Abstract
This study investigated anti-inflammatory effects and possible mechanisms of Chikusetsusaponin IVa (Chi IVa), one of the main bioactive components in saponins from
Introduction
Inflammation is a series of vascular and cellular reactions that occur in response to pathogen invasion and/or tissue injury. 1 As part of the machinery to remove injurious stimuli and to initiate the healing process, inflammation plays a critical role in the progression of many diseases, including cardiovascular diseases, cancer, and autoimmune diseases. 2 It is well-established that the activation of macrophages and/or monocytes provides the first line of defense in host immunity and plays a central role in regulating inflammation via enhanced production of inflammatory mediators and cytokines, which include tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6). 3 These cytokines contribute to defense mechanisms of the host immunity in response to external invasion, but they may induce immuno-pathological disorders when macrophages are activated in excess. 4 Due to the complexity of the pathophysiology of inflammatory diseases, efforts have focused on identifying novel anti-inflammatory drugs that alleviate the inflammatory process at early stages of pro-inflammatory cytokine gene expression. 5
Chikusetsusaponin IVa (Chi IVa), an oleanane-type triterpenoid saponin, is considered to be a major component of SPJ. 13 In previous studies, Chi IVa was shown to possess antithrombotic, anti-viral, and anti-apoptotic effects in Fas-mediated cell death.13–15 However, at a single compound level, the anti-inflammatory effects and possible mechanisms of Chi IVa actions are not fully elucidated. Since activation of macrophages and their subsequent synthesis of TNF-α, IL-1β, and IL-6 play important roles in the occurrence and development of inflammation, we chose to explore the expression of inflammatory cytokines, NF-κB activation, and phosphorylation of ERK, JNK, and p38 in human THP-1 macrophages following LPS exposure in order to identify the anti-inflammatory effects of Chi IVa and its possible mechanism(s) of action.
Material and methods
Chemicals and reagents
Chi IVa (98% purity) was purchased from Sichuan Weikeqi Biological Technology Co., Ltd. (Chengdu, PR China). Phorbol 12-myristate 13-acetate (PMA) and lipopolysaccharide (LPS) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Human IL-1β, IL-6, and TNF-α ELISA kits were purchased from Dakewe Biotech Co., Ltd. (Shenzhen, PR China). Real-time PCR Master Mix and ReverTra Ace qPCR RT kits were purchased from Toyobo Life Science Department (Shanghai, PR China). Trizol® Reagent was purchased from Life Technologies Corporation (Shanghai, PR China). CCK8 and BCA protein assay kits were purchased from The Beyotime Institute of Biotchnology (Haimen, PR China). Total protein extraction kits and nucleoprotein extraction kits were purchased from Key GEN Biotech (Nanjing, PR China). Anti-human p-JNK, JNK, p-p38 MAPK, p38 MAPK, and COX-2 antibodies were purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA). Anti-human NF-κB (p65), p-ERK, and ERK antibodies were purchased from Bioworld Technology, Inc. (St. Louis Park, MN, USA). An anti-human iNOS antibody was purchased from Abcam, Inc. (Cambridge, MA, USA). Anti-human Lamin B antibody, anti-human β-actin antibody, horseradish peroxidase (HRP)-labeled goat-anti-rabbit, rabbit-anti-goat, and goat-anti-mouse IgG antibodies were purchased from Wuhan Boster Bio-engineering Co., Ltd. (Wuhan, PR China).
Cell culture and treatment
THP-1 human monocyte-like cells were obtained from the cell culture center of the Chinese Academy of Medical Sciences and cultured in DMEM medium (Gibico, Shanghai, PR China) supplemented with 15% heat-inactivated fetal bovine serum (FBS,Gibico, Shanghai, PR China), 100 U/mL penicillin, and 100 μg/mL streptomycin (Gibco-BRL, Gaithersburg, MD, USA). For the induction of monocyte macrophage differentiation, THP-1 cells were seeded in 96-well tissue culture plates at a density of 2 × 105 cells/mL for cell viability, and in 6-well tissue culture plates at a density of 1.2 × 106 cells/well for enzyme-linked immunosorbent assays (ELISA), real-time-PCR, and western blot analysis. The cells were pre-treated with PMA (100 nM) for 72 h to induce macrophage differentiation. 16 Differentiated cells adhered to the plate bottom, whereas undifferentiated monocytic cells were removed by washing with culture medium. Then, the cells were stimulated with LPS (1 μg/mL), with or without increasing concentrations of Chi IVa for 24 h at 37°C in the presence of 5% CO2. Cells were collected for real-time PCR and western blot, and culture supernatants were used for enzyme-linked immunosorbent assays (ELISA).
