Abstract
The aim of the present study was to assess the analgesic activity of the macrolide antibiotic tilmicosin at dose levels of 20 and 40 mg/kg of body weight, subcutaneously, against chemical- and thermal-induced acute pains, using acetic acid-induced writhing, formalin-induced pain, hot-plate, and tail-flick models in mice. Tilmicosin showed a dose-dependent significant decrease in the number of writhes in the acetic acid-induced writhing test and significant decrease in hind paw-licking time in the late phase of the formalin test. However, it did not cause any significant changes in the reaction times to heat stimuli in the hot-plate and tail-flick models. In chemically-induced pains, both dose levels of tilmicosin showed significant effects compared to those of the corresponding standard peripheral analgesic, acetylsalicylic acid (200 mg/kg of body weight, subcutaneously) being 26.37 ± 2.88 and 43.64 ± 3.85% vs. 73.35 ± 1.44% in acetic acid test; and 19.23 ± 3.85 and 44.90 ± 1.80% vs. 73.63 ± 2.39% in the late phase of formalin test, respectively. These results may indicate that tilmicosin possesses a significant peripheral but not central analgesic potential that may be beneficial in symptomatic relief of pain when it is used in therapy, in addition to its well-established antibacterial effect.
Introduction
Pain is an established result in almost all disease conditions. Although unpleasant, yet it serves as a warning of disease or a threat to the body. The control of pain, in addition to combating the specific cause of the disease, is an important issue in therapy for safety and comfort of animal and human patients.
Pain is generated in the spinal cord and brain by nociceptive input. Injuries to the peripheral nervous system, spinal cord, or brain can lead to the report of pain, even in the absence of noxious stimulus. 1 Chemical and thermal stimuli are the two main pathways inducing acute pain via different neurobiological mechanisms. Pain stimuli are sensed by nociceptors at free nerve endings. The body of the bipolar afferent first-order neuron lies in a dorsal root ganglion. Nociceptive impulses are conducted through either unmyelinated (C-fibers), whose conduction velocity is relatively slow, in the range of 0.2–2.0 ms; or myelinated axons (Aδ-fibers), whose conduction velocity is relatively fast, in the range of 5–30 ms. The free endings of Aδ fibers respond to intense pressure or heat stimuli, while those of C-fibers respond to chemical stimuli (prostaglandins, histamine, bradykinin, etc.) arising from tissue injury, trauma, or inflammation. 2 The role of nociceptors and ion channels in thermal-induced acute pain is not exactly the same as in chemical-induced acute pain. 3 Therefore, both pathways should be targeted in analgesic studies.
An analgesic is any drug or agent that can achieve analgesia or relief from pain as a symptom, without affecting its cause.4,5 The word is derived from Ancient Greek (an-, “without”) + (álgēsis, “sense of pain”) from (álgos, “pain”), meaning “without feel of pain”. Analgesic drugs act in various ways on the peripheral and central nervous systems. They are numerous including, non-steroidal anti-inflammatory drugs (NSAIDs) such as the salicylates, and opioid drugs such as morphine. The severity and nature of pain as well as the response to other medications determine the choice of the most suitable analgesic agent. 6
In addition to the well-documented, standard analgesic drugs, some other drugs may have analgesic potentials in addition to their main pharmacological actions. This may carry the benefit of synergism when these drugs are combined with the standard ones rendering, sometimes, therapy more effective and agreeable.
Tilmicosin is a macrolide antibiotic with the chemical name of 20-deoxo-20-(3,5-dimethyl piperidin-1-yl) desmycosin. Macrolide class of antibiotics contains a macrocyclic lactone ring in their molecular structure; tilmicosin contains a 16-member one. Their pharmacokinetic properties of having a high volume of distribution allow for a smaller and a single dose to be administered to reach a high concentration in the target tissue. Macrolides, including tilmicosin, are bacteriostatic and work by invading the cell membrane of sensitive bacteria and binding to the 50s ribosome subunit, preventing protein synthesis; translocation between the 50s and 30s ribosomes is interrupted, causing early detachment and thus creating of incomplete peptide chains.
7
Tilmicosin has been developed in an injectable form for use in cattle and sheep to treat respiratory infections (10 mg/kg of body weight); and as a feed premix for swine (200–400 mg/kg feed) for 10–21 days, equivalent to 8–20 mg/kg of body weight per day.
8
It is an effective antimicrobial for Gram-positive and some Gram-negative bacteria, as well as atypical bacteria as
