathway. 10 ROS, such as superoxide, hydrogen peroxide, hydroxyl radicals, and nitrogen species, are responsible for 60% of the overall damage caused by BPA and IR. ROS may directly alter proteins and organelles, but it can also create hydroxyl radicals that damage DNA in an iron-dependent way (Fenton reaction). Because of the absence of repair, cells often have thousands of variant copies and are more vulnerable to damage than nuclear DNA. mtDNA damage is a DNA damage-associated molecular pattern that causes inflammation, immunological responses, and cell death, and is significantly linked to immune-related lung disorders.11,12
These findings suggest that long-term BPA and IR exposure have a negative effect on lung function, implying that BPA can modify the development of lung illnesses, leading to the development of lung disorders. However, there is little evidence to support this idea, and the underlying processes remain unknown. Therefore, according to the previous studies,7,10 the current study aimed to look into the effects of toxic pollutants like BPA and ionizing radiation stressors on rat lung health. Also, a proposed mechanism for their deleterious effect on the lung tissue was revealed.
Materials and methods
Chemicals
The bisphenol-A from Sigma Chemical Co., Nasr City, Cairo, Egypt, was obtained in the form of rutin hydrate. Reagents and other chemicals applied in this study were of analytical grade.
Animals
It was reported that BPA is a xenoestrogen compound; therefore, the current study was carried out on 5-week-old Wister female rats (150 ±20 g), obtained from the National Centre for Radiation Research and Technology (NCCRT), Cairo, Egypt. The rats were housed in cages and maintained a 12-hour light-dark cycle. They were given a week to acclimate to the ambient conditions before beginning the experiment, and they were fed regular food pellets containing all the nutritional ingredients, as well as ample quantities of water ad libitum.
Irradiation procedure
Gamma Cell-40 (137Caesium), a biological irradiator produced by Canada Ltd, was used to perform the irradiation at the National Center for Radiation Research and Technology (NCRRT) in Cairo, Egypt. Animals were placed in a plastic sample tray with a lid and support was provided for use in the sample cavity. Rats were whole-body-exposed to fractionated doses of gamma radiation of 2 Gy every week of the experimental course, 10 up to a total dose of 12 Gy given at a dose rate of 0.68 Gy/min.
Investigational design
The animals were randomly divided into four experimental groups (n = 10) and treated in parallel as follows: 1-Control group: untreated normal rats, 2-BPA group: Rats received BPA (150 mg/kg body weight/day) intraperitoneally (IP), dissolved in corn oil, for 6 weeks13. 3-IR group: Rats were exposed to IR at 2 Gy/week up to 12 Gy for 6 weeks, 4-BPA + IR group: Rats received BPA as in group 2 following with IR for 6 weeks as in group 3. The rats fasted overnight following the last dose of BPA treatment. Blood samples were drawn from the hearts of each animal while they were under mild anesthesia with diethyl ether. After allowing blood to coagulate for 15 min, it was centrifuged at 3000 rpm for 15 min. Animals were killed through cervical dislocation immediately after blood collection; lung tissues were promptly dissected, cleaned with ice-cold saline, allowed to dry, and weighed. The left lobes of lung tissue were preserved in 10% formalin prepared in phosphate-buffered saline (PBS) for histological analysis. To produce a 20% w/v homogenate, a weighted portion of each lung was homogenized with ice-cold PBS. To eliminate cells, the homogenate was spun at 4000 rpm for 5 min at 4°C in a cooling centrifuge to remove cell debris. After that, the aliquots were stored at −80°C until the day of the analysis. Bronchoalveolar lavage was performed with 500 µl saline through a cannula put into the trachea, and the bronchoalveolar lavage fluid (BALF) was extracted for biochemical markers determination. The lung tissues were collected for additional biochemical investigation after the lavage.
Estimation of oxidative stress and inflammatory markers
In lung tissues, malondialdehyde (MDA) content, 14 and the total level of nitric oxide (NO) 15 , were determined. Also, tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin 1 beta (IL-1β) were estimated in lung tissues using rat ELISA kits (R&D Systems).
