Abstract
Background
This study aimed to establish a nomogram to distinguish advanced- and early-stage lung cancer based on coagulation-related biomarkers and liver-related biomarkers.
Methods
A total of 306 patients with lung cancer and 172 patients with benign pulmonary disease were enrolled. Subgroup analyses based on histologic type, clinical stage, and neoplasm metastasis status were carried out and multivariable logistic regression analysis was applied. Furthermore, a nomogram model was developed and validated with bootstrap resampling.
Results
The concentrations of complement C1q, fibrinogen, and D-dimers, fibronectin, inorganic phosphate, and prealbumin were significantly changed in lung cancer patients compared to benign pulmonary disease patients. Multiple regression analysis based on subgroup analysis of clinical stage showed that compared with early-stage lung cancer, female (P < 0.001), asymptomatic admission (P = 0.001), and total bile acids (P = 0.011) were negatively related to advanced lung cancer, while C1q (P = 0.038), fibrinogen (P < 0.001), and D-dimers (P = 0.001) were positively related. A nomogram model based on gender, symptom, and the levels of total bile acids, C1q, fibrinogen, and D-dimers was constructed for distinguishing advanced lung cancer and early-stage lung cancer, with an area under the receiver operating characteristic curve of 0.919. The calibration curve for this nomogram revealed good predictive accuracy (P−Hosmer−Lemeshow = 0.697) between the predicted probability and the actual probability.
Conclusions
We developed a nomogram based on gender, symptom, and the levels of fibrinogen, D-dimers, total bile acids, and C1q that can individually distinguish early- and advanced-stage lung cancer.
Introduction
Lung cancer (LC), which remains the leading cause of cancer incidence and mortality worldwide, 1 usually shows similar imaging features to benign pulmonary disease (BPD), and clinical symptoms appear in the late stages. 2 According to several guidelines,3,4 individualized management of the pulmonary nodule is based on the probability of malignancy, which is estimated based on clinical experience or a validated risk prediction calculator. Moreover, the treatment strategies are completely different between a malignant nodule (early- vs. advanced-stage LC) and a benign nodule. 5 Therefore, identification and staging of LC among patients with pulmonary nodules become a great challenge in early screening.
Hypercoagulability is often associated with tumor initiation and progression, via promoting cancer cell proliferation, invasion, and immune escape.6,7 Tumor cells can activate systemic coagulation through multiple pathways to induce hemostatic and fibrinolytic abnormalities, which in turn promote cancer angiogenesis and metastasis. 8 Previous retrospective research has shown that elevated platelet count (PLT), fibrinogen (FIB), and D-dimers (DD) can predict poor prognosis in several cancer types,9–11 including LC. 12 Complement C1q was first identified in 1961 and described as 11 s thermolabile serum protein. 13 It participates in immune hemolysis and activates the classical pathway of the complement as the first subcomponent of the C1 complex. 14 Evidence from biology experiments showed that C1q might act as a cancer-promoting factor in the tumor microenvironment. 15 In addition, a study published in 2022 showed that the elevated level of C1q was associated with poor prognosis of non-small cell lung cancer (NSCLC) based on bioinformatic databases. 16 Bile acids (BA) are products of cholesterol metabolism that contribute on immune responses, glucose regulation, and energy homeostasis. 17 Research has found that BA may exert anti-cancer effects by accumulating natural killer T cells that fight cancer cells. 18 In contrast, BA might also serve as signaling molecules that involve in multiple cancer-related signaling pathways, such as promoting NSCLC cell migration and invasion through Janus kinase/signal transducer and activator of transcription (JAK2/STAT3) signaling pathway. 19 The abnormalities of BA in cancers have been found by several clinical studies,20–22 while studies focused on BA and LC were limited.19,23,24 As mentioned above, although the application of coagulation-related biomarkers and liver-related biomarkers in LC has been explored, study on those biomarkers as potential LC staging predictors in patients with pulmonary nodules is limited in the real world.
Nomograms, as a simple statistical predictive model, are frequently utilized for various disease risk prediction and prognosis evaluation, especially for cancers.25,26 Nowadays, several nomogram models for discriminating pulmonary benign nodules and malignancy have been established.27–29 However, blood hypercoagulability plays an important role in the development of LC, while few nomogram models based on pretreatment serum coagulation-related biomarker features have been developed for accurately distinguishing between early and advanced-stage LC yet to date. Furthermore, there are few studies exploring the application of C1q and total bile acids (TBA) in the diagnosis and clinical staging of LC. In this study, we comprehensively assessed the differences in the expression of preoperative circulating coagulation-related biomarkers, liver-related biomarkers between LC and BPD patients. Then, we established a nomogram model to distinguish advanced- and early-stage LC patients.
Methods
Participants characteristic
In this retrospective study, 478 participants were enrolled from March 2022 to September 2022 at Renmin Hospital of Wuhan University, including 306 patients with LC and 172 patients with BPD. The following were inclusion criteria: (a) patients who were first admitted to hospital with a space-occupying lung lesion without radiotherapy, chemotherapy, or surgery before admission; (b) patients without other malignancies; and (c) patients that were histologically or cytologically confirmed as LC or BPD. The exclusion criteria were: (a) the patient had metastatic or secondary LC; (b) patients with incomplete clinical laboratory data; and (c) patients with acute inflammatory or auto-immune disease. This study was approved by the Ethics Committee of the Renmin Hospital of Wuhan University.
