Abstract
Introduction
Diagnosis and monitoring of metastatic colorectal cancer (mCRC) depend on diagnostic imaging. Circulating carcinoembryonic antigen (CEA) can be analyzed but no optimal, non-invasive biomarker exists. Circulating collagen IV (COL IV) is a promising biomarker in patients with colorectal liver metastases (CLM). This study aimed to evaluate COL IV and other cancer-related and stroma-derived proteins as biomarkers for mCRC.
Materials & methods
Plasma COL IV and 10 other proteins were analyzed with ELISA and Luminex multiplex assays.
Results
mCRC patients have elevated levels of circulating COL IV, CEA, interleukin-8 (IL-8), hepatocyte growth factor (HGF), cytokeratin-19 fragments (CYFRA 21-1), osteopontin (OPN), and migration inhibitory factor (MIF) compared to primary CRC (pCRC) patients. COL IV is elevated in mCRC patients compared to healthy individuals. Levels of COL IV, CEA, OPN, CYFRA 21-1, IL-8, and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) were dependent on the metastatic site. OPN, CEA, and HGF are very good at discriminating between mCRC patients and pCRC controls. COL IV is very good at distinguishing between mCRC patients and healthy controls. The combination of OPN + CEA is superior at detecting mCRC than CEA alone. High HGF and COL IV levels correlate to poor prognosis.
Conclusion
OPN, CEA, and HGF are potential biomarkers for mCRC. COL IV is a potential biomarker for CLM. The combination of OPN with CEA is superior to CEA alone in detecting mCRC. Levels of circulating proteins depend on metastatic localization, implying that a combination of markers is better than single markers in detecting mCRC disease. High levels of COL IV and HGF have potential prognostic value.
Keywords
Introduction
Colorectal cancer (CRC) is the second most common cause of cancer-related death worldwide, 1 mainly attributed to metastatic disease. 2 Metastases develop in about 50% of primary CRC (pCRC) patients, 3 and patients with colorectal liver metastases (CLM) constitute approximately 70–80% of all patients with metastatic CRC (mCRC).4,5
To achieve a long-lasting cure for mCRC patients’ surgery is key but is mainly performed on CLM patients with limited disease. Evidence is weak or lacking for surgical procedures in other metastatic locations.3,6,7 Approximately 15–25% of CLM patients are eligible for surgery3,8,9 giving them a 5-year overall survival (OS) of 30–50%.10–13 Treatment of advanced metastatic disease or otherwise inoperable patients is limited to therapy aiming to slow disease progression and minimize symptoms—so-called palliative treatment.3,6 Patient outcomes are thus highly dependent on early detection of metastases.
Diagnosis and monitoring of mCRC and resected pCRC patients depend on diagnostic imaging.3,6 Measurement of carcinoembryonic antigen (CEA) can be applied, but recommended use is limited to detecting local and distant recurrence.3,6,14 Circulating CEA is not elevated in all mCRC patients, with a reported sensitivity of 66.7% in detecting single metastases. 15 Circulating CEA is especially sensitive in detecting liver metastases (LM) and around 80% of CLM patients present with high levels.16–18 Elevated CEA is also present in 45% of pCRC patients15,19,20 as well as with other malignancies, conditions, and in smokers.21–23 The low sensitivity and specificity of CEA15,16,24 make it suboptimal as a single biomarker, highlighting the need for new biomarkers for early diagnosis of mCRC.
Collagen is a vital component of the tumor stroma 25 and type IV collagen (COL IV) is a potential biomarker for CLM.26–29 Increased COL IV expression is observed in the stroma of CLM26,30–32 and LM of different origins.30,33 Elevated levels of circulating COL IV (cCOL IV) in mCRC patients correlate to poor prognosis and hepatic tumor burden,26,27,29,34 and the combination of cCOL IV with CEA is better than either protein alone at detecting CLM. 27 Also, cCOL IV is elevated with other cancer types.30,33,35,36 We have previously shown that COL IV is expressed in liver and bone metastases of breast cancer patients and that metastatic breast cancer patients have elevated cCOL IV compared to primary breast cancer patients and healthy controls. 33 We also observed that cCOL IV was superior to conventional biomarker CA 15-3 at detecting metastatic breast cancer. 33
We hypothesized that cCOL IV is a marker of general metastatic disease and that combining COL IV with other biomarkers could improve the diagnosis of mCRC disease compared to a single biomarker. A combination of biomarkers that can detect mCRC at an early stage could potentially increase the number of CLM patients eligible for resection, lead to an earlier onset of therapy, and improve OS. Optimal biomarkers could be used to monitor recurrence after surgery and disease status under oncological therapy. Hence, a new low-cost, non-invasive, and optimal biomarker for mCRC detection could have a high clinical impact. The overarching aim of this study was to examine the biomarker potential of cCOL IV and 10 other cancer-related proteins in detecting mCRC. The first aim was to analyze cCOL IV in mCRC patients and controls and to subsequently study the levels of all 11 proteins in mCRC patients and controls. The second aim was to analyze the diagnostic values of all biomarkers and whether combining two biomarkers was more accurate in detecting mCRC than a single marker. The third aim was to compare the levels in patients with LM versus those with non-hepatic metastatic disease. Lastly, we aimed to investigate the potential prognostic value of all proteins in mCRC patients.
Materials and methods
Ethics statement
Informed consent was obtained from all patients and the Swedish Ethical Review Board approved this study (DNR 09-175M) in compliance with the Helsinki Declaration of 1975.