Determination of gastrin-releasing peptide
Rat gastrin-releasing peptide (GRP) secreted protein was measured in the lung homogenate by an ELISA kit (ab133049) from Abcam, Cambridge, UK, USA, which performs the same previously mentioned technique.
Measurement of lactate dehydrogenase, myeloperoxidase, matrix metalloproteinases, and transforming growth factor- 1β in the BAL fluid
The levels of matrix metalloproteinases MMP-2 and MMP-9 were quantified using the following ELISA kits from My BioSource, Inc., San Diego, CA, USA. The activity of lactate dehydrogenase (LDH) was measured via the method of Horecker and Kornberg. 16 Following the manufacturer’s instructions, the level of Transforming Growth Factor β1 (TGF-β1) was estimated using an ELISA kit (My BioSource, Inc., San Diego, CA, USA) and myeloperoxidase (MPO) activity was described. 17
Measurement of Monocyte chemoattractant protein, Intercellular adhesion molecule-1, and E-Cadherin in the lung tissue
In lung homogenates, monocyte chemoattractant protein (MCP-1) was determined using an ELISA Kit (Rat MCP-1/CCL2 Cat. RAB0058), Sigma Aldrich. Fibrinogen was measured using a Rat ELISA Kit (Cat. b108845), Abcam, USA. Intercellular adhesion molecule-1 (ICAM-1) and E-Cadherin (E-Cad) levels were quantified according to the manufacturer’s instructions using a Rat ELISA Kit MyBioSource Europe SA Brussels, Belgium (Cat. MBS843373 and MBS761750, respectively)
Use of RT-PCR for the determination of G-protein-coupled estrogen receptor (GPER) and epidermal growth factor receptor (EGFR) gene expression.
Primer sequences used in RT-PCR table.

2X SYBR Green PCR Master Mix (Qiagen, Cat. No. 204,143), 900 nM of each primer, and 2 µL of cDNA made up the reaction mixture, which had a total volume of 25 µl. A first step at 95°C for 5 min was followed by 40 cycles at 95°C for 20 s, 60°C for 30 s, and 72°C for 20 s. By the ΔΔCt method, the relative expression of the real-time reverse transcriptase PCR products was investigated. The relative expression was calculated relative to the housekeeping gene by the equation: fold induction = 2−(ΔΔCt). Where ΔΔ Ct = Ct [gene of interest (unknown sample)-Ct housekeeping gene (unknown sample)]−[Ct gene of interest (calibrator sample)−Ct housekeeping gene (calibrator sample). 18
Western immunoblotting analysis of ERK1/2, KRAS, MEK1\2 and C‐Jun proteins in lung tissue homogenate
Lung tissue protein was extracted using TRIzol reagent and protein concentration was quantified in accordance with a previous procedure. 19 Twenty grams of protein per lane were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to PVDA membranes using 10% acrylamide gels. Membranes were incubated at room temperature for 2 h with a blocking solution (5% non-fat dried milk in 10 mM Tris-HCl, pH 7.5, 100 mM NaCl, and 0.1% Tween 20), then incubated overnight at 4°C with a primary antibody toward extracellular signal-regulated kinase (ERK)1/2, Kirsten rat sarcoma virus (KRAS), mitogen-activated protein kinase kinases 1 and 2 (MEK1/2), and C‐Jun with a dilution factor of 1:2000, and β-actin was used as a reference protein. After washing 3 times in washing buffer, the membrane was incubated with the secondary monoclonal antibody conjugated to horseradish peroxidase at room temperature for 2 h, and the washing buffer was used to the membrane 4 times. The membrane was developed and visualized by chemiluminescence using the InvitrogenTM detection kit (Catalog # AHO1202) according to the manufacturer’s protocols, then exposed to X-ray film. Quantification of proteins was carried out using a scanning laser densitometer (Biomed Instrument Inc., USA).
