Abstract
The aim of this study was to investigate the possible protective effects of diallyl trisulfide (DATS) against naphthalene-induced oxidative and inflammatory damage in the livers and lungs of mice. Elevated serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels showed significant hepatic damage after the challenge with 100 mg/kg naphthalene. Hepatic malondialdehyde (MDA) contents and the activity of myeloperoxidase (MPO) increased significantly, accompanying a decrease in the hepatic activity of total superoxide dismutase (SOD) and glutathione (GSH) levels after the naphthalene damage. In addition, the serum levels of nitric oxide (NO), tumor necrosis factor α (TNF-α), and interleukin 8 (IL-8) increased significantly in the groups damaged with naphthalene. The main parameters related to oxidative stress and inflammatory responses in the lungs, including the NO, MPO, and GSH contents, were determined, and the histopathological and immunohistochemical changes in the lung and liver tissues were also observed. In the DATS-treated groups, all of the oxidative and inflammatory damage in the serum, liver, and lung tissues were significantly prevented.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) have been listed as priority pollutants. Naphthalene (NAP), one of the most toxic PAHs, is commonly found in waste water.1,2 Due to its chemical stability and lipophilic nature, NAP biodegradation is difficult, and NAP is easily accumulated and biomagnified in the food chain. The kinetics of NAP metabolism in microsomes and the main metabolites have been described. 3 Many studies have been performed in order to investigate the toxicity of NAP and its metabolites in experimental animals by various routes of administration. The ability to cause pulmonary, hematologic, and ocular damage has been confirmed.4–6 In addition, NAP-induced genotoxicity has also been reported. 7 Due to its volatility, NAP might cause irritation following inhalation.8,9 NAP-induced lung injury affects the incidence and severity of several pulmonary diseases. 10 The inflammatory response is used to evaluate lung damage.11,12 A previous study characterized the cytotoxicity to the cells of both target (lung, nasal epithelium) and non-target (liver) organs following exposure to high concentrations of NAP. 13 Another study demonstrated that NAP exposure results in respiratory tract tumors in mice and rats. 14 There is evidence that NAP can induce oxidative stress as shown by measurements of hepatic lipid peroxidation and glutathione (GSH) depletion.15,16
Diallyl trisulfide (DATS) is a major organosulfur compound found in the alliaceae family members, including garlic. It displays a wide variety of internal biological activities.17–19 Some findings indicate that DATS protects against hyperglycemia-induced ROS-mediated apoptosis during diabetes treatment. 20 DATS dramatically attenuates acute ethanol-induced liver injury and mitochondrial dysfunction. The increases of hepatic GSH levels and the elevation of the antioxidant enzyme activities account for the preventive effects. 21 The anti-inflammatory properties of DATS have been evaluated, and the mechanisms might be related to the inflammatory signaling mediated by NF-κB and STAT3. 22
The aim of this study was to evaluate the serum, liver, and lung tissue alterations induced by the toxic effects of NAP and to investigate whether DATS was beneficial in preventing or reducing the various damage. The protective effects of DATS against naphthalene-induced liver injury were examined by testing serum hepatic enzymes of ALT, AST, and LDH, as well as the oxidative enzyme of SOD. Serum cytokines of nitric oxide (NO), tumor necrosis factor α (TNF-α), and interleukin 8 (IL-8) were assayed to explore the anti-inflammatory activities of DATS. The prevention potentials of DATS on oxidative damage in the liver tissues were evaluated by the determination of SOD, MDA, MPO, and GSH while MPO, NO, and GSH were assayed for the lung tissues. Finally, the histopathological and immunohistochemical analyses of lung and liver tissue injury were also conducted to detect morphometrical changes in the lung and liver.
Materials and methods
Animals and experimental groups
Kunming mice (weight range, 18–20 g) of either sex were purchased from the Center for New Drug Evaluation of Shandong University, Jinan, PR China. The mice were housed in a room at a mean constant temperature of 25°C with 50–60% relative humidity and a 12-h light/dark cycle, and they had free access to food and water. The mice were maintained under these conditions for at least 1 week before the experiments.