PMA-induced differentiation of THP-1 macrophages and CCK8 assay for THP-1 cell viability
THP-1 cells (105 cells/mL) were seeded in 96-well plates and differentiation was induced by PMA (100 nM) for 72 h in a 37°C and 5% CO2 incubator. Then, different concentrations of Chi IVa were added. Twenty-four hours later, supernatants were evaluated by CCK8 kit, according to the manufacturer’s instructions.
Real-time PCR
Total RNA was extracted and dissolved in RNA-free water and quantified using UV-clear microplates. Then, single-strand cDNA was synthesized from 2 μg total RNA by using ReverTra Ace qPCR RT Kit. Real-time PCR was performed using Realtime PCR Master Mix. The primers used were as follows: forward 5’-GGT- CGG-AGT- CAA-CGG-ATT-TG-3’ and reverse 5’-GGA-AGA-TGG-TGA- TGG-GAT-TTC-3’ for GAPDH; forward 5’-CTG-CTG-CAC-TTT-GGA-GTG-ATC-3’ and reverse 5’-GGT- TCG-AGA-AGA-TGA-TCT-GAC-TG-3’ for TNF-α; forward 5’-TGA-ACT-ACG- TCC-TGT-CCC-CT-3’ and reverse 5’-CTC-TTC-TCT-TGG-GTC-TCC-GC-3’ for iNOS; forward 5’-AGG-TCC-AAA-TCT-TGC-CTG-GG-3’ and reverse 5’-ATC-TGG- AGG-GGT-AGG-CTT-GT-3’ for COX-2; forward 5’-AAT-GAT-GGC-TTA-TTA-CAG- TGG-CA-3’ and reverse 5’-GCT-GTA-GTG-GTG-GTC-GGA-GAT-T-3’for IL-1β; forward 5’-GTG-AAA-GCA-GCA-AAG-AGG-CA-3’ and reverse 5’-TTG-GGT-CAG-GGG-TGG- TTA-TT-3’ for IL-6. All samples were measured in triplicate. Differences in gene expression were calculated using the 2-(△△CT) method.
Measurement of IL-6, IL-1β, and TNF-α
The cells were plated in 6-well plates and treated with LPS (1 μg/mL) alone or co-treated with Chi IVa for 24 h. IL-6, IL-1β, and TNF-α was detected in culture supernatants by ELISA kits, according to the manufacturers’ instructions.
Western blot assays
Total cell protein and the nucleoprotein fraction was extracted by protein extraction kits according to the manufacturer’s instructions. Protein concentrations were determined using the Enhanced BCA Protein Assay Kit (Beyotime Biotechnology, Nanjing, PR China). Samples of cell lysates were separated by 12% SDS-PAGE and then transferred onto nitrocellulose membranes. After being placed in blocking buffer, the membranes were incubated with the following primary antibodies: rabbit polyclonal anti-p-ERK (1:600 dilution), rabbit polyclonal anti-ERK (1:600 dilution), mouse monoclonal anti-p-p38MAPK (1:300 dilution), mouse monoclonal anti-p38MAPK (1:600 dilution), mouse monoclonal anti-JNK(1:800 dilution), mouse monoclonal anti-p-JNK (1:700 dilution), rabbit polyclonal anti-iNOS (1:200 dilution), goat polyclonal anti-COX-2 (1:300 dilution), rabbit polyclonal IκBα (1:300 dilution), rabbit polyclonal p-IκB-α (1:300 dilution), rabbit polyclonal NF-kB p65 (phospho S536) (1:500 dilution), rabbit polyclonal TLR4 (1:300 dilution), rabbit polyclonal anti-p65 (1:700 dilution), and mouse monoclonal anti-β-actin (1:1000; BOSTER Biotechnology, Wuhan, PR China). Then, goat anti-rabbit or goat anti-mouse -HRP-labeled secondary antibodies were used at a 1:40,000 dilution. The protein bands were visualized by the ECL kit. Then the intensities of the protein bands were analyzed by BandScan 4.3 software. β-actin protein was used as the internal control.