Data collection
The demographic and clinical information was collected from electronic medical records, including age, gender, symptom, smoking status, and histologic type and tumor metastasis status of LC patients. In addition, the tumor node metastasis (TNM) staging system were applied for confirming tumor stage based on the eighth edition guideline. 30 A total of 14 biomarkers were detected in this study, involving indictors relating to coagulation and liver function. All samples of the participants were obtained through intravenous collection on the first day of hospitalization. Six coagulation-related biomarkers were tested by Sysmex XE-2100 automatic blood cell analyzer and CA7000 automatic coagulation analyzer (Sysmex), including PLT, thrombin time (TT), FIB, FIB degradation products (FDP), DD, and antithrombin-Ⅲ (AT-Ⅲ). Siemens ADVIA 2400 biochemical analyzer was applied for detecting liver biomarkers, including alanine aminotransferase, aspartate aminotransferase (AST), TBA, prealbumin (PA), fibronectin (Fn), complement C1q, calcium, and inorganic phosphate (IP).
Statistical analysis
All continuous variables were tested for normality by the Kolmogorov–Smirnov test method; normally distributed continuous variables were presented as mean ± standard, while the non-normal continuous variables were presented as medians (interquartile ranges), and categorical variables were presented as frequencies (percentage). The T-test was applied to compare the difference of the normally and skewed distributed continuous variables in two groups; otherwise, Mann–Whitney U test was conducted. The comparison of categorical variables in two groups were tested by Chi-square test. Spearman analysis was performed to test the correlation between all indexes. Additionally, multivariable logistic regression analysis was applied to identify the risk factors for adenocarcinoma, advanced-stage LC, and neoplasm metastasis in the subgroup analyses.
Furthermore, a nomogram model was built based on the multivariable logistic regression analysis for distinguishing both advanced- and early-stage LC. The performance of the nomogram model was evaluated by receiver operating characteristic (ROC) curves and the area under the ROC curve (AUC), calibration plot, Hosmer–Lemeshow test, and decision curve analyses (DCA). In addition, an internal validation was performed by 1000 bootstrap resamples analysis.