These mice were divided into nine groups, each comprising 10 animals (5 females, 5 males): Blank, without any treatment group; DATS, DATS only at a dose of 40 mg/kg p.o.; Pre-1, pre-treated with DATS for 7 days at doses of 20 mg/kg p.o.; Pre-2, 40 mg/kg p.o.; Pre-3, 80 mg/kg p.o.; Nap, NAP only at a dose of 100 mg/kg p.o.; and Co-1, Co-2, and Co-3, DATS at doses of 20, 40, or 80 mg/kg p.o., respectively, after the treatment of naphthalene. DATS and NAP were both dissolved in soybean oil. At 6 h after the NAP challenge, the mice were killed by decapitation, and blood was collected. The serum and tissue (liver and lung) samples were stored at −80°C before the analyses. Part of each lung tissue sample was embedded in paraffin for histological assessment. All animals received humane care, and the study protocols were conducted according to EU Directive 2010/63/EU for animal experiments.
Tissue sample preparation
The liver and lung tissue samples were homogenized with ice-cold physiological saline (10%, wt%, 1 g tissue in 10 mL of physiological saline) in an Ultra Turrax tissue homogenizer. The homogenate was centrifuged at 2500 rpm, and the supernatants were diluted to an appropriate concentration with physiological saline. The supernatant was analyzed for total protein using the bicinchoninic acid (BCA) method.
Biochemical analysis
The serum AST, ALT, and lactate dehydrogenase (LDH) levels were measured to assess hepatic function. The assays were performed according to the ELISA kit instructions (Nanjing Jiancheng Bioengineering Institute). The mean value was calculated from the measurements performed at least in triplicate.
Determination of SOD activity
The SOD activity in the serum samples was determined using the microplate spectrophotometer method, and the serum SOD activity was expressed as U/mL. Hepatic SOD was measured using commercial detection kits according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute). The SOD activity was expressed as U/mg protein.
Determination of MDA
The MDA content was determined using the TBA method, which results in the formation of a red compound. The maximum absorbance of the compound was measured at 532 nm. The liver tissue samples were determined without dilution. The MDA levels were assayed as an indication of the products of lipid peroxidation by monitoring thiobarbituric acid-reactive substance formation. The lipid peroxidation as indicated by the MDA results was expressed as nmol/mg protein.
Determination of TNF-α and IL-8
The serum TNF-α and IL-8 levels were determined using commercial ELISA kits (Shanghai Xitang Bioengineering Institute) according to the instructions of the manufacturer. TNF-α and IL-8 concentrations were calculated from standard curves. The mean value was calculated from measurements performed at least in triplicate.
Determination of NO levels
The levels of NO were determined by an assay for nitrite. The procedure followed the instructions of the total nitric oxide assay kit (Nanjing Jiancheng Bioengineering Institute). The optical density of the assay samples was measured spectrophotometrically at 540 nm. The serum NO level was determined using the same method. The NO level was expressed as μM/g protein.
Measurement of MPO activity
The tissue MPO activity was assessed according to the instructions of the assay kit (Nanjing Jiancheng Bioengineering Institute). One unit of enzyme activity was defined as the amount of MPO present that caused a decomposition of 1 μM H2O2 at 37°C and was expressed in U/g tissue.
Measurement of GSH activity
The tissue GSH activity was assessed according to the instructions of the assay kit (Nanjing Jiancheng Bioengineering Institute). The results are expressed as μmoL/g protein.
Histological preparation
Samples of the lung and liver tissues were fixed in 10% formaldehyde and processed routinely for embedding in paraffin. Paraffin sections were stained with hematoxylin and eosin (H&E) and examined under a light microscope. Microscopic analyses were performed by experienced histologists unaware of the treatment to which the animal had been subjected.
Immunohistochemistry
Immunohistochemical staining from paraffin-embedded lung and liver tissues was performed in accordance with a routine procedure. The sections (10 μm) were mounted on glass slides. The sections were deparaffinized and incubated in 3% H2O2 for 10 min to quench the endogenous peroxidase activity. After blocking with normal goat serum for 20 min, the sections were incubated with anti-MPO or anti-NF-κB p65 primary antibodies (1:100, Beijing Biosynthesis Biotechnology Co., Ltd.) at 4°C overnight, followed by washing with PBS and incubating with goat anti-rabbit antibody at 37°C for 30 min. Then, they were developed using the DAB Envision System according to the manufacturer’s protocol (Nanjing Jiancheng Bioengineering Institute). All incubations were performed in a humidified chamber at room temperature.
Statistical analysis
Statistical analysis was carried out using GraphPad Prism 6.0. The groups of data were compared using analysis of one-way ANOVA followed by Tukey’s multiple comparison tests. Values of P <0.05 were regarded as significant. All data are expressed as means ± SD.