Usually, prescriptions for an inflammatory infectious disease include potent anti-inflammatory analgesic-antipyretic drugs in addition to the antibacterial base of the prescription. It will be of good value if that antibacterial base has, in addition, a pain-relieving effect.
Therefore, the objective targeted in the current study was to assess the analgesic potential of tilmicosin on the two types of acute pain induced by thermal and chemical stimuli using different pain models in mice.
Material and methods
Tilmicosin
Tilmicosin is structurally related to tylosin, having the chemical formula (C46H80N2O13) with a molecular weight of 869.15. Physically, it is freely soluble (1500 mg/L or greater) in organic solvents (hexane, acetone, acetonitrile, chloroform, dichloromethane, ethyl acetate, methanol, tetrahydrofuran); water solubility is temperature- and pH-dependent, but is 566 mg/mL at pH 7 and 25°C. Tilmicosin was obtained as the patent preparation Pneumotac® (ADWIA, 10th of Ramadan City, Egypt) that is a subcutaneous therapy for pneumonia and other respiratory diseases in cattle and sheep, formulated as 100 mL amber glass vials containing 333.828 mg tilmicosin phosphate/mL, equivalent to 300 mg tilmicosin/mL. The drug solution was further diluted in sterile water to adjust dose volumes as 0.3 mL diluted solution equivalent to 20 (small dose) and 40 (large dose) mg/kg of body weight of mice.
Chemicals and equipment
Acetic acid and formalin were from PARK scientific Ltd (UK). Acetylsalicylic acid (ASA) was obtained as Aspegic® 500 mg powder for injection (Amriya Pharmaceutical Industries, Egypt). Morphine sulphate was obtained as MSI injectable solution, 10 mg/mL (Mundipharma GmbH, Germany). Other routinely used chemicals were locally purchased and they are of an analytical grade. The used hot-plate was Ceran® 500 model (Germany).
Experimental animals
A total of 80 male albino mice weighing 25–30 g were used for the present study. Animals were housed in polypropylene cages with a suitable bedding material under controlled environmental condition of temperature (25°C), humidity (60%), and 12-h light/dark cycle. All animals were maintained on standard pellet diet and water
Experimental design
All animals were screened for normal responsiveness to pain by probing them with thermal stimuli; and only normally responsive ones were introduced into the study. A parallel design was followed in these experiments. Animals were randomly divided into four main groups (20 in each), each main group was further divided into four subgroups (n = 5 in each) and labelled appropriately. First and second main groups were assigned to analgesic assessment against chemical stimuli; while the third and fourth main groups were assigned to assessing analgesic activity against thermal stimuli. The first main group was used for acetic acid-induced writhing test; within this main group, the first and second subgroups received a single small and large doses of tilmicosin (equivalent to 20 and 40 mg/kg, SC, respectively); the third subgroup received ASA (200 mg/kg, SC) as a standard for peripheral analgesics; while the fourth subgroup received sterile water as control. The second main group was assigned to formalin test where animals received various treatments as given in the first main group and then, 1 h later, challenged with formalin diluted solution (2.5%), 20 μL, SC, in the dorsal surface of the right hind paw. The third main group was assigned to the hot-plate test 1 h after treating animals with either tilmicosin (small or large dose) or morphine sulphate (5 mg/kg, SC) as a standard for central analgesics, or vehicle. The fourth main group was subjected to tail-flick test 1 h after treating animals as in the third main group. The control treatment involved administration to mice of the same volume of the vehicle (0.3 mL of sterile water, SC) and their data were recorded as control results to which both test and standard drugs were compared to. The analgesic investigative tests are described below.
Acetic acid-induced writhing test
The test was adopted according to the model described by Koster and Anderson 9 with minor modifications. An hour post the different treatments described above, 0.55% acetic acid (10 mL/kg) was injected intraperitoneally. Animals were placed in individual Perspex cylinders for observation where the number of writhing movements (stretching of abdominal muscle and hind limbs as a pain behavior) was counted for 25 min, beginning 5 min after the injection of acetic acid into each mouse. The mean value for each subgroup was calculated and both test and standard records were compared with that of the control. Percent of inhibition or analgesia was calculated from the following equation:

Formalin-induced pain test
The method described by Hunsakar and Fasmer 10 was followed. Mice received different treatments as described earlier and used in the test 1 h later. Twenty microliters of 2.5% formalin were injected into the dorsal surface of the right hind paw using a microsyringe with a 26-gauge needle. Nociception was evaluated immediately after the injection of formalin and quantified based on the total paw-licking time in the early phase (phase 1, 0–5 min) and the late phase (phase 2, 20–30 min). The total time of each phase was measured for each subgroup using a stop watch and recorded. Records of both test and standard drugs were compared to that of the control group. Percent of inhibition or analgesia was calculated in each phase from the following equation:

Hot-plate test
The hot plate test was carried out according to the model described by Woolfe and MacDonald 11 at a fixed temperature of 55°C on mice of the third main group. After treating animals as described, they were placed into individual Perspex cylinders on the heated plate; and response to the thermal stimulus was defined as licking of a paw or jumping. The time in seconds between the contact with the stimulus and reaction was recorded as the “response latency”. The latencies were determined four times: 1, 2, 3, and 4 h after administration. A “cutoff” time of 30 s was applied to prevent excessive paining or tissue damage to mice. The percentage of maximal possible effect (%MPE) was calculated using the following formula:

Tail-flick test
The tail-flick test was carried out using a thermostatic water bath with a temperature fixed at 55°C according to the principle described by Janssen et al. 12 on mice of the fourth main group. After the described treatments, an animal was restrained in a fit-size mouse holder with tail extending out. The terminal part (3 cm) of each mouse’s tail was immersed in the hot water bath and the time in seconds taken to flick the tail (brief vigorous movement away from the stimulus) was recorded. The reaction times of all mice were recorded 1, 2, 3, and 4 h post administrating the vehicle/test drug/standard drug. A “cutoff” time of 15 s for the tail to flick was applied to avoid further pain and tissue injury and MPE% was calculated as in the hot-plate test.
Statistical analysis
Results are expressed as mean ± standard error of the mean of five observations (n). Differences between control and treated groups were tested for significance using a one-way analysis of variance (ANOVA).
Results
Animals receiving tilmicosin 20 or 40 mg/kg of body weight showed no signs of toxicity; however, abnormal localized tender swellings were developed after subcutaneous injection of both tested doses. Results of the adopted analgesic tests were recorded as described below.
In the hot-plate test, tilmicosin at the two tested dose levels failed to increase the latency of responses from 1 h to 4 h after treatment (
Effects of tilmicosin (20 and 40 mg/kg bw, SC) and morphine sulphate (5 mg/kg bw, SC) on latency of nociceptive response induced in the hot-plate test (mean ± SEM; n = 5).

Significantly different from control (
SW, sterile water; TLD, tilmicosin large dose; TSD, tilmicosin small dose.

The percentage of maximal possible effect (%MPE) of morphine (5 mg/kg, SC) and tilmicosin (20 and 40 mg/kg, SC) in the hot plate test (mean ± SEM; n = 5).
Effects of tilmicosin (20 and 40 mg/kg bw, SC) and morphine sulphate (5 mg/kg bw, SC) on latency of nociceptive response induced in the tail-flick test (Mean ± SEM; n = 5).

Significantly different from control (
SC, subcutaneously; SW, sterile water; TLD, tilmicosin large dose; TSD, tilmicosin small dose.

The percentage of maximal possible effect (%MPE) of morphine (5 mg/kg, SC) and tilmicosin (20 and 40 mg/kg, SC) in the tail flick test (mean ± SEM; n = 5).
In the acetic acid-induced writhing test, compared with vehicle treatment, tilmicosin at the tested two dose levels decreased the number of writhing movements in a dose-dependent manner (
Effects of tilmicosin (20 and 40 mg/kg, SC) and acetylsalicylic acid (ASA; 200 mg/kg, SC) on the writhing responses induced by acetic acid (10 mL/kg of 0.55% solution, IP) in 25 time periods.

Significantly different from control (
SC, subcutaneously; SW, sterile water; TLD, tilmicosin large dose; TSD, tilmicosin small dose.