The IBM SPSS Statistics 26.0 (IBM Corporation, Armonk, NY, USA) was applied for the statistical analyses. R studio v4.1.1 was used to construct the nomogram model. All P values were two sided and P < 0.05 was considered to be statistically significant.
Results
Clinical characteristic of participants
The basic demographics and clinical characteristics of study participants are shown in Supplementary Table 1. Briefly, a total of 306 LC patients and 172 BPD patients were enrolled. No significant differences were found between the LC group and BPD group regarding gender (50.3% male in LC, 59.3% male in BPD, P−value = 0.059), and smoking status (65.7% smoker in LC, 71.5% smoker in BPD, P−value = 0.431). We found that patients with LC were more likely to be older (59.10 ± 11.40 years for LC, 56.90 ± 11.41 years for BPD, P−value = 0.044),
Of the LC group, 57.8% of patients had respiratory symptoms on admission, such as cough, expectoration, and chest tightness, etc.; 72.9% were adenocarcinoma; 13.1% were squamous cell carcinoma; and 6.8% were small cell lung cancer (SCLC); and the remaining were other NSCLC. According to the TNM stage of LC, 60.5% patients were categorized as early-stage LC (57.2% for stage I and 3.3% for stage II), and 30.1% were categorized as advanced-stage LC (9.8% for stage III and 20.3% for stage IV). In addition, tumor metastasis occurred in 23.5% of LC patients.
Alteration of coagulation-related biomarkers and C1q in LC patients
The comparison of several biomarkers between the LC and BPD groups is shown in Table 1. The results showed that the concentrations of C1q, FIB, DD, Fn, and IP were significantly positively associated with LC patients compared to BPD patients. Additionally, the serum level of PA was significantly negatively associated in LC patients (Figure 1(a)).
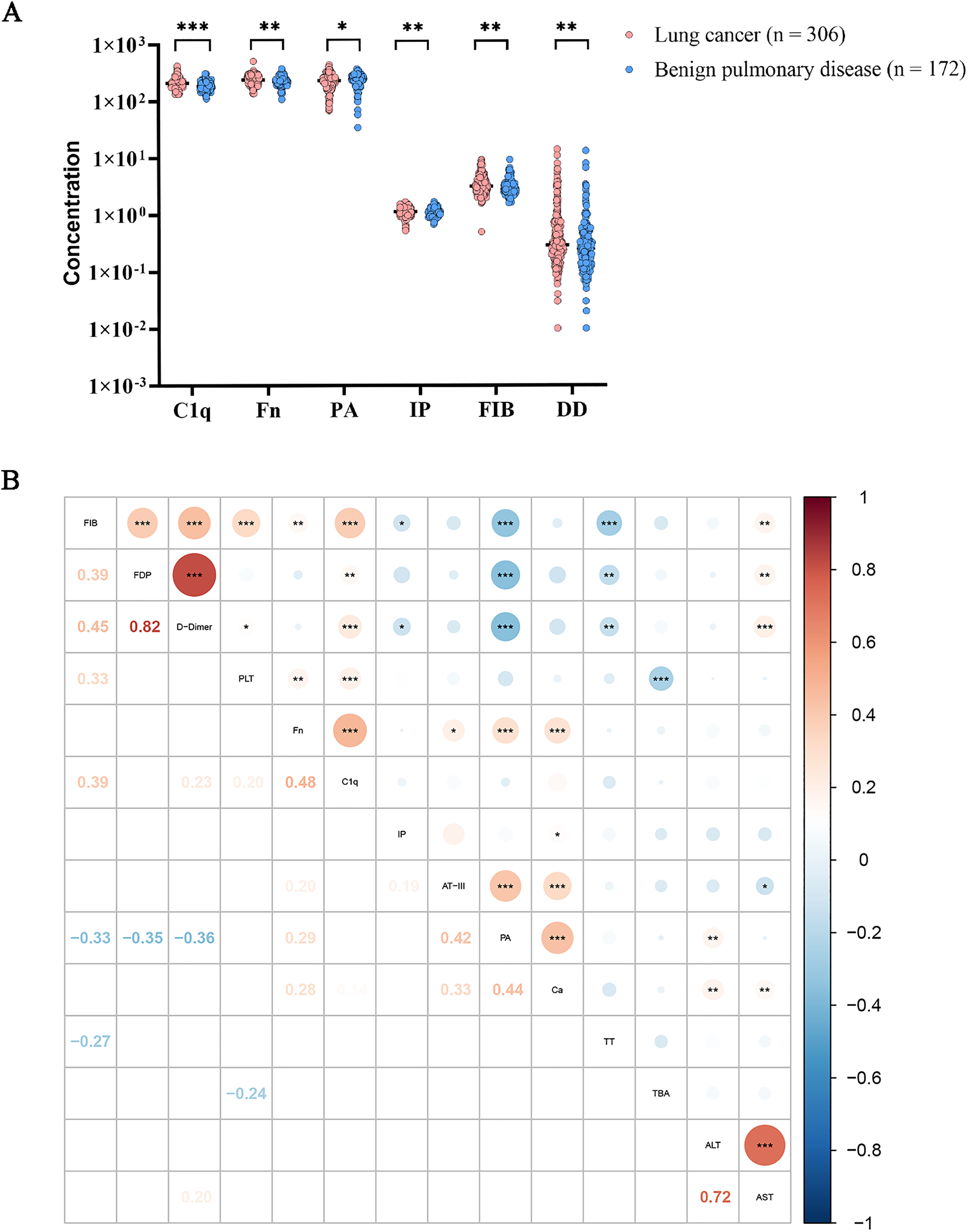
The expression profile of coagulation- and liver-related biomarkers in the LC and BPD groups. *: P < 0.05, **: P < 0.01, ***: P < 0.001. (a) The indicators differed among LC and BPD groups. (b) The correlation expression profile between coagulation- and liver-related biomarkers in LC group.
The different biomarkers between the LC and BPD groups.
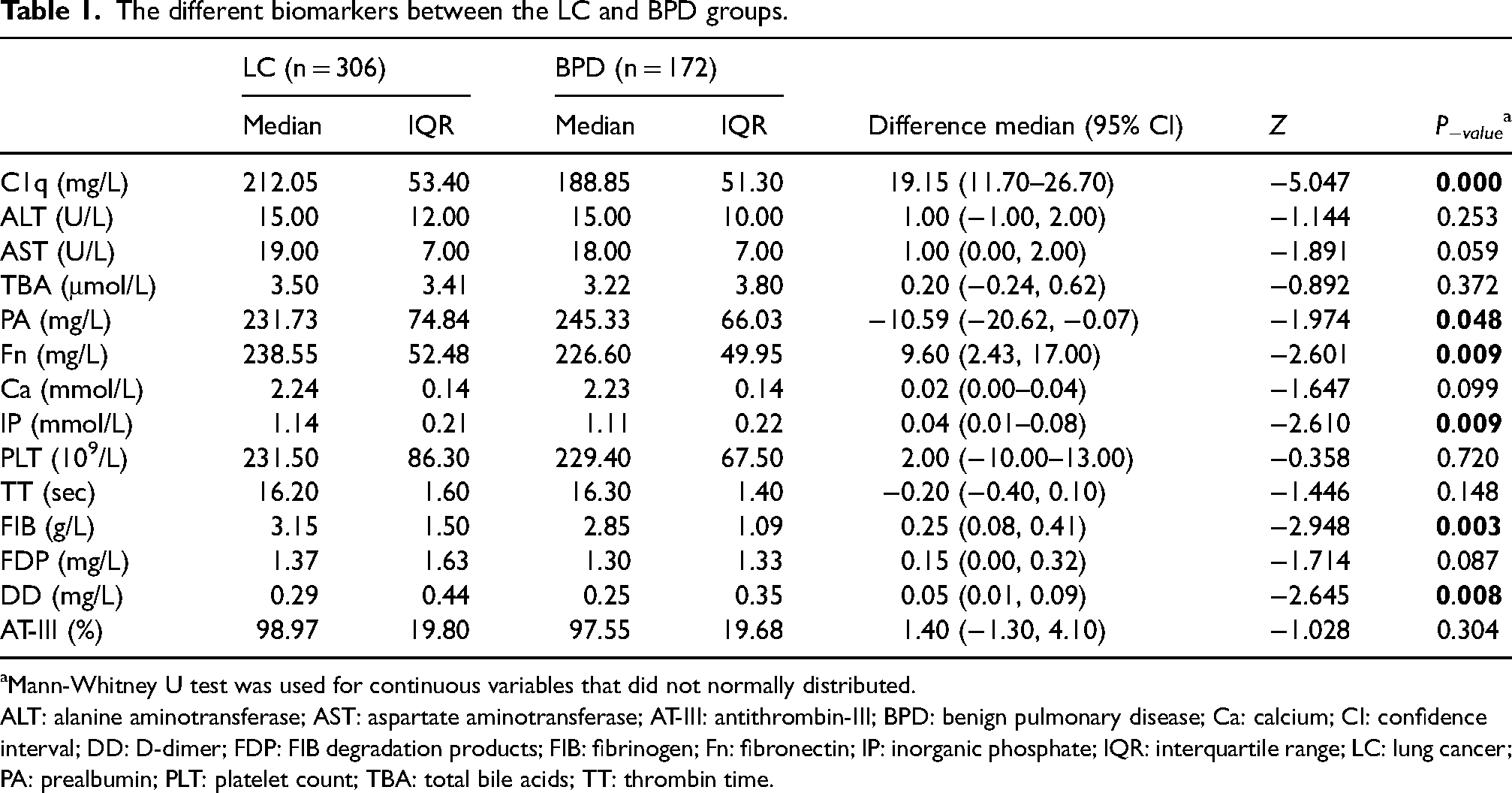
Mann-Whitney U test was used for continuous variables that did not normally distributed.