Results
Biochemical analysis
The results of the current study revealed that the various serum enzymes were strongly affected by NAP treatment, while upon treatment with DATS at either low or high doses, a significant improvement were achieved. ALT and AST levels were significantly increased in the NAP groups compared to the Con groups (P <0.001). Treatment with DATS caused a significant reduction in both ALT and AST levels compared to the NAP group. Significant differences in the SOD and LDH activities in the serum were detected after the treatment with NAP compared with the Con group (P <0.05 and P <0.001, respectively). The animals that received DATS showed a significant improvement in these antioxidant parameters toward the normal value of the controls. The soybean oil treatment did not influence the serum parameters. All of these results are shown in Table 1.
Serum ALT, AST, SOD, and LDH levels of different groups.
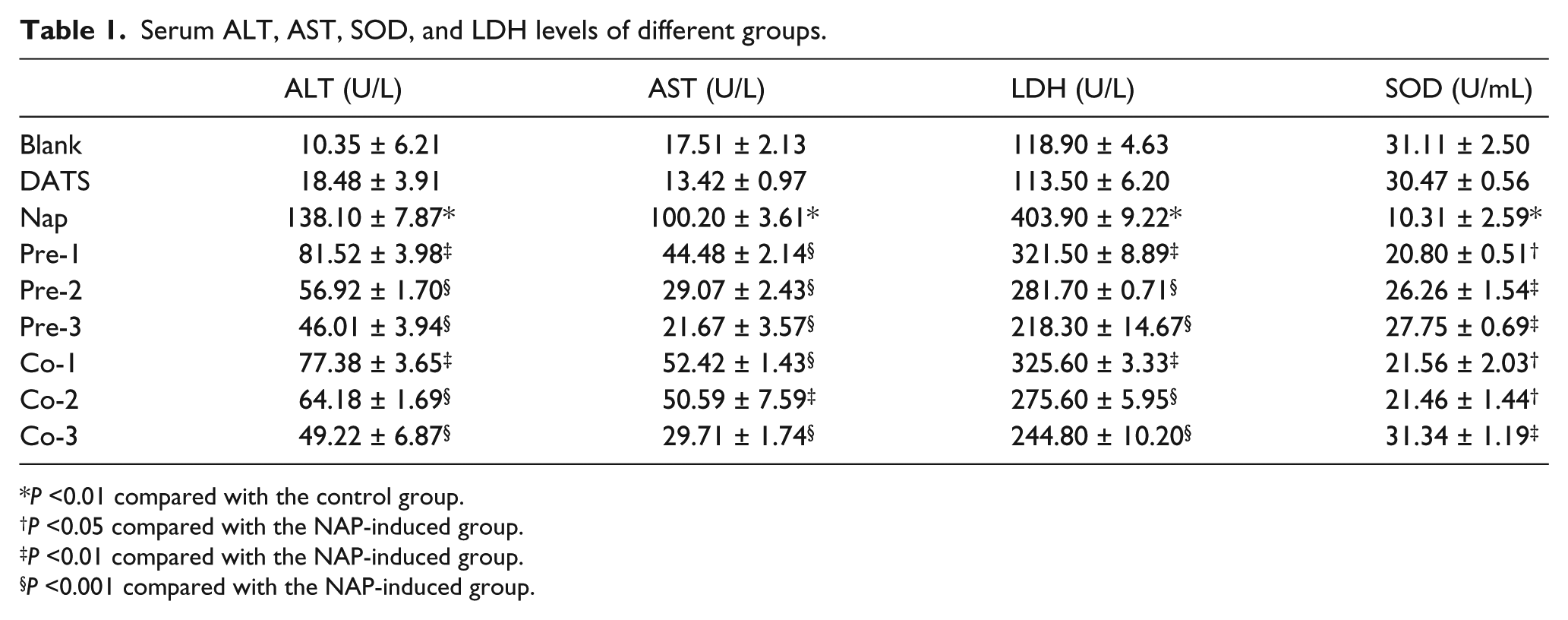
P <0.01 compared with the control group.
P <0.05 compared with the NAP-induced group.
P <0.01 compared with the NAP-induced group.
P <0.001 compared with the NAP-induced group.