Histopathological investigation
Lung tissue samples have been treated with a 10% formaldehyde solution and integrated by conventional methods in paraffin. Hematoxylin and eosin (H&E) staining were used on 3 µm sections. The stained sections were examined under a light microscope. 20
Statistical analysis
A one-way analysis of variance was used to calculate variations in the mean of variables throughout all experimental groups (ANOVA). The findings have been expressed as mean ± SE. SPSS statistical software version 20 for Windows (SPSS ® Chicago, IL, USA) was used to analyze all test findings. At p < 0.05, all variations were statistically significant.
Results
MDA and NO levels in the lung tissues of rats exposed to BPA/IR.
MDA and NO levels in lung tissues and LDH and MPO levels in the BAL fluid of lung rats exposed to BPA/IR.
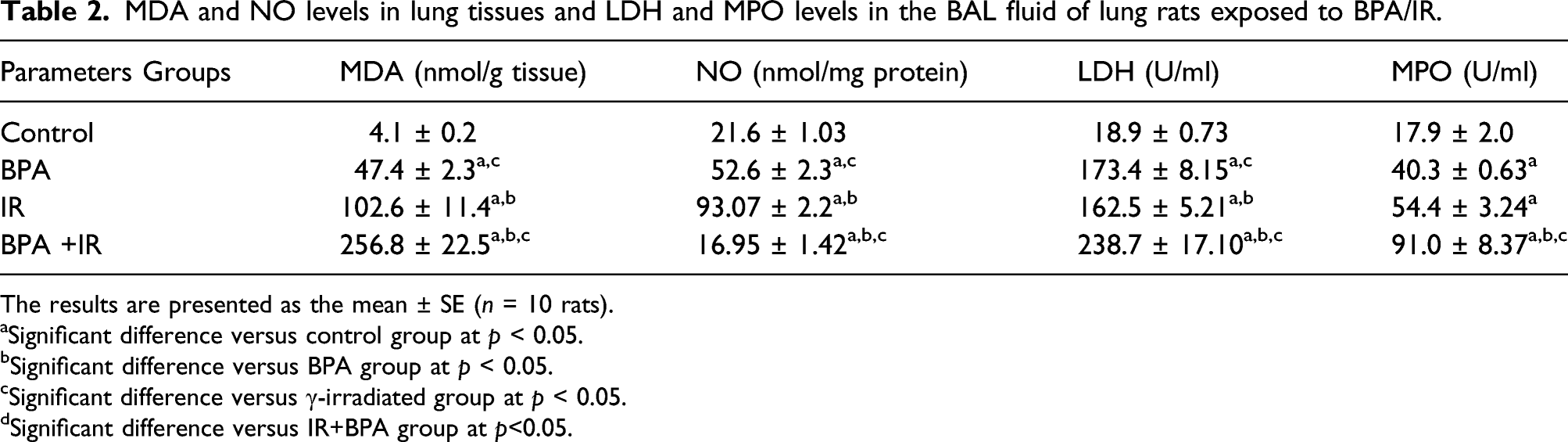
The results are presented as the mean ± SE (n = 10 rats).
aSignificant difference versus control group at p < 0.05.
bSignificant difference versus BPA group at p < 0.05.
cSignificant difference versus γ-irradiated group at p < 0.05.
dSignificant difference versus IR+BPA group at p<0.05.
LDH and MPO levels in BAL fluid were lower in rats exposed to BPA/IR
In the BPA or IR treated groups, the LDH and MPO activity content were found to vary significantly from the control group. The increase in LDH and MPO levels in the IR + BPA group was highly significant (p < 0.001) as compared to the BPA or IR groups, making the combination exposure a pivotal factor in elevating lung injury (Table 2).
TGF-1β levels in the BALF fluid, as well as TNF-a, IL-6 and IL-1β levels in the lungs of rats exposed to BPA/IR
TGF-1β in BAL fluid of lung and TNF- α, IL-6 and IL-1β levels in the lungs of rats exposed to BPA/IR.

