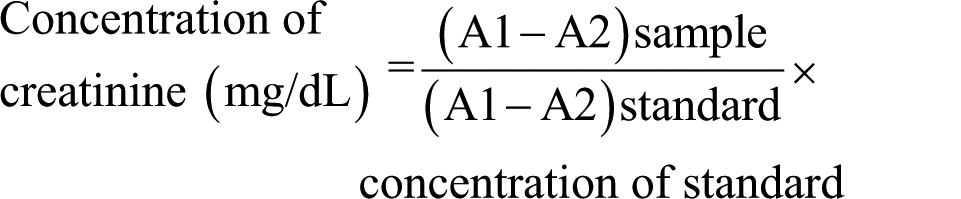Abstract
Diabetes patients suffer from chronic disorders in the metabolism due to high blood sugar caused by anomalies in insulin excretion. Recently, vanadium compounds have been prepared and functionalized to decrease the level of hyperglycemia. Vitamin A boosts beta cell activity; therefore, the lack of this vitamin plays a role in the development of type 2 diabetes. The aim of this article focused on the synthesis of a new anti-diabetic drug formed from the complexation of a vanadium(IV) salt with vitamin A. Vitamin A acts as a unidentate chelate through the oxygen of its –OH group. The vanadium(IV) compound is surrounded by two vitamin A molecules. The [VO(vitamin A)2(H2O)2] compound was synthesized in a binary solvent system consisting of MeOH/H2O (1:1 ratio) in alkaline media at pH = 8. This compound was characterized using Fourier transform infrared spectra (FT-IR), electronic spectra (UV–vis), effective magnetic moment, electron spin resonance (ESR), scanning electron microscopy (SEM), transmission electron microscopy (TEM), and thermal analysis (thermogravimetry (TG)–differential thermal analysis (DTA)). Anti-diabetic efficiency for the vanadium(IV) compound was assessed in streptozotocin (STZ)-induced diabetic mice. The results of the animal studies demonstrate the ability of the vanadium(IV) complex to act as an anti-diabetic agent, as measured by improvements of lipid profile, antioxidant activity (superoxide dismutase), malondialdehyde (MDA), glutathione, methionine synthase, and kidney and liver functions.
Introduction
Oral dosage of vanadyl(IV) sulfate salt improved the insulin sensitivity in insulin-independent diabetes mellitus patients. 1 Additionally, a vanadyl sulfate oral dose also improved hepatic/peripheral insulin efficiency in insulin-independent diabetes patients. 2 Moreover, the vanadyl(IV) sulfate salt improves muscle and hepatic insulin efficiency for type 2 diabetes. 3 Several different coordination compounds containing vanadyl(IV) ions have been studied in vitro and in vivo as insulin-mimetic agents.4–6 Sakurai et al.7,8 synthesized a colored vanadium(IV)bis(1-oxy-2-pyridinethiolato) chelate and showed that this compound acted as an insulin-mimetic agent in vitro and in vivo in insulin-dependent diabetes (type 1) following daily intra-peritoneal (i.p.) injection or oral administration. Vanadyl(IV) form is less toxic than vanadate ions and is more active in biological cells.9,10
The increase in the number of diabetes patients and others who have a predisposition to diabetes is due to population increase, aging, obesity, and lack of exercise. 11 In 1985, the World Health Organization (WHO) reported that 30 million people worldwide had diabetes, and this number increased to approximately 135 million in 1995 and continued to grow until eventually reaching 217 million in 2005. The WHO estimates the number of diabetes cases will rise to approximately 366 million by 2030, 12 with the majority of diabetic patients residing in poor countries. 13 The increase in blood glucose levels is due to the inability of liver or peripheral tissues to metabolize glucose in addition to the activation of gluconeogenesis in liver and kidney.14,15 Lipidemia is a significant risk factor for cardiovascular complications in type 2 diabetes patients at all ages.16–18 Additionally, high blood sugar causes many adverse effects on human blood vessels, such as impairment of the coronary artery, kidney, nerve, and retina.19,20 There are a number of literature reports on vanadium(IV) chelates that have been prepared and studied as agents for reducing hyperglycemia.21,22 These compounds are important due to their biological activity and the limited side effects of vanadium compounds.23–25
Vitamin A is thought to play an important role in islet regulation and pancreas development. 26 In obese mice, vitamin A supplementation resulted in reduced hyperglycemia and hyperlipidemia. 27 This highlights the importance of vitamin A in disease biology and calls for an exploration of the molecular mechanism of action in therapy. 28 This study reports the synthesis of a vanadium(IV) complex with Vitamin A in a 1:1 MeOH/H2O solvent system as a new insulin-mimetic agent and assessment of its anti-diabetic activity compared to vanadium(IV) sulfate toward the aim of developing a more effective and less toxic agent.
Materials and methods
The analytically pure chemicals (VOSO4·xH2O and vitamin A (Figure 1) and solvents (methanol and dimethylsulfoxide (DMSO)) were used as received from Aldrich Company without further purification.