Study design
This study was divided into two cohorts. In Cohort I, plasma levels of COL IV were analyzed from mCRC patients receiving palliative chemotherapy, patients undergoing adjuvant therapy after pCRC surgery and healthy controls. In Cohort II, COL IV, CEA, TNF-related apoptosis-inducing ligand (TRAIL), interleukin-6 (IL-6), interleukin-8 (IL-8), hepatocyte growth factor (HGF), stem cell factor (SCF), cytokeratin-19 fragments (CYFRA21-1), osteopontin (OPN), fibroblast growth factor 2 (FGF2) and macrophage migration inhibitory factor (MIF) were analyzed in the plasma from mCRC patients receiving palliative chemotherapy and in preoperative plasma samples from pCRC patients. The proteins were chosen, except for COL IV, based on the availability of a predetermined multiplex assay with cancer-related tumor and stroma-derived proteins. In Cohort II, proteins significantly elevated with mCRC compared to controls were analyzed in combination with COL IV and each other.
Patients and plasma sample collection
For Cohort I, cCOL IV was measured in mCRC patients (n = 25) diagnosed as palliative between 2010 and 2012. For Cohort II, circulating levels of 11 proteins were measured in mCRC patients (n = 60) diagnosed as palliative between the years 2010 and 2019.
The inclusion criteria for these cohorts were: mCRC patients diagnosed as palliative and treated at Umeå University Hospital; patients participating in Uppsala-Umeå Comprehensive Cancer Consortium (U-CAN) project; available plasma samples stored in the Umeå U-CAN biobank. Exclusion criteria for this study were: no available plasma samples as stated above; patient diagnosed with another type of cancer; patient passed away < month after resection due to non-cancer-related disease; non-characterized metastatic location at the time of the sample. Plasma samples analyzed with Cohort I were excluded from Cohort II.
Controls for Cohort I were plasma samples from 118 healthy individuals collected between 1990 and 2007 within the Västerbotten intervention program 37 and plasma collected between 2010 and 2012 from 43 pCRC patients under adjuvant therapy. Controls for Cohort II were preoperative plasma samples from 40 patients treated at Umeå University Hospital for pCRC between 2011 and 2015. These plasma samples were collected before the resection of the primary tumor. Staging of metastatic disease in patients at the time of blood sampling was established by computed tomography and/or magnetic resonance imaging. Clinical data were collected from patients’ charts. Blood samples were drawn by peripheral venous puncture and plasma was aliquoted and stored at −80 °C until analysis.
See Supplementary Figure 1 and Supplementary Table 1 and 2 for a flowchart of included and excluded patients and patient characteristics.
Analysis of circulating stromal proteins in patients with metastatic colorectal cancer
Circulating COL IV in mCRC-patients and controls were measured with a Collagen IV EIA kit (Sensitivity 0.1 ng/ml, Argutus Medical, Dublin, Ireland), a sandwich-ELISA kit with two monoclonal antibodies directed against the 7S and the collagenous domain of COL IV. 38 Samples were run in duplicates and analyzed according to the manufacturer's protocol. The difference between duplicate samples was not allowed to be more than 12.5%.
Circulating TRAIL, IL-6, IL-8, HGF, SCF, CYFRA 21-1, OPN, MIF, CEA, and FGF-2 in mCRC-patients and controls were measured with Millplex MAP Human Circulating Cancer Biomarker Magnetic Bead Panel 1 (HCCBP1MAG-58 K, EMD Millipore Corporation, Billerica, MA, USA), The manufacturers’ protocol was followed and the samples were analyzed in duplicates where the difference between samples was not allowed to be more than 12.5%.
Statistical analysis
The Mann–Whitney and Kruskal–Wallis tests were used to compare circulating levels of analytes between mCRC patients and controls and between patients with LM and non-hepatic metastatic disease. The difference in the presence of primary tumors between patients with LM and patients with non-hepatic metastases was analyzed with the chi-square test. Differences in gender, location of the primary tumor (colon/rectum), and age between patients and controls were analyzed with a chi-square test, T-test and Mann–Whitney test. The correlation between age and cCOL IV was analyzed with multiple linear regression. Values that were low and out of range (<OOR) were replaced with the value of the lowest detected value divided by 2. High values and values out-of-range (>OOR) were replaced with the highest detected value. Extrapolated values were treated as normal values. Kaplan–Meier survival curves were used to analyze OS for mCRC patients and differences in survival were calculated by the log-rank test. The patient cohorts were dichotomized based on the median circulating protein value. Survival data were collected from the Swedish death registry. The study endpoint was death from any cause. Receiver operating characteristic (ROC) analysis was applied to calculate the area under the curve (AUC) to establish the potential of analytes to discriminate between patients and controls. Logistic regression was used to correct for age and ROC analysis was repeated. Combined ROC curves were constructed and the difference between single and combined ROC was analyzed using the ROCCOMP function in STATA. ROCCOMP reports summary statistics for each curve and provides a test for the equality of the area under the curves utilizing an algorithm. GraphPad Prism version 9 (GraphPad Software, San Diego, CA, USA), MATLAB version 9.11.0.1769968 (R2021b) (The Mathworks, Inc. Natick, MA, USA) and STATA version 15 (STATA Corp, TX, USA) was used. A P-value < 0.05 was considered statistically significant.
