Abstract
Introduction
Autism spectrum disorders (ASDs) are neurodevelopmental diseases characterized by communication inabilities, social interaction impairment, repetitive behavior, as well as learning problems. Although the exact mechanism underlying this disease is still obscure, researchers believe that several factors play a significant role in its development and pathogenesis. Some authors have reported an association between adipokines family and autism. C1q/TNF-related protein-1 (CTRP1) is a member of the adipokines family, and we hypothesized that this adipokine might have an influential role in the pathogenesis of ASDs. Since there is no specific marker for screening the disease, we evaluated CTRP1 as a potential marker for achieving this purpose.
Methods
Blood samples were collected from 82 (41 ASDs boys, 41 healthy boys as controls) children aged 5–7 years old. CTRP1 gene expression and CTRP1 serum level were measured by quantitative realtime-PCR and enzyme-linked immunosorbent assay methods, respectively.
Results
It was found that CTRP1 is significantly elevated in autistic children in comparison to healthy controls, both at the gene expression level, as well as at the serum level; demonstrating a good diagnostic value with a good range of sensitivity and specificity for detecting ASDs.
Conclusion
CTRP1 expression is elevated in ASDs boys aged 5–7 years old, suggesting a role for this adipokine in ASDs pathophysiology. Also, receiver operating characteristic curve analyses revealed that this adipokine could be utilized as a diagnostic biomarker for differentiating ASDs patients from healthy individuals along with other recently proposed biomarkers.
Introduction
Autism spectrum disorders (ASDs) encompass a complex set of mental symptoms which are determined by verbal difficulties, restricted interests, impaired social communication, repetitive behaviors, and lack of sympathy which leads to severe introversion.1,2 The prevalence of ASDs in the general population is estimated to be nearly 1.85%, and approximately affects males four times more than females 3 ; but in Iran, the prevalence of autism is estimated to be lower in comparison with western countries.4,5 Although the etiology of this disease is poorly understood, 6 investigators believe that a combination of genetic, environmental, and immunological factors might contribute to the ASDs onset.7-10 Several studies have been conducted and various hypotheses were made to elucidate the exact mechanisms involved in ASDs pathogenesis. 1 Most recent studies propose glutamate excitotoxicity, oxidative stress, and mitochondrial dysfunction as the major contributing factors in ASD pathogenesis.11-13 Glutamate serves as an important neurotransmitter in the central nervous system, which exerts neuro exciting and stimulating effects upon secretion from the nerve terminals and engaging with its glutaminergic receptors on adjacent nerves. 14 It has been shown that patients with ASD as well as mice models for ASD have higher serum glutamate in comparison to healthy individuals or their glutamate receptors do not function properly15,16; on the other hand, the function of γaminobutyric acid (GABA) which is another neurotransmitter, was also found to be impaired in ASDs; this neurotransmitter is involved in either excitatory or inhibitory signaling in the CNS, depending on the concentration of chloride ions; such that high levels of intracellular chloride ions contribute to excitatory stimulations. 17 If the cellular transporters of chloride ions are over-expressed or over-activated, their persistent functioning results in excess chloride concentration inside the nerve cells and maintains the excitatory state. 18 These findings suggest a glutamate/GABA imbalance which in turn contributes to excitatory/inhibitory imbalances in synaptic nerves19-22; this mechanism which results in inflammation of the target nerves could be underlying neuroinflammatory conditions and nerve damage in many psychiatric and neurodegenerative disorders, as well as in ASDs patients.23-25 Accordingly, novel approaches have been proposed for targeting and lowering glutamate in ASD patients or modulating the corresponding receptors.26,27 Another recently proposed underlying etiology in ASDs pathogenesis is excessive oxidate stress in CNS of the ASDs patients, which is associated with higher blood markers of oxidative stress among ASDs patients in comparison to healthy individuals 28 ; this excessive oxidative stress may result from genetic disorders, environmental pollutions, malnutrition, or defects in compensatory mechanisms involved in modulating oxidative stress.29-32 Mitochondrial dysfunction is also proposed to be involved in ASDs pathophysiology and mitochondrial stress markers were found to be higher in ASDs patients 33 ; this kind of disorder is associated with decreased cellular energy (ATP) as a result of malfunctioning electron transfer chain (mitochondrial respiration) and increased ROS and other oxidative stress markers 34 ; the resulting consequences mostly affect cells with higher energy consumption such as nerves (with lots of transporter molecules consuming ATP) which leads to neuronal inflammation and nerve damage. besides, well-known DAMPS such as cardiolipin and mitochondrial DNA are responsible for neuronal inflammation in response to environmental or intrinsic genetic disorders. 