Abstract
Background
Typically, the overexpressed keratin 7 (KRT7) is considered a validated therapeutic target and prognosis marker in bladder cancer. However, the crucial roles of KRT7 in the clinical prognosis and immune microenvironment in bladder cancer remain unclear.
Methods
Initially, the expression levels of KRT7 in public databases were analyzed that is,Tumor Immune Estimation Resource (TIMER) 2.0 and Gene Expression Profiling Interactive Analysis (GEPIA). Further, the clinical tissue samples from patients (n = 10 pairs) were collected to confirm the expression trends of KRT7 and detected by immunohistochemistry (IHC) analysis. Meanwhile, the relationship between KRT7 and the prognosis of bladder cancer patients was analyzed by Kaplan–Meier plotter estimation and Cox regression analysis. Finally, TIMER 2.0 and IHC staining analyses were performed to calculate the infiltration abundances of three kinds of immune cells in eligible bladder tumor samples.
Results
The TIMER 2.0 and GEPIA datasets suggested the differences in the expression levels of KRT7 in tumors, in which KRT7 was significantly upregulated in bladder cancer. The KRT7 expression was closely associated with patients’ gender, tumor histologic subtypes, T status, and American Joint Committee on Cancer stages. Notably, the increased KRT7 indicated poor overall survival and disease-free survival rates. Moreover, KRT7 expression could be responsible for immune infiltration in the cancer microenvironment of the bladder. Finally, the high expression level of KRT7 increased the presence of regulatory T cells (Tregs) but reduced the infiltration of CD8+ T and natural killer cells.
Conclusion
KRT7 as a biomarker potentiated the prediction of bladder cancer prognosis and the immune microenvironment.
Introduction
Bladder cancer has emerged as the fourth most common cancer in the world, 1 characterized by gross or microscopic hematuria. Although bladder cancer is heavily influenced by geography and aging, cigarette smoking is the most prevalent manifestation, contributing to the increasing incidence rates. 2 Recently, several advancements have been evidenced by exploring various treatment strategies against bladder cancer. Along this line, non-muscle invasive bladder cancer patients mainly choose transurethral resection or en bloc bladder tumor removal (EBRT), depending on subtype. 3 Contrarily, radical removal together with concomitant meticulous pelvic lymph node dissection, referred to be golden standard treatment, is often applied for the muscle invasive bladder cancer patients. 4 In addition to various kinds of surgeries, cisplatin-based chemotherapy is the first-line therapeutic option after surgical procedure. Nevertheless, the recurrence and metastasis attributes remain significant challenges due to the 5-year survival rate of about 15%. 5 In recent times, the immune checkpoint blockade has been explored, in which the contemporary clinical trials proved the unexpected response of immune checkpoint inhibitors in bladder cancer. 6 Nevertheless, further investigations of immune therapy are still under progress.
Keratin 7 (KRT7), one of the keratin genes, belongs to type II cytokeratin. 7 Along this line, KRT7 has been identified as a prognostic biomarker in various types of cancers, such as pancreatic, 8 gastric, 9 lung, and breast cancers. 10 The abnormal expression of KRT 7 could influence cancer cell growth, metastasis, and chemoresistance, 11 which could be related to the poor prognosis of patients. Hitherto, several reports indicated the role of KRT7 as a prognosis marker in transitional cell carcinoma (TCC) of the bladder. 12 Nevertheless, no comprehensive explorations have been made focusing on the clinical validation of KRT7 in bladder cancer.
Tumor-infiltrating T lymphocytes (TILs) are the main tumor killer cells, including cytotoxic CD8+T cells, CD4+T cells, natural killer (NK) cells, and regulatory T cells (Tregs). 13 One case reported that the bladder cancer was enriched with Tregs CD4+T cells, lacking CD8+T cells in the microenvironment. 14 Along this line, some reports indicated that KRT7 could participate in immune cell infiltration in pancreatic and ovarian cancers.7,15 Motivated by these considerations, this study explores the role of KRT7 expression in altering the presence of TILs in the bladder cancer microenvironment.
Methods
TIMER 2.0 and GEPIA data analysis
In this study, we have employed the public online datasets; that is, Tumor Immune Estimation Resource (TIMER) 2.0 (http://timer.cistrome.org/), to analyze the differential expression of KRT7 in a variety of cancers and explore the relationship between KRT7 and immune cell infiltration. In addition, the Gene Expression Profiling Interactive Analysis (GEPIA) (http://gepia.cancer-pku.cn/) was used to screen the messenger RNA (mRNA) expression of KRT7 in cancer and normal tissues of patient tissue samples.
Clinical specimens and immunohistochemical analysis
For this study, a total of 100 patients with bladder cancer were recruited following the guidelines of the Ethics Committee of The Third Affiliated Hospital of Zhejiang Chinese Medicine University (Zhejiang, China).