Results
Metastatic CRC patients have high levels of circulating COL IV, OPN, CYFRA 21-1, IL-8, HGF, CEA, MIF, and low levels of TRAIL
Analysis of Cohort I showed that cCOL IV was significantly elevated in mCRC patients (n = 25) compared with healthy controls (n = 118) (P < 0.0001) (see Table 1 and Supplementary Figure 2(a)). There was no significant difference in cCOL IV between mCRC patients and controls under adjuvant therapy (n = 43) (P = 0.0814) possibly reflecting ongoing wound-healing in pCRC patients since samples are withdrawn within a relatively short time after surgery. 39 There was no difference in gender between patients and either of the control groups or in age between mCRC patients and controls under adjuvant therapy. The healthy controls were significantly younger than the mCRC patients (P < 0.0001). There was a significant correlation between age and cCOL IV in healthy controls where cCOL IV increased with age (P < 0.0001). The difference in cCOL IV between mCRC patients and healthy controls remained after adjusting for age (P < 0.0001).
Circulating levels of COL IV in cohort I and cohort II.
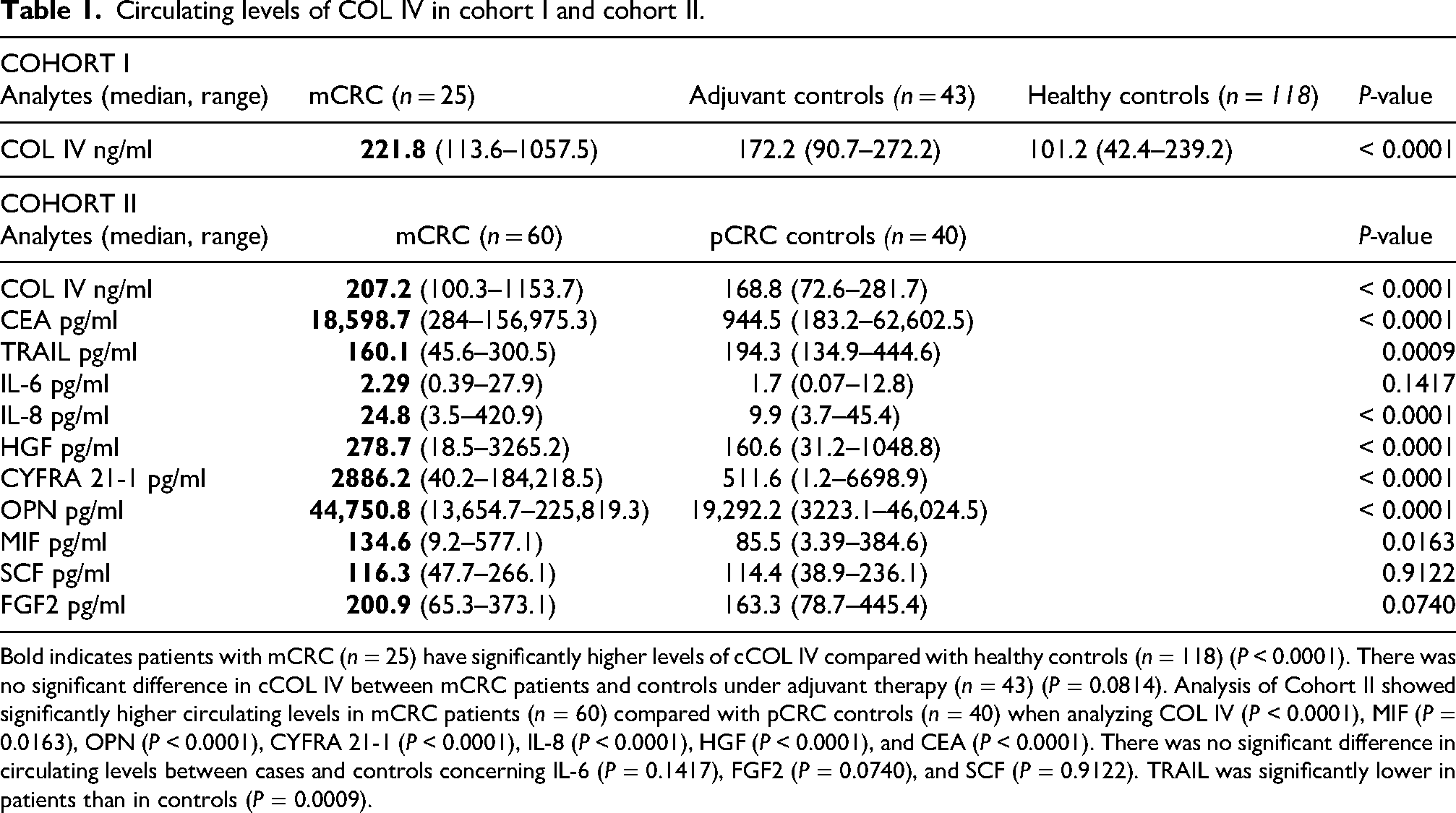
Bold indicates patients with mCRC (n = 25) have significantly higher levels of cCOL IV compared with healthy controls (n = 118) (P < 0.0001). There was no significant difference in cCOL IV between mCRC patients and controls under adjuvant therapy (n = 43) (P = 0.0814). Analysis of Cohort II showed significantly higher circulating levels in mCRC patients (n = 60) compared with pCRC controls (n = 40) when analyzing COL IV (P < 0.0001), MIF (P = 0.0163), OPN (P < 0.0001), CYFRA 21-1 (P < 0.0001), IL-8 (P < 0.0001), HGF (P < 0.0001), and CEA (P < 0.0001). There was no significant difference in circulating levels between cases and controls concerning IL-6 (P = 0.1417), FGF2 (P = 0.0740), and SCF (P = 0.9122). TRAIL was significantly lower in patients than in controls (P = 0.0009).
