Abstract
The aim of this study was to evaluate the concentrations of CD45pos leukocytes in the semen samples of infertile patients with low testicular volume (TV) compared to subjects with normal TV. The testis was considered normal in size when it had a volume between 15 and 25 cm3, low–normal with a volume between 10 and 12 cm3 and hypotrophic when the volume was <10 cm3. The patients with low testicular volume (<10 cm3) showed significantly higher concentrations of CD45pos leukocytes compared to other groups (P <0.05). The correlation analysis showed the presence of a positive linear relationship between CD45pos leukocytes and the percentage of immature germ elements (r = 0.88; P <0.05) and between CD45pos leukocytes and the percentage of spermatozoa with phosphatidylserine externalisation (r = 0.90; P <0.05) as well as a negative linear relationship between the percentage of spermatozoa with normal morphology and the seminal CD45pos leukocyte concentrations ( r = −0.75; P <0.05). The results of this study showed that patients with low testicular volume (<10 cm3) have significantly increased CD45pos leukocyte concentrations associated with increased percentages of immature germ elements, spermatozoa with signs of early apoptosis and spermatozoa with abnormal morphology.
The testis consists of two structurally distinct compartments: the interstitial cell or Leydig cell compartment and the seminiferous tubules compartment, which contains germ cells and Sertoli cells. These two compartments are responsible for the two main testicular actions: the endocrine and the exocrine functions (production of androgens and spermatozoa). In clinical practice, testicular volume (TV) can be estimated by a physical examination using an orchidometer, but it can be more objectively assessed by an ultrasound examination, which is less affected by factors such as scrotal effusions, varicose veins, intra-scrotal masses, testicular pain, and distinction with the adjacent anatomical structures. It has been widely shown that TV reflects its function including both endocrine (androgen decline) and reproductive activity (abnormal sperm parameters). The testicular functional characterisation in relation to different TV is traditionally correlated to parameters such as sperm concentration, motility, morphology, gonadotropins and total testosterone serum concentrations.1,2
We recently published a paper on the quality of some important bio-functional parameters of patients with reduced testicular volume, in particular their sperm DNA fragmentation, the mitochondrial membrane potential, the compaction of chromatin and finally the apoptosis of spermatozoa. 2 However, in the literature there is a paucity of data on another important aspect which is the characterisation of the chronic inflammatory response in the semen of these patients.
The testis is considered an immunoprivileged organ because it is able to tolerate autoantigens from germ cells that appear after puberty when immunocompetence is already established. Multiple mechanisms to prevent autoimmune disease in the testis involve: (1) an immunological blood testis barrier: a structure that limits the access of germ cell antigens to interstitial immune cells and the passage of antibodies from interstitium to tubular lumen; (2) secretion of immunosuppressive factors mainly by macrophages, Sertoli, peritubular and Leydig cells; (3) a scarce number of T cells (mainly suppressor CD8+ cells); and (4) the presence of regulatory T cells which have been postulated as modulators of immune response acting through local and systemic mechanisms. The delicate equilibrium between immunoprivilege and inflam-mation is modulated by cytokines playing a dual role as immunosuppressors and proinflammatory mediators. The existence of several T cell subsets in normal testicular interstitium of human and rodents has been established. These cells exhibit a wide variety of functions, including regulation of immune response and innate immunity modulation. 3
In clinical practice the finding of reduced testicular volume in adulthood with no apparent cause could reflect a sign of impaired pubertal maturation and the semen analysis is an indispensable tool for the evaluation of testicular function (Figure 1).

Importance of the low testicular volume in clinical practice: traditional and unconventional aspects.