The immunofluorescence staining was performed as follows. Initially, the tissues were sectioned into thin slices and then deparaffinized with xylene and rehydrated in graded ethanol. Further, the citrate buffer was used to block the slides and were incubated with KRT7, CD4, CD8, Foxp3, and CD56 antibodies (Abcam, 1:200, UK) overnight at 4°C. Further, the slides were incubated with secondary antibodies for another 1 h at room temperature. The expression of each signal was further examined by the 3,3′-diaminobenzidine (DAB) Detection Kit (Abcam, UK). The immunohistochemistry (IHC) staining was scored as 0 (no staining or membranous staining in less than 10% of tumor cells), 1+ (faint/barely visible membranous staining in at least 10% of cells, or staining in parts of their membrane), 2+ (weak to moderate complete or basolateral membranous staining in at least 10% of tumor cells), 3+ (strong complete or basolateral membranous staining in at least 10% of tumor cells). 16 Eventually, the IHC score from 2 to 3 was regarded as the high expression group.
Statistical analysis
All the experiments were repeated thrice, and data were displayed in mean ± SD. The statistical significance analysis among different experimental groups was performed using the GraphPad Prism 8.0 by Student's t-test or two-way analysis of variance (ANOVA), considering a P-value of < 0.05 as statistically significant.
Results
Expression of KRT7 in bladder cancer
Initially, the KRT7 expression data from The Cancer Genome Atlas (TCGA) was obtained by accessing the TIMER 2.0 database. It was observed from the results that the KRT7 mRNA levels were upregulated in various cancers, such as colorectal, cervical, endocervical, cholangio carcinoma, esophagus, kidney, ovarian, and lung cancers, compared