Analysis of Cohort II showed significantly higher circulating levels in mCRC patients (n = 60) compared with pCRC controls (n = 40) when analyzing COL IV (P < 0.0001), MIF (P = 0.0163), OPN (P < 0.0001), CYFRA 21-1 (P < 0.0001), IL-8 (P < 0.0001), HGF (P < 0.0001), and CEA (P < 0.0001) (see Table 1 and Supplementary Figure 2). The results of CEA align with previous research and the use of CEA in health care as an indicator of metastatic disease. However, only 67% of CLM patients had a CEA value above clinical cut-off (5 ng/ml) indicating that measuring CEA is not enough for detecting metastases in the clinic. TRAIL was significantly lower in patients than in controls (P = 0.0009) which is coherent with the role of TRAIL as an inducer of apoptosis in tumor cells and a suppressor of metastasis.40–42 There was no significant difference in circulating levels between cases and controls concerning IL-6 (P = 0.1417), FGF2 (P = 0.0740), and SCF (P = 0.9122). There was no difference between mCRC patients and pCRC controls in terms of age (P = 0.1191), gender (P = 0.1416), or location of the primary tumor (P = 0.8671). The percentage of values out of range and extrapolated within the patient and control group are presented in Supplementary Table 3.
Circulating COL IV, CEA, OPN, and HGF distinguish mCRC patients from controls
Cohort I: cCOL IV was very good at discriminating mCRC patients from healthy controls (AUC = 0.90, P < 0.0001) (see Table 2 and Figure 1(a)). The discriminative potential of cCOL IV remained after adjusting for age (AUC = 0.76, P < 0.0001). Circulating COL IV did not discriminate patients from adjuvant controls (AUC = 0.63, P = 0.08) (see Supplementary Figure 3(a)), which might reflect ongoing wound healing in adjuvant controls. 39
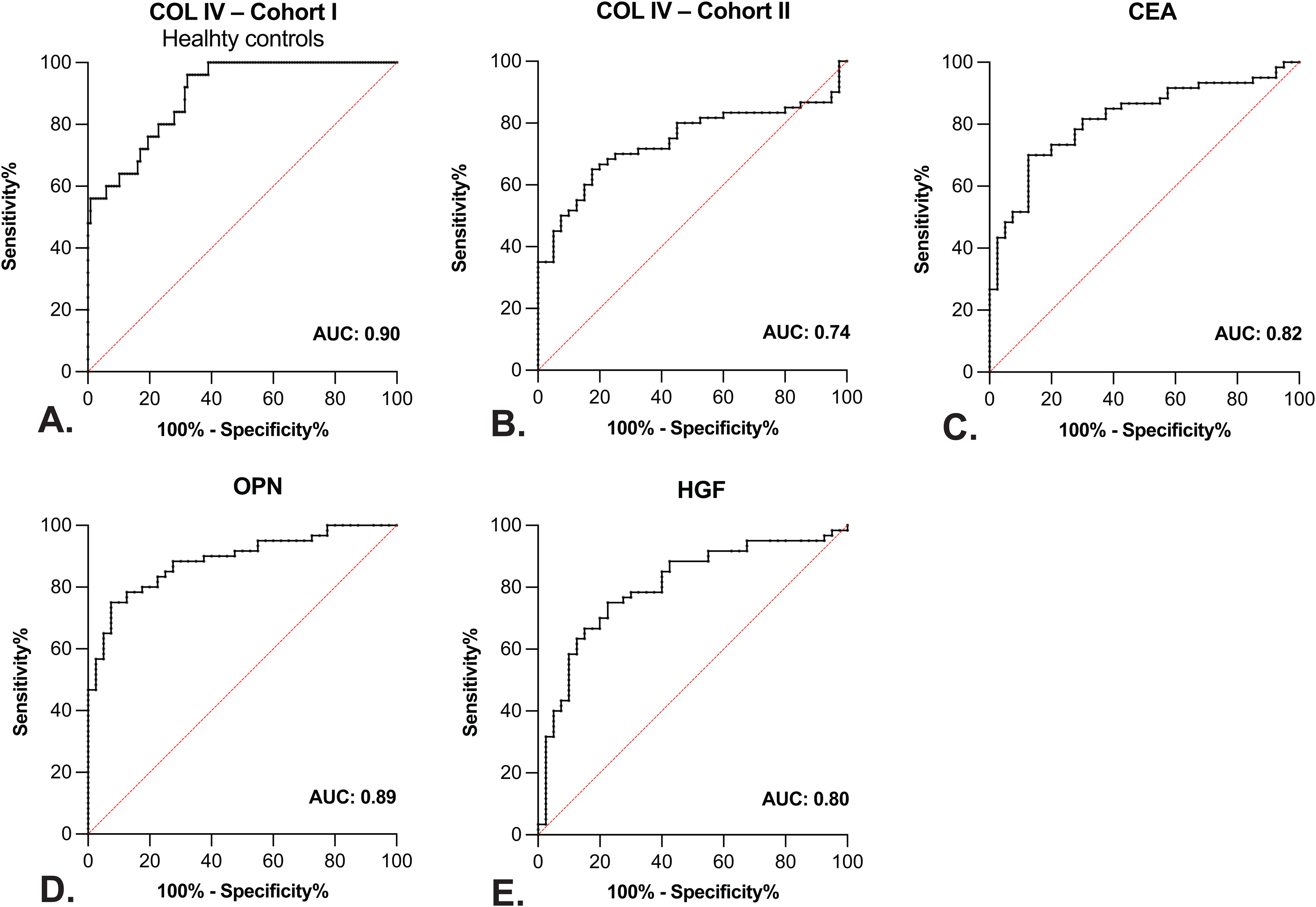
ROC curve analysis of circulating type IV collagen (COL IV), carcinoembryonic antigen (CEA), osteopontin (OPN), and hepatocyte growth factor (HGF). (a) In Cohort I, the area under the curve (AUC) of 0.90 indicates that COL IV is very good at discriminating between patients with metastatic colorectal cancer (mCRC) and healthy controls (P < 0.0001). (b) to (e) In Cohort II, (b) COL IV was good at discriminating between mCRC patients and pCRC controls (AUC = 0.74, P < 0.0001). (c) CEA (AUC = 0.82, P < 0.0001), (d) OPN (AUC = 0.89, P < 0.0001), and (e) HGF (AUC =0.80, P < 0.0001) were very good at discriminating patients from controls.