ALT: alanine aminotransferase; AST: aspartate aminotransferase; AT-III: antithrombin-III; BPD: benign pulmonary disease; Ca: calcium; CI: confidence interval; DD: D-dimer; FDP: FIB degradation products; FIB: fibrinogen; Fn: fibronectin; IP: inorganic phosphate; IQR: interquartile range; LC: lung cancer; PA: prealbumin; PLT: platelet count; TBA: total bile acids; TT: thrombin time.
Furthermore, the correlation expression profile between coagulation- and liver-related biomarkers in the LC group is shown in Figure 1(b). The analyses revealed that coagulation-related biomarkers were almost related to several liver-related biomarkers. Among these, the level of C1q was positively related to PLT (r = 0.20; P < 0.001), FIB (r = 0.39; P < 0.001), DD (r = 0.23; P < 0.001), and FDP (r = 0.15; P < 0.01). The concentration of PA was negatively related to FIB (r = −0.33; P < 0.001), DD (r = −0.36; P < 0.001), and FDP (r = −0.35; P < 0.001), and positively related to AT-Ⅲ (r = 0.42; P < 0.001). TBA was negatively related to PLT (r = −0.24; P < 0.001). In addition, the level of AST was positively related to FIB (r = 0.18; P < 0.01), DD (r = 0.20; P < 0.001), and FDP (r = 0.18; P < 0.01), and negatively related to AT-Ⅲ (r = −0.13; P < 0.05).
Subgroup analysis of biomarkers levels in LC groups
To explore the difference of the 14 biomarkers in LC patients mentioned above, subgroup analyses based on histologic type, clinical stage, and neoplasm metastasis status were carried out. The results showed that age, gender, and symptom status were all significantly different among the three subgroups (Table 2). Additionally, liver-related markers (C1q, AST, TBA, and PA), and coagulation-related markers (PLT, TT, FIB, FDP, DD, and AT-III) were almost changed (Table 2). We further performed multivariable regression analyses to explore the independent factors in each subgroup focusing on markers with differential expression (P < 0.100) (Table 3).
Subgroup analysis of biomarkers levels in patients with LC.
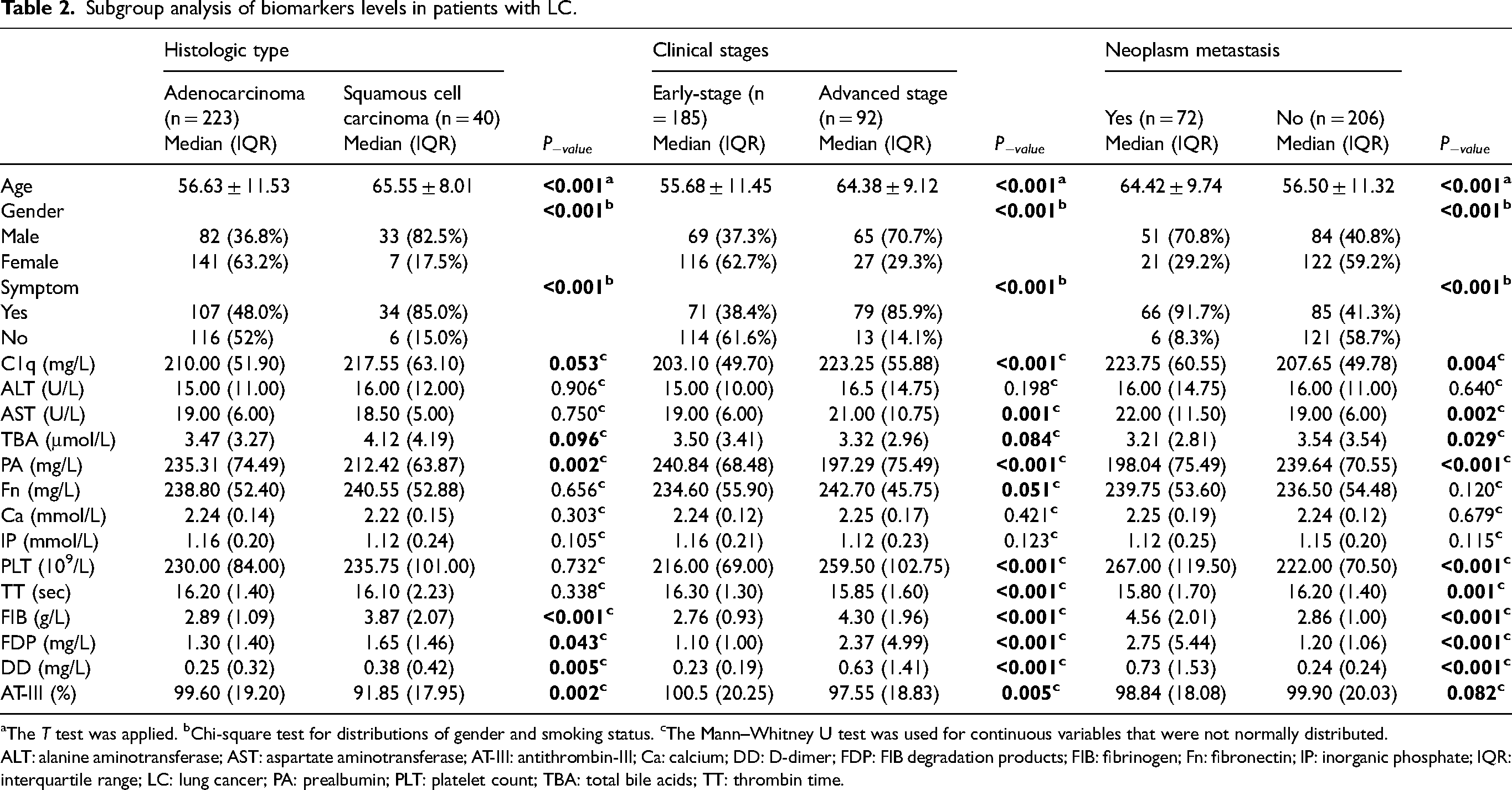
The T test was applied. bChi-square test for distributions of gender and smoking status. cThe Mann–Whitney U test was used for continuous variables that were not normally distributed.
