Abstract
Background:
Primary immunodeficiencies (PIDs) are generally characterized by recurrent infections; however they may be complicated by other clinical disorders such as allergy, autoimmunity, and lymphoproliferation. In particular, autoimmunity may be the first manifestation of the disease in patients with low serum immunoglobulins (Ig) levels. Here we describe a group of patients that share features of immunodeficiency and autoimmunity.
Materials and Methods:
All patients went through a complete T and B cell subset characterization and a B cell function analysis in the peripheral blood by flow-cytometry. B cell proliferation and plasma cell differentiation was measured,
Results:
Immunological and molecular analysis did not lead to the identification of known molecular defect typical of Hyper IgM syndrome. A comparative study of the peripheral blood B cell subsets between patients and healthy donors showed that in patients with autoimmune manifestations all circulating B cells expressed high amounts of surface IgM.
Conclusions:
These results suggest that the increased IgM expression on circulating B cells, reflecting B cell activation, might identify a clinical condition characterized by hyper IgM serum levels of unknown molecular defects, associated with susceptibility to infections and autoimmunity.
Keywords
Introduction
Primary immunodeficiencies (PIDs) are generally characterized by recurrent infections; however they may be complicated by other clinical disorders such as allergy, autoimmunity, and lymphoproliferation. 1 In particular, autoimmunity may be the first manifestation of the disease in patients with disturbances of serum immunoglobulin (Ig) levels, such as common variable immunodeficiency (CVID), selective IgA defect, or hyper-IgM (HIGMs) syndrome.
While in CVID patients mutation analysis of the B-cell activating factor (BAFF) receptor, transmembrane activator (TACI), has revealed a direct correlation between the presence of TACI mutations2,3 and increased susceptibility to develop splenomegaly, immune thrombocytopenic purpura (ITP), and autoimmune hemolytic anemia, 4 for HIGM no genetic predisposition has been described.
HIGMs, also known as class-switch recombination (CSR) defects, are rare PIDs characterized by normal/increased IgM and low/normal IgG serum levels. 5 At least seven different forms of HIGMs have been identified with either X-linked or autosomal (either recessive or dominant) inheritance and a wide range of clinical manifestations mainly represented by increased susceptibility to opportunistic infections and a broad spectrum of autoimmune manifestations.6,7
The most common HIGM form, HIGM type 1, is caused by mutations of the CD40L and is responsible for about 70% of all HIGM.5,8 HIGM1 patients suffer from recurrent infections caused by bacteria, viruses, and fungi often associated to neutropenia (44–60%), but not always linked to the presence of circulating anti-neutrophil antibodies. 9
Patients with CD40 defect (HIGM type 3, HIGM3) have a disease phenotype similar to HIGM1 and may develop autoimmune disorders during the course of disease.10,11
HIGM type 6 and 7 syndromes involve mutations in the NF-kB modulator (NEMO) and IkBα genes, respectively, and are characterized by the presence of hypohidrotic ectodermal dysplasia. 12
Finally HIGM2-4 and 5 are PIDs associated with intrinsic B-cell defects involving mutations in CSR and somatic hyper-mutation (SHM) enzymes. HIGM2 is associated with mutations in activation-induced cytidine deaminase (AICDA) gene;
13
patients often present lymph node enlargement and suffer of recurrent bacterial infections of the upper respiratory tract; autoimmunity is frequently present.
14
HIGM5 is clinically similar to HIGM2 but is caused by mutations in another member of the CSR machinery, the uracil-DNA-glycosylase (UNG) gene.
15
HIGM4 is the last reported syndrome, clinically resembling to a milder form of HIGM2, but whose gene defect is still unknown.
16
Finally, another rare CSR defect has been described in Europe, involving the
We performed immunological and molecular analyses in nine patients, all characterized by hyper IgM, mild infections, and/or autoimmune manifestations, aimed at evaluating whether these cases may be classified among one or more of the above reported defects.
Material and methods
Patients and Healthy controls: Nine individuals with high serum IgM were followed and treated at the S. Andrea University Hospital outpatient Allergy, Clinical Immunology and Rheumatology Division. Patients reported variable clinical manifestations, ranging from arthritis to mild infections. A detailed description of the patient’s main demographic and serological features is presented in Table 1. Fifty healthy blood donors, herein named healthy controls (HCs) (Blood Transfusion Unit, Ospedale Pediatrico Bambino Gesù, Rome) were used as controls for the flow-cytometry analysis.
Demographic, clinical, and serologic characteristics of the studied patients.