Structure of retinol (vitamin A) compound.
Synthesis of binuclear vanadium(IV) compound
The new neutral vanadium(IV) compound, [VO(vitamin A)2(H2O)2], has a molar ratio 1:2 (V(IV):vitamin A). This compound was synthesized by reacting VOSO4·xH2O (1 mmol, 0.163 g) and vitamin A (2 mmol, 0.572 g) in 40 mL (1: 1 MeOH/H2O) mixed solvent. This mixture was adjusted to pH 7–8 using 0.1 M ammonia solution. The neutralized mixture was refluxed in a water bath at 45°C for 1 h. The dark green precipitate was isolated, washed three times with a small amount of CH3OH, and dried under vacuum over P2O5. The [VO(vitamin A)2(H2O)2] solid compound was obtained in 69% yield. Elemental analysis was performed, and the data are reported as %C, %H, and %V. Calcd: C, 71.30; H, 9.27; V, 7.56. Found: C, 71.21; H, 9.14%; V, 7.43.
Chemical analyses
Elemental analyses were performed using a Perkin Elmer CHN 2400 Elemental Analyzer. A Jenway 4010 conductivity meter was used for determining molar conductance of samples that were dissolved in DMSO solvent at 1.0 × 10−3 mol/cm3 concentration. A UV2 Unicam UV−Vis Spectrophotometer with quartz cell (1.0 cm path length) was used to scan the binuclear vanadium(IV) compound over the wavelength range of 900−200 nm. A Bruker Fourier transform infrared (FT-IR) spectrophotometer was used over the range of 4000–400 cm−1 to study the coordination mode of vitamin A with vanadium(IV) ion. The effective magnetic moment of the vanadium(IV) complex was obtained using a Gouy magnetic balance at room temperature. A Jeol JES-FE2XG electron spin resonance (ESR) spectrometer was used at a frequency of 9.44 GHz to collect the ESR spectrum for [VO(vitamin A)2(H2O)2]. Scanning electron microscopy (SEM) was performed with a Quanta FEG 250 system to collect images of the sample surface. Thermal analyses (thermogravimetry (TG)–differential thermogravimetry (DTG)) were performed on a Shimadzu 50−H analyzer under inert gas from 25°C to 800°C.
Biological analyses
Animals
Male MFI mice (weighing 25–35 g) were purchased from National Research Center in Cairo (Egypt). The treatment group of mice (N = 30) were injected with streptozotocin (STZ) at a dosage of 50 mg/kg body weight for the induction of diabetes, and a control group of mice (N = 10) were left untreated. A blood glucose level of >450 mg/dL was considered an indicator of diabetes induction. Diabetes induction in the mice was determined by measuring blood glucose level, and the diabetic mice were classified into three groups. The first group was not treated and named +Ve control, the second group was treated with VOSO4·xH2O (100 mg/kg dosage) as a single dose, and the third group was treated with the binuclear vanadium(IV) compound, [VO(vitamin A)2(H2O)2]. After 1 month, the mice were anesthetized using diethyl ether, and the blood specimens were collected in heparinized tubes. The other blood samples were collected into anticoagulant free tubes for serum separation. These samples were centrifuged for 10 min at 3000 r/min. The clear serum was collected and preserved in a freezer at −20°C until analyses were performed. The experimental data are reported as the mean ± standard deviation (SD). Statistical analysis was performed and indicated a significant difference between two means. The blood samples also underwent the following analyses: blood glucose level, creatinine serum, superoxide dismutase (SOD), glutamate pyruvate transaminase (GPT) serum, blood glucose-6-phosophate dehydrogenase (G-6-PD), lactate dehydrogenase (LDH), and hemoglobin (% Hb). Experimental diabetes was induced in 18 h fasted mice by single i.p. injection of STZ in a dose of 50 mg/kg, freshly prepared in cold 0.1 M citrate buffer (pH 4.5). STZ-injected mice were provided with a 5% glucose drinking solution for the first 24 h to ensure survival. Normal control group was injected with citrate buffer alone. Animals were considered diabetic when their blood glucose level exceeded 220 mg/dL and were included in the study after 72 h of STZ injection.
Estimation of glucose level in blood
The level of glucose in the blood was calculated using Spinreact kits. A 10 µL of serum sample was incubated in the presence of 1 mL of kit reagent at approximately 377°C for 10 min, and the absorbance was measured at 450 nm using a Bio-system 320 spectrophotometer and compared with a blank tube. The percentage of glucose was calculated using the following equation
Estimation of serum creatinine