Blank, no treatment; DATS, DATS only; NAP, naphthalene; Pre-1, Pre-2, and Pre-3, pre-treatment with DATS for 7 days, at DATS doses of 20, 40, and 80 mg/kg, respectively; Co-1, Co-2, and Co-3, after treatment with DATS for 7 days, the doses of DATS were 20, 40, and 80 mg/kg, respectively.
Determination of SOD, MPO, MDA, and GSH levels in the liver tissues
The levels and/or activities of SOD, MPO, MDA, and GSH in liver tissues were determined to evaluate how DATS functions to protect the liver tissue from oxidative damage. The results indicated significant cytotoxicity in the hepatic tissue of the NAP-fed mice. Moreover, treatment with NAP resulted in a significant decrease (P <0.001) in the SOD activity in the liver, which was accompanied by a significant increase in the levels of MDA (Figure 1a).

Determination of SOD (a), MDA (b), MPO (c), and GSH (d) levels in the liver tissues. Blank, no treatment; DATS, only DATS; NAP, naphthalene (model); Pre-1, Pre-2 and Pre-3, pre-treatment with DATS for 7 days, at doses of 20, 40, and 80 mg/kg, respectively; Co-1, Co-2, and Co-3, post-treatment with DATS for 7 days, the doses of DATS were 20, 40, and 80 mg/kg, respectively. #P <0.01 compared with the blank group; *P <0.05 compared with the NAP (model) group; **P <0.01 compared with the NAP (model) group; ***P <0.001 compared with the NAP (model) group.
The mean level of MDA, a major degradation product of lipid peroxidation, was increased in the liver tissues after NAP administration compared with the blank group (P <0.001; Figure 1b). In the DATS-treated groups (both pre- and post-treatment), there were markedly decreases in the MDA levels compared with the NAP group (P <0.05–0.001).
NAP administration increased the MPO activity in the liver compared with the blank group (P <0.05–0.001), and DATS treatment reversed this effect (P <0.05–0.001; Figure 1c).
The mean GSH levels of liver samples in the naphthalene-induced group were significantly lower than that of the blank group (P <0.01–0.001; Figure 1d). In all the DATS-treated groups (pre- and post-treatment), GSH levels were significantly elevated compared to the naphthalene-damaged group (NAP, P <0.05–0.001).
Determination of TNF-α, IL-8, and NO
The levels of IL-8 and TNF-α in the serum were determined to investigate the effect of DATS on the NAP-induced inflammatory response. Oral administration of NAP caused significant increases in the levels of the pro-inflammatory cytokines IL-8 and TNF-α compared with those in the normal control group. Pre-treatment of the mice with DATS (60 mg/kg) significantly decreased the levels of IL-8 and TNF-α in the serum compared with the levels in mice treated with NAP.
An overproduction of NO is thought to contribute to the NAP-induced mucosal damage. Therefore, we measured the total nitrate concentration as a marker for NO production in the lung tissue and serum. As shown in Table 2, administration of NAP resulted in significantly (P <0.001) increased levels of NO in the serum and lung tissue compared to normal mice. Treatment with DATS prior to or after the NAP administration showed a significant dose-dependent decrease in NO levels compared with the NAP group.
Serum NO, IL-8, and TNF-α levels of the various groups.