Statistical analysis
All data were expressed as mean ± standard deviation (SD). A database was set up with the SPSS 11.0 software package (SPSS Inc., Chicago, IL, USA). Differences among groups were analyzed by one-way analysis of variance (ANOVA), followed by Student–Newman–Keuls test, when applicable. Resulting
Results
PMA-induced differentiation of THP-1 macrophages and effects of Chi IVa on THP-1 cell viability
To determine the toxicity of Chi IVa on THP-1 cells, the cell viability was assayed by a CCK8 assay. As shown in Figure 1b, we found that Chi IVa, at a dose of 50–200 μg/mL, did not display any cellular toxicity against THP-1 cells over the 24-h treatment period, which allowed us to exclude non-specific cytotoxicity as a possible explanation for any decrease in cytokine output. We also observed the changes of some macrophage surface markers, such as CD14, CCR7, and CD80 molecules on PMA differentiated and undifferentiated THP1 cells by flow cytometry method. And we found that increased expression of CD14, CCR7, and CD80 molecules on these cells was followed by PMA stimulation (Figure 1c).

Chemical structure of Chi IVa and effects of Chi IVa on the cell viability of THP-1 cells. Chemical structure (a) and cell viability (b) was assessed by CCK8 assays, and the results are expressed as the percentage of surviving cells over the THP-1 cells after treatment with different concentrations of Chi IVa (0, 50, 100, and 200 μg/mL). Data represent means ± SD for three independent experiments.
Effects of Chi IVa on LPS-stimulated COX-2 production in THP-1 cells
To evaluate the effects of Chi IVa on COX-2 expression, THP-1 cells were induced to differentiate into macrophages through pre-treatment with PMA; the resulting cells were then were stimulated with LPS (1 μg/mL) with or without selected concentrations of Chi IVa for 24 h. The protein levels of COX-2 were increased by LPS alone. However, Chi IVa significantly decreased COX-2 production in a dose-dependent manner (Figure 2a and b). Moreover, to investigate the mechanism responsible for the inhibitory effect of Chi IVa on COX-2 production, mRNA expression was evaluated by real-time PCR. We found that the expression of COX-2 mRNA was markedly enhanced in the presence of LPS alone; Chi IVa (50–200 μg/mL) significantly reversed these abnormal changes compared to the LPS group (

Effects of Chi IVa on COX-2 production in LPS-stimulated THP-1 cells. The level of iNOS were determined by western blot. (a) Representative western blot result for three experiments, (b) means ± SD for three independent experiments. The expression of COX-2 mRNA (c) was assessed by real-time PCR. ##
Effects of Chi IVa on LPS-stimulated iNOS production in THP-1 cells
Compared to the untreated group, LPS-stimulated THP-1 cells showed significantly increased iNOS production. In contrast, Chi IVa (50–200 μg/mL) reduced iNOS production at the protein level, which was dose-responsive (Figure 3a and b).