ALT: alanine aminotransferase; AST: aspartate aminotransferase; AT-III: antithrombin-III; Ca: calcium; DD: D-dimer; FDP: FIB degradation products; FIB: fibrinogen; Fn: fibronectin; IP: inorganic phosphate; IQR: interquartile range; LC: lung cancer; PA: prealbumin; PLT: platelet count; TBA: total bile acids; TT: thrombin time.
Multivariable regression analyses of the risk factors of adenocarcinoma, advance stage LC, and neoplasm metastasis.
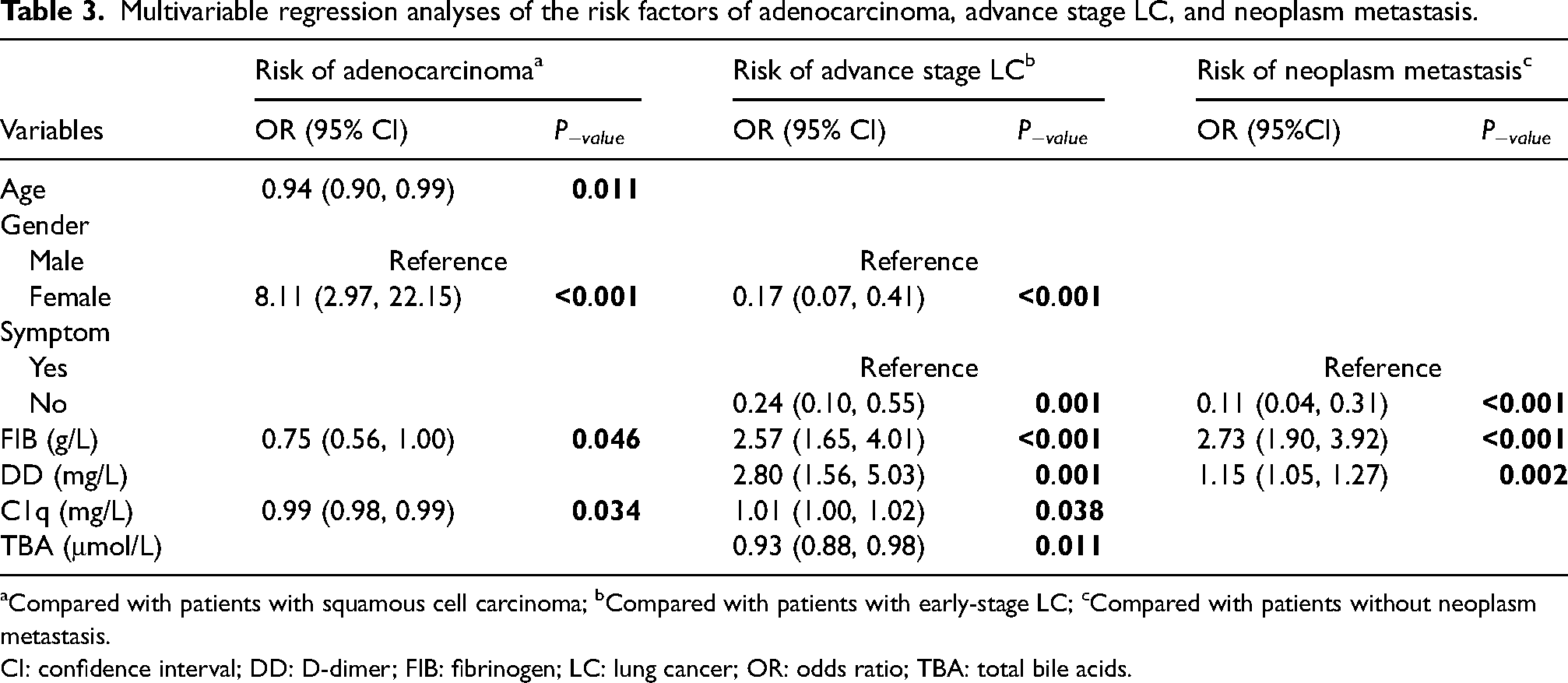
Compared with patients with squamous cell carcinoma; bCompared with patients with early-stage LC; cCompared with patients without neoplasm metastasis.
CI: confidence interval; DD: D-dimer; FIB: fibrinogen; LC: lung cancer; OR: odds ratio; TBA: total bile acids.