Normal values: IgM 30–130 mg/dl; IgG 700–1600 mg/dl; and IgA 30–270 mg/dl.
CCS, corticosteroid; SSA, sulphasalazine.
Flow cytometry analysis: Peripheral blood lymphocytes (PBLs) were isolated and stained with the appropriate combination of fluorescent labeled antibodies as previously described.18,19 All analyses were performed on a FACSCanto (Becton and Dickinson, Sunnyvale, CA, USA) interfaced to a PC FACSDiva software – 100,000 events per sample were analyzed.
B cell proliferation and plasma cell differentiation: Patients and controls PBLs (n = 10) were labelled with 5-chloromethylfluorescein diacetate at the final concentration of 0.1 µg/mL (CellTracker CMFDA, Molecular Probes, Eugene, OR, USA) and cultured at 2–3×105 cells per well in 96-well plates (Becton Dickinson) in complete RPMI 1640 (Gibco BRL) supplemented with 10% heat inactivated fetal bovine serum (FBS, Hyclone Laboratories, Logan, UT, USA) for 7 days with 2.5 µg/mL CpG-ODN (Hycult Biotechnology, Uden, The Netherlands). B cell proliferation, differentiation, and antibody secreting cell generation were measured on day 7 by flow cytometry 20 and antibody secretion by ELISA.
ELISA: Ig concentration, in the culture supernatant, was quantified by ELISA as previously described. 19 Serum BAFF concentration was also quantified by ELISA as following: plates coated with 2.5 µg/mL purified goat anti-human BAFF (PeproTech, London, UK) were incubated overnight at 4°C. Serum samples were diluted 1:3 starting with undiluted sample and BAFF detected with biotinylated goat anti-BAFF (PeproTech). Optical density was measured on a microtiter plate reader at 450 nm and BAFF concentration was calculated by interpolation with the standard curve done using a recombinant BAFF (PeproTech). Due to the sensitivity of our ELISA, values under 0.3 ng/mL were considered background.
Semi-quantitative PCR for AID and UNG: Total RNA was extracted from patients and HCs (n = 5) PBLs stimulated for 5 days with 2 µg/mL anti-CD40 (Calbiochem, Merck KGaA, Darmstadt, Germany) and 100 units/mL of IL-4 (PeproTech) as previously described, 21 using the RNeasy Mini Kit (Qiagen). Reverse transcription was performed with SuperScript™ II Reverse Transcriptase (Invitrogen) using the oligo dT primers for the first strand cDNA synthesis. Expression levels of the targeted mRNA were quantified by semi-quantitative PCR. PCR products separated on agarose gels stained with GelRedTM (Biotium, CA, USA) and images acquired using Fluor-S MultiImager MAX and quantified by densitometry using Quantity One 4.1.1 (Bio-Rad Laboratories, CA, USA). The following PCR protocols were used: housekeeping gene HPRT1 using the forward 5’-CAGCCCTGGCGTCGTGATTAGT-3’ and the reverse primer 5’-CCCTGTTGACTGGTCATTACAAT-3’ (94°C for 30 s, 60°C for 30 s, 72°C for 45 s for 35 cycles); AICDA using the forward 5’- TAGACCCTGGCCGCTGCTACC- 3’ and reverse primer 5’- CAAAAGGATGCGCCGAAGCTGTCTGGAG-3’ (94°C for 30 s, 65°C for 30 s, 72°C for 45 s for 35 cycles); UNG2 using the forward 5’-ATCGGCCAGAAGACGCTCTA-3’ and the reverse primer 5’-ACAGCAGCTTCTCAAAGGCCA-3’ (94°C for 30 s, 65°C for 30 s, 72°C for 2 min for 40 cycles).
Statistical analysis
Data were analyzed using StatView statistical program for MacIntosh (StatView Software, San Diego, CA, USA) and
Results
Patient description
Patients 1 and 2 presented a diagnosis of primary anti-phospholipid syndrome (PAPS) with positivity for anti-cardiolipin (aCL) and anti-β2-glycoprotein I (anti-β2GPI I) antibodies. Note that in both patients autoantibodies were only of IgM isotype whereas IgG were within the normal range. The first was a 36-year-old woman that suffered two consecutive spontaneous miscarriages, while the second was a 56-year-old man who had retinal thrombosis. At the time of the analysis both were treated with acetylsalicylic acid 100 mg/day. Blood tests showed a mild increase in erythrocyte sedimentation rate (ESR) (32 and 28 mm/h, respectively), the presence of antinuclear antibodies (ANA) detected by standard indirect immunofluorescence test (both patients, 1:160), and a net increase in serum IgM, without a significant decrease in IgG and IgA values (Table 1).