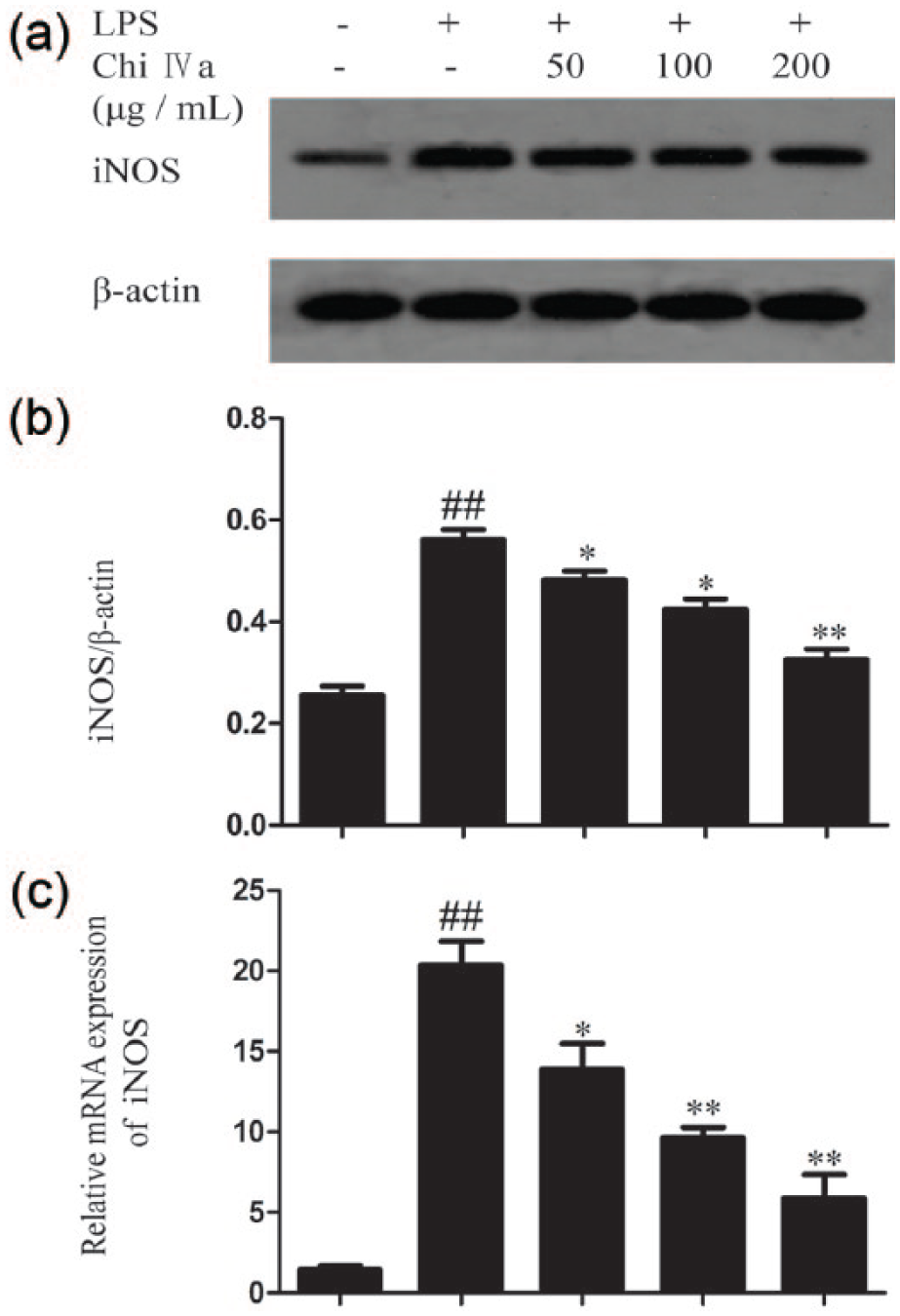
Effects of Chi IVa on iNOS production in LPS-stimulated THP-1 cells. The level of iNOS were determined by western blot. (A) Representative western blot result for three experiments, (b) means ± SD for three independent experiments. The expression of iNOS mRNA (c) was assessed by real-time PCR. ##
As for iNOS mRNA levels, there was a significant increase in the LPS-treated group relative to the untreated group (
Effects of Chi IVa on LPS-stimulated TNF-α, IL-1β, and IL-6 production in THP-1 cells
As shown in Figure 4, the expression of TNF-α, IL-1β, and IL-6 in the supernatants of LPS-stimulated THP-1 cell cultures was significantly increased compared to the control group (

Effects of Chi IVa on TNF-α, IL-1β, and IL-6 production in LPS-stimulated THP-1 Cells. The levels of TNF-α (a), IL-1β (b), and IL-6 (c) in the culture medium were measured by enzyme-linked immunosorbent assay (ELISA).The expressions of TNF-α, IL-1β, and IL-6 mRNA (d) was assessed by real-time PCR. The values shown are the means ± SD of three independent experiments. ##
To further detect changes in TNF-α, IL-1β, and IL-6 expression in LPS-stimulated THP-1 cells, real-time PCR was used. As shown in Figure 3, TNF-α, IL-1β, and IL-6 mRNA expression in THP-1 cells was markedly increased upon exposure to LPS alone (
Effects of Chi IVa on NF-κB activation in LPS-stimulated THP-1 cells
NF-κB is a key transcription factor involved in iNOS, COX-2, and TNF-α gene expression in LPS-stimulated macrophages. To further understand how Chi IVa influences anti-inflammatory effects in THP-1 cells, we investigated whether Chi IVa also had an inhibitory effect on the expression of NF-κB. Our results demonstrate that LPS treatment strongly decreased p65 in the cytoplasm and increased it in nuclei in PMA-differetiated-THP-1 cells; Chi IVa (50–200 μg/mL) treatment significantly prevented this cytoplasm- nuclei shift at protein level compared to the LPS-treated group (
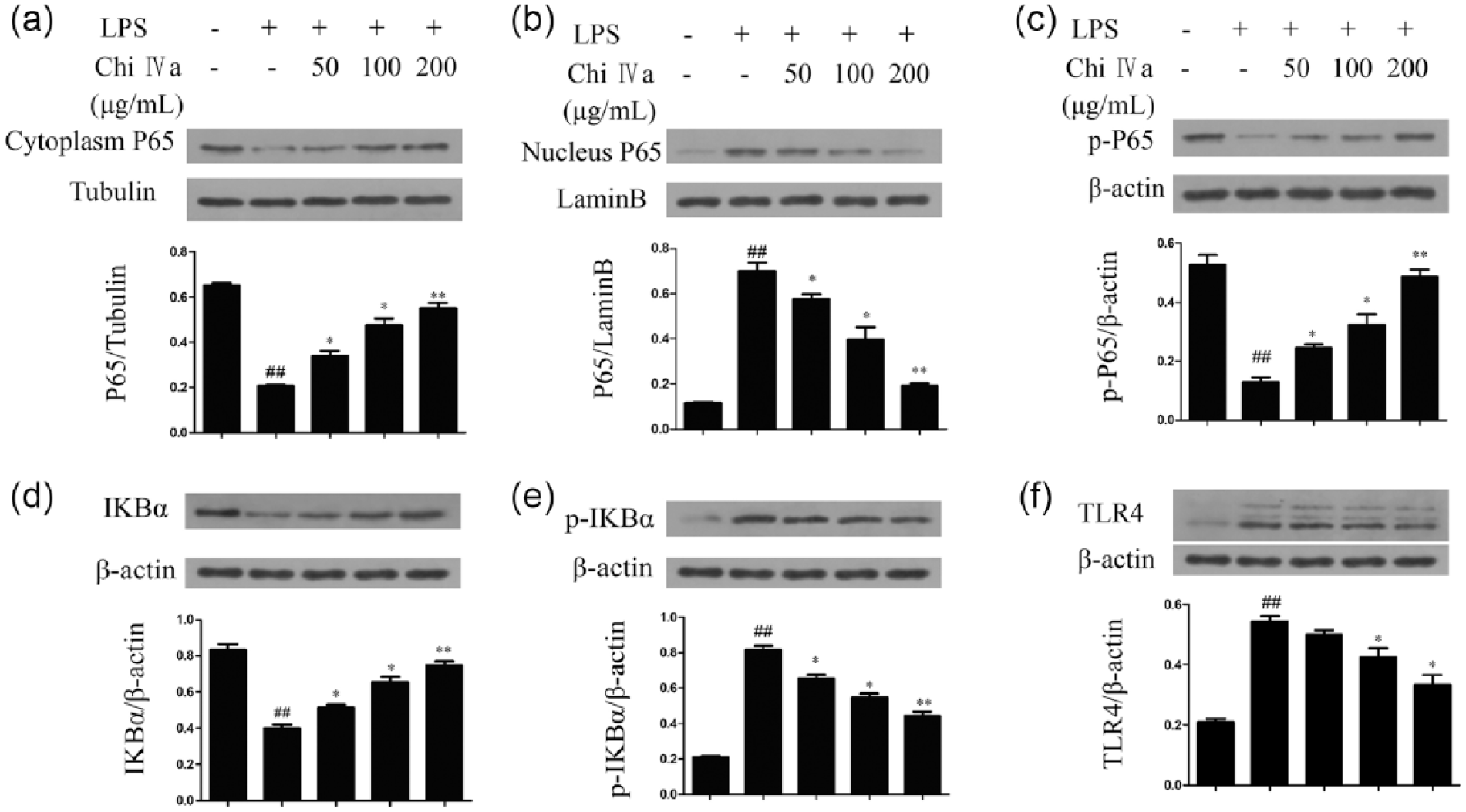