to normal tissues. Specifically, an abnormally high expression was observed in bladder cancer (P < 0.05) (Figure 1(a) and (b)). Further, we analyzed the KRT7 expression in the GEPIA database in the tissues extracted from bladder cancer (n = 404) and healthy tissues (n = 28). The results indicated that KRT7 expression was significantly higher in tumor tissues compared with normal bladder tissues (Figure 1(c)).
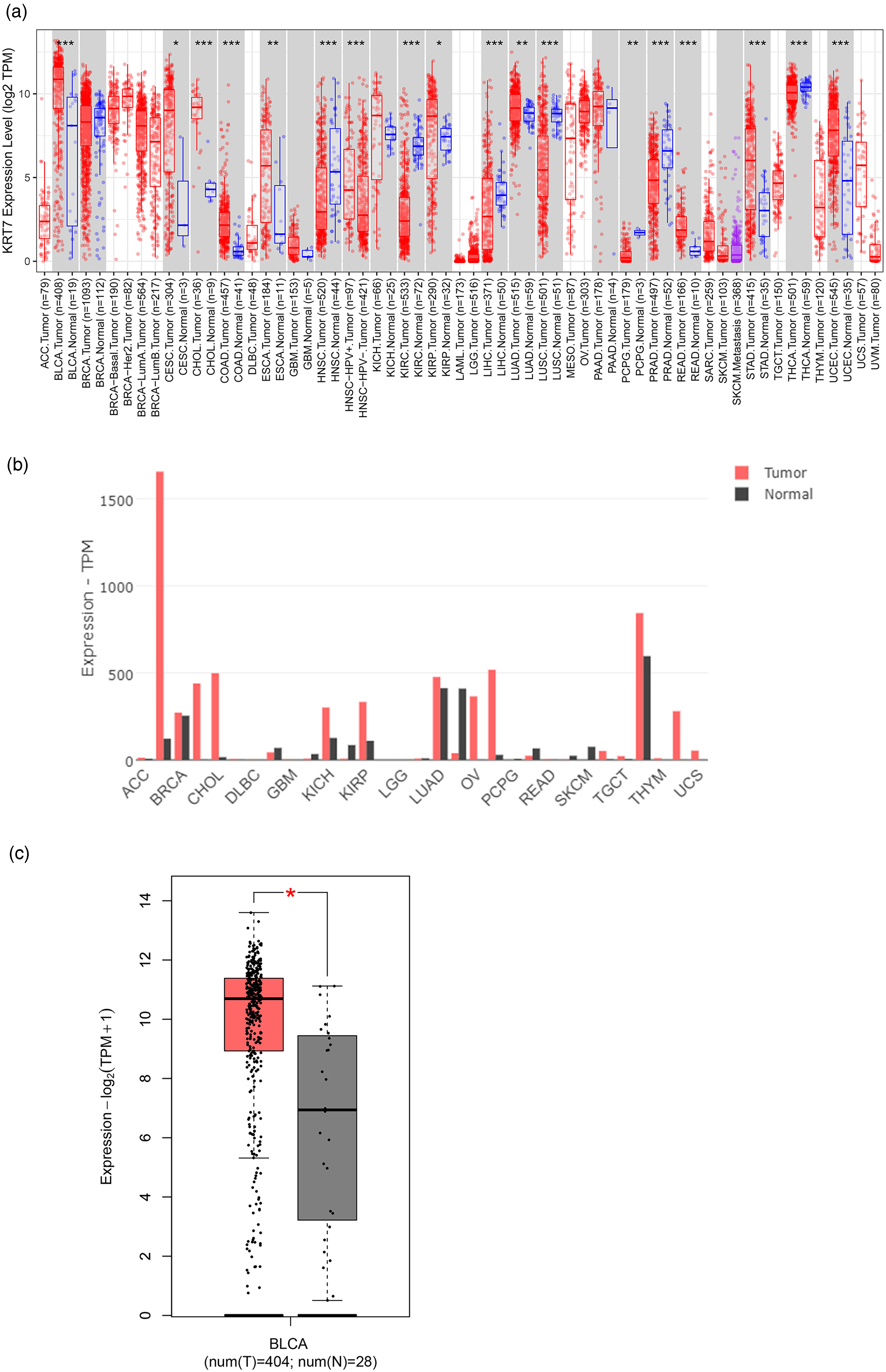
Expression of KRT7 in bladder cancer. (a) and (b) TIMER 2.0 and GEPIA databases present the mRNA expression levels of KRT7 in cancers and normal tissues. (c) The GEPIA database presents the expression level of KRT7 in bladder cancer tissues and normal tissues. * indicates P < 0.05, ** defines P < 0.01, and *** signifies P < 0.001, compared to the normal treatment group.
Clinical features of KRT7 expression
To confirm the KRT7 expression in clinical samples, 10 pairs of tumor and normal tissues from patients with bladder cancer were collected and subjected to IHC staining analysis. Compared with normal tissues, the expression levels of KRT7 protein were tremendously higher in tumor sections (Figure 2(a)). Accordingly, the enhanced trend of KRT7 protein expression in tumor tissues was consistent with the mRNA levels in TIMER 2.0 and GEPIA databases.
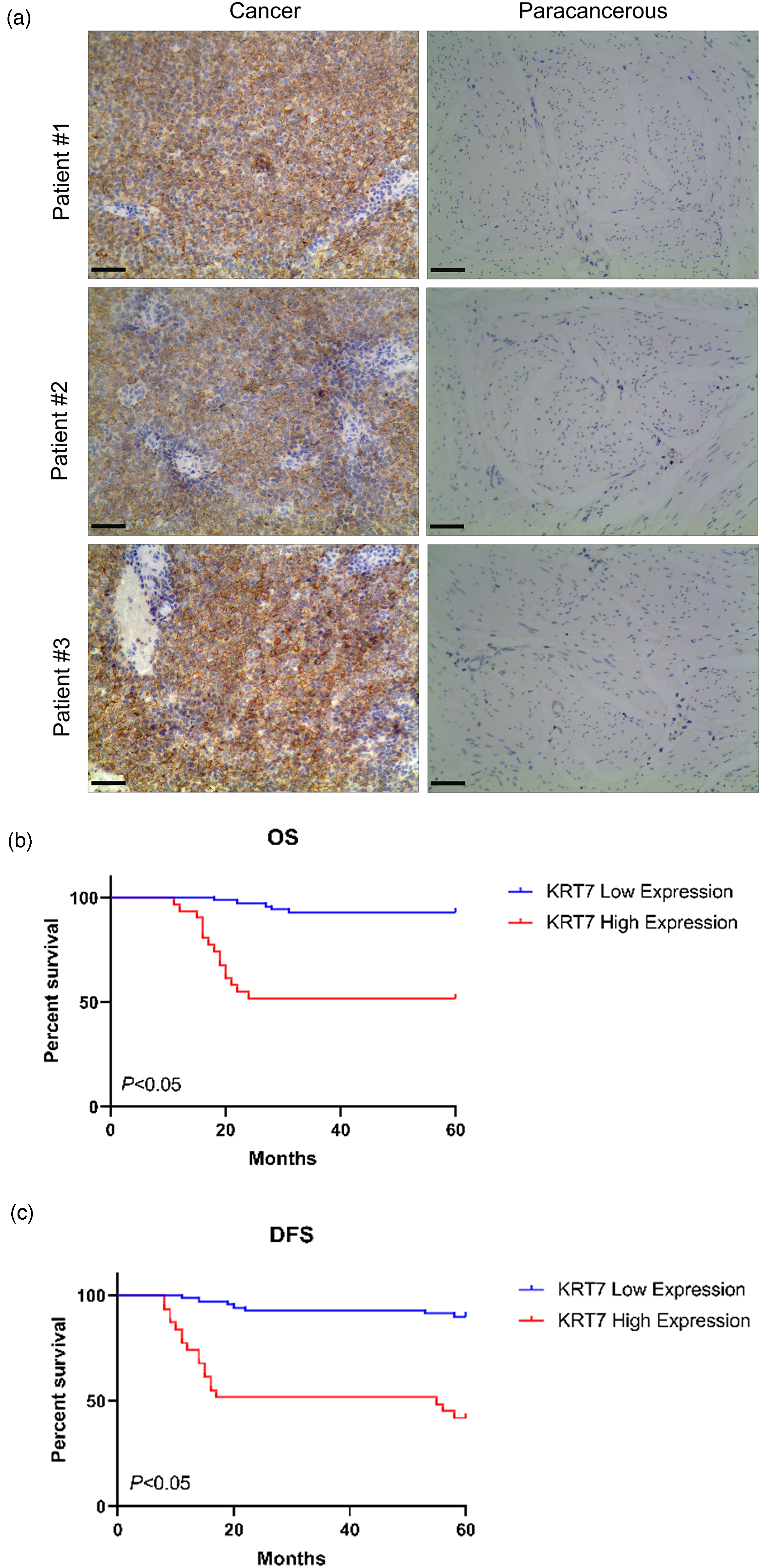
Expression of KRT7 associated with OS and DFS in bladder cancer patients. (a) The IHC analysis shows the expression levels of KRT7 protein in clinical patient tissues; the left pictures indicated tumor tissues and the right ones indicated normal tissues (×200). The KM survival curves assess the (b) OS and DFS rates (c) of patients with KRT7 high or low expression. * indicates P < 0.05, compared to KRT7 low expression group.