Area under the curve (AUC) for circulating COL IV, CEA, OPN, HGF, IL-8, CYFRA 21-1, MIF, TRAIL, and combinations of COL IV, CEA, OPN, and HGF.
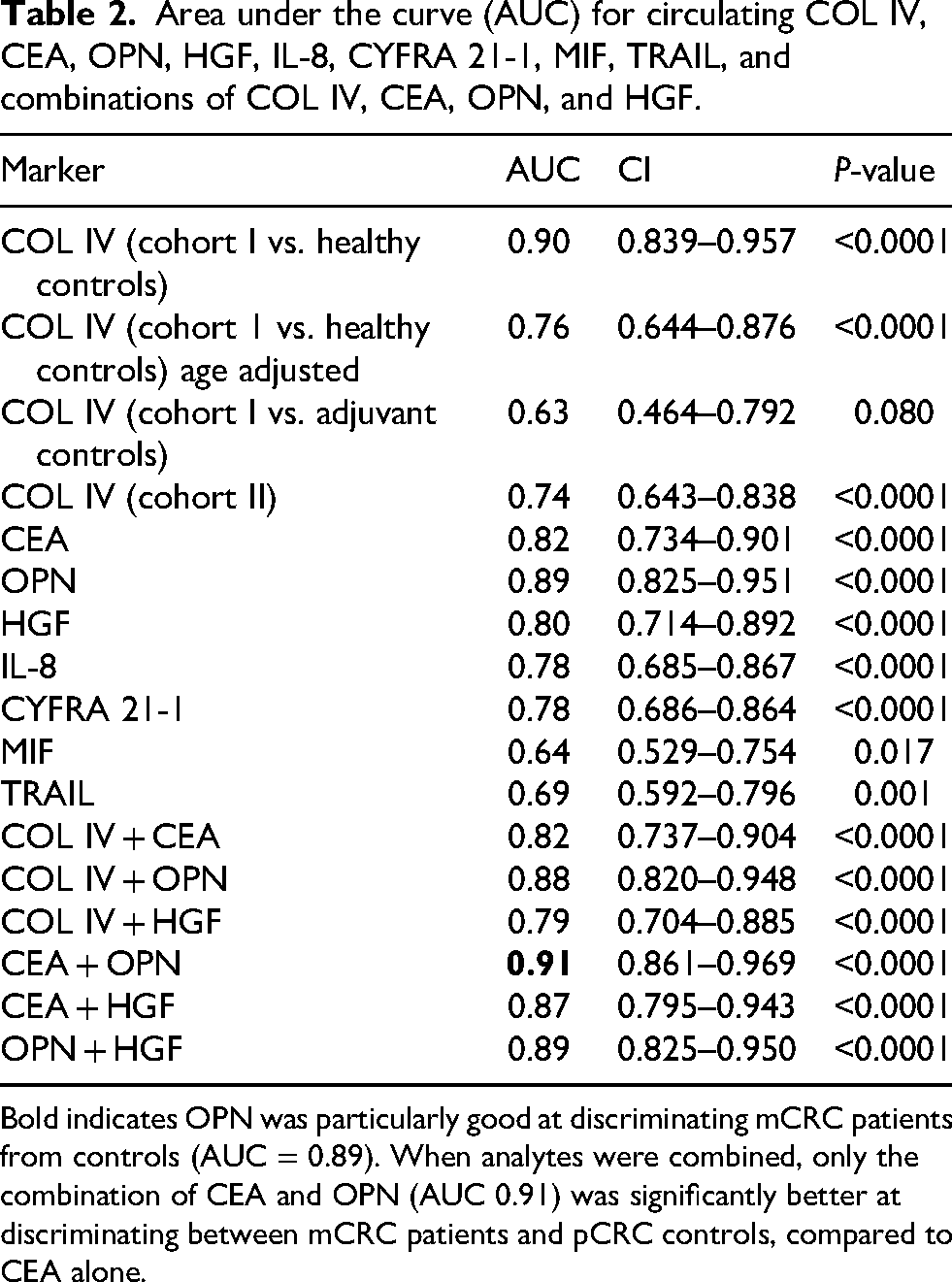
Bold indicates OPN was particularly good at discriminating mCRC patients from controls (AUC = 0.89). When analytes were combined, only the combination of CEA and OPN (AUC 0.91) was significantly better at discriminating between mCRC patients and pCRC controls, compared to CEA alone.