Effects of Chi IVa on NF-κB activation in LPS-stimulated THP-1 cells. THP-1 cells were stimulated with LPS (1 μg/mL) and different concentrations of Chi IVa (0, 50, 100, and 200 μg/mL) for 24 h. The cytoplasm P65 (a), nucleus P65 (b), phosphor-p65 (c), IkBα (d), total p-IkBα (e), and TLR4 (f) were then detected by western blot. (a–f) Representative western blot result for three experiments and means ± SD for three independent experiments. ##
Effects of Chi IVa on phosphorylation of MAPK in LPS-stimulated THP-1 cells
To further investigate the mechanism of Chi IVa-mediated inhibition of LPS-stimulated THP-1 activation, the effects of Chi IVa on the activities MAPK (p38, ERK, and JNK) were evaluated. Western blot analysis showed that LPS stimulation resulted in phosphorylation of ERK, JNK, and p38 MAPK in THP-1 cells, while Chi IVa (50–200 μg/mL) suppressed the phosphorylation of ERK, JNK, and p38 MAPK (

Effects of Chi IVa on phosphorylation of MAPK in LPS-stimulated THP-1 cells. Cells were stimulated with LPS (1 μg/mL) and different concentrations of Chi IVa (0, 50, 100, and 200 μg/mL) for 24 h. The cells were then collected and extracted. Western blot analysis was performed to detect p-JNK, total JNK, total p38 MAPK, p-p38 MAPK, p-ERK, total ERK, and β-actin. (a) Representative western blot result for three experiments, (b–d) means ± SD for three independent experiments. ##
Discussion
Macrophage activation plays an essential role in the generation, development, and resolution of inflammation.
17
Macrophages can produce large amounts of inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, in response to various inflammatory stimuli, especially LPS.
18
This results in enhanced macrophage activation and can therefore result in significant amplification of the inflammatory process.
19
Consequently, macrophages and their products have become a focal target for therapies aimed at alleviating inflammatory diseases. Recently, a growing body of evidence suggests that some Traditional Chinese Medicines (TCMs), from a wide variety of sources, show promise as inflammation dampening agents that possess low toxicity levels.