Patient 3, a 27-year-old woman, presented asthenia, polyarthritis, and fingernail bacterial infection. She referred having suffered from recurrent respiratory infections requiring periodical antibiotic treatment (approximately 4 episodes/year) since childhood. Routine blood analyses revealed a marked neutropenia (300 cells/µL) with lack of anti-neutrophil serum antibodies, mild increase in ESR (32 mm/h) and in C reactive protein (CRP) (10 mg/L). To try to reduce neutropenia six intravenous IgG (IVIG) cycles (1.5 g/kg body weight; Flebogamma, Grifols), 2 months apart, were administered, with initial good results. Unfortunately from the fifth cycle onwards therapy lost efficacy and was abandoned. Serum levels of rheumatoid factor and ANA were below detection levels, IgG and IgA were normal, but serum IgM was increased six-fold as compared to the normal upper limit.
Patients 4 and 5 referred to a 54-year-old woman and her 20-year-old daughter; both suffered from recurrent upper respiratory and urinary tract infections, requiring periodical antibiotic treatment (approximately 4 episodes/year). Inflammatory parameters were normal and the screening for autoantibodies (ANA, RF, ENA) was negative. The only serum alteration was the increased value of IgM, with IgG at the lowest limit of the normal range.
Patient 6, a 58-year-old man, although not belonging to the previous family, shared the same clinical and serological phenotype.
Patients 7, 8, and 9 presented mild recurrent skin (furunculosis) and/or upper respiratory tract infections resolved with antibiotic treatment, associated with different autoimmune manifestations. In particular, patient 7, a 51-year-old woman, was diagnosed with psoriatic arthritis (PsA) and initially treated with methotrexate then with leflunomide and currently with corticosteroids; she also had a mild persistent pancytopenia (also present before PsA diagnosis) not belonging to hematologic malignancies. Patient 8 was a 42-year-old woman with persistent arthritis in both knees; the screening for autoantibodies (ANA, RF, CCP, ENA) was negative, but she had a mild increase in inflammatory parameters (ESR, 40; RCP, 9 mg/L). Inflammation was confirmed by ultrasonography that showed moderate synovitis of the knees. She is under salazopyrin (SSA) treatment (1 g/day) with a good control of pain and function; as for patient 7, IVIG cycles are scheduled. Patient 9 was a 52-year-old woman suffering from mild piastrinopenia and ANA positivity; she is currently receiving IVIG (10–20 g/cycle; Flebogamma, Grifols) monthly with a good outcome.
Peripheral T cell compartments
The increased serum IgM associated with recurrent infections suggested the diagnosis of HIGMs in our patients, most of all suffering of systemic autoimmune manifestations. We first analyzed the peripheral T cell compartment and found that the frequency of T-cells (CD3pos cells) was similar to age-matched HCs, without any significant alteration on the frequency of CD4 and CD8 T-cells or on the CD4/CD8 ratio (normal ratio, 1.5). Memory T cells, CD3posCD45ROpos, were roughly half of the total T cells as usually found in the peripheral blood of HCs. T regulatory cells (Treg, CD4posCD25posCD127low) were lower than in HCs, but the reduction was not statistically significant (Table 2).
Frequency of peripheral blood T cell subsets in patients with or without autoimmunity and in healthy controls. Values were calculated after staining using fluorescent surface makers and FACS analysis. Values for healthy controls (HC) represent the mean ± standard deviation of 50 specimens (age range, 25-60 years).

CD4posCD25posCD127low cells.
CD3posCD45ROpos cells.
Tmem HC vs. patients with autoimmunity
All patients upregulated CD40L upon CD3/CD28 stimulation (data not shown) confirming that T cells are able to provide T cell help to B cells, while serum levels of class-switched Ig were at the lower normal limit (Table 1).
Peripheral B cell compartments
Transitional, mature, IgM, and switched memory B cells were identified in the peripheral blood by using the combination of CD24, CD38, CD27, and IgM surface markers. 22
A non-significant reduction in the frequency of CD19pos cells in patients with autoimmunity (9.0 ± 3% of total lymphocytes) as compared to patients without autoimmunity (15.4 ± 4.2%) and HCs (17.1% ± 7.9%) was also observed (Table 3). The relative frequencies of transitional (tr-CD24bright CD38brightCD27negIgMhigh), mature (mat-CD24inter CD38interCD27negIgMpos), and memory B cells (mem-CD24brightCD38negCD27posIgpos) varied among patients (data not shown and Figure 1a).