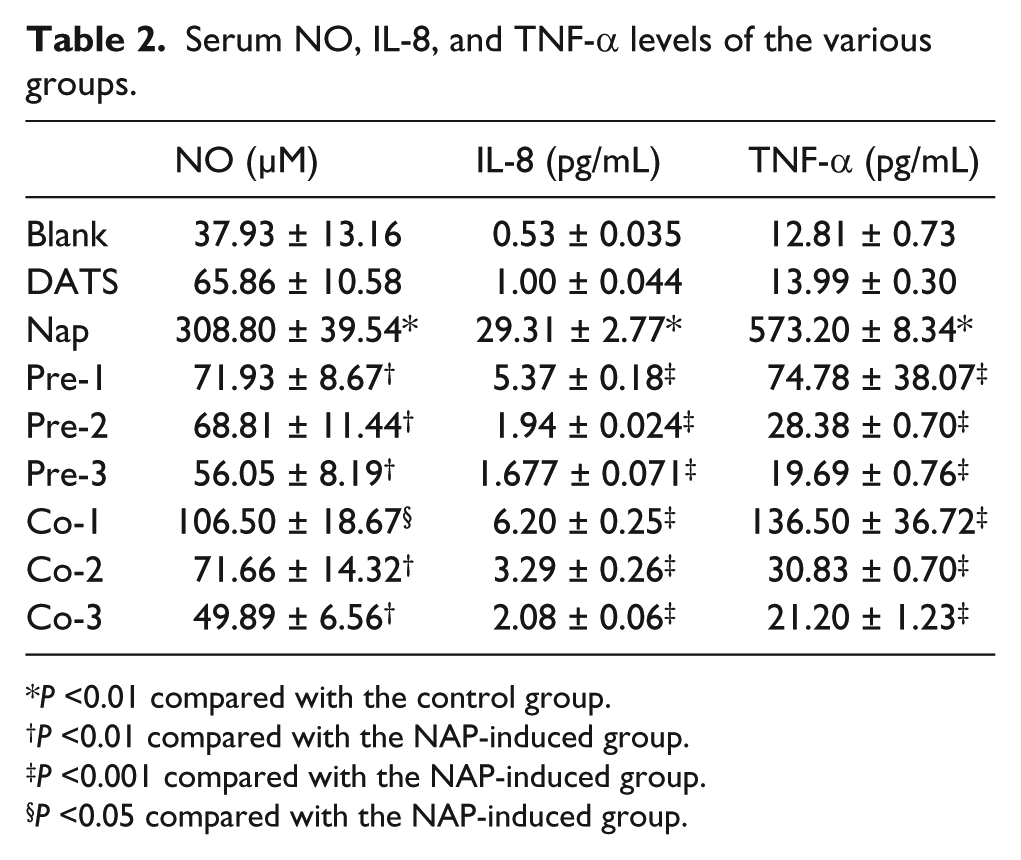
P <0.01 compared with the control group.
P <0.01 compared with the NAP-induced group.
P <0.001 compared with the NAP-induced group.
P <0.05 compared with the NAP-induced group.
Blank, without any treatment; DATS, DATS only; NAP, naphthalene only; Pre-1, Pre-2, and Pre-3, pre-treatment with DATS for 7 days, at doses of 20, 40, and 80 mg/kg, respectively; Co-1, Co-2, and Co-3, post-treatment of DATS for 7 days, at doses of 20, 40, and 80 mg/kg, respectively.
Measurement of MPO, NO, and GSH levels in the lungs
Administration of NAP significantly increased the activity of MPO, a marker of neutrophil infiltration into the lung mucosa, in comparison to the normal control group (Figure 2a). Treatment with DATS significantly reduced the levels of MPO compared to those in the NAP group (P <0.05). As shown in Figure 2b, the increased NO level in the lung tissues was reversed by the treatment of DATS, and the effects of the low and high doses of DATS (20 mg/kg and 80 mg/kg) were both statistically significant (P <0.05). The mean GSH levels of lung samples in the naphthalene-induced group were significantly lower than that of the blank group (P <0.01–0.001; Figure 1d). In all the DATS-treated groups (both pre- and post-treatment), GSH levels were significantly elevated compared to the NAP-damaged group (NAP, P <0.05–0.001).

Determination of MPO (a), NO (b), and GSH (c) levels in the long tissues. Blank, no treatment; DATS, only DATS; NAP, naphthalene (model); Pre-1, Pre-2, and Pre-3, pre-treatment with DATS for 7 days, at doses of 20, 40, and 80 mg/kg, respectively; Co-1, Co-2, and Co-3, post-treatment with DATS for 7 days, the doses of DATS were 20, 40, and 80 mg/kg, respectively. #P <0.01 compared with the blank group; *P <0.05 compared with the NAP (model) group; **P <0.01 compared with the NAP (model) group; ***P <0.001 compared with the NAP (model) group.
Histological analysis of lungs
Light microscopic examination of the H&E-stained sections from the sham group revealed normal lung architecture in which the spongy structure of the lung appeared with thin inter-alveolar septa and normal clear alveoli (Figure 3a). Inducible minimal inflammatory cell infiltration was detected in the lung sections from this group and was only observed occasionally.

Histopathological analyses of normal (blank) (a), NAP-damaged (model) (b), DAST pre-treated (c), and DATS post-treated (d) lung tissues. Normal, no treatment; NAP-damaged, naphthalene (model); DATS pre-treated, pre-treatment with DATS for 7 days at a dose of 80 mg/kg; DATS post-treated, post-treatment with DATS for 7 days at a dose of 80 mg/kg. (#): loss of the normal architecture of the lung with extensive infiltration by inflammatory cells; (★): marked thickening of the inter-alveolar septa; (*): areas of irregular air spaces with destroyed inter-alveolar septa alternating with narrowed alveoli (↙); (•) extravasations of blood in alveolar spaces.