Multivariable regression analyses indicated that the occurrence of adenocarcinoma was significantly related to age (odds ratio (OR) 0.94; 95% CI 0.90, 0.99; P = 0.011), female (OR 8.11; 95% CI 2.97, 22.15; P < 0.001), and levels of C1q (OR 0.99; 95% CI 0.98, 0.99; P = 0.034) and FIB (OR 0.75; 95% CI 0.56, 1.00; P = 0.046) compared with squamous cell carcinoma. Additionally, there were six factors significantly correlated to the risk of advanced-stage LC compared to early-stage LC. Among these, female (OR 0.17; 95% CI 0.07, 0.41; P < 0.001), the admission with symptomatic status (OR 0.24; 95% CI 0.10, 0.55; P < 0.001) and the level of TBA (OR 0.93; 95% CI 0.88, 0.98; P = 0.011) showed a negative correlation with advanced-stage LC. The levels of C1q (OR 1.01; 95% CI 1.00, 1.02; P = 0.038), FIB (OR 2.57; 95% CI 1.65, 4.01; P < 0.001), and DD (OR 2.80; 95% CI 1.56, 5.03; P = 0.001) were positively associated with advanced-stage LC. Moreover, when compared to LC without neoplasm metastasis, patients with neoplasm metastasis showed admission with symptoms (OR 0.11; 95% CI 0.04, 0.31; P < 0.001), and higher levels of FIB (OR 2.73; 95% CI 1.90, 3.92; P < 0.001) and DD (OR 1.15; 95% CI 1.05, 1.27; P = 0.002).
Nomogram model for distinguishing early- and advanced-stage LC patients
According to the results of multivariable regression analysis, a nomogram model was constructed for distinguishing early- and advanced-stage LC (Figure 2(a)). The nomogram model was based on six indicators, including gender, symptom, the levels of TBA, C1q, FIB, and DD. Each variable was given a certain score on a point scale from 0 to 100, and a total score could be calculated by adding the score of each variable. Finally, the total score could be mapped to the total point scale axis, we then could distinguish patients between early and advanced-stage LC.
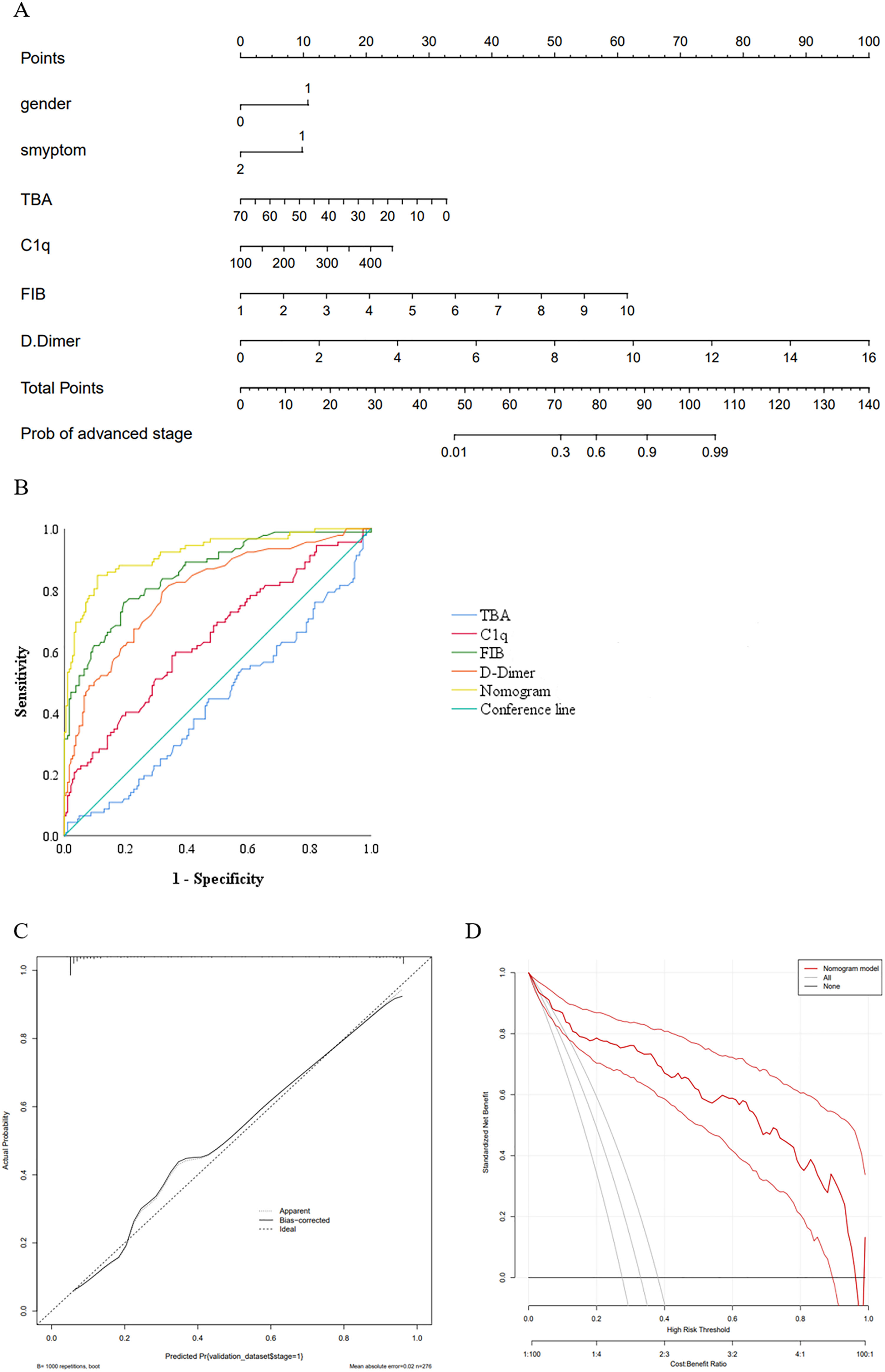
Construction and evolution of a nomogram for distinguishing early- and advanced-stage LC. (a) Nomogram model. Gender (1: male, 0: female), symptom (1: yes, 2: no). (b) The ROC curves of the nomogram for advanced-stage LC risk. (c) Calibration curves of the model. The dotted line represents the performance of the nomogram model, while the solid black line conducts bias corrected by bootstrapping (1000 repetitions), and the dashed line represents the reference line where the nomogram would be. (d) The DCA of the nomogram for advanced-stage LC risk.