35 Due to the heavy burden of ASDs in terms of financial considerations as well as the excess negative impact on families and societies,8,36,37 lots of attempts have been made by scientists of the field in order to introduce effective treatments as well as discovering sensitive and specific biomarkers, capable of diagnosing ASD at early stages of life, since the sooner the ASD is caught the better the outcome of the interventions will pursue. 38 Despite the recent advances in the field of biomarkers and pathophysiology of ASDs, no such biomarkers nor effective medicinal interventions have been employed so far in clinical settings, and the available treatments are only limited to those interventions managing the ASD complications and improving quality of life of the patients to an extent. 39
Since the discovery that ASD might have an inflammatory etiology, among the others, some researchers of the immunology field have focused on finding the immunological aspects of ASDs pathogenesis. 40 The majority of studies conducted so far have been considering inflammatory cytokines and there are a few studies that have addressed the immuno-metabolic interactions in ASD pathogenesis.41,42
Among the inflammatory mediators are the adipokines which are chemicals mainly secreted by the adipose tissue as well as some immune cells and subsequently impact immune responses in health and diseases such as ASD. 43 The alteration of some adipokines in ASD has been reported in a limited number of studies, including downregulation of adiponectin, 44 upregulation of leptin, 45 and downregulation of Fatty acid-binding protein 4. 46
Among the recently discovered adipokines, are the C1q/TNF-related protein family (CTRP) which are implicated in various inflammatory conditions and contribute to different functions such as regulation of metabolism pathways as well. The first member of this family, namely CTRP1, has been studied in some clinical conditions associated with metabolism and inflammation such as diabetes, dyslipidemia, atherosclerosis, cancer, etc.47-54
To our best knowledge, no study has investigated CTRP1 in ASD yet. According to the previous studies indicating the dysregulation of adipokines in patients with ASDs and also the improper formation of brain neuronal junctions in presence of inflammation55-57; we aimed to investigate the expression of CTRP1 in ASD preschool children presenting to Ali-Asghar children hospital, Tehran, Iran. both at the gene expression level and protein level; and in case a significant difference was observed between the ASD group and healthy controls, evaluate the diagnostic value of CTRP1 in differentiating ASD children from their healthy counterparts.
Furthermore, regarding the many different roles of CTRP1 in metabolic and cardiovascular disorders, investigating this adipokine in ASD children who are frequently suffering from such disorders, could be utilized as a prognostic factor too.58,59
Purpose of current study
Since detection of proper diagnostic and prognostic markers will lead to more effective treatment and intervention, we aimed to determine the level of CTRP1 in patients with ASDs in comparison with healthy subjects, a marker that may be useful for both diagnostic and prognostic assessments.
Method
Patients
Demographic characteristics of children with ASD.

Comorbidities in ASD children.

RNA extraction and cDNA synthesis
0.5 mL blood was collected from both the ASD group and control group into an EDTA container. Total RNA was isolated from whole blood using RNAzol-BD according to the manufacturer’s instructions. The concentration and quality of total RNA was assessed by Thermo Scientific™ NanoDrop 2000-spectrophotometer as well as gel electrophoresis. Then the resulting RNA samples were immediately reverse transcribed into cDNA using a cDNA synthesis kit (Takara, Japan) Based on the manufacturer’s protocol. cDNA stocks were stored at −20°C before qRT-PCR.
Quantitative Real-Time PCR
The qRT-PCR was conducted using SYBR green master mix (Amplicon, Canada) and specific primers as follows; CTRP1 forward primer: (5′-TGAGGGATAGGTGGACCCTG-3′), CTRP1 reverse primer: (CTCGCAGCTGTGACTCAGAA) GAPDH forward primer: (GTCTCCTCTGACTTCAACAGCG) and GAPDH reverse primer: (ACCACCCTGTTGCTGTAGCCAA). The gene expression of CTRP1 and GAPDH as the housekeeping gene was measured using the Rotor gene-Q Real-Time PCR system (Qiagen, Germany). The cycling profile consisted of a 15-min initial denaturation followed by 45 cycles of repeating 20-s denaturation and 40-s annealing/extension step. The relative changes in gene expression were calculated using the 2−ΔΔCT method and were compared using the unpaired T-test. CTRP1 gene expression data were normalized using the GAPDH gene as the internal control gene. The gene expression measurement by qPCR was performed in triplicates.