NAP-induced cellular damage was found in lung tissues as indicated by structural degeneration, vasocongestion, and edema, accompanied by inflammatory cell infiltration. In the NAP group, histological changes were variable among the animals. The H&E-stained sections revealed a marked infiltration of inflammatory cells. The destroyed inter-alveolar septa alternated with narrowed alveoli. Furthermore, thickening of the inter-alveolar septa and large irregular alveolar spaces were noted, which are signatures of damage to the lung tissue (Figure 3b).
Although pre-treatment with DATS (80 mg/kg) did reduce the scores, the differences were not significant (Figure 3c). Examination of various sections of the DATS post-treated group (80 mg/kg) revealed that most of the changes that were observed in the NAP-treated group decreased but did not disappear completely. The lung alveoli were lined by a normal alveolar epithelium. The inter-alveolar septa were thinner than those in the NAP group. The histopathological severity of the lung injury was significantly reduced by DATS (Figure 3d).
Histological analysis of livers
The histological feature of liver sections from the normal group indicated a normal cellular architecture with healthy hepatic cells and sinusoidal spaces (Figure 4a). The NAP model group showed multiple and extensive areas of hepatocellular necrosis and the obvious increase in inflammatory cell infiltration (Figure 4b). In addition, massive fatty degeneration, cellular necrosis, moderate increase of inflammatory cell, and loss of cellular boundaries were observed. The features of radiating to hepatic cords disappeared as shown in Figure 4b. In contrast, the liver slices of the liver-injured mice treated with DATS showed a significant histopathology improvement (Figure 4c and 4d). The liver architectures with less fatty degeneration, cell necrosis, and inflammatory infiltration in both pre- and post-treated groups. The histological results are consistent with the liver enzyme analyses as shown in the results sections of Biochemical analysis and Determination of SOD, MPO, MDA, and GSH levels in the liver tissues.
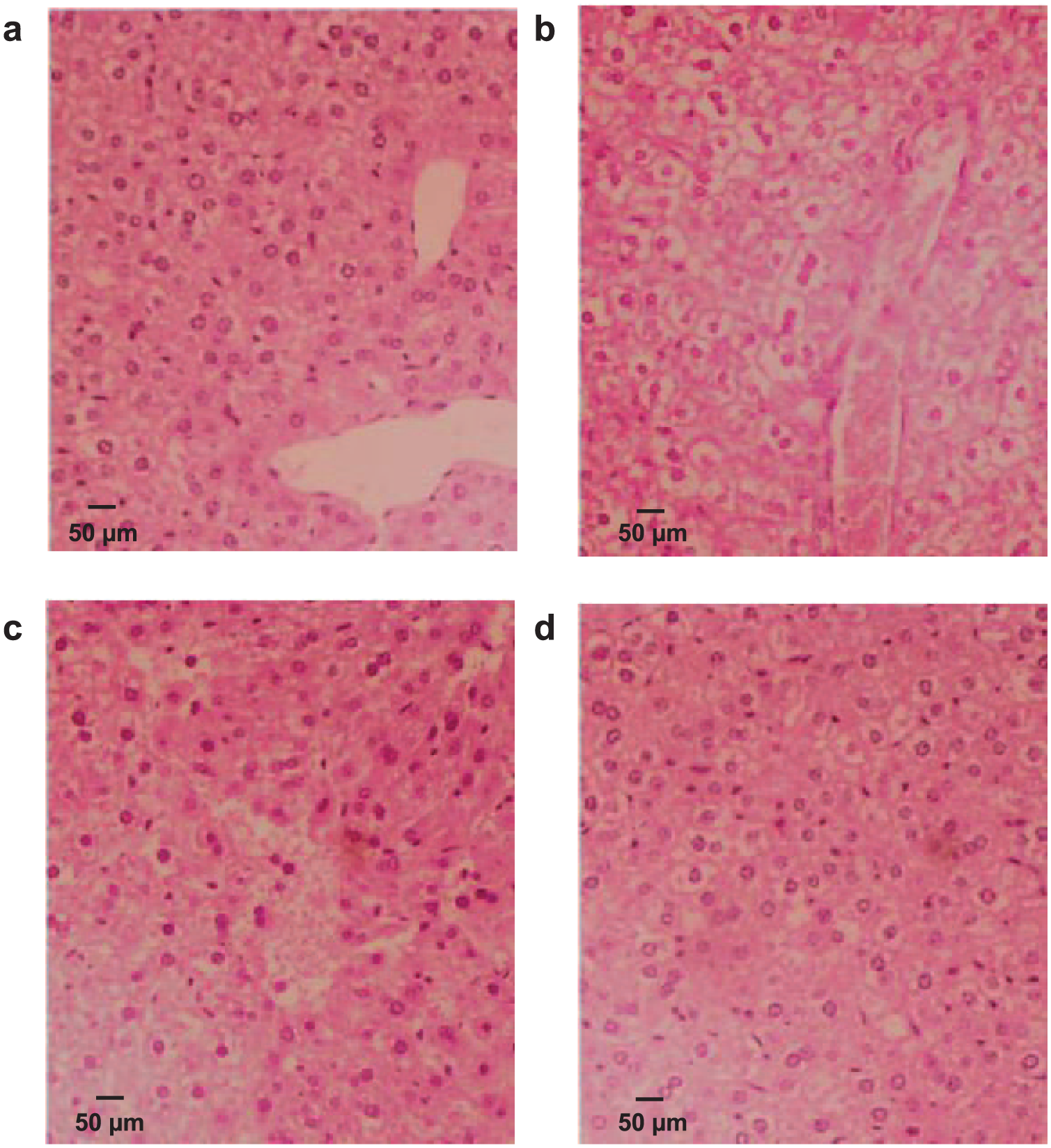
Histopathological analyses of normal (blank) (a), NAP-damaged (model) (b), DAST pre-treated (c), and DATS post-treated (d) liver tissues. Normal, no treatment; NAP-damaged, naphthalene (model); DATS pre-treated, pre-treatment with DATS for 7 days at a dose of 80 mg/kg; DATS post-treated, post-treatment with DATS for 7 days at a dose of 80 mg/kg.
Immunohistochemical analyses