The evaluation of leukocytes in the semen represents a fundamental tool for the quantification of the inflammatory response, in particular acute inflammation. However this parameter does not evaluate the chronic inflammatory response. 4
It is not possible to identify reliably leukocytes by conventional sperm staining techniques. The peroxidase method is sufficient for quantification of granulocytes, but immunocytology is the gold standard for the detection of all leukoyte populations in semen. Granulocytes are the most prevalent leukocyte type in semen (50–60%), followed by macrophages (20–30%) and T-lymphocytes (2–5%). 4
CD45 (lymphocyte common antigen) is a receptor-linked protein tyrosine phosphatase that is expressed on all leucocytes, and which plays a crucial role in the function of these cells. On T cells the extracellular domain of CD45 is expressed in several different isoforms, and the particular isoform(s) expressed depends on the particular subpopulation of cells, their state of maturation and whether or not they have previously been exposed to antigens. 5
Therefore the aim of this study was to evaluate a specific unconventional sperm parameter: the concentrations of CD45pos leukocytes in the semen of patients with low TV compared to subjects with normal TV.
Materials and methods
Forty infertile patients with an age range of 23–45 years (mean, 25.0 ± 5.0 years) and a BMI of 20.0–30.0 kg/m2 (mean, 25.0 ± 3.0 kg/m2) were consecutively enrolled for the clinical work-up of male infertility (Table 1) and were classified into three different categories according to the different testicular volume measured by ultrasound. Twenty fertile men age matched (pregnancy-induced <12 months) represented the control group.
Investigations carried out on the patients for the evaluation of the clinical condition of male infertility.

Exclusion criteria
Primary scrotal disease identified by ultrasound evaluation: microlithiasis, inhomogeneous testi-cular echotexture, testicular cancer, varicocele, hydrocele, abnormal testicular arterial haemodynamic (systolic peak velocity and resistance index); clinical history of cryptorchidism, varicocelectomy, eversion of the tunica vaginalis, head trauma, endocrine abnormalities (increased gonadotropins, reduced serum total testosterone, hyperprolactinaemia, increased oestrogen concentration), systemic diseases (kidney disease, liver disease, diabetes mellitus), cigarette smoking, alcohol use, concomitant use of other drugs during the previous 6 months, leukocytospermia, male accessory gland inflammatory disease, and positive seminal, urine and/or urethral swab cultures.
Scrotal ultrasound evaluation
The scrotal ultrasound evaluation was performed in two phases: the first with the patient in a supine position (with the penis resting on the suprapubic region) and the second in an upright position to evaluate reflux along the pampiniform plexus, testicular pain, testicular malposition or the extent of any fluid collections. The examination was performed with a GX Megas Esaote (Esaote SpA, Genova, Italy) device, equipped with linear, high-resolution and high-frequency (7.5–14 MHz) probes dedicated to the study of soft body areas, with colour Doppler for detecting slow flow and a scanning surface of at least 5 cm. Testicular volume was calculated automatically by the ultrasound machine using the ellipsoid formula (length × width × thickness × 0.52). The testis was considered normal in size when it had a volume in the range of 15–25 cm3, low–normal with a volume in the range of 10–12 cm3 and hypotrophic for a volume of less than 10 cm3. The parenchymal echostructure was considered normal in the presence of thin, densely packed and homogeneously deployed echoes. The presence of a finely inhomogeneous echopattern and weakly hypo- or hyper-echogenic areas was considered indicative of primary testicular disease. During the Doppler evaluation, flow was detected at the level of the spermatic artery and the testicular artery and its branches. The velocity analysis was considered normal in the presence of low resistance, a prolonged systolic phase, flow maintenance during diastole and a low resistance index (IR: 0.62). Systolic flow speed along the centripetal arteries was considered normal if it was lower than 15 cm/s and/or in the range of 4–12 cm/s. 6
Hormone measurements
Hormonal evaluations were performed by electrochemiluminescence with Hitachi-Roche equipment (Cobas 6000, Roche Diagnostics, Indianapolis, IN, USA). The reference intervals were as follows: LH, 1.6–9.0 mIU mL−1; FSH, 2.0–12.0 mIU mL−1; 17β-estradiol, 8.0–43.0 pg mL−1; total testosterone, 2.8–8.0 ng mL−1; and prolactin, 4.0–15.0 ng mL−1. Blood sampling was performed at 08:00, after at least 8 h of sleep. The determination of serum LH and prolactin was repeated after an interval of 30 min.