Inhibition % produced by tilmicosin (20 and 40 mg/kg, SC) and acetylsalicylic acid (ASA; 200 mg/kg, SC) against the writhing responses induced by acetic acid (10 mL/kg of 0.55% solution, IP), (mean ± SEM; n = 5, *significantly different from control,
Effects of tilmicosin (20 and 40 mg/kg, SC) and acetylsalicylic acid (ASA; 200 mg/kg, SC) on the nociceptive responses induced by formalin (20 μL of 2.5% solution, SC in the dorsum of the hind right paw).
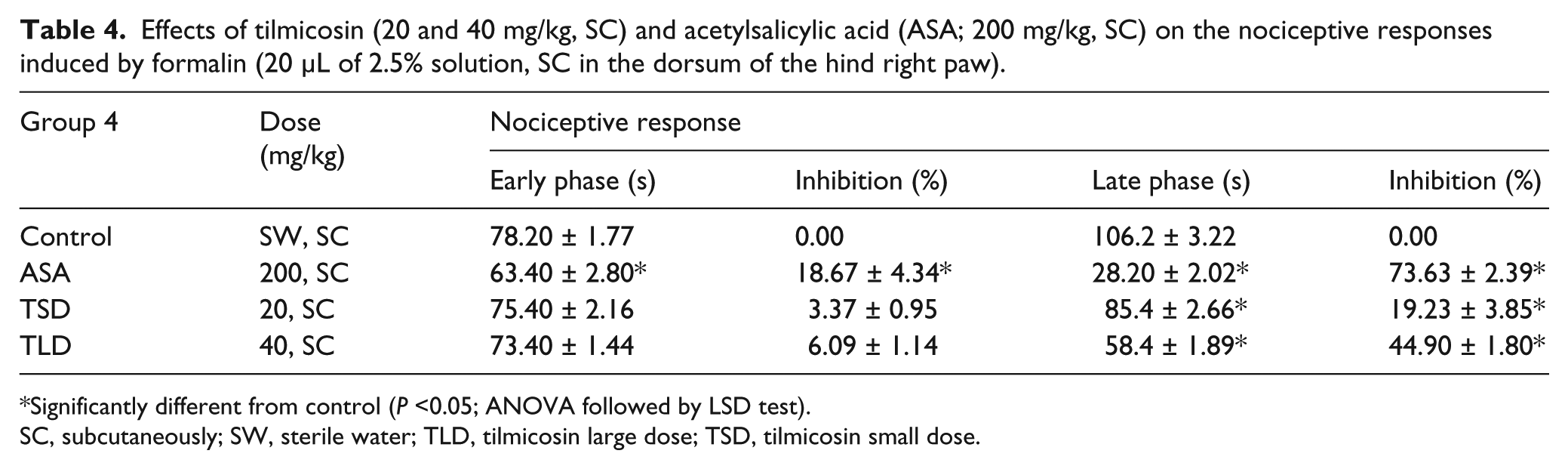
Significantly different from control (
SC, subcutaneously; SW, sterile water; TLD, tilmicosin large dose; TSD, tilmicosin small dose.
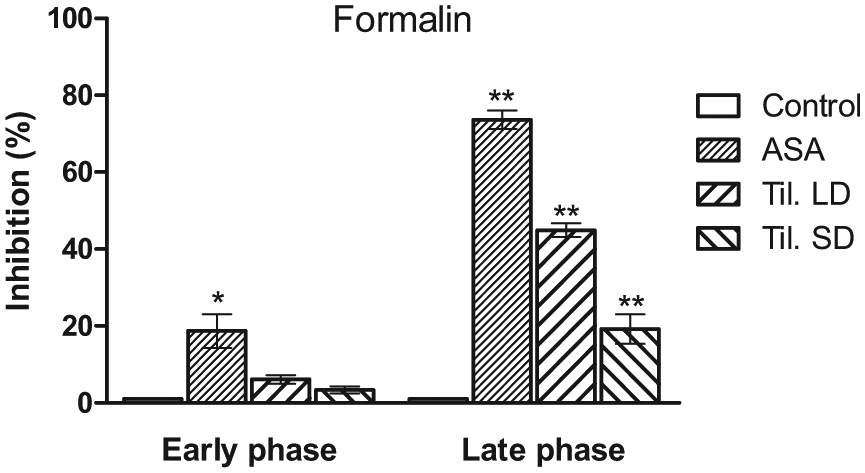
Inhibition % produced by tilmicosin (20 and 40 mg/kg, SC) and acetylsalicylic acid (ASA; 200 mg/kg, SC) against the nociceptive responses induced by formalin (20 μL of 2.5% solution, SC in the dorsum of the hind right paw), (mean ± SEM; n = 5, * and **Significantly different from control at the early and late phases, respectively;
Discussion
Macrolides are a long-used class of antibiotics which still play an important role in the chemotherapy of infectious diseases. Their effectiveness in infections caused by intracellular pathogens was the basis for the development of newer derivatives with improved tolerance, antimicrobial activity, and pharmacokinetics. 13 Nevertheless, the ability of the intracellular accumulation of this family of drugs 14 may also alter host cell functions with a new interest in their therapeutic potential other than infections. 15
The macrolide antibiotic tilmicosin is a tylosin derivative being used in treatment of respiratory diseases in different animal species including cattle, 8 horse, 16 swine, 17 sheep, 18 goat, 19 rabbit, 20 and turkey. 21
Although the inflammatory modulating effects of parent macrolides, particularly erythromycin, have been documented and reviewed, 22 yet there is no, for our information, any data about the analgesic potential of tilmicosin.
Pain, although a discomforting sensation, constitutes an alarm that ultimately may help to protect the organism by triggering reactions and inducing learned avoidance behaviours, which, as a result, may limit damaging affected tissues. Sherrington
23
introduced the term nociception (from the Latin
In the present study, we report that tilmicosin attenuated chemical-induced, but not thermal-induced, acute pain in mice. This can be explained on the basis that the mechanistic pathways of thermal- and chemical-induced acute pains including receptors and ion channels are somehow different. For example, 5-HT1 receptors, except for the 5-HT1A subtype, are involved in the spinally mediated antinociception induced by thermal noxious stimuli. 25