Frequencies of peripheral blood B cells that proliferated and differentiated in secreting cells, after stimulation for 7 days with CpG. Values for healthy controls (HC) represent the mean ± standard deviation of 10 specimens (age range, 25–60 years).
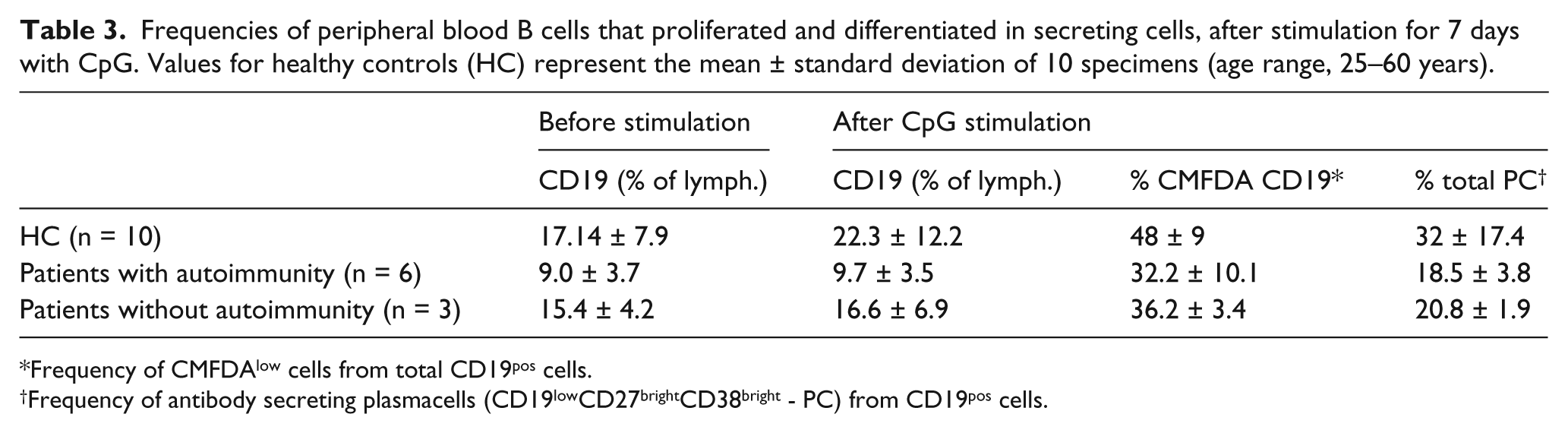
Frequency of CMFDAlow cells from total CD19pos cells.
Frequency of antibody secreting plasmacells (CD19lowCD27brightCD38bright - PC) from CD19pos cells.

Characterization of the peripheral blood B cell. Cells were stained for CD24, CD38, and IgM, and analyzed by flow-cytometry.
The analysis of CD19pos cells for IgM expression showed that mean fluorescence intensity (MFI) was significantly higher in patients with autoimmunity than in patients without autoimmunity and in HCs (MFI = 83,900, 38,500, and 21,700, respectively;
We have previously shown that B cells express TLR9 and that CpG induces TLR9 dependent B cell proliferation and antibody secretion; thus CpG stimulation is a good tool to measure,

Plasma cell differentiation and Ab secretion in CpG stimulated PBLs.
A normal CD40 expression on B cells was found (data not shown), confirming that CD40-CD40L crosstalk between T-B and DC-B cells is not compromised.
All patients showed normal or increased AID mRNA and UNG mRNA expression (Figure 3). Because AID and UNG are enzymes that mediate class-switching and SH and patients were able to produce IgG and IgA both

Expression of AID and UNG mRNA in peripheral blood mononuclear cells activated with anti-CD40 and IL-4 for 5 days. Graph represents the fold increased mRNA expression of AID and UNG in the overall patients (mean + SD). Note that intensity of specific PCR products was measured by densitometry and reported to the unit (arbitrary densitometry value obtained from triplicates using a pool of stimulated PBMCs from five healthy controls).