Enzyme-linked immunosorbent assay
The blood samples were obtained from the antecubital vein. After clotting was performed at room temperature, the serum samples of the patients and healthy volunteers were isolated by centrifugation (15 min, 1500g). For the next step, serum samples were stored at −70°C until the time of assessment. CTRP1 serum levels were analyzed by an enzyme-linked immunosorbent assay (ELISA) kit (Elabscience, USA) in duplicates. The minimum detection limit for CTRP1 was 3.13 ng/mL.
Statistical analysis
All statistical analyses were performed using SPSS software (22.0; IBM Corporation, Chicago, IL, USA). p values ≤ 0.05 were considered significant. In addition, GraphPad Prism software version 8.0 (GraphPad Software, Inc., La Jolla, CA, USA) was used for plotting the data. The comparisons were made using the unpaired T-test.
There is no community involved in this study.
Results
Gene expression of CTRP1
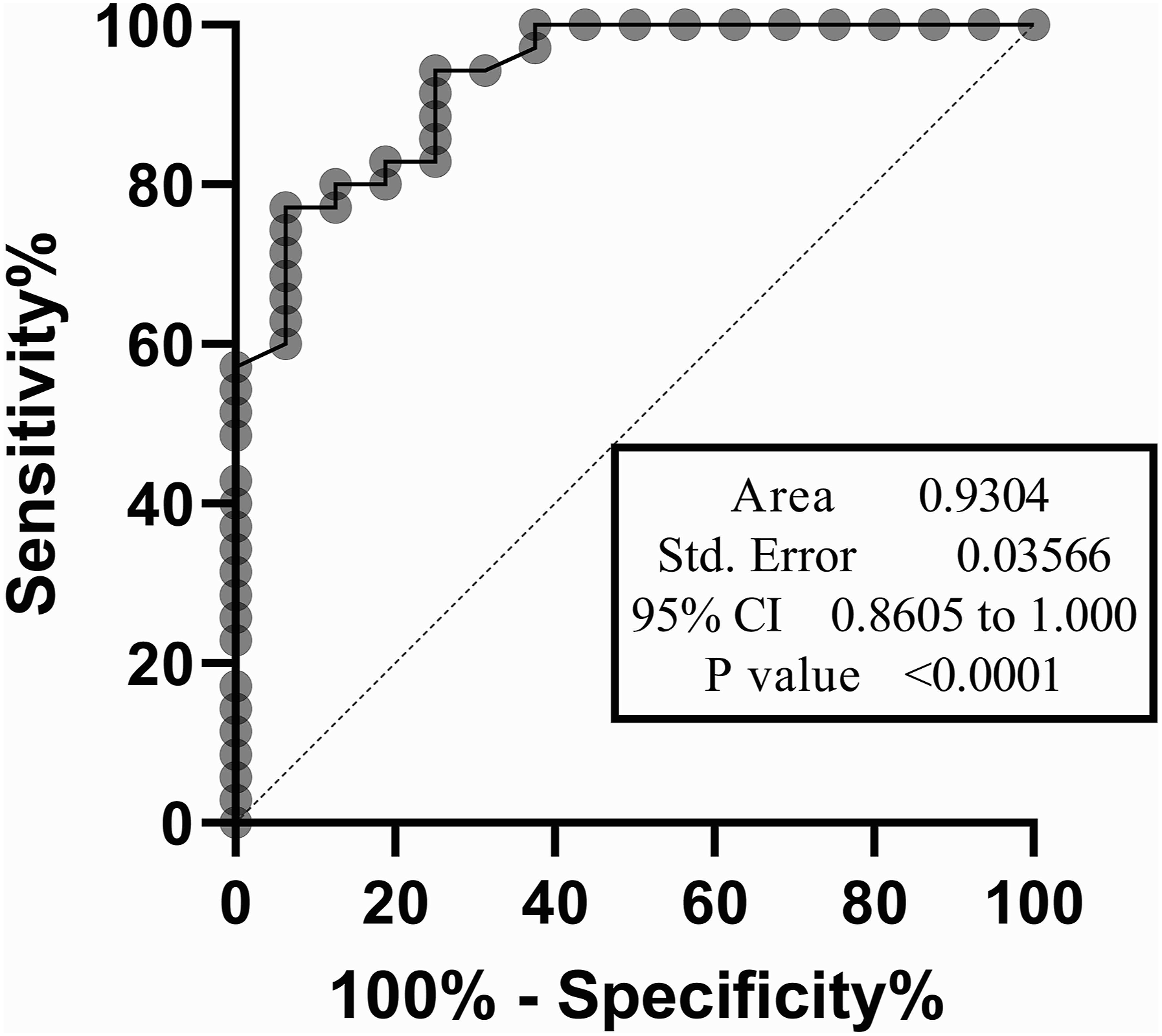
We evaluated the gene expression of CTRP1 both in the ASD group and normal healthy control group. The statistical analysis demonstrated that CTRP1 gene expression was significantly upregulated in the ASDs boys compared with healthy controls (p-value < 0.0001), as represented in Figure 1. Also, ROC curve analysis was performed to evaluate the diagnostic value of CTRP1 gene expression for differentiating ASDs from healthy individuals (Figure 2). An area under the curve (AUC) of 0.93 with a p value ≤ 0.0001 was obtained. The sensitivity and specificity of the selected cutoff point were ≈73% and ≈94%, respectively. Comparison of CTRP1 Relative Gene Expression between ASD children and Healthy Control Group using T-test. The mRNA expression level was normalized to the GAPDH level and displayed as relative expression. The data are represented as mean ± SD. (**** = p-value < 0.0001). ROC Curve plot for gene expression data. This plot indicates the capability of the CTRP1 gene expression as a diagnostic tool with an area under the curve of 0.9304 and a p-value ≤ 0.0001.
CTRP1 protein level
For further investigation, we evaluated the CTRP1 protein serum level in patients and healthy control groups. Our results showed that the CTRP1 protein level was higher among ASDs children rather than healthy controls (p-value < 0.0001), represented in Figure 3. Also, ROC curve analysis was performed on ELISA data, in order to assess the diagnostic value of CTRP1 serum protein levels in ASDs (Figure 4). The resulting sensitivity and specificity of the selected cutoff point were ≈84% and 97%, respectively. Comparison of CTRP1 Protein Level between ASDs Patients and Healthy Control Group. CTRP1 protein levels were evaluated using ELISA assay on participants' blood samples. The values are represented as mean ± SD. (**** = p-value < 0.0001). Roc curve plot for ELISA data. This plot demonstrates the capability of serum CTRP1 level in differentiating ASD children from healthy controls. As it is illustrated an area under the curve of 0.9528 with a p-value lower than 0.0001 was calculated in ROC analysis.

Discussion