The results are presented as the mean ± SE (n = 10 rats).
aSignificant difference versus control group at p < 0.05.
bSignificant difference versus BPA group at p < 0.05.
cSignificant difference versus γ-irradiated group at p < 0.05.
dSignificant difference versus IR+BPA group at p < 0.05.
Levels of MCP-1, fibrinogen, ICAM-1 and E-Cad in the lungs of rats exposed to BPA/IR
MCP-1, fibrinogen, ICAM-1 and E-Cad levels in the lungs of rats exposed to BPA/IR.
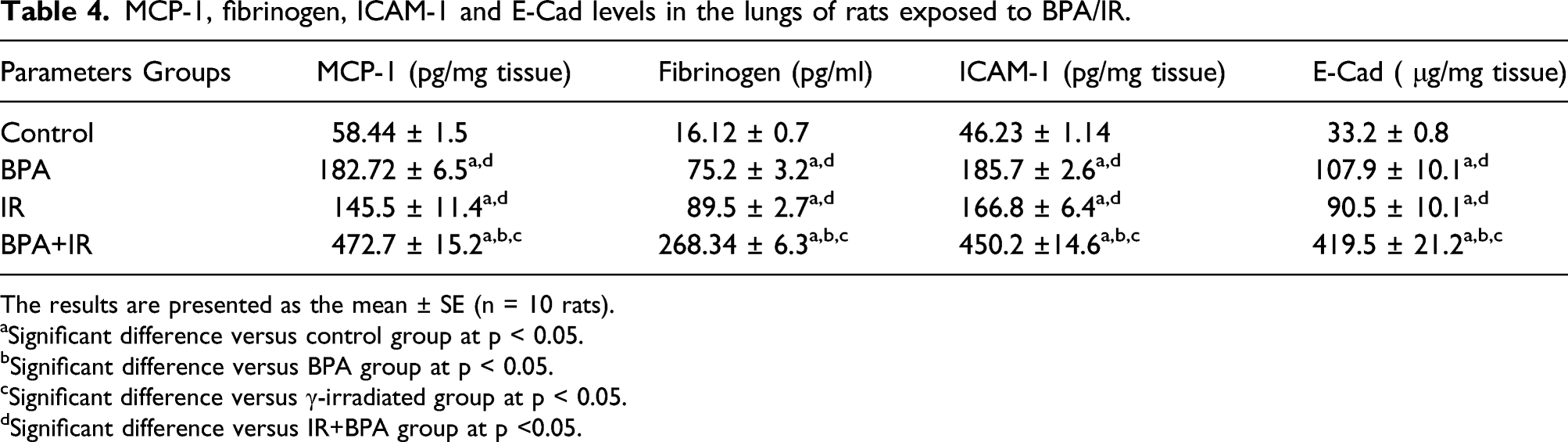
The results are presented as the mean ± SE (n = 10 rats).
aSignificant difference versus control group at p < 0.05.
bSignificant difference versus BPA group at p < 0.05.
cSignificant difference versus γ-irradiated group at p < 0.05.
dSignificant difference versus IR+BPA group at p <0.05.
The tumor marker GRP was elevated in the lungs of rats exposed to BPA/IR
Estrogen receptors are thought to have a crucial role in lung cancer development and growth. Previous research has shown that both the ERa and the GPER can mediate the estrogenic effects of BPA and other external environmental. GPER is involved in a number of carcinogenesis processes, including angiogenesis, cellular migration, and adhesion.