Semen analysis
Semen samples were collected by masturbation into a sterile container after 2–7 days of sexual abstinence and were transported to the laboratory within 30 min after ejaculation. According to the 2010 World Health Organization (WHO) guidelines, each sample was evaluated for seminal volume, pH, sperm count, progressive motility, morphology and round cell concentration. 7
Conventional measurement of seminal leukocytes and immature germ cells Seminal leukocytes: Peroxidase test
The working solution used for the test was obtained by adding 1 μL of H2O2 to 20 μL of a 0.09% 3,3’-diaminobenzidine tetrahydrochloride stock solution (DAB, ISOPAC, Sigma, Milan, Italy) in 40% ethanol. In each assay, 20 μL of semen was incubated with 20 μL of working solution in an Eppendorf tube for 5 min at room temperature. Before setting up the slide, 40 μL of PBS was added. Peroxidase-positive cells were marked by yellow-brown-red staining, while peroxidase-negative cells remained colourless. At least 100 round cells were counted using an optical microscope at 400X magnification, and the percentages of peroxidase-positive and negative cells were evaluated. The total leukocyte count was expressed in millions/ml of semen.
Immature germ cell evaluation in semen
Spermatid identification was conducted on the basis of the following parameters: colouration, size, core shape and size (approximately 5 μM), absence of intracellular peroxidase and the absence of leukocyte-specific antigen (see cytofluorimetric assessment section). Morphologically, multinucleated spermatids were distinguished from polymorphonuclear leukocytes by the presence of a pink colour in contrast to the bluish colour of polymorphonuclear leukocytes.
Cytofluorimetric assessment
The analysis was carried out with an EPICS XL Flow Cytometer (Coulter Electronics, IL, Milan, Italy), equipped with an argon laser at 488 nm and three fluorescence detectors: green (FL-1 at 525 nm), orange (FL-2 to 575 nm) and red (FL-3 at 620 nm). For each sample, 100,000 events were measured at a low flow velocity and analysed using Sistem II™, version 3.0. The following unconventional parameters were investigated: (1) sperm mitochondrial membrane potential (MMP); (2) chromatin compaction degree after staining with propidium iodide (PI); (3) phosphatidylserine externalisation (PS) (early apoptosis index) after staining with annexin V and PI; and (4) sperm DNA fragmentation by TUNEL.
MMP evaluation
An aliquot containing 1 × 106 sperm/mL was incubated with JC-1 (5,5’,6,6’-tetrachloro-1,1’,3,3’-tetraethylben zimidazolylcarbocyanine iodide) (Space Import Export, Milan, Italy) in the dark for 10 min at a temperature of 37°C. At the end of the incubation period, the cells were washed with PBS and analysed.
Degree of chromatin compaction
PI staining was performed after cell membrane permeation to allow fluorochrome penetration of the nucleus. In brief, 1 × 106 spermatozoa were incubated with LPR DNA-Prep Reagent containing 0.1% cyanide potassium, 0.1% NaN3, non-ionic detergents, salts and stabilisers (Beckman Coulter, IL, Milan, Italy) in the dark at room temperature for 10 min. They were then incubated with Stein DNA-Prep Reagent containing 50 µg/mL of PI (<0.5%), RNase A (4 KUnits/mL) and <0.1% NaN3 (Beckman Coulter, IL, Milan, Italy) in the dark at room temperature. Flow cytometric analysis was performed after 30 min. In this analysis, FL3 was the only detector used.
PS externalisation
Staining with annexin V and PI was performed using the Annexin V-FITC Kit (Sigma Chemical, Perth, Australia). An aliquot containing 0.5 × 106 spermatozoa/mL was suspended in 0.5 mL of buffer containing 10 μL of annexin V-FITC plus 20 μL of PI and incubated for 10 min in the dark. After incubation, the sample was immediately analysed. Signals were detected by FL-1 (FITC) and FL-3 (PI) detectors. The cell population in question was identified in forward scatter (FSC) vs. side scatter (SSC) plots. Different staining patterns enabled the identification of different cell populations: FITC negative and PI negative indicated live cells, FITC positive and PI negative indicated apoptotic cells, and FITC positive and PI positive indicated late-phase apoptosis or necrosis.