Additionally, the results of the ROC analysis are showed in Figure 2(b). The AUC of the nomogram model was 0.919 (95% CI 0.882, 0.956; P < 0.001) and 0.927 (95% CI 0.924, 0.929; P < 0.001) for internal validation, presenting excellent discrimination of the nomogram model compared with a single biomarker (Supplementary Table 2). The calibration curve for this nomogram revealed good predictive accuracy (P−Hosmer−Lemeshow = 0.697) between the predicted probability and the actual probability (Figure 2(c)). Furthermore, the DCA demonstrated the benefits and clinical utility of the predictive power of our nomogram. The result indicated that if the threshold probability is between 0.01 and 0.96, the model showed better clinical benefit (Figure 2(d)).
Discussion
In this retrospective study, we comprehensively explored the different expression profile of coagulated- and liver-related biomarkers in patients with LC and BPD. The results showed that the concentrations of C1q, Fn, IP, PA, FIB, and DD were significantly changed in LC patients compared to BPD patients. In addition, multivariable regression analyses indicted that compared with early-stage LC, females and patients with higher TBA levels had a reduced risk of advanced-stage LC, whereas admission with symptoms and higher C1q, FIB and DD levels were positively related to advanced-stage LC. Furthermore, the nomogram model for distinguishing patients between early- and advanced-stage LC, which, based on gender, symptom, and the levels of C1q, FIB, DD, and TBA, exhibited excellent discrimination function with AUC = 0.919, and the model showed better clinical benefit if the threshold probability was between 0.01 and 0.96.
FIB can produce a fibrous network structure with PLT, providing a scaffold for the formation of cancer thrombi and favorable conditions for tumor metastasis. Guan et al. 31 reported that elevated levels of FIB can promote the adhesion between tumor cells and vascular endothelial cells, thereby infiltrating surrounding tissues, promoting the infiltration and metastasis of malignant tumor cells. FIB can also form a protective cover on tumor cells, promoting tumor cells not to be recognized and killed by immune cells in the body. Wang et al. 32 reported that fibrinogen alpha (FGA), one of the three polypeptide chains of FIB, had low expression in human LC tissues. Furthermore, they identified a direct interaction of FGA with integrin ɑ5 as well as FGA-integrin signaling that regulated the AKT-mTOR signaling pathway in A549 cells. Also, FGA knockout may increase tumor growth and metastasis through activation of AKT signaling in an A549 xenograft model. Our results show that there is a significant difference in serum FIB levels between patients with advanced- and early-stage LC, which was consistent with the results reported by Liu et al. 33 and Zhang et al., 34 indicating that the higher the clinical stage of LC the higher its FIB levels. However, Li et al. 35 found that there was no statistically significant difference in serum FIB concentration between different tumor stages, which may be due to the differences in the number of patients with early, middle, and advanced disease among the selected patients. From this, it can be seen that FIB, components, and their derivatives seem to play different roles in the occurrence and development of cancer. Therefore, more research on the mechanism is urgently needed to explore the specific role of FIB in LC.
DD is a regional marker reflecting the hypercoagulability and high fibrinolysis in vivo. However, increasing clinical evidence shows that the coagulation system of patients with malignant tumors is remarkably abnormal, and the level of DD is significantly elevated. 36 The mechanism may be that tumor cells or tissues around necrosis trigger the release of tissue factors, and activate coagulation factors X and XI in the external coagulation system and fibrinolysis system, resulting in the local production of plasmin to directly degrade the extracellular matrix, giving tumor cells the ability of invasion and metastasis. In this process, the released inflammatory mediators damage the vascular endothelial cells, leading to a hypercoagulable state of blood, which makes tumor cells adhere to the vascular wall. The hypercoagulable state of tumor patients not only increases the risk of venous thromboembolism, but also promotes the distribution of blood vessels around the tumor, enriches the blood supply of tumor tissue, and promotes tumor recurrence and metastasis. Through a meta-analysis of seven studies involving a total of 1488 LC patients and 328 control groups, Biruk et al. 37 concluded that the DD value was significantly higher in LC patients (standardized mean difference) 3.10; 95% CI 2.08, 4.12; P < 0.000) when compared with the health control group. In addition, Li et al. 36 found that patients with a high level of plasma DD have a worse overall survival prognosis (hazard ratio (HR) = 1.742; 95% CI 1.542, 1.969; P < 0.001; I2 = 72.1%) compared with these patients who have a normal plasma DD; elevated plasma DD is also a risk factor of progression-free survival in LC (HR = 1.385; 95% CI 1.169, 1.641; P = 0.003; I2 = 69.1%). Similar to our findings, Chen et al. 38 showed that the level of DD was associated with tumor stage; the level of neuron-specific enolase; the presence of distant metastasis, hyponatremia, and the progression of patients. These results might provide an important clue for high plasma DD levels as an independent factor of poor prognosis in patients with LC. Nonetheless, more prospective cohort studies and mechanism studies are warranted to verify and explore.