Cohort II: circulating CEA (AUC = 0.82, P < 0.0001), OPN (AUC = 0.89, P < 0.0001), and HGF (AUC = 0.80, P < 0.0001), were very good at discriminating between mCRC patients and pCRC controls (see Table 2, Figure 1(c) to (e)). OPN was particularly good at discriminating mCRC patients from controls, aligning with previous studies.43,44 OPN can promote metastatic progression43,44 and high plasma OPN after curative CRC surgery correlates to metastatic development and lower disease-free survival. 45 HGF has a hepato-regenerative effect on liver injuries 46 and high circulating levels in CRC patients have been linked to metastatic disease, poor prognosis, and resistance to targeted treatment.47–49 Circulating COL IV (AUC = 0.74, P < 0.0001), IL-8 (AUC =0.78, P < 0.0001), and CYFRA 21-1 (AUC =0.78, P < 0.0001) were good at discriminating between mCRC patients and pCRC controls (see Table 2, Figure 1(b), and Supplementary Figure 3(b) and (c)). MIF (AUC = 0.64, P = 0.017) and TRAIL (AUC =0.69, P = 0.001) could discriminate between cases and controls at a satisfactory level (see Table 2, Supplementary Figure 3(d) and 3(e)). The ability of cCOL IV to distinguish patients from controls varied with the cohorts, probably due to cCOL IV being better at distinguishing patients from healthy controls than primary CRC patients.
Combinations of COL IV, CEA, HGF, and OPN (all Cohort II) were analyzed based on AUC > 0.8; see Table 2 and Figure 1. The AUC values were not significantly increased by combining analytes, except for the combination of OPN and CEA compared to CEA alone (see Table 2 and Figure 2). Combining COL IV with CEA did not improve the discriminative power contrasting with a previous observation. 27 This could be explained by the already relatively high AUC for single biomarkers in this study, and in the previous study, the discriminatory power of COL IV was analyzed in CLM patients and compared with healthy controls. Here, COL IV was better at discriminating mCRC patients from healthy controls than pCRC controls. This study included patients with non-hepatic metastases while the previous study focused on CLM patients, and as discussed below, LM seems to be associated with higher COL IV levels.
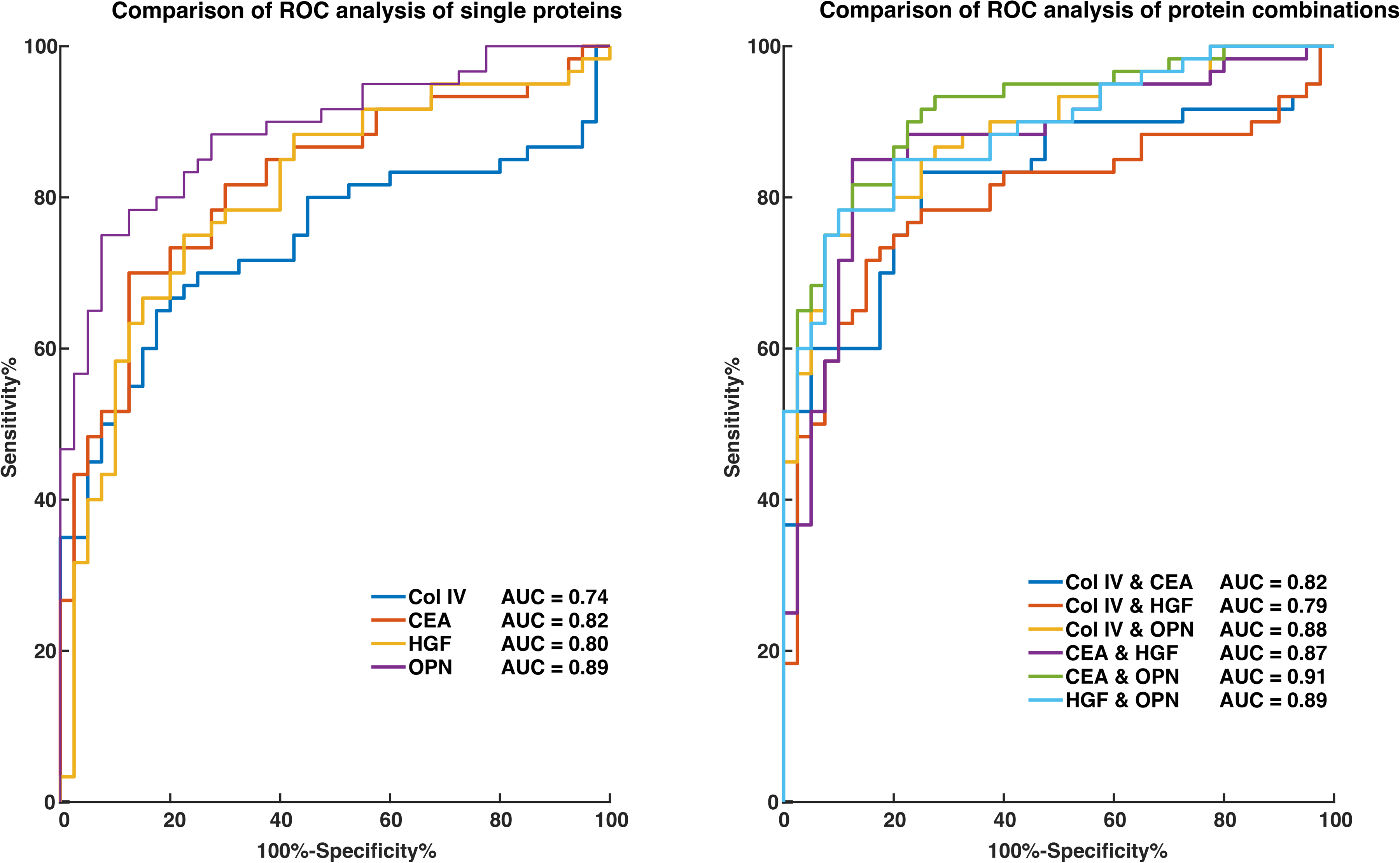
ROC curves with type IV collagen (COL IV), carcinoembryonic antigen (CEA), hepatocyte growth factor (HGF), osteopontin (OPN) and combinations of mentioned proteins for discriminating between metastatic colorectal cancer patients and controls with primary colorectal cancer. Single ROC analysis shows that OPN is superior to other proteins at discriminating between cases and controls. A study of combined ROC curves showed that the combination of CEA and OPN is significantly superior to CEA alone (P = 0.017) but not to OPN alone.