The overproduction of iNOS and COX-2 in response to bacterial LPS is often associated with inflammatory disorders resulting from high levels of production of both nitric oxide (NO) and prostaglandin E2 (PGE2). In LPS-activated macrophages, the iNOS enzyme produces copious amounts of NO, an important messenger molecule that when too abundant can contribute to inflammatory diseases, including atherosclerosis, rheumatoid arthritis, and septic shock; NO has functions in vascular regulation, host immune defense, and other systems.20,21
Likewise, COX-2, another inducible enzyme, generates PGE2 by catabolism of the substrate lipid arachidonic acid; the production of PGE2 can stimulate pain, inflammation, and fever at inflammatory sites. 21 Therefore, limiting the activation of iNOS and COX-2 may effectively reduce pro-inflammatory levels of PGE2 and NO to minimize the detrimental effects of excessive/unresolved inflammation, representing an effective therapeutic strategy for the prevention of inflammatory diseases. Our results suggest that Chi IVa targets both iNOS and COX-2 at both the mRNA and protein level in a dose-dependent manner in LPS-stimulated THP-1 macrophages .To our knowledge, this is the first report to demonstrate that Chi IVa can effectively inhibit iNOS and COX-2 and/or modulate inflammation.
Furthermore, excessive synthesis of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) plays a critical role in acute inflammation and chronic inflammatory diseases, such as atherosclerosis, liver disease, and inflammatory arthritis.22,23 In this study, we demonstrated that Chi IVa can inhibit the production of pro-inflammatory cytokines by inhibiting of expression/translation of pro-inflammatory mediators in LPS-stimulated THP-1 macrophages. As the dose of Chi IVa increases, all of these changes became much stronger.