Further, the relationship between the expression of KRT7 and the clinical characteristics of bladder cancer was evaluated. To explore these aspects, we collected the clinical features of patients (n = 100) and analyzed the data by Chi-square test. It was observed from the results that high expression of KRT7 was positively correlated with patients’ gender, tumor histologic subtypes (non-papillary or papillary), T status (T1–T2, T3–T4), and American Joint Committee on Cancer (AJCC) stages (I–II, III–IV) (P < 0.05). On the other hand, the high expression of KRT7 was not related to age, smoking history, N status (N0, >N0), and M status (M0, M1) (Table 1).
A summary displays the correlation of KRT7 expression and clinical features in bladder cancer (n=100).
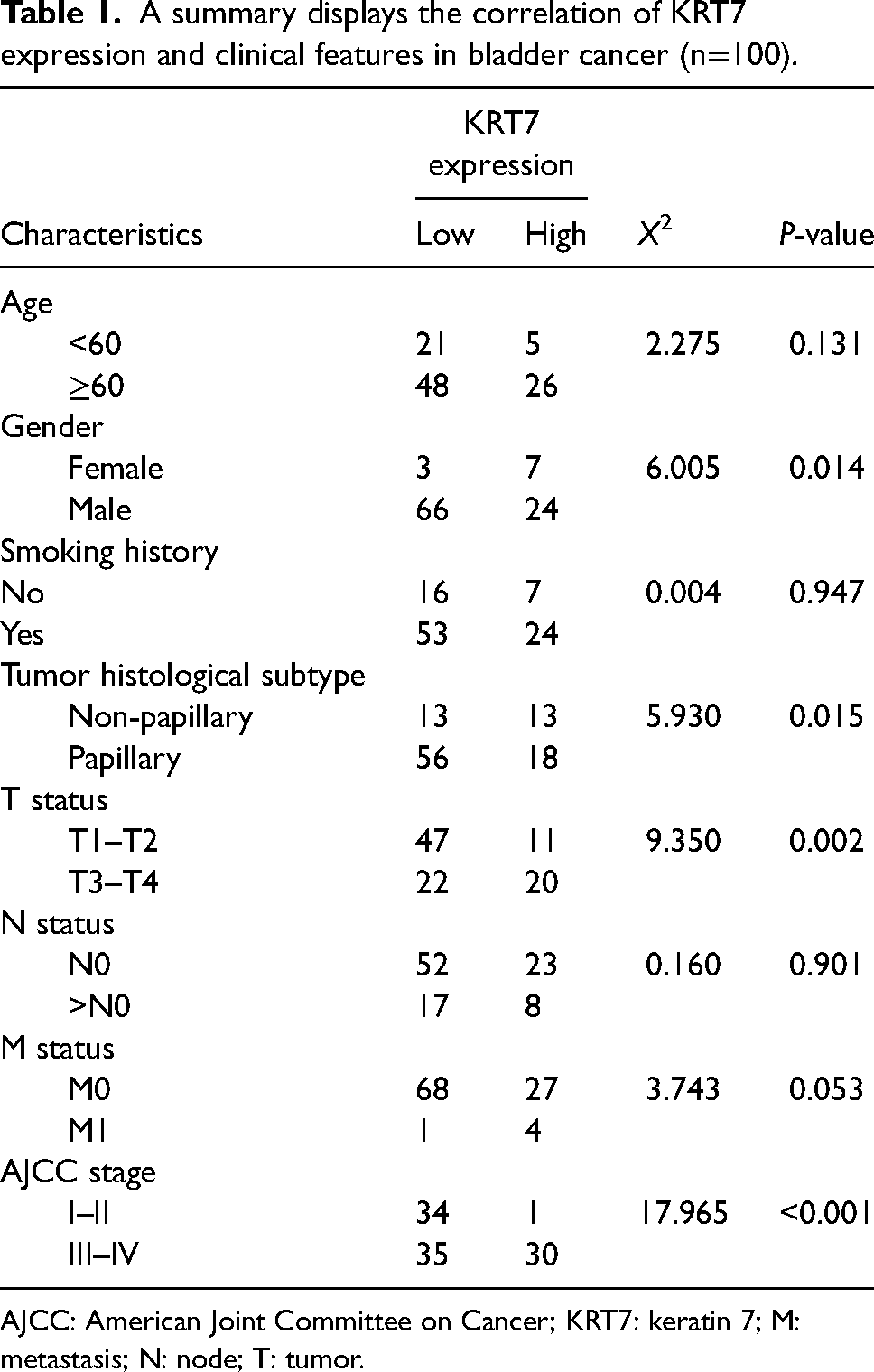
AJCC: American Joint Committee on Cancer; KRT7: keratin 7; M: metastasis; N: node; T: tumor.
