Abstract
Numerous studies have reported the clinical value of alkaline phosphatase (ALP) and its bone-specific isoforms (bone-specific alkaline phosphatase (BAP)) in breast cancer. The purpose of this meta-analysis was to summarize the prognostic value of serum ALP and BAP in breast cancer, especially focused on bone metastasis and survival. PRISMA guidelines were followed to conduct this review. Observational studies were searched in PubMed, Cochcrane Library and EMBASE to January 1, 2022. Data were extracted to explore the prognostic value of ALP and BAP. The quality of the included studies was assessed and the outcome effects were evaluated. Subgroup and sensitivity analyses were performed to explore the potential sources of heterogeneity. Publication bias was assessed. There was a total of 53 studies with 22,436 patients included. For the primary outcome of survival, high levels of both ALP and BAP were associated with short survival time. The hazard ratio of high ALP level on overall survival was 1.72 (95% CI 1.37, 2.16, P < 0.001). For the secondary outcomes, a high ALP level (not BAP) was detected in breast cancer compared with healthy controls, and high levels of both ALP and BAP were risk factors for bone metastasis, while ALP (not BAP) was a risk factor for non-bone metastasis. This study showed that high levels of both serum ALP and BAP were associated with metastasis (BAP was associated with bone metastasis) and survival in breast cancer. The biomarkers could provide useful information for the early diagnostic assessment and monitoring in the follow-up of breast cancer patients.
Keywords
Introduction
Breast cancer (BC) is the most common female malignancy worldwide and is one of the leading causes of morbidity and mortality.1–3 Metastases, especially bone metastases, play an important role.4,5 With the continuous improvement of early diagnostic methods and comprehensive treatments, the survival of BC has significantly increased. 6 Nevertheless, distant metastasis is still a major cause for mortality, and early prognostic assessment and treatment is still required. 4
Alkaline phosphatase (ALP) is a nonspecific enzyme that hydrolyzes phosphate in alkaline medium and its total level reflects the combined activity of several isozymes found in the bone, liver, kidney, and intestinal lining.1,7 Its bone-specific isoforms (bone-specific alkaline phosphatase, BAP) originates in osteoblasts that release large amounts of the enzyme when bone repair activity occurs, for example bone metastases.8,9 In the last few decades, hundreds of studies have reported the clinical value of ALP/BAP in BC and a significant increase in serum ALP was observed, which had been demonstrated to predict bone and liver metastases. 10
Although studies have shown that ALP and BAP are predictors of metastasis and related to prognosis in BC,3, 4,10–13 the prognostic values reported in some studies are still controversial,11,14–17 and no systematic review has been performed. The purpose of this meta-analysis was to summarize the prognostic value of ALP/BAP in BC, with special focus on the predictive value of bone metastasis and survival.
Methods
Search strategy and screening
PRISMA guidelines were followed to conduct this review. We searched PubMed, Cochrane Library and EMBASE (all to January 1, 2022) for studies exploring the prognostic value of ALP and BAP in BC. The search strategy for PubMed was “(Alkaline Phosphatase [All Fields] OR ALP [All Fields] OR BSAP [All Fields] OR “Alkaline Phosphatase” [MeSH Terms]) AND (“breast neoplasms” [MeSH Terms] OR (“breast” [All Fields] AND “neoplasms” [All Fields]) OR “breast neoplasms” [All Fields] OR (“breast” [All Fields] AND “cancer” [All Fields]) OR “BC”[All Fields])”.
After duplications were excluded, two independent reviewers screened titles and abstracts to determine relevance to the present analysis. If unclear, a publication would be included. The full text of each study was reviewed. In case of discrepancies, a third reviewer would be involved. To be included, studies had to explore the prognostic value of serum ALP/BAP in BC patients. Exclusion criteria were studies not relevant to the meta-analysis, insufficient outcome data reported, studies with inappropriate patients, and studies with no full text.
Data extraction
Two independent reviewers reviewed the full text of each publication for study characteristics, study quality, and primary and secondary outcomes. The primary outcome was the risk effects of high ALP and BAP levels on overall survival (OS) and progression-free survival (PFS)/disease free survival (DFS). The secondary outcomes were the ALP/BAP levels in BC compared with healthy controls and the risk effects of high ALP/BAP level on bone metastasis and non-bone metastasis in BC. The hazard ratio (HR)/odds ratio (OR) with 95% confidence interval (CI) to primary and secondary outcomes could be calculated by univariate analysis, the multivariable Cox proportional hazards model, or logistic regression analysis adjusted for other risk factors.
Quality of including studies
The quality of the included studies was independently assessed by two reviewers. A standardized scale for observational studies was modified to suit the present study. With a total of 8 points, the scale ranked the quality of the included studies from aspects of study design, sample size, data analysis, and presentation.
Statistical analysis
A meta-analysis was performed using the Review Manager Software (version 5.4, Thomson Research Soft, Carlsbad, CA, USA) and STATA Software (version 11.0, Stata Corp, College Station, TX, USA). For dichotomous variables, OR with 95% CI was utilized to calculate the pooled effects of individual data. For continuous variables, the mean difference and the standardized mean difference (SMD) were used. For survival analysis, generic inverse variance of risk ratio with 95% CI on a log scale was utilized. The heterogeneity analysis was performed using the chi-square test and expressed in I2 statistics. A random-effects model was chosen over the fixed-effects model. The subgroup analysis was performed in order to decrease substantial heterogeneity. The publication bias was assessed by funnel plot, Egger's linear regression, and Begg's test. A sensitivity analysis was performed to evaluate the influence of a single study on overall effect by sequential omission of individual studies. A probability of P < 0.05 was regarded as statistically significant.
Results
Included studies
The stepwise literature review procedure is shown in Figure 1. We eventually included 53 studies (29 cohorts, 20 case-control studies, and 4 cross-sectional studies). As shown in Table 1, there were only 3 studies of prospective design. The included studies were undertaken in 23 countries and 1 study was of international design. There were 11 studies undertaken in China, 4 studies each in the USA, the UK and India. The sample size ranged from 24 to 3798 patients, with a total of 22,436 patients. For the patient characteristics, 7 studies were undertaken among BC patients and healthy controls, 5 studies of BC with bone metastasis, 7 studies of BC with non-bone metastasis, 5 studies of BC with specific type, and 29 studies of non-specific BC. The treatments were mentioned in 21 studies, which included surgery, hormone therapy, chemotherapy, and radiotherapy. The follow-up periods were mentioned in 28 studies and the median ranged from 6 months to 8 years. Serum ALP levels were in 44 studies and BAP levels were in 14 studies (5 studies reported both ALP and BAP results).