The serum level of creatinine was estimated by forming a colored creatinine picrate complex (1:1 ratio) in alkaline media. The color of creatinine picrate complex is proportional to the concentration of creatinine. A total of 10 µL of clear serum was added to 1 mL of sodium hydroxide:picric acid solution (1:1 mixture). Next, equal volumes of the creatinine/picric acid reagent and creatinine buffer were mixed. In total, 3.0 mL of working reagent was added to test tubes, and 0.1 mL of sample was added to the appropriate tube. Distilled water was added to the reagent blank followed by mixing. All tubes were transferred into a water bath at 37°C for 15 min. The absorbance values were measured at 510 nm in a spectrophotometer after standardizing the instrument with the blank sample. The room temperature absorbance measurements were called the A1 readings. After the first reading, 0.1 mL of acetic acid reagent was added to each tube followed by mixing. The solutions were allowed to stand for 5 min before reading absorbances a second time (A2 readings). The absorbance values for both standard and unknown were used to calculate creatinine values using the following equation
Estimation of blood SOD
The SOD activity was calculated using a biodiagnostic kit. A 0.5 mL of EDTA blood was washed four times with normal saline. The washed erythrocytes were resuspended in 2.0 mL cold redistilled water, mixed and then let stand at 4°C for 15 min. A total of 100 µL of this lysate was mixed with the appropriate reagent and the absorbance was measured at 560 nm for 5 min for both control (∆A) and sample (∆A). The % of inhibition was calculated according to the following equation
Results
Micro-analytical and physical discussions
The new binuclear vanadium(IV) compound is dark green in color and possesses thermal stability. The vitamin A acts as a unidentate ligand and binds to the vanadium(IV) ions through the oxygen of the hydroxyl group following deprotonation. The chemical analyses of the solid vanadium(IV) complex support the 1:2 (V(IV):vitamin A) stoichiometric ratio. The molar conductance of the vanadium(IV) compound measured in DMSO was 20 Ω−1 cm2 mol−1, which indicates non-electrolytic behavior.
29
The conductance value gives information regarding the ionization status of metal–organic compounds.
29
Thus, the conductance data suggest that no
Infrared investigation
The infrared spectrum of the new vanadium(II)–vitamin A compound is shown in Figure 2. The FT-IR spectrum of free vitamin A has some distinguishing bands at several frequencies. The spectrum of vitamin A has a broad band at ~3400 cm−1 that is attributed to stretching vibrations of the O−H (alcoholic) group. The wavenumbers in the range of 2950−2825 cm−1 are assigned to stretching vibrations of aliphatic C−H (CH2 and CH3) groups. 30 The bands exhibited at 1590 and 1451 cm−1 are due to −CH2 bending vibration, and the band at 1382 cm−1 corresponds to symmetric C−H stretching vibrations. The bands at 1359 and 1073 cm−1 are due to C−O stretching vibrations and O−H bending motions. The absorption band at 885 cm−1 is due to the =C−H group. 30 The peak in the spectrum of free vitamin A exhibited at 1628 cm−1 is assigned to stretching vibrations of C−C. The FT-IR spectrum of the vanadium(IV) compound is similar to the spectrum of free vitamin A, but there are a few changes which are attributed to chelation. The disappearance of the −OH frequency and the shifting of ν(C−O) and δ(O−H) to lower wavenumbers (1339 and 1031 cm−1) suggest the participation of the alcoholic −OH group of vitamin A in chelation of vanadium(IV) metal ion. This interpretation is consistent with the ionized −OH group coordinating vitamin A as a unidentate ligand. 30 The new band exhibited at 964 cm−1 in the vanadium(IV) compound is attributed to the stretching vibration of the oxovanadium group V = O. 30 There are two other new bands at 582 and 537 cm−1 attributed to V−O stretching vibrations. The presence of two water molecules inside the coordination sphere is suggested by the presence of broad bands at 3378 and 3153 cm−1 caused by asymmetric and symmetric vibration motions of −OH group. The distinctive bending bands of water molecules δb(bend), δr(rock), δt(twist), and δw(wag) are exhibited at 1630, 851, 582, and 645 cm−1, 30 respectively. Some vibrational motions of vitamin A are overlapped with the angular deformation motions of coordinated water molecules.