Correlation between KRT7 and the prognosis of bladder cancer
Considering the expression levels and the diagnostic roles of KRT7, we further explored the KRT7 expression in patients’ tissues to elucidate its role in affecting patients’ overall survival (OS) and disease-free survival (DFS). The survival analysis was performed on bladder cancer cases (n = 100). As shown in Figure 2(a), the KRT7 expression levels were increased in patients’ cancer tissues over the paracancerous tissues. Moreover, the Kaplan–Meier (KM) plotter analysis indicated that only half of patients with higher KRT7 expression could reach the survival rate of patients with low KRT7 levels (P < 0.05), suggesting that higher KRT7 could lead to poorer prognosis in both OS and DFS rates (Figure 2(b) and (c)). To further assess the prognostic value of KRT7 expression, the univariate and multivariate analyses were carried out using the Cox regression model. The results showed that tumor histologic subtype and high KRT7 expression were closely related to poor outcomes of bladder cancer in patients concerning both OS and DFS rates (Supplemental Table S1 and S2). Together, these findings indicated that the upregulated KRT7 could be an important risk factor for poor prognosis of bladder cancer.
Correlation between KRT7 expression and immune infiltration cells
In the tumor microenvironment, the status of TILs often influences the therapeutic responses and eventual survival outcomes of cancer patients. 17 Recently, a case report demonstrated that high KRT7 expression was associated with the suppressive immune microenvironment in pancreatic cancer. 18 Considering these reports, we hypothesized that KRT7 could influence the infiltration of immune cells in bladder cancer. The TIMER2.0 database was employed to illustrate profiles of KRT7 associated with the infiltration of various immune cells. Interestingly, it was observed that the KRT7 expression was positively related to the purity (Rho = 0.208, P = 5.58E-05) and the presence of Tregs (CIBERSORT: Rho = 0.107, P = 4.00E-02) (Figure 3(a)). In addition, the highly expressed KRT7 was negatively correlated with the infiltration levels of CD8+T cells (TIMER: Rho = −0.142, P = 6.46E-03) and NK cells (MCPCOUNTER: Rho = −0.143, P = 6.08E-03) (Figures 3(b) and (c)). The abundance of Tregs and the loss of CD8+T and NK cells indicated the suppressive status of anti-tumor immunity. Together, these results revealed that KRT7 could play a vital role in bladder cancer immune infiltration.