NF-κB is an important transcription factor that regulates various genes involved in immune and inflammatory responses. 24 As a key transcription factor, NF-κB induces expression of iNOS, COX-2, and various inflammatory cytokines, such as IL-1β, IL-6, and TNF-α. 25 Prior to activation, NF-κB is in an inactive form bound to inhibitory IκB subunits within the cytoplasm. When triggered by LPS binding to toll-like receptor 4 (TLR-4), IκBα and IκBβ are degraded, which results in the translocation of specific NF-κB subunitsto the nucleus to activate gene transcription. 26 In our study, we found that p65 decreased in the cytoplasm and increased in nuclei in PMA-differetiated-THP-1 cells after LPS treatment. Chi IVa was able to revise this process in a dose dependent manner. Moreover, we also observed that increased p-IkBα and decreased total IkBα in PMA-differetiated-THP-1 cells following LPS treatment were inhibited by Chi IVa. From these data, it suggested that the Chi IVa could effectively downregulate inflammatory cytokine expression in LPS-activated THP-1 macrophages, which may be associated with the potency of Chi IVa to inhibit NF-κB pathway.
In the macrophage response to LPS, the first step is the combination of LPS and the LPS-binding protein (LBP).The LPS –LBP complex can be recognized by TLR4 and trigger the TLR4 downstream signaling cascade, including the MyD88-dependent and MyD88 independent pathway. In the MyD88-dependent pathway, recruitment of MyD88 to TLR4 is followed by signaling cascade involving the interleukin-1-receptor-associated kinases (IRAKs), among which IRAKs-1, -2, and -4 promote the signal transduction while IRAK-3 inhibits the signal transduction. Subsequently, the MyD88-dependent pathway culminates in the activation of MAPK and NFκB. 27 In our study, we found that Chi IVa was able to suppress the increasing expression of TLR4 primed by LPS. These data suggest that Chi IVa might regulate the expression of TLR4 to inhibit NF-κB and MAPK activation. However, further studies need to investigate whether Myd88 or IRAKs are the potential targets for Chi IVa.
The activation of NF-κB is also regulated by various cellular kinases, including the mitogen-activated protein kinase (MAPK) family, which is classified into at least three components: extracellular signal-regulated kinases 1/2 (ERK 1/2), c-Jun N-terminal kinase (JNK), and p38 MAPK. 28 As one of the most important intracellular signaling cascades in macrophages, MAPKs have been implicated in the release of pro-inflammatory cytokines in LPS-induced inflammatory responses. 29 To investigate whether these pathways are involved in the molecular mechanism(s) of Chi IVa-mediated anti-inflammatory effects in THP-1 macrophages, we explored the levels of phosphorylation of ERK, JNK, and p38 MAPK. The levels of phosphorylation of ERK, JNK, and p38 MAPK were upregulated in THP-1 macrophages treated with LPS, but were downregulated in cells also treated with Chi IVa. As the dose of Chi IVa increased, these changes became stronger. These results suggest that Chi IVa might also affect the MAPK signaling cascade in LPS-stimulated THP-1 cells.
Other transcriptional factors, in addition to NF-κB, such as activation protein 1 (AP-1), signal transducer and activator of transcription 2 (STAT2), and cyclic AMP-responsive element-binding protein 1 (CREB1) are able to mediate LPS-induced inflammation. 30 These transcriptional factors also regulate the expression of pro-inflammatory genes, including TNF-α, COX-2, and iNOS. 31 Therefore, additional studies are needed to investigate the relationships among the various signaling pathways and transcription factors involved in the Chi IVa-induced inhibition of pro-inflammatory gene expression.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation of China (NSFC81100282, NSFC81030007, NSFC81171558), the National twelfth ‘five years’ project in Science and Technology (2013ZX10002003), Hubei Province’s Outstanding Medical Academic Leader Program, Program for Innovative Research Team of Chinese Ministry of Education (IRT1131, IRT14R20), China Postdoctoral Science Foundation (No. 2013M531700).