Circulating levels of COL IV, CEA, OPN, CYFRA 21-1, IL-8 and TRAIL correlate with metastatic site
There were significantly higher levels of cCOL IV in CLM patients compared to patients with non-hepatic metastases (Cohort I: P = 0.0004, Cohort II: P = 0.0012) and compared to healthy controls (P < 0.0001) and pCRC controls (Cohort II: P < 0.0001) (see Table 3 and Supplementary Figure 4(a) and (b)). Patients with LM more often had their primary tumor present at the time of blood sample (see Supplementary Table 4), which could explain the observed elevations in cCOL IV. However, previous studies report contradicting results regarding whether pCRC causes elevations in cCOL IV.26,29,34 Moreover, earlier reports on high cCOL IV with CLM indicate that LM is the likely source of high levels observed in this study, even though the primary tumor might contribute.
The difference in circulating analytes between patients with liver metastases and those with non-hepatic metastases.
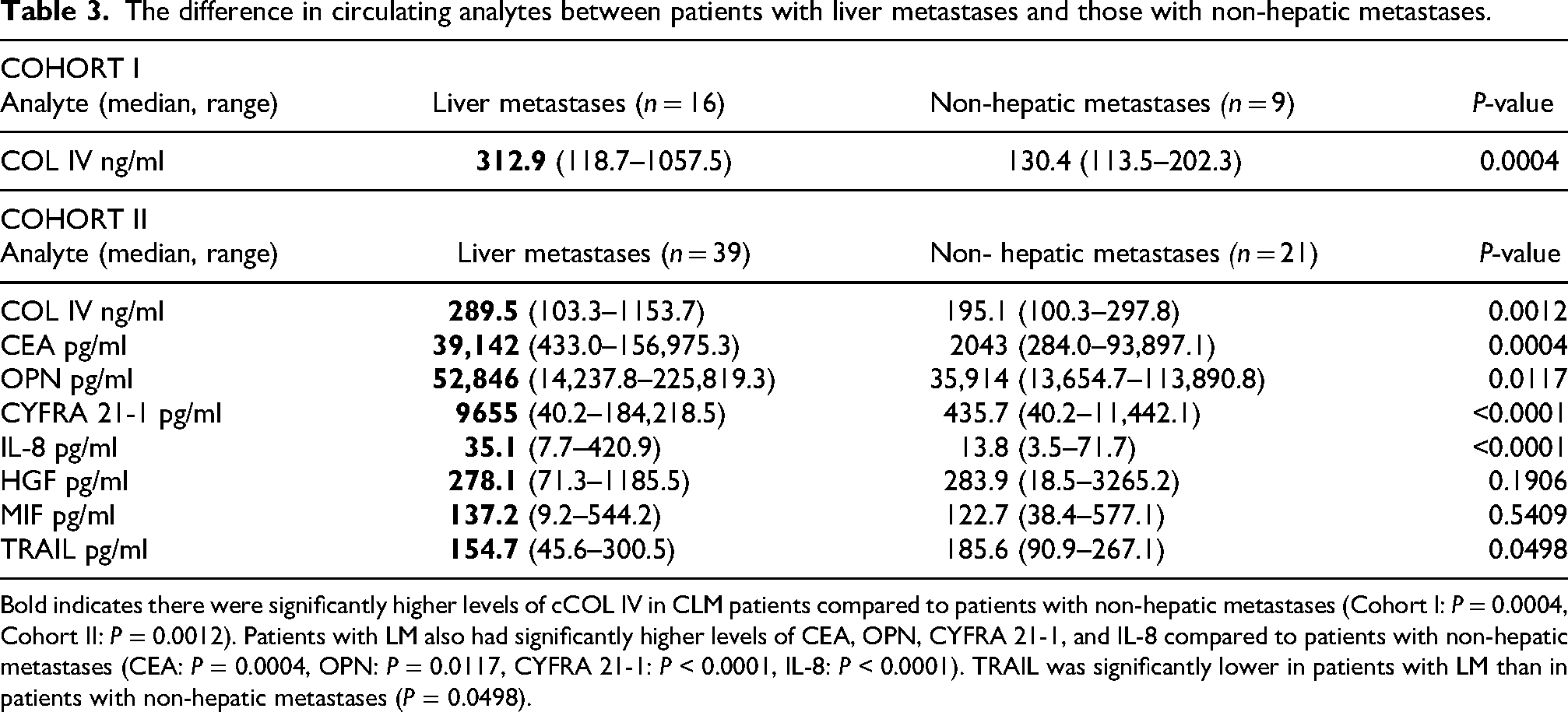
Bold indicates there were significantly higher levels of cCOL IV in CLM patients compared to patients with non-hepatic metastases (Cohort I: P = 0.0004, Cohort II: P = 0.0012). Patients with LM also had significantly higher levels of CEA, OPN, CYFRA 21-1, and IL-8 compared to patients with non-hepatic metastases (CEA: P = 0.0004, OPN: P = 0.0117, CYFRA 21-1: P < 0.0001, IL-8: P < 0.0001). TRAIL was significantly lower in patients with LM than in patients with non-hepatic metastases (P = 0.0498).