Acetic acid-induced writhing, a visceral pain model, and formalin-induced pain, a cutaneous pain model are chemical stimuli tests that are commonly used for the evaluation of a general analgesic activity. In these models, pain is generated indirectly via irritating affected peripheral tissues and releasing from it endogenous mediators, including prostaglandins, bradykinin, serotonin, histamine, and substance P. These inflammatory mediators cause pain by stimulating peripheral nociceptive neurons, and are sensitive to NSAIDs and to narcotic analgesics as well. NSAIDs can inhibit cyclooxygenases in peripheral tissues, thus, interfere with the mechanism of transduction in primary afferent nociceptors via inhibition of the synthesis of prostaglandins.26,27
From the results presented in this study, the suppression of acetic acid-induced writhing and formalin-induced paw-licking (in the second phase) by tilmicosin was comparable to those of the standard drug, ASA, although its effect was lesser. The results indicated that tilmicosin may possess anti-nociceptive activity through reducing the synthesis of mediators involved in the nociceptive response, especially prostaglandins by inhibition of cyclooxygenases.
Unlike peripherally acting analgesics that act by blocking the generation of impulses at nociceptor site of pain, centrally acting analgesics raise the threshold of pain, and alter the physiological response to pain. 28 Failure of tilmicosin to change the reaction time against thermal-induced pain (unlike the standard drug morphine) in hot-plate and tail-flick tests indicate that it may not have any central analgesic effects.
Results of the formalin test, in particular, demonstrate that the two phases in the test may have different nociceptive mechanisms. It is suggested that the early phase is due to a direct effect on nociceptors (hence named the neurogenic phase) and that prostaglandins do not play an important role during this phase. However, the late phase seems to be an inflammatory response with inflammatory pain (hence named the inflammatory phase) that can be inhibited by anti-inflammatory drugs as ASA and, here, by the tested drug tilmicosin as well. Data, in addition, may indicate that ASA seems to have actions independent on their inhibition of prostaglandin synthesis as they also have effects on non-inflammatory pain of the first phase of formalin test as described previously. 29
In conclusion, data of the present study may indicate that tilmicosin has the potential of being a peripherally acting analgesic in addition to its antibacterial activity. This may have the benefit of synergism between tilmicosin and the concurrently administered analgesics and gives more explanation to its overall efficacy in respiratory inflammatory diseases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