Autism spectrum disorders are among the top new concerns of the new world, which affects nearly 1.9% of people living in western countries. 3 It is estimated that the ASD prevalence in Iran is lower than the western societies, but there is no certain data about this condition in Iran. 60 Also, this disease which has a male predisposition affects pupil boys 4 times more than pupil girls. 60 This condition which is mainly characterized by verbal difficulties, repetitive behaviors, introversion, and impaired social interactions among the others, is not curable; but the complications could be managed, and hence a much better social life could be achieved through psychotherapy or certain medications if ASD is caught at infancy. 62 Despite this, ASD diagnosis is usually delayed until school age especially in mild to moderate cases of ASDs63,64; since the early signs, such as repetitive movements or delayed talking might be misleading and considered normal by the parents or caregivers, or it may be misdiagnosed with attention deficit hyperactivity disorder (ADHD).65-68 Although early diagnosis of ASD even in infancy is crucial for the proper management of this condition, there are limited to no accepted clinical biomarkers for its accurate diagnosis; instead, the current global approach being practiced for ASD diagnosis is majorly made based on interviews and standard questioners such as the DSM5 rather than laboratory tests.10,65 Since the awareness for ASD has been raised in the world and especially in developing countries, finding a reliable diagnostic biomarker has been the focus of researchers in the field. 69
The current study was conducted in order to evaluate the diagnostic value of CTRP1 in ASD; given the contribution of the CTRP family in the modulation of inflammatory responses in previous studies, we hypothesized that the first member of this family, CTRP1, could be dysregulated in patients with ASD. As we had a collaboration with the psychiatry department of Ali-Asghar children’s hospital, Tehran, Iran, we sought to evaluate the gene expression level of CTRP1 as well as its serum protein level in children presenting to this hospital who were diagnosed with ASD. Accordingly, the measurements were performed and the results obtained from 41 ASD boys aged from 4 to 7 were compared to a group of 41 healthy and normally developing children. After statistical analysis, It was interestingly observed that the level of CTRP1 is significantly elevated in ASDs children both at the gene expression level and also at the protein level, in comparison with the healthy controls. In order to evaluate the diagnostic value of CTRP1, the receiver operating characteristic (ROC) curve was plotted and it was demonstrated that the gene expression of this adipokine, as well as its serum levels, are of a good diagnostic value for ASD diagnosis, both of which have an area under the curve of more than 0.9 with a p-value lower than 0.0001; also, the ROC curve analyses showed an accepted sensitivity and specificity in either case. At this point, we could propose a role for CTRP1 in ASD pathogenesis as it was shown to be significantly elevated in ASD children. The exact mechanism of CTRP1 contribution to ASD remains to be elucidated which of course necessitates further research. The majority