DNA fragmentation analysis
DNA fragmentation analysis was performed using a commercially available kit (Mebstain Apoptosis, Beckman Coulter, IL, Milan, Italy). To obtain the negative control, TdT was omitted from the mixing reaction. The positive control was obtained by pretreating spermatozoa with 1 mg/mL of deoxyribonuclease I without RNase A at 37°C for 60 min before staining. Debris was removed using the same procedure. Light-scatter and fluorescence data were obtained at a fixed setting on a logarithmic scale. The percentage of sperm cells stained with FITC was determined using the FL-1 detector of the flow cytometer.
Leukocytes flow cytometric analysis
To perform the absolute leukocyte count, 100 μL of each liquefied semen sample was incubated with a mixture containing Syto-16 green fluorescent nucleic acid stain to identify the spermatozoa and exclude debris (final concentration 200 nM, Molecular Probes, Eugene, OR, USA), 7-Amino-Actinomycin D (7-AAD Via-Probe, BD Pharmingen, San Diego, CA, USA) to assess viability, anti-CD45-APC (pan-leukocyte antigen) to recognise white blood cells, and anti-CD16-PE for PMN recognition. The addition of 100 μL of Flow-CountTM Fluorospheres (Beckmann-Coulter, Fullerton, CA, USA) at a 1034 beads/mL allowed the determination of the absolute leukocyte count by flow cytometry. After incubation in the dark for 20 min at room temperature, 1 mL of PBS was added, and the sample was analysed by flow cytometry. For each test, 100,000 events were acquired.
All patients signed informed consent. The study was approved by the internal committee of the Institute. The patients of the treated group signed an additional consent for off-label use of the drug.
Statistical analysis
The results were reported as the mean ± SEM. The data were analysed using a one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test. A correlation analysis was conducted by evaluating the Pearson correlation coefficient to assess possible covariance linearity between CD45pos leukocytes and the other parameters. Statistical analyses were performed using SPSS 9.0 for Windows (SPSS Inc., Chicago, IL, USA). A P value less than 0.05 was accepted as statistically significant.
Results
Table 2 shows the comparative patient data following classification by testicular volume. Patients with VT <10 cm3 showed a significant reduction of all conventional sperm parameters compared to controls and patients with testicular volume in the range of 12–25 cm3. Moreover, these patients showed significant alterations of hormonal and flow cytometry parameters compared to other examined groups.