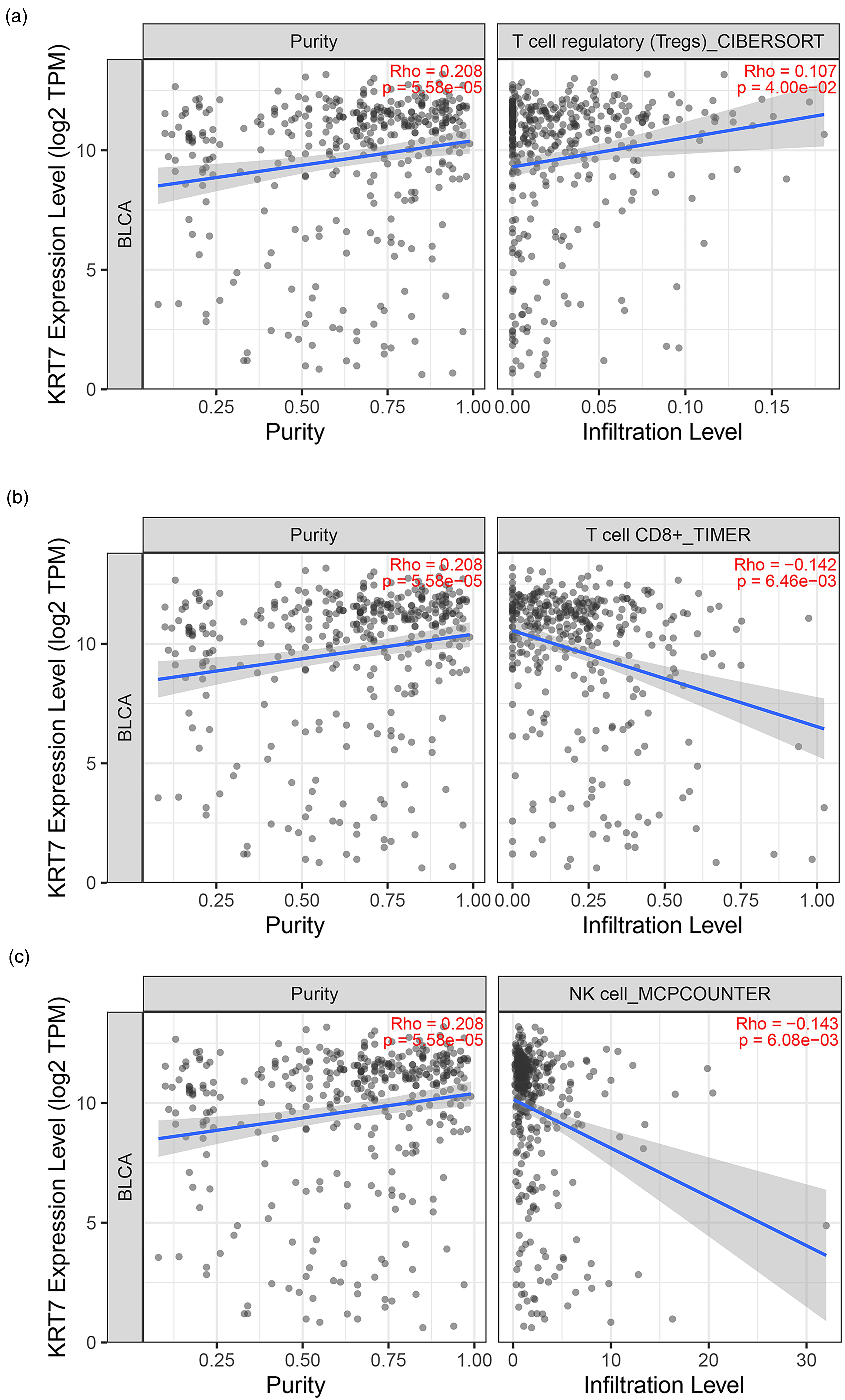
Expression of KRT7 modulated immune infiltration cells in bladder cancer. High expression of KRT7 is positively associated with Treg cells (a) but is negatively related with CD8+ T cells (b) and NK cells (c).
Correlation of KRT7 with the expression of immune cell markers
Considering the aforementioned results, the KRT7 expression levels could be closely related to the presence of different cohorts of immune cells, including CD8+T, NK, and Tregs. To further confirm the relationship, different immune cell markers were comprehensively examined by IHC staining analysis. As depicted in Figure 4(a), KRT7 was highly expressed in human bladder cancer tissues. In addition, Tregs maker Foxp3 was higher in the same tissue section. Conversely, the decrease of Foxp3 matched the reduced expression level of KRT7. On the other hand, the expression levels of markers of main tumor killer cells, such as CD4+T, CD8+T, and NK cells, were also altered with the KRT7 expression. Interestingly, it was observed that KRT7 showed a negative relationship with tumor-killer cell markers; that is, CD4, CD8, and CD56. Accordingly, we emphasized that the highly expressed KRT7 could lead to low levels of these markers (Figure 4(b) to (d). Combined with KRT7 IHC scores and different cohorts of immune cell markers, the high KRT7 group of tissues showed a positive relationship with the Foxp3 IHC score. Nevertheless, the higher KRT7 samples showed lower proportions of CD4 and CD8 levels, suggesting a strong correlation between KRT7 expression and infiltration of immune cells in bladder cancer.