Patients with LM also had significantly higher levels of CEA, OPN, CYFRA 21-1, and IL-8 compared to patients with non-hepatic metastases (CEA: P = 0.0004, OPN: P = 0.0117, CYFRA 21-1: P < 0.0001, IL-8: P < 0.0001) and pCRC controls (CEA: P < 0.0001, OPN: P < 0.0001, CYFRA 21-1: P < 0.0001, IL-8: P < 0.0001). TRAIL was significantly lower in patients with LM than in patients with non-hepatic metastases (P = 0.0498) and pCRC controls (P = 0.0001) (see Table 3 and Supplementary Figure 4). CEA and OPN were the only proteins that were significantly elevated in patients with non-hepatic metastases compared to pCRC controls (see Supplementary Figure 4(c) and (d)), which could be related to their high AUC values. HGF had the third-highest AUC and showed no difference regarding the metastatic site. Our results imply that OPN, CEA, and HGF, are increased in mCRC patients regardless of metastatic site, while high levels of COL IV, CYFRA 21-1, MIF, and IL-8 are more dependent on LM.
High levels of COL IV and HGF correlate to poor prognosis
A significant correlation between high cCOL IV levels and shorter survival time was observed in Cohort I (n = 25, P = 0.011) (see Supplementary Figure 5(a)). The median survival time in the group with low cCOL IV (<221.8 ng/ml) was 21 months compared to 9.5 months in the group with high cCOL IV (>221.8 ng/ml). No prognostic value of cCOL IV was observed for Cohort II (P = 0.09, data not shown). The discrepancy between cohorts might be explainable by a higher tumor burden in Cohort I since two minor studies on cCOL IV and tumor burden indicate a correlation between high cCOL IV and tumor burden.26,34 Here we did not establish patients’ tumor burden, which could impact cCOL IV and prognostic and diagnostic evaluation. We also did not have the exact details of the oncological treatment of the mCRC patients, which can affect the prognostic results.
The remaining proteins showed no prognostic value except for HGF, where high levels (> 278.7 pg/ml) correlated to poor prognosis (P = 0.02) (see Supplementary Figure 5(b)).
Discussion & conclusion
Colorectal cancer is the second most common cause of cancer-related death worldwide, 1 mainly attributed to metastatic disease. 2 Patients with mCRC, predominantly CLM, can be cured with surgery but early diagnosis is essential which today is dependent on imaging. There is no optimal biomarker for mCRC and a circulating, low-cost, and sensitive biomarker for early detection of mCRC could potentially increase the number of CLM patients eligible for resection, lead to earlier onset of therapy, and improve OS. Optimal biomarkers could also be used to monitor recurrence after surgery and disease status under oncological therapy and ideally be used as predictive biomarkers to personalize treatment. It could also potentially replace, at least partly, the expensive imaging modalities.
This paper aimed to test the hypothesis that cCOL IV, in combination with other cancer-related proteins, could increase the potential of discriminating between patients with metastases and those without. The study is limited by the small amount of mCRC patients and lack of information regarding tumor burden and oncological treatment; nevertheless, the reality is that palliative metastatic patients are a heterogeneous patient group. Our results indicate that OPN and HGF are potential biomarkers for mCRC and that cCOL IV is a potential biomarker for CLM. The combination of OPN with CEA is superior to CEA alone in detecting mCRC disease. We also show that biomarkers for metastatic disease depend on metastatic localization, where circulating levels of COL IV, CEA, OPN, CYFRA 21-1, IL-8, and TRAIL in mCRC patients, depended on the presence of LM. These results imply that a combination of biomarkers more accurately captures the general mCRC disease in patients compared to a single biomarker.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155251329590 - Supplemental material for Type IV collagen, carcinoembryonic antigen, osteopontin, and hepatocyte growth factor as biomarkers for liver metastatic colorectal cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155251329590 for Type IV collagen, carcinoembryonic antigen, osteopontin, and hepatocyte growth factor as biomarkers for liver metastatic colorectal cancer by Moa Lindgren, Ingrid Ljuslinder, Pär Jonsson and Hanna Nyström in The International Journal of Biological Markers
Footnotes
Acknowledgments
We thank Christina Lundin for her skilled technical assistance and Björn Tavelin for statistical support.
Grant support
This study was supported by grants from Cancerforskningsfonden Norrland, the Wallenberg foundations/Knut and Alice Wallenberg's stiftelse, Västerbotten County Council, Cancerfonden, Cancerforskningsfonden Norrland, Umeå University, and Stiftelsen JC Kempe Memorial Foundation Scholarship Fund.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Swedish Ethical Review Board (DNR 09-175 M). Informed consent was obtained from all participants prior to their inclusion in the study. All data were collected anonymously and securely, ensuring participant confidentiality.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data can be shared upon reasonable request to the (Hanna Nyström) author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