Laboratory data of the enrolled patients (n = 40 patients) according to the different testicular volumes.

P <0.05 vs. controls.
P <0.05 vs. TV 12–25 cm3 and controls.
P <0.05 vs. TV 15–25 cm3 and controls.
No significant differences were detected with regard to the following parameters: age, BMI and duration of infertility (for infertile patients).
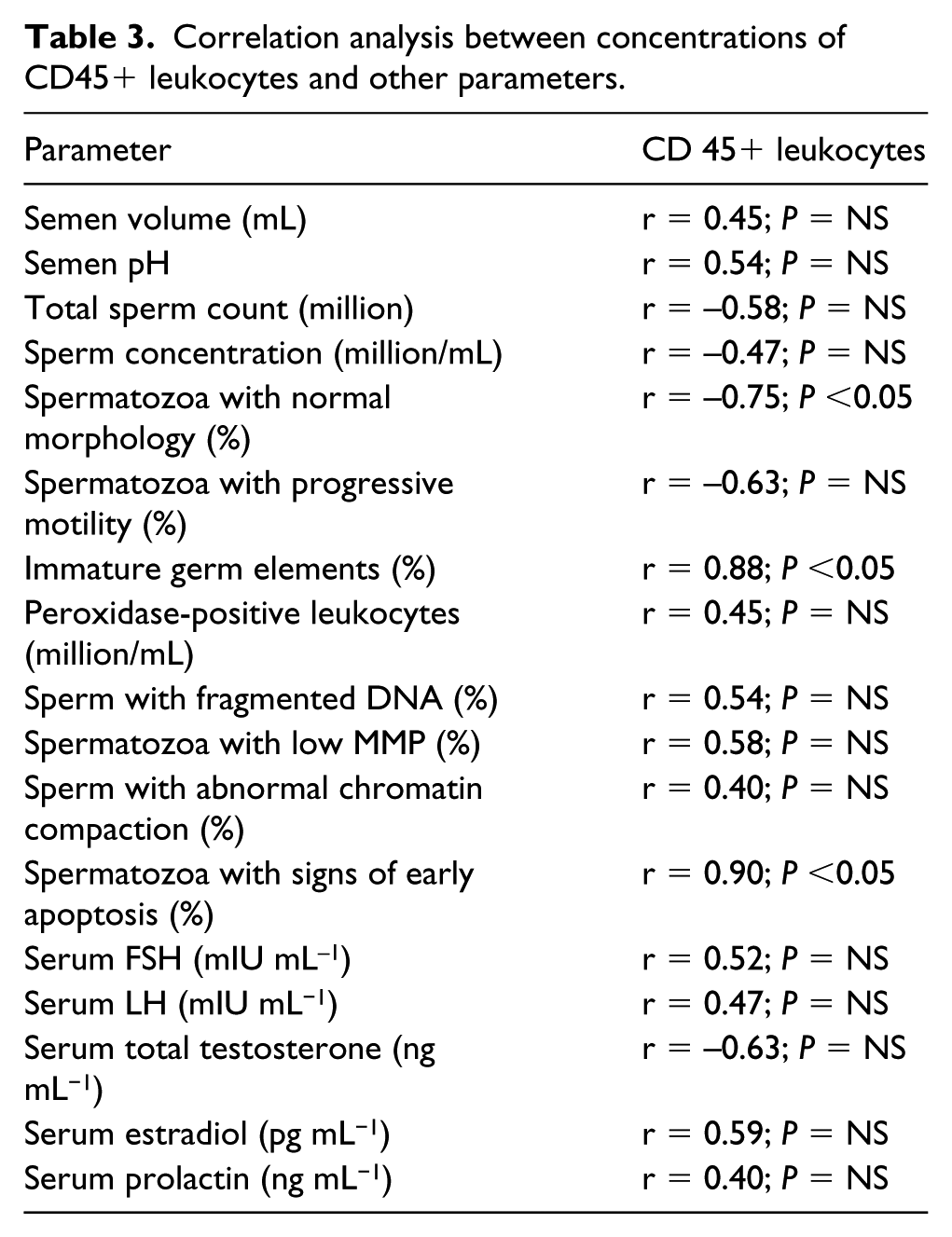
The correlation analysis (Table 3), showed a positive linear relation between CD45pos leukocytes and the percentage of immature germ elements and between CD45pos leukocytes and the percentage of sperm with PE externalisation. Finally, the correlation analysis showed a negative linear relation between the percentage of spermatozoa with normal morphology and the conentrations of CD45pos leukocytes in the semen.
Correlation analysis between concentrations of CD45+ leukocytes and other parameters.
A significant relation was observed between TV with BMI (r = −0.75; P <0.05) and age (r = −0.75; P <0.05), respectively. No significant relation was observed between the concentrations of CD45pos leukocytes in the semen with BMI (r = −0.56; P = NS) and age (r = −0.59; P = NS), respectively.
Discussion
Our data suggest that patients with low TV (<10 cm3) in the absence of ultrasound-detectable scrotal abnormalities and endocrine signs of functional primitive testicular impairment and with normal concentrations of peroxidase-positive leukocytes (neutrophils) have significantly increased CD45pos leukocyte concentrations in the semen. This may be related to an increase of immature germ cells, a greater percentage of spermatozoa with signs of early apoptosis, and significant teratozoospermia. The same parameter did not show any statistically significant correlation with BMI and age. However, according to our recent experience, 8 obesity could have a negative impact on male fertility as confirmed by the presence of a negative correlation between BMI and TV.
Several studies have shown that the biological fate of leukocytes in semen is not unique. Antimicrobial action is not the only function of these cells. Their presence in the semen is also aimed at facilitating the elimination of immature germ cells via an endogenous anti-inflammatory response.