FT-IR spectrum of vanadium(IV)–vitamin A new compound.
UV–vis, ESR, and magnetic discussions
Free vitamin A has three UV absorption bands at 250, 310, and 375 nm due to π→π* and n→π* electronic transitions. 31 After binding to vanadium(IV) ion, there are some shifts in the two types of electronic transitions due to the new electronic configuration of the vanadium(IV) compound. At room temperature, the vanadium(IV) compound has an effective magnetic moment of 1.63 B.M. which is in the range of spin values corresponding to one unpaired electron for the vanadium atom. 32 The diffuse solid reflectance spectrum of the vanadium(IV) compound has three distinguishing bands at 13,793, 16,667, and 22,936 cm−1 attributed to 2B2→2E, 2B2→2B1, and 2B2→ 2A1 electronic transitions, respectively, for square pyramidal geometry around V(IV) ion (Figure 3). The experimental ESR spectrum of the [VO(vitamin A)2(H2O)2] complex is presented in Figure 4. The g┴(1.945) < g║(2.089) values are in agreement with those of square pyramidal complexes possessing C4V symmetry with the unpaired electron in the dxy orbital. 33

Proposed structure of new synthetic vanadium(IV) compound.

ESR spectrum of [VO(vitamin A)2(H2O)2] compound.
Scanning and transmission electron microscopes
The morphological surface of the vanadium(IV)−vitamin A compound was studied using SEM (Figure 5). The micrograph of [VO(vitamin A)2(H2O)2] compound reflects a well sintered and homogeneous surface. The transmission electron microscopy (TEM) image was scanned using a JEOL JEM 1400 transmission electron microscope with an accelerating voltage of 120 kV. Figure 6 shows a TEM image of the [VO(vitamin A)2(H2O)2] nanoparticles synthesized, showing that the particles have spherical shapes with sizes varying from 10 to 200 nm.

SEM micrograph of [VO(vitamin A)2(H2O)2] compound.