of literature data are focused on the role of CTRP1 in metabolic and cardiovascular diseases and to our best knowledge, this is the first study that assessed the gene expression and serum levels of CTRP1 in ASD children. From the previous studies in other conditions, it is suggested that CTRP1 is increased in response to inflammatory cytokines such as IL-1β and IL-6; this adipokine functions through engaging with Adiponectin receptor1 and promotes inflammation through upregulation of pro-inflammatory cytokines such as TNF-α or IL-653,57; moreover, CTRP1 is released from immune cells such as monocytes and macrophages and these cells are also affected by CTRP1 through upregulation of NF-κB and P38 MAPK signaling pathways resulting in increased TNFα and adhesion molecules, for enhancing inflammation; furthermore, it promotes M1 phenotype in macrophages through activating TLR4 signaling as well.70-72 Besides, CTRP1 is capable of activating AKT-PI3K-mTOR pathways and as a result, the P53 levels are decreased which leads to malignancy and inflammation.52,73-75 AMPK, SIRT1, FOXO1, and IRS-1 are also affected by CTRP1; these factors not only contribute to metabolism hemostasis but also are implicated in various inflammatory diseases and malignancies, hence CTRP1 is considered as a link between lipid metabolism and inflammation.72,76-79 There are a few studies regarding the role of adipokines in ASD pathophysiology which demonstrate the aberrant levels of adipokines such as leptin, ghrelin, and adiponectin or their corresponding receptors in patients suffering from ASD80,81; the findings from those studies support the hypothesis that dysregulation of adipokines resulting from either improper diet or certain medications such as risperidone, could contribute to ASD pathogenesis.82-86 Even dysregulation of adipokines during pregnancy is proposed to increase the relative risk of developing ASD in the newborn child.87,88 In line with previous studies demonstrating the dysregulation of adipokines in ASD patients, our data also indicate that there might be an imbalance of adipokines in ASD children even in the absence of any identifiable metabolic or inflammatory diseases.44-46,89 Regarding the high incidence of obesity due to different reasons such as sedimentary life, certain medications, or much desire for food, it has been proposed that ASD children are predisposed to metabolic diseases or dyslipidemia, resulting in aggravation of their inflammatory profile and worsening their symptoms.90,91
In this regard, Maekawa. M et al., recently have demonstrated the downregulation of fatty acid-binding protein 4 (FAB4) in preschool ASD children which was further supported by gene sequencing analysis and mice models knockout for FAB4 gene which showed Autism-like symptoms, suggesting an adipose-brain axis in ASD; they have also proposed FAB4 as a convenient biomarker in ASD. 46 Although we did not assess other inflammatory cytokines along with CTRP1 to test whether there is a correlation between them in ASD children participating in our study, it could be proposed that other inflammatory cytokines might be dysregulated in the ASD group, which needs further evaluations.