In the normal control group, mice lung and liver tissue cells showed only weak p65 staining in the cytoplasm and no nuclear staining. The p65 nuclear staining was increased in the liver and lung tissue macrophages of the NAP group compared with the normal control group, as shown in Figure 5a and 5c. The p65 expression intensity in the DATS-treated groups was significantly inhibited compared with the NAP (model) group.

Immunohistochemical analyses of liver MPO-positive cells (a), liver tissue NF-κB p65 (b), lung MPO-positive cells (c), and lung tissue NF-κB p65 (d). Control, no treatment; NAP, naphthalene damaged (model); Co-3, post-treatment with DATS for 7 days at a dose of 80 mg/kg.
We observed a significant increase in liver and lung levels of MPO in the NAP-damaged group compared to the control group as shown in Figure 5b and 5d. The treatment study data show that DATS markedly reduced the MPO activity induced by NAP in the lung and liver tissues.
Discussion
Naphthalene, an environmental toxicant, has been reported to induce oxidative changes in the livers, kidneys, lungs, and brains of mice. 15 Naphthalene exposure is associated with several toxic manifestations in tissues, with the lungs being the most sensitive. 23 The formation of hydroxyl radical (·OH) is one of the mechanisms for the NAP-induced oxidative damage. 24
Garlic extracts that include S-allyl cysteine sulfoxide, a bioactive compound of garlic, prevented the reduction of hepatic SOD activities in diabetic rats. 25 Another study showed that aged garlic extract has antioxidant and protective effects against cisplatin-induced oxidative stress and changes in parameters of blood cells and liver structure in rats. 26 Similar results have been found in the studies of aqueous garlic extract. 27 The organosulfur compounds in garlic and onions exert their effects on human health via multiple different functions, including antioxidant, anti-inflammatory, and antibacterial properties. These active compounds may interact with sulfur-containing enzymes, thus inhibiting the oxidation of fatty acids and preventing the formation of pro-inflammatory factors and bacterial growth. 28
In the present study, the protective effects of DATS in mice were analyzed following challenge with NAP. Administration of DATS (20, 40, and 80 mg/kg) decreased the production of IL-8, TNF-α, and NO in serum, which indicated the anti-inflammatory effect of DATS. Treatment with DATS also decreased the levels of MDA and GSH and improved the activity of SOD and MPO in the tissues, suggesting an antioxidant activity of DATS. Additionally, the parameters from the liver and lung tissues showed protection by DATS against the NAP-induced oxidative and inflammatory damage.