TEM image of [VO(vitamin A)2(H2O)2] compound.
Thermal analyses
The thermal decomposition curves (TG/differential thermal analysis (DTA)) of the [VO(vitamin A)2(H2O)2] complex are shown in Figure 7. The thermal decomposition process takes place in two exothermic steps by DTA. These decomposition steps occur in the temperature range of 100°C–500°C at DTA = 350 and at 450°C are attributed to thermal fragments of vanadium(IV) compound with loss of two coordinated water molecules with an experimental mass loss 87.37% (theoretical = 87.69%). The final residual product that is thermally stable from 500°C (steady state) through 800°C is vanadium(IV) oxide, VO2, with experimental TG data showing 12.63% (theoretical = 12.31%). The experimental and theoretical data are in good agreement.
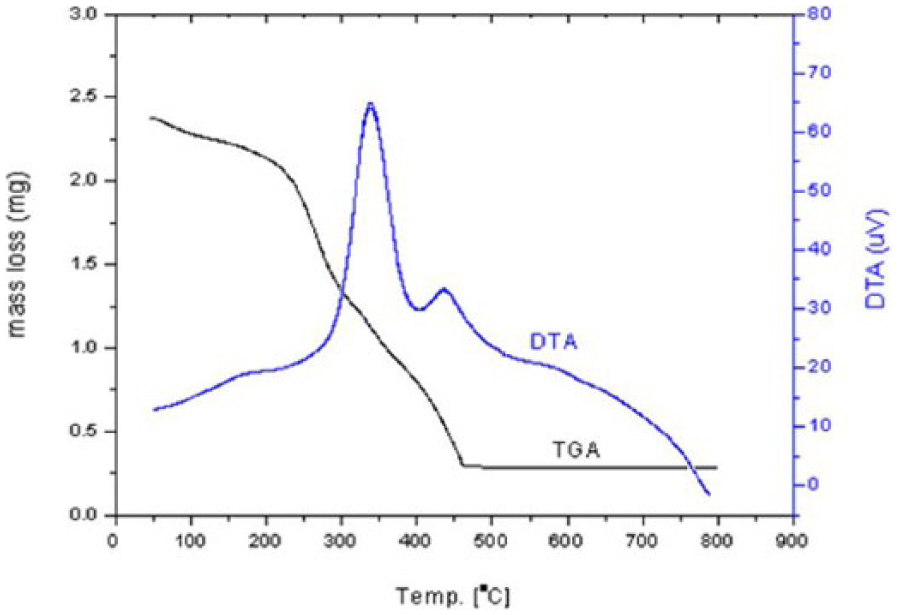
TG and DTA diagrams of [VO(vitamin A)2(H2O)2] compound.
Test of insulin resistance
Four experimental animal groups are classified as follows: Group I is a negative control (without treatment by both ZTZ or vanadium(IV) sulfate, Group II is a positive control (treatment with STZ only), Group III consists of diabetic rats treated with vanadium(IV) sulfate at 100 mg/kg body weight dosage, and Group IV consists of diabetic mice treated with the vanadium(IV) vitamin A complex at a dose of 100 mg/kg body weight.
Blood glucose level
The blood glucose level for the different experimental groups are shown in Figure 8. The results show that blood glucose level is significantly decreased (33%) from 472.75 mg/dL (positive control group and Group II) to 317.22 mg/dL in Group III (treated with vanadium(IV) sulfate at 100 mg/kg dose) and is decreased to a greater extent (44%) to 262.78 mg/dL in the case of Group IV (treated with vanadium(IV) vitamin A complex with 100 mg/kg dose).

Mean(±)SE for the blood glucose level (mg/dL) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
GPT enzyme activity
The GPT enzyme provides a measure of liver function to assess liver cell damage. 34 The presence of GPT at high levels indicates liver tissue damage. The GPT enzyme activity in the different animal experimental groups is shown in Figure 9. The experimental data show that the activity of GPT enzyme is increased from 142.17 U/dL in untreated diabetic animals (Group II) to 154.46 U/dL in Group III animals treated with vanadium(IV) sulfate. This activity decreased to 117.92 U/dL in the case of Group IV animals that were treated with the vanadium(II) vitamin A complex.

Mean(±)SE for the GPT serum (U/dL) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
SOD enzyme
The SOD enzyme is an essential enzyme in the antioxidant system, so increased SOD activity indicates a positive effect of an antioxidant agent in living systems. Figure 10 shows that the treatment of diabetic mice with vanadium(IV) sulfate and the vanadium(IV) vitamin A complex with low dose moderately increases the antioxidant activity by SOD activity.

Mean(±)SE for the SOD superoxide dismutase (U/dL) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
Hemoglobin levels
The hemoglobin levels of the different experimental groups (GI−GIV) are presented in Figure 11. The results show only a slight effect on the hemoglobin level in all experimental animal groups treated with the vanadium(IV) compound at different doses.