There are some major limitations in our study that we were unable to address, first of all, based on previous national studies, the minimum sample size was estimated to be near 90; given the ASD prevalence of roughly 0.63 among children at the preschool age (5–7 years old). But we could only collect samples from 50 ASD children, of which 9 individuals were excluded due to other complications mentioned in Table 2. On the other hand, we assessed the history of children presented to the hospital so as to exclude those with metabolic diseases or inflammatory conditions; unfortunately, the majority of the ASD children were diagnosed with metabolic conditions such as hyperlipidemia, thyroid problems, or diabetes. Since at this stage, we only aimed to investigate the fluctuations in CTRP1 as a result of ASD itself not resulting from other conditions. Furthermore, due to the lack of collaboration with the expert and well-experimented nurses and psychiatrists in other centers we could not collect more samples from different areas in Tehran or even in Iran. Selection of the control children also was a challenge; we would like to randomly choose among the children who were screened positive for ASD and then were not confirmed at the referral hospital, but only a few parents were eager to enroll their children in the study, as a result, we could only test the blood samples from healthy normal developing children who were referred to the hospital laboratory with normal laboratory findings and no known history of the mentioned illnesses as the exclusion criteria. Assessing other factors and inflammatory cytokines or laboratory parameters along with the CTRP1 could be very beneficial in the current study but due to the limited fund allocated to our study as well as a short deadline from the university to complete it, we were unable to conduct more evaluations. Also, comparing CTRP1 diagnostic value with other proposed biomarkers could enhance the scientific merit of the study. On top of that, we only assessed the aberrant expression of CTRP1 in ASD boys, due to the very low number of girls presenting to our center; we could also evaluate the role of CTRP1 in ASD pathophysiology and determine whether it is a causal factor contributing to ASD or it is aberrantly expressed in ASD children as a result of associated complications. We did every effort to our best knowledge so as to minimize the confounding factors, by excluding the ASD children who suffered from obesity or other comorbidities such as inflammatory diseases. We only had access to this very narrow age group (4–7 years old), because in the past few years a national screening has been conducted for ASD among preschool children in Iran. There is no precise estimation of ASD prevalence in Iran in other age groups, but the limited number of previous studies suggest that the prevalence of ASD in Iran is lower than in western countries, and we do not know yet if this difference is due to limited screening for various age groups and results from lack of proper diagnosis, or it is actually lower in Iran. Anyway, despite the limitations discussed here, we hope that this preliminary study fascinates other researchers and clinicians and inspires their future studies. Further studies are required to thoroughly assess the contribution of CTRP1 in ASD pathophysiology in both sexes and across different ages, especially in infants which is quite necessary for obtaining a definite conclusion; also, other members of the CTRP family are yet to be investigated.
Evaluation of CTRP1 level not only could be considered as a novel biomarker in the cytokine profile of ASD patients but also might serve as a suitable prognostic factor for cardiovascular and metabolic disorders in these patients. 92 Also, employing mice models of autism and knockout mice for CTRP1 could help gain a better insight. 93
Conclusion
In this study, it was demonstrated that CTRP1 relative gene expression, as well as its serum concentration, was higher in ASD boys at the age of preschool in comparison to healthy control boys matching their age. Regarding the good AUC in ROC curve analyses and corresponding acceptable sensitivity and specificity, it might be concluded that the evaluation of CTRP1 gene expression and/or serum concentration could be utilized as a biomarker along with other proposed biomarkers to facilitate the accurate diagnosis of ASD, especially at infancy.
Footnotes
Acknowledgments
We would like to appreciate Mrs. FatemehYusefi Laksari for her support and help in blood sampling.
Authors contributions
HR: sample collection and hypothesis development, PB: sample preparations, performed all lab experiments, and statistical/bioinformatics analyses, prepared the manuscript. FN was involved in the study design and primer design. MHS: patients gathering and diagnosis. MEA: helped with the study design as well as editing the manuscript. MS: helped with the study design as well as editing the manuscript. NM has designed the study and provided the study materials; also, she has guided all the study procedures from the first idea to the preparation of the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported bya grant from the Iran University of Medical Sciences (96013030675).
Ethics approval and consent to participate
All patients filled a consent form confirmed by the ethics committee of Iran University of medical sciences (ethic code: IR. IUMS.FMD.REC.1397.100).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Informed consent
Written informed consent was obtained from all subjects before the study.