From the laboratory point of view, the routine practice only includes a peroxidase test for leukocyte measurement as recommended by the WHO manual. 7 However, this method is only able to detect granulocytes. 4 Conversely, flow cytometry allows the identification of all leukocyte sub-populations present in the seminal fluid.9,10 In particular, granulocytes represent the predominant component (range, 50–60%), followed by macrophages (range, 20–30%), T lymphocytes (range, 2–5%) and B lymphocytes. 4 . Finally, leukocytospermia (>1 million/mL peroxidase-positive leukocytes in the semen) is present in approximately 10–20% of infertile patients.11,12
The significance of the presence of leukocytes in the semen is very controversial. In fact, some authors have not observed any relationship between seminal leukocytes and sperm damage, highlighting that, physiologically, the presence of low concentrations of white blood cells in the semen is associated with improved fertilisation and better results in assisted reproduction techniques. 11 In contrast, other studies have shown that leukocytes have a co-factor role in male infertility, 12 indicated by the high prevalence of leukocytospermia in infertile patients, the association between leukocytospermia and oligozoospermia, asthenozoospermia, alteration of functional tests and/or a high failure rate in fertilisation cycles.
In the absence of clinical symptoms, the true origin of leukocytes in the semen is difficult to understand. Their origin from the epididymis has been confirmed by studies performed using the vasectomy model,13,14 with evidence of a local inflammatory response following the procedure. However, in patients with leukocytospermia, seminal citric acid concentration (a marker of prostate function) appears to be lower, suggesting that prostatitis is the main cause of their presence in semen. 15 Moreover, a large percentage of semen samples with leukocytospermia could result negative to microbiological tests. In fact, bacteriospermia and elevated seminal leukocytes are prevalent, but not statistically associated. 12 However, the mere presence of leukocytes can alter sperm quality.16,17
Several previous studies have shown that leukocytes play a physiological role in the immune surveillance and the removal of abnormal spermatozoa.18–20 Although the mechanisms have not been clarified, some authors have shown a close relationship between leukocytospermia and morphological and functional sperm alterations, including teratozoospermia and sperm necrosis.18–20 Accord-ing to these studies, high leukocyte concentrations in the ejaculate compromise spermatozoa structural integrity, resulting in alterations of the acrosome, nucleus and tail.
The production of free oxygen radicals by leukocytes could represent the connecting element between leukocytospermia and infertility. Free oxygen radicals interfere with the physiological processes related to reproduction, and it is known that a free oxygen radical increase may compromise male fertility. 21
However, the relationship between the presence of leukocytes and spermatozoon structural alterations may not only be a cause-effect relationship; seminal alterations may also represent a stimulus for leukocyte production. In this regard, Aziz and colleagues 20 showed that semen leukocyte concentration is significantly correlated with structural alterations of the sperm acrosome, the presence of cytoplasmic droplets, and structural defects of the tail using an index named the ‘DFI sperm deformity index’. These observations suggest that leukocytospermia may be associated with impaired spermatozoon structural integrity. Data concerning the spontaneous variation of leukocyte concentration in the semen of asymptomatic infertile men 22 appear to further support this hypothesis since spermatogenesis is not a static phenomenon. It is also interesting that the increase of M540 bodies (round elements without a nucleus that express apoptotic markers) in the semen is related to ultrasound alterations in the testicular region; in particular, a negative relationship between TV and the M540 body concentration in semen has been reported. 23
The causes of TV reduction with no clear clinical signs remain unclear because of the absence of testicular disease identified by scrotal ultrasonography. In this regard, it is important to emphasise that some testicular morphostructural alterations may be undetectable by physical and/or instrumental examination. In particular, chronic orchitis is difficult to clinically diagnose because of its asymptomatic course. However, a histological examination can detect inflammatory lymphocytic peritubular infiltrate in many patients in the absence of relevant scrotal pathology associated with TV and sperm count reduction.24,25
Unfortunately, we could not perform a histological examination in the patients enrolled in this study. This aspect should be evaluated in subsequent studies.
In conclusion, the results of this study showed that patients with low TV (<10 cm3) have a significantly increased CD45pos leukocyte concentration that is associated with increased percentages of immature germ cells and spermatozoa with signs of early apoptosis and abnormal morphology.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