21
As shown in Figure 1, the levels of GRP and gene transcript of GPER were elevated significantly in IR and BPA relative to the control group (p < 0.05). An additional increase in the IR+BPA relative to the relevant IR and BPA group values was observed. The GRP/GPER signaling factors reinforce the MMP-2 and MMP-9 in lung rats exposed to BPA/IR. (a, c, d) GPR and MMP2 and 9 protein levels were determined by ELISA technique. (b) Relative gene expression of GPER mRNA was detected by RT-PCR and quantified by scanning densitometric analysis and normalized to housekeeping gene β-actin mRNA expression. a) Significant difference versus control group at p< 0.05. b) Significant difference versus BPA group at p< 0.05., c) Significant difference versus γ-irradiated group at p<0.05. d) Significant difference versus IR+BPA group at p< 0.05.
Levels of MMPs in the BALF fluid of rats exposed to BPA/IR
The matrix metalloproteinases are zinc-dependent endopeptidases that play an important role in the migration and invasion of various types of cancer cells. We then detected the protein level of MMP-2 and 9 in the rats treated with BPA and/or exposed to IR. The results revealed that BPA/IR can significantly increase the levels of MMP-2 and MMP-9 (p < 0.05) compared to the control group. Therefore, it can be suggested that GPER gene mediates the BPA-induced enhancement of MMPs in lung cancer cells. (Figure 1).
Levels of EGFR, KRAS, MEK, and ERK1/2 in the lung tissue of rats exposed to BPA/IR
BPA activates ERK rapidly in breast cancer cells line via the GPER/EGFR transduction route, and stimulate the rate of estrogenic GPER-mediated signaling genes.
22
A Western blot analysis was used to determine the effects of BPA or IR on the phosphorylation of ERK1/2, KRAS, MEK, ERK1/2, and p-C-Jun in lung tissue after exposure to BPA or/and IR. As shown in Figure 2, upregulation of phosphorylation (activation) of the epidermal growth factor receptor (EGFR), KRAS, MEK, and ERK1/2 in rats exposed to BPA and/or IR compared to the control group (p < 0.05). Our findings show that the combined toxic stressors BPA and IR have the greatest ability to phosphorylate the EGFR, KRAS, MEK, and ERK1/2 in lung tissue via GPER/EGFR signals. BPA and IR activate EGFR and phosphorylation of KRAS/ERK and signaling pathways cascades to promote cell growth. (a) EGFR mRNA folds change, relative protein expression levels of (b)p-KRAS, (c) p-MEK, (d) p-ERK1/2, (e) and p-c‐Jun in lung tissue cells following treatment with BPA and IR were distinguished by western blot analysis. (f) Relative quantification of western blot analysis is presented in bar charts. The results are presented as the mean ± SE (n = 10 rats). a) Significant difference versus control group at p< 0.05. b) Significant difference versus BPA group at p< 0.05, c) Significant difference versus γ-irradiated group at p≤ 0.05. d) Significant difference versus IR+BPA group at p< 0.05.
Histological alterations in the lung rat exposed to BPA/IR
The lung tissues in the control rat group Figure 3(a) showed normal lung architecture, thin inter-alveolar septa, folded columnar epithelial cells of the bronchiole, clearly seen alveolar sacs, normal pulmonary vessels, and normal fibrous tissue distribution. On the other hand, animals exposed to IR (Figure 3(b)) showed a thickened wall of the bronchiole with moderate congestion of perialveolar capillaries and mononuclear cell infiltration, mainly macrophages and lymphocytes, in the alveolar spaces. Alveolar edema and mild hyperplasia of the bronchial epithelial lining with increased numbers of goblet cells. The animals given a toxic dose of bisphenol (Figure 3(c)) developed lung edema and mononuclear cell infiltration. Multiple focal emphysematous areas accompanied by giant alveoli formation were seen. Squamous metaplasia of the bronchial epithelial lining to stratified squamous epithelium, which appeared as a fungated mass in the bronchial lumen. Marked thickening of the inter-alveolar septa and bronchiolar walls was detected. Diffuse inflammatory cell infiltration was noticed. Congested blood vessels appeared with a markedly thickened wall. Extravasated RBCs in the lumen of alveoli and the interstitium were also seen. Animals exposed to a combination of BPA and IR (Figure 3(d) & (e)) showed limited-stage small-cell lung carcinoma, which appeared as a peribronchial aggregation of relatively small epithelial cells, with scant cytoplasm, ill-defined cell borders, finely granular nuclear chromatin, and