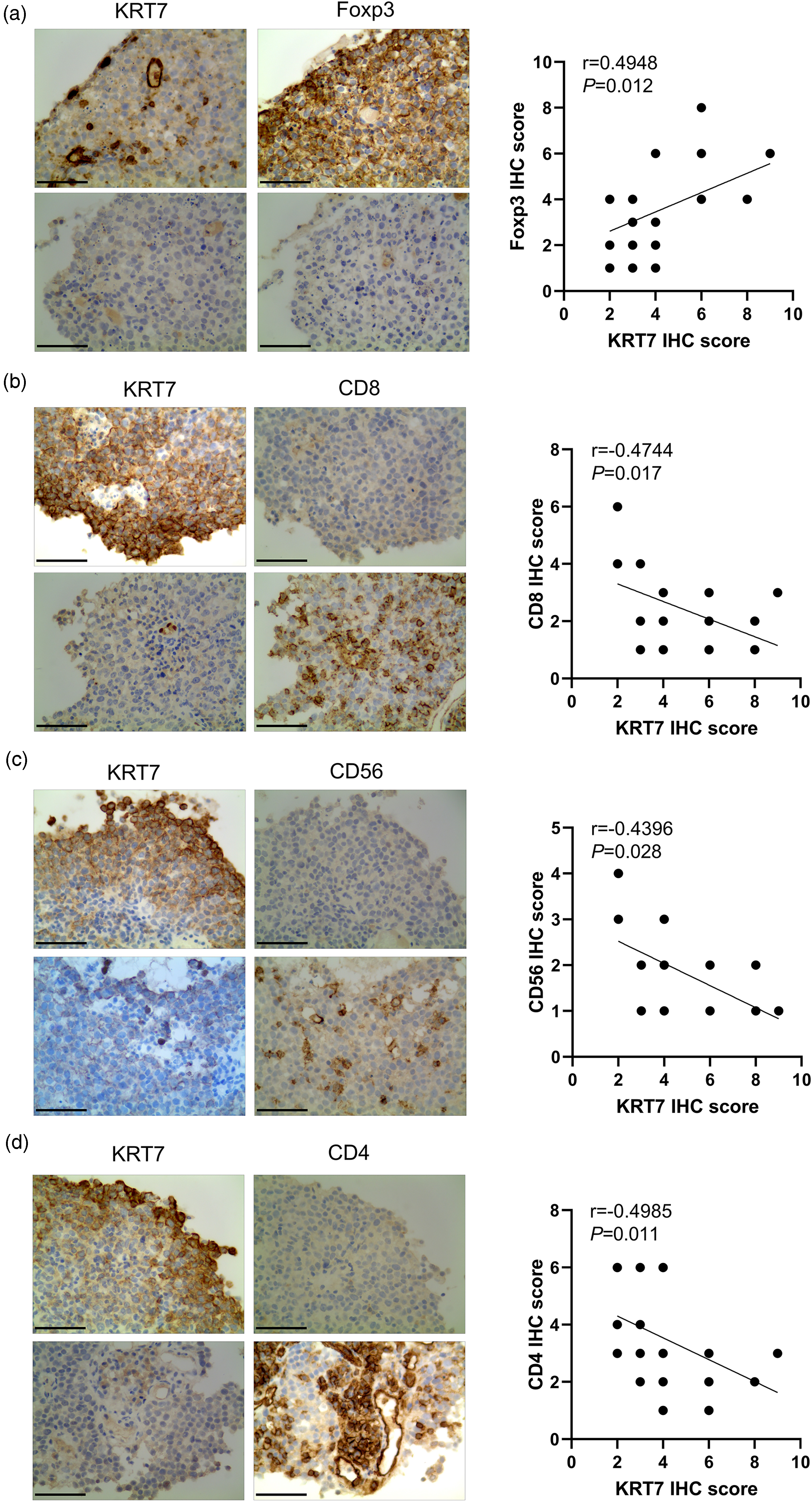
Correlation of KRT7 with the expression of immune cell markers. (a to (d) Sequential pathological sections and immunohistochemical staining analysis detect the expression of KRT7 and Foxp3, CD4, CD8, and CD56 in tumor tissues (n = 25). The data show the correlation of IHC scores of KRT7 compared to that of Foxp3, CD4, and CD8. Magnification ×200, * signifies P < 0.05, ** denotes P < 0.01, and *** indicates P < 0.001.
Discussion
Bladder cancer, a common cancer of the urinary system, is categorized into non-muscle invasive and muscle-invasive cancer, in which the former type accounts for 70% of all cases. 1 Owing to the advancements in medical technology, the overall 5-year survival rates reached 48%–60%. 19 Nevertheless, metastatic bladder cancer still shows a poor prognosis. In recent times, immunotherapy has garnered a great deal of interest from researchers to act against bladder cancer. Nonetheless, the tumor microenvironment displays an immunosuppressive status, leading to the lack of response of immune checkpoint inhibitors and enriched immune gene expression. Therefore, novel biomarkers associated with cancer immune cell infiltration are urgently needed.
The Keratin gene family belongs to the largest intermediate filament (IntFil) genes. These genes were classified into six classes by Dr. Steinert in the 1990s. 20 Among them, type II cytokeratin genes (KRT1-KRT8) are typically expressed in the epithelial cells of internal organs, blood vessels, hair, and nails. 21 Recently, tremendous advancements in research have evidenced the increased number of KRT genes in cancers. In one case, KRT1 was identified as a molecular marker for drug targeting in breast cancer. 22 In another case, it was demonstrated that KRT4 was abnormally expressed in oral cancer, 23 while KRT5 was reported as a core gene in metastatic melanoma via bioinformatics analysis. 24 Motivated by these findings, this study identified Keratin 7 (KRT7) as a novel prognosis biomarker in bladder cancer.