Mean(±)SE for the hemoglobin (g/dL) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
Glucose-6-phosphate dehydrogenase
Glucose-6-phosphate dehydrogenase (G-6-PD) converts glucose-6-phosphate to 6-phosphoglucono-δ-lactone and is the rate-limiting enzyme of this metabolic pathway. G-6-PD shortage is a genetic condition in which red blood cells (RBCs) mold when the body is exposed to certain drugs or the stress of infection. 35 The activity of G-6-PD enzyme in different experimental groups is presented in Figure 12. The results show that the enzyme activity decreases with oral administration of the vanadium(IV) complex at different doses from 3635.74 U/g hemoglobin (Group II) to 3158.78 U/g (Group III) and 3525.56 U/g (Group IV), respectively. This is remarkable side effect of administration of vanadium(IV) compound.

Mean(±)SE for the glucose-6-phosphate dehydrogenase (G-6-PD) (U/L) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
LDH activity
The LDH enzyme is an indicator of tissue breakdown. LDH is present at high levels in RBC and can act as a marker of hemolysis. 36 The activity of LDH in the experimental groups of animals is shown in Figure 13.

Mean(±)SE for the lactate dehydrogenase (LDH; U/L) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
Serum creatinine levels
Serum creatinine levels are an indicator of renal function, and increasing creatinine levels indicate late-stage kidney failure. 37 The creatinine levels of experimental groups are shown in Figure 14.

Mean(±)SE for the creatinine serum (mg/dL) for groups (I−IV). Group I: untreated control group, Group II: diabetic mice group (treated with STZ at a dose of 50 mg/kg), Group III: diabetic mice (treated with VOSO4·xH2O at a dose of 100 mg/kg), and Group IV: diabetic mice (treated with binuclear vanadium(IV) compound at a dose of 100 mg/kg).
Discussion
Free radicals are reactive chemical species which cause harmful oxidation to living beings by attacking macromolecules such as carbohydrates, lipids, nucleic acids, and proteins. Normal physiological function requires a critical balance in the generation of oxygen free radicals and antioxidant defense systems used by living organisms to activate and protect themselves against toxic free radicals.38,39 A decrease in the oxidation/antioxidant balance creates a condition known as oxidative stress. Recent studies have not specified that the overproduction caused by high blood sugar seems to be a main factor in the development of diabetes complications. Superoxide overproduction increases with increases in nitric oxide (NO) levels and upon the formation of the strong oxidant peroxynitrite and activation of poly (adenosine diphosphate-ribose) polymerase, which in turn further affect the pathways implicated in the development of diabetes-related complications. 40
Hyperglycemia-induced overproduction of superoxide is the linkage between high glucose and the pathways responsible for hyperglycemic damage. In fact, diabetes is typically associated with increased generation of free radicals and/or impaired antioxidant defense qualifications, representing a central contribution of reactive oxygen species in the onset, progression, and pathological consequences of diabetes. 41 The vanadium(IV) salts can be used under oral administration as hypoglycemic agents against STZ-induced diabetes mellitus in experimental animals. Efficiency of vanadium(IV) compound was monitored by different biochemical parameters such as GPT, SOD, creatinine, and hemoglobin.
In this work, the blood glucose level data show that oral administration of the new vanadium(IV) vitamin A compound is effective as a hypoglycemic agent. The GPT activity data suggest that treatment with the vanadium(IV) vitamin A complex has a low liver toxicity compared to untreated Group II diabetic animals. The hemoglobin levels data suggest that the vanadium(IV) compound has a low toxicity in living system tested. In comparison of the LDH activity data with the positive control, there are only slightly significant effects after treatment with the vanadium(IV) complex at different doses. This negligible effect suggests that the vanadium(IV) complex has a low side effect on the RBC. It was found that the vanadium(IV) complex at different doses had no influence on the creatinine levels. These results suggest that the vanadium(IV) compound has no side effects on the kidneys in experimental mice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The project was financially supported by King Saud University, Vice Deanship of Research Chairs.