an absence of nucleoli. The neoplastic cells showed criteria of low malignancy, which were characterized by cellular and nuclear pleomorphism with frequent mitotic figures. The bronchial epithelial lining revealed hyperplasia of goblet cells, which was accompanied by occlusion of the bronchial lumen by thick basophilic mucous. A–E: Photomicrograph of H & E stained lung sections in all rat groups. (a) An image of the control group reveals normal lung architecture with thin alveolar septa, normal bronchioles (b), and blood arteries to the bronchioles (bv). (b) An image from the IR group shows a thickened bronchiole wall with mononuclear cell infiltration and mild hyperplasia of the bronchial epithelial lining arrow, (c) an image from the BPA group shows squamous metaplasia of the bronchiolar epithelial lining with marked thickening of its wall and diffuse inflammatory cell infiltration. (d) and (e) images of the BPA+IR group show peribronchial aggregation of relatively small epithelial cells with scant cytoplasm. The arrow indicates a neoplastic mass with cellular and nuclear pleomorphism and frequent mitotic figures.
Discussion
Evermore research indicates that BPA might cause cancer, particularly malignancies linked to estrogen. Lung cells include estrogen receptors, which stimulate their growth. 23 According to the previous studies,7,10 which found that BPA-induced lung injury in rats, the present study revealed that exposure to BPA along with ionizing radiation interactions influences cellular responses and induces small cell lung cancer (SCLC) in female rats confirmed by bio and histology analysis. The estrogen receptor GPER mediated this effect through which the BPA/IR prompted activation signals GPCR/EGFR/KRAS/ERK1/2 stimulates upregulation of MMP-2 and MMP-9. In our estimation, this is the first research to demonstrate that BPA/IR may cause SCLC in female rats.
Similar to the above, female mice prenatally treated with BPA and DMBA showed a comparable rise in tumor development. 24 BPA has been related to malignancy in a few scientific studies. For case in point, BPA has been thought a hazardous cause for breast and prostate cancer. 25
In the study, GRP over-secretion in the combined group significantly promotes parameters of IR and BPA-induced pulmonary carcinogenesis. In parallel with this, many lines of investigational indication have suggested that GRP may act as a growth factor in many types of cancer. Also, the increase in the activity of MPO level as a marker of neutrophil infiltration with the detected obvious rise in LDH activity as a marker of cell damage or death substantiates lung damage. Besides, the elevated levels of TNF-α, IL-6, IL-1β, and TGF-β in lung tissues indicate that cytokines stimulate the progress and widespread of the epithelial lung cells by altering the release of lung fibroblasts, causing worsening of tissue injury and deterioration. It is worthy to note that rats' lung impairment was exacerbated when they were exposed to IR and BPA. Chronic inflammation is a response to an alteration induced by IR, BPA, or combination revilement, which functions to eliminate the source of the harm and restore the physiological state of the impressed tissue. However, it triggers cellular events that can promote the malignant transformation of cells and carcinogenesis. Several inflammatory mediators have been shown to take part in both the initiation and progression of cancer.26,27 Even though GRP and GRPR cell surface receptors trigger inflammatory pathways, they interfere with other relevant growth factor pathways such as TGF-1β, which promotes matrix deposition and encourages the secretion and activation of the proteolytic enzymes MMP-2 and MMP-9. 22
The increase in inflammatory cytokines stimulates the expression of adhesion molecules in cancer patients leading to the interaction between metastatic cells and target organs. In the tumor microenvironment, the interactions between adhesion molecules and inflammatory cells play an important role in activating premalignant cells transformation, migration, and spreading of carcinoma cells. 28
In the present investigation, lung tissue was found to secrete different levels of inflammatory cytokines. MCP-1 was significantly secreted cytokines by lung cells. Increases in the production of MCP-1 were found to be associated with elevated levels of adhesion molecules (ICAM-1 and E-Cad). This finding suggests a role for MCP-1/adhesion signaling pathway in BPA/IR-induced SCLC in rats.