KRT7, a member of type II cytokeratin genes, 25 is involved in cancer cell proliferation, migration, and invasion of various cancers. 26 In one case, KRT7 was upregulated in pancreatic cancer in which KRT7 silencing could diminish the growth of cancer and reduce the proliferation and invasiveness of tumor cells. 27 In a recent study, KRT7 controlled lung and breast cancer oncogenesis and apoptosis. 10 By screening TIMER 2.0 and GEPIA datasets, the current research study indicated that KRT7 was overexpressed in bladder cancer tissues. To confirm the expression of KRT7, the levels of KRT7 in patients’ tumor tissues and paracarinoma tissues were detected by IHC analysis. The results demonstrated that the KRT7 expression was significantly increased, which was consistent with the trend in TCGA pan-cancer databases. The KM plotter survival analysis demonstrated that the survival rates of patients with low expression of KRT7 were higher than that of patients with high levels of KRT7 in terms of both OS and DFS rates.
Furthermore, information on 100 cases of bladder cancer patients in clinics was collected to examine the correlation between KRT7 and the clinical features of cancer patients. It was observed that the high KRT7 expression was closely related to gender, tumor histologic subtypes, T status, and AJCC stages. In addition, the Cox regression analysis showed that KRT7 could act as a diagnostic and prognostic risk factor in bladder cancer patients. These findings indicated that KRT7 played an important role in the progression of bladder cancer.
Up to now, several reports indicated that different immune cell abundances played critical roles in the progression of bladder cancer. 28 On the one hand, the high expression of cytotoxic cells (i.e., CD8+ T, CD4+T, and NK cells in the tumor immune microenvironment) is a sign of a favorable prognosis. 29 On the other hand, it was observed that the tumor samples with high infiltration levels of Tregs showed poor outcomes. 30 Accordingly, several reports demonstrated that KRT7 overexpression was associated with immune cell infiltration in pancreatic and breast cancers.8,15 Nevertheless, the roles of KRT7 in bladder cancer are yet to be reported. In this study, the TIMER 2.0 database was employed to clarify the relationship between KRT7 and immune cell abundances in bladder cancer. It was observed that high expression of KRT7 was positively related to the presence of Tregs and negatively correlated with CD8+T and NK cells.
Furthermore, the clinical tumor tissues were collected to confirm the relationship between KRT7 and immune infiltration by IHC analysis. The staining results showed that upregulation of Tregs marker Foxp3 was observed in tumor tissues and correlated with high levels of KRT7 expression. Conversely, the levels of CD4+ T, CD8+ T, and CD56+ NK cells were decreased in tumor samples with KRT7 overexpression. Together, these findings suggested that KRT7 expression might participate in the formation of an inhibitory immune microenvironment in bladder cancer. However, our study is largely based on bioinformatics analysis and is limited by prelimilary verification studies via only immunohistochemical experiments. Future experiments have to be performed to further validate the expression and regulatory role of KRT7 using cell and animal models.
Conclusion
In summary, the potential roles of KRT7 in bladder cancer prognosis and immune infiltration were identified using the bioinformatics analysis. Moreover, this study confirmed that KRT7 was highly expressed in bladder cancer. The overexpression of KRT7 was closely associated with clinical patients’ features, considering it as an important risk factor. Importantly, KRT7 was involved in the immune cell infiltration in the tumor microenvironment, indicating a new molecular target in bladder cancer therapy.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231224798 - Supplemental material for Clinical significance of KRT7 in bladder cancer prognosis
Supplemental material, sj-docx-1-jbm-10.1177_03936155231224798 for Clinical significance of KRT7 in bladder cancer prognosis by Jun Song, Ye Wu, Zhongming Chen, Dong Zhai, Chunpei Zhang and Shizhan Chen in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155231224798 - Supplemental material for Clinical significance of KRT7 in bladder cancer prognosis
Supplemental material, sj-docx-2-jbm-10.1177_03936155231224798 for Clinical significance of KRT7 in bladder cancer prognosis by Jun Song, Ye Wu, Zhongming Chen, Dong Zhai, Chunpei Zhang and Shizhan Chen in The International Journal of Biological Markers
Footnotes
Abbreviations
Acknowledgments
Not applicable.
Authors’ contributions
Conception and design: JS, YW. Data analysis and interpretation: JS, ZMC, DZ. Manuscript writing: JS, YW, CPZ, SZC. Final approval of manuscript: All authors. JS and YW contributed equally as the first authors to this study.
Consent for publication
Not applicable.
Data availability
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Written informed consent was obtained from each patient included in the study, and the study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the Ethics Committee of The Third Affiliated Hospital of Zhejiang Chinese Medicine University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