B lymphocyte stimulator (BLyS also known as BAFF, CD257) is a B cell survival factor that has been often found increased in the serum of patients suffering from CVID associated to autoimmunity.23–25 Because our patients showed atypical features of HIGM associated with autoimmunity or not we measured serum BAFF and found that in all patients serum BAFF was not significantly higher than in HCs (normal levels <0.02 µg/mL – range, 0.0003–0.02 µg/mL vs. 0.05 µg/mL – range, 0.001–0.05 µg/mL, respectively).
Discussion
PIDs are characterized by frequent, recurrent, or persistent infections; however, they can also be associated with autoimmunity. 26 In particular, in patients with partial antibody deficiency such as CVID, IgA defect, or HIGM, autoimmunity may represent the first signs of disease.
In the current study, nine patients with high serum IgM levels and almost normal IgG-IgA, suffering from autoimmune manifestations and/or mild respiratory infections are reported.
HIGM1 and 2 were immediately excluded since our patients were able to express CD40L on activated T cells and CD40 on B cells. In addition patients were not suffering of ectodermal dysplasia, thus most likely they are neither carrying mutations in the X-linked NEMO nor in the IκBα (NFKBIA) gene.27–29 These observations strongly suggested that crosstalk between T and B cells can occur and that T cell help to B cells can be provided during the germinal center formation. The impossibility of attributing already known molecular defects to our patients, together with the clinical aspects fitting those described by Imai et al., pushed us to consider HIGM4 as the possible cause of disease. 16 As in HIGM4, in fact these patients suffered from recurrent non-life-threatening bacterial infections from childhood, often associated with autoimmunity and they did not present opportunistic infections. However, we could not make a definitive diagnosis of HIGM4 because patients showed only a mild reduction of IgG with normal levels of serum IgA and do not fit the widely accepted HIGM classification criteria. 30
Autoimmunity coexists with immunodeficiency in 20–30% of HIGM and CVID patients but the pathogenesis remains vague and still not well identified. Several groups have analyzed this apparent paradoxical phenomenon 31 and proposed a role for different molecules/cell subsets32,33 in promoting autoimmunity, among which reduced frequency of regulatory T cells 34 increased levels of circulating BAFF 23 and AID dysfunction.
Six patients presented different autoimmune disorders associated with increased serum IgM. Patents 1 and 2 suffered from PAPS with IgM antibodies against both a-CL and anti-B2GPI-I; Granel et al. described a positive correlation between increased serum IgM levels and a-CL IgM antibodies in a large cohort of patients. 35 Overall these observations suggest a potential relationship between B cell activation and a pathogenic role for IgM autoantibodies.
Patients 3 and 7 presented the more critical autoimmune profile with severe arthritis and marked neutropenia / moderate pancytopenia, associated with mild recurrent respiratory infections. Neutropenia is the most frequent autoimmune pathology associated to HIGM, often without detectable levels of autoantibodies, being IVIG treatment able to restore neutrophil count. 36 Patient 8 suffered from oligoarthritis and patient 9 showed mild thrombocytopenia associated to ANA positivity.
The six patients, previously discussed, showed an increased expression of surface IgM, typical of activated B cells. During B cell development at the periphery, IgM is down-modulated. This occurs when recently generated bone marrow B cells enter the spleen where B cell maturation progress from transitional to mature stage. Later on increased IgM expression is characteristic of IgM memory B cells that represent an “atypical” memory B cell pool containing pre-activated cells able to secrete polyreactive IgM, the so called natural antibodies.
19
High expression of IgM on patient B cells reveals a dysfunction on the mechanisms controlling cell activation. In fact, these patients produced IgM secretory cells more efficiently than class-switched plasma cells in an
We observed that B cell activation, in patients, generated a potent upregulated response of AID and UNG molecules. These molecules have the dual role of CSR in the BCR locus and induce BCR hyper-mutation aiming at the production of high affinity antibodies.
38
In all patients, CSR was not very efficient because circulating switched memory B cells were reduced and when stimulated with CpG
Although, in this study we accomplished an extensive analysis of our patient’s immune system, initially aiming at a diagnosis of HyperIgM, we were unable to attribute to any of them any previously described defects that characterize this group of syndromes.
In conclusion, we reported a case series of nine patients characterized by high levels of serum IgM with almost normal IgG-IgA and an immunodeficient / autoimmune clinical profile that to our knowledge does not fit currently used diagnosis criteria of PID; moreover our results suggest that higher expression of surface IgM on B cells is associated with the development of autoimmunity in these patients.
Footnotes
Acknowledgements
We thank Dr C Pioli for critical review of the manuscript. MM Rosado has been partly supported by a Mérieux Research Starting Grant, Institut Merieux, Lyon, France.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