In addition to coagulation-related biomarkers, the complement system also plays a crucial role in tumor development. 39 As one of the main regulators of cancer immunity, C1q can activate the classic complement pathway via binding to immunoglobulin-M, playing an important role in immune regulation, inflammatory regulation, and maintaining body balance. Research has found that activated complement systems can inhibit tumor formation by promoting acute inflammation and lysis of tumor cells and stimulate the body to produce chronic inflammation and suppress immune responses to promote tumor growth, depending on the type of cancer. 40 High levels of C1q can be used as a worse prognostic indicator for the overall survival of basal breast cancer, cervical cancer, and HER2-positive breast cancer. Although C1q has been found as a cancer-promoting factor in the tumor microenvironment, there are still limited studies on the relationship of C1q and LC. As for adenocarcinoma, C1q promotes the proliferation, invasion, and angiogenesis of tumor cells. 41 Liu et al. 42 found that phosphate monoesters can inhibit the binding of C1q to LC cells, thus increasing the expression of phospholipid component phosphate monoesters in LC cell membranes can delay the progression of LC. In addition, Fan et al. 43 also discovered that C1q is highly expressed in LC stroma and vascular endothelial cells and plays a role in promoting tumor growth by promoting tumor cell adhesion, cell migration, cell proliferation, angiogenesis, and metastasis. Moreover, a study based on bioinformatic databases showed that the elevated level of C1q was associated with poor prognosis of SCLC. 16 We found that the level of pretreatment serum C1q was significantly positively related to patients with LC compared with BPD, and could be an independent factor for the risk of advanced-stage LC patients, which is consistent with prior studies.
BA are synthesized in the hepatocytes as primary BA, excreted into the bile duct and then into the intestinal lumen to produce secondary BA, and reabsorbed by the hepatic portal vein back to the liver. BA metabolism is commonly studied in the context of hepatobiliary and intestinal diseases, and its association with pulmonary diseases has been rarely reported. 44 In recent years, studies have shown that the higher BA level of bronchoalveolar lavage fluid in patients with respiratory disease is related to a higher incidence of combined gastroesophageal reflux disease. 45 It has also been shown that BA receptors such as farnesoid X receptor and Takeda G protein-coupled receptor 5 are expressed to varying degrees in lung tissue.46,47 In patients with pneumonia, BA receptors can suppress inflammation by inhibiting the activation of the NF-κB pathway; lipopolysaccharide and certain pro-inflammatory cytokines can also induce the downregulation of BA receptors in the lung, resulting in a corresponding decrease in BA entering the peripheral blood. 48 In terms of LC, BA receptors in lung tissue activate cell cycle protein-D1, which promotes tumor growth, and stimulate cancer cell proliferation by activating the JAK2/STAT3 signaling pathway. 49 With the occurrence of tumors, LC patients have more lung tissue BA receptors and a corresponding increase in BA entering peripheral blood. In our study, TBA was associated with malignant tumor metastasis; we also explored that compared with early-stage LC, the TBA level was positively related to advanced-stage LC by multivariate linear analysis. With the rapid development of metabolomics, BA profiles will play an important role in the early diagnosis of tumors, the exploration of mechanisms of occurrence, and the development of individualized treatment plans.
The strength of this study was that we systematically analyzed the application value of pretreatment coagulation-related indicators and liver-function-related indicators of lung space occupying patients in the differential diagnosis and staging of LC and BPD. We also established a nomogram model for distinguishing advanced LC in patients with early-stage LC with excellent discrimination and calibration. However, there were some limitations of this study. First, this study was retrospective and required further external validation with a prospective cohort study to have sufficient evidence to verify the findings. Second, all enrolled patients came from a single institution and the number of patients included is not large enough. To expand the scope of its clinical application, further multicenter studies on populations from different countries and regions are necessary. Third, our research lacked some routinely available data of clinical cancer-related biomarkers. Constructing a prognostic model using both the factors identified in our model and other lab tests explored by other studies would thus be beneficial in creating an even more accurate predictive model.
Conclusions
We established a nomogram model that was available for an individualized quantitative implement to distinguish early- and advanced-stage LC patients. The nomogram might assist clinicians to make treatment recommendations such as clinical trials and hospice management.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155241229454 - Supplemental material for Application of a nomogram from coagulation-related biomarkers and C1q and total bile acids in distinguishing advanced and early-stage lung cancer
Supplemental material, sj-docx-1-jbm-10.1177_03936155241229454 for Application of a nomogram from coagulation-related biomarkers and C1q and total bile acids in distinguishing advanced and early-stage lung cancer by Tingting Long, Xinyu Zhu, Dongling Tang, Huan Li and Pingan Zhang in The International Journal of Biological Markers
Footnotes
Author contributions
Conception and design: Pingan Zhang and Huan Li. Administrative support: Pingan Zhang and Dongling Tang. Provision of study materials or patients: Tingting Long. Collection and assembly of data: Tingting Long, Xinyu Zhu, and Dongling Tang. Data analysis and interpretation: Tingting Long, Xinyu Zhu, and Dongling Tang. Manuscript writing: All authors. Final approval of manuscript: All authors. TingtingLong and XinyuZhu contributed equally to this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Approval of the research protocol by an Institutional Reviewer Board: This study was approved by the Ethics Committee of the Renmin Hospital of Wuhan University.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hubei Province, National Natural Science Foundation of China, Science Foundation of Renmin Hospital of Wuhan University, (Grant Number 2019CFC846, 81773444, WJ2023M073).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