Tumor cell proliferation, invasion, and malignant transformation are hypothesized to be caused by cancer-related coagulation disorders. Fibrinogen plays an important role in the pathogenesis of cancer, according to a number of studies. Fibrinogen can act as a bridge to connect tumor cells and platelets when they dissociate from the central focus and enter the circulation, which helps tumor cells adhere to distant organs and enhances tumorigenesis. 29 In the present study, a significant increase in the level of fibrinogen in rats exposed to BPA/IR may suggest its role in the progression of lung carcinoma.
Radiation has triggered pulmonary neuroendocrine cell hyperplasia, mainly attributed to the overproduction of GRP secretion, which then mediates chronic inflammatory accumulation leads to pneumonitis and fibrosis, which lead to plastic epithelial cells invasion 26. In the current study, the results show that the increase in GRP and TGF- β1 causes a significant increase in MMP-2 and MMP-9. Estrogen receptors are considered to have a crucial role in lung cancer development and metastases. GPER is involved in a number of carcinogenesis processes, including angiogenesis, cellular migration, and adhesion. 30
Our results indicate that GPER is involved in the signaling action provoked by BPA and IR and may stimulate cancer progression. Despite the felicitous synergism between BPA and IR in angiogenesis development, BPA plus IR treated rats revealed fortuitous upregulation in gene expression of GPER mRNA and EGFRm RNA and caused activation of KRAS transducer protein that can promote p-MEK 1/2 and p-ERK1/2 expression levels release concurrently with phosphorylated-C-Jun, which plays a role in cell survival from toxic stresses and the up-regulation of cell growth.31,32 In the present study, we found that BPA can trigger GPER-mediated signaling in the lungs of rats. In this context, the estrogen receptor GPER was previously revealed to arbitrate the BPA-dependent rapid activation of intracellular signaling. 22
Lung cancer is an aggressive disease characterized by early metastatic spread and rapid development. It requires a more detailed understanding of SCLC to develop more effective therapeutic approaches. Unique features dictated by both the cell of origin and the genetic lesion shape the resulting tumors. 33 Therefore, detailed insight into signaling pathways in the cells and molecular changes that lead to their transformation may help to identify better ways to inhibit SCLC progression. Yet, our research has some limitations; utilization of a variety of in vitro and in vivo model systems helps to uncover key driver mutations and identify environmental pro-oxidant pollutants like chemicals and irradiation. An in-depth analysis of the genetic alterations that take place within the tumor cells upon the acquisition of the alternation is of the utmost importance. Thus, expansion of a currently limited tumor tissue collection is urgently needed, with samples taken longitudinally, representing different stages of disease progression. Conclusion: The current study characterized the molecular consequences of lung exposure to ionizing radiation and prolonged administration of BPA, which causes deleterious aggravating lung damage and leads to tumor development. Further, GPER/EGFR/KRAS/ERK1/2 signals mediated the BPA-induced lung cell carcinoma and upregulation of MMPs. This study provided new insight, for the first time, into tumorigenesis and metastasis for lung cancer development stimulated by BPA and IR. Therefore, the use of BPA should be prohibited in plastic synthesizing industries. Also, there is a need for some more comprehensive studies to unravel the effects of BPA at a molecular level in various cancers. Considering the potential for BPA to cause cancer could increase awareness among scientists and the manufacturing industry about the importance of looking for alternatives to BPA.
Footnotes
Acknowledgments
Sincerely, the authors acknowledge Dr. Ahmed Osman
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Animals’ handling and anesthesia procedures were based on the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines (serial: 18-2019). The experiment was conducted and approved according to the guidelines of the Ethical Committee of the Faculty of Veterinary Medicine, Benha University (Approval number; BUFVT145/22).