The assessment of serum oxidative stress was performed by analyzing the activity of the hepatic enzymes AST, ALT, and LDH, and antioxidative enzyme of SOD. The NAP-induced increase in the levels of the enzymes AST, ALT, and LDH in the serum confirmed that damage had been inflicted on the cellular plasma membranes, which might have compromised their integrity. 29 The antioxidant enzyme SOD contributes to the biological defense by playing a key role in scavenging the toxic intermediate of incomplete oxidation. 30 High levels of SOD activity might protect the human body against oxidative stress, consistent with the potential health benefits. A reduction in the activities of antioxidant enzymes is associated with increases in the levels of reactive oxygen species (ROS), leading to initiation and propagation of lipid peroxidation (LPO). The increased levels of LPO are also an important index of oxidative injury. 31 MDA, an end product of lipid peroxidation, has frequently been used as a standard biomarker of lipid peroxidation in vivo and is a commonly applied index of oxidative damage. 32
NO is an inflammatory mediator, which has a dual function in inflammation. Studies have shown that NO seems to have both pro-inflammatory and anti-inflammatory properties. 33 Nitric oxide effects in airways are influenced by the activity of nitric oxide synthase (iNOS). Inducible iNOS, which produces large amounts of NO, is active during the inflammatory process. 34 Some cytokines including IL-1, TNF-α, and IFN-γ can induce the iNOS activity in various cells, leading to enhanced NO formation. 35 The release of inflammatory cytokines such as TNF-α, IL-1β, IL-6, and IL-8 are potential factors that may indicate a mounting inflammatory response. 36 It was noted that the levels of IL-8 and TNF-α, which are well-established molecular biomarkers of inflammation, were also dysregulated in the serum of NAP-exposed mice in our study. The inflammatory cell infiltration was also shown in the histopathological observation of the lungs of the NAP-treated mice. The histopathological changes were reduced in the DATS-treated groups, especially in the pre-treatment groups. Moreover, the results of the histopathological observation support the results of the measurements of anti-inflammatory parameters from the sera and tissues.
MPO is an enzyme that is correlates significantly with the cellular oxidative damage in inflamed tissues. MPO activity is commonly used to estimate in the initiation and progression of acute and chronic inflammatory diseases.37,38 Our present findings show that NAP exposure resulted in an enhancement of the MPO activity in the livers and lungs and DATS inhibited the expression of MPO. The content determination data are consistent with the results of the immunohistochemistry.
NF-κB is considered to an inducible transcriptional factor involved in the production of pro-inflammatory cytokines.NF-κB is widely expressed by all cells. Over-expression of NF-κB p65 is associated with the inflammatory response.39,40 Therefore, inhibiting NF-κB activation appears to be beneficial for controlling hyper-inflammatory responses. Previous studies have shown that the inflammatory cytokine levels in the sera and the inflammatory gene expression in the lung tissues were inhibited consistent with the reduced expression of NF-κB p65.41,42 The results of this study suggest that the suppressive effects of DATS on NF-κB p65 were involved in the effects related to decreased levels of cytokines, thereby exhibiting protective effect on lung and liver injury.
Conclusion
DATS treatment significantly inhibited MDA production accompanied by changes in the activities of serum ALT, AST, and LDH, indicating a reduction in lipid peroxidation and cellular injury, thus protecting the liver tissues against NAP-induced oxidative damage. These data suggest that DATS demonstrated antioxidative effects, which may be associated with the changes in the activities of antioxidant enzymes (GSH, SOD, and MPO) in vivo. This study also showed that DATS inhibited the production of serum TNF-α and IL-8 by suppressing the infiltration of inflammatory cells in lung tissues. This effect seems to be mediated by inhibition of neutrophil infiltration, reducing the overproduction of MPO and NO and elevation of GSH in lung tissues. In addition, the results from the histopathological and immunohistochemical analyses of the liver and lung tissues are in consistent with the enzymatic and cytokine data described above. These results suggest that DATS may be a promising agent against naphthalene toxicity by protecting tissues against oxidative and inflammatory damage. However, further studies are needed to explore the protection mechanism of DATS on the molecular levels.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the fund from National “Major Science and Technology Project–Prevention and Treatment of AIDS, Viral Hepatitis, and Other Major Infectious Diseases” (Grant no. 2013ZX10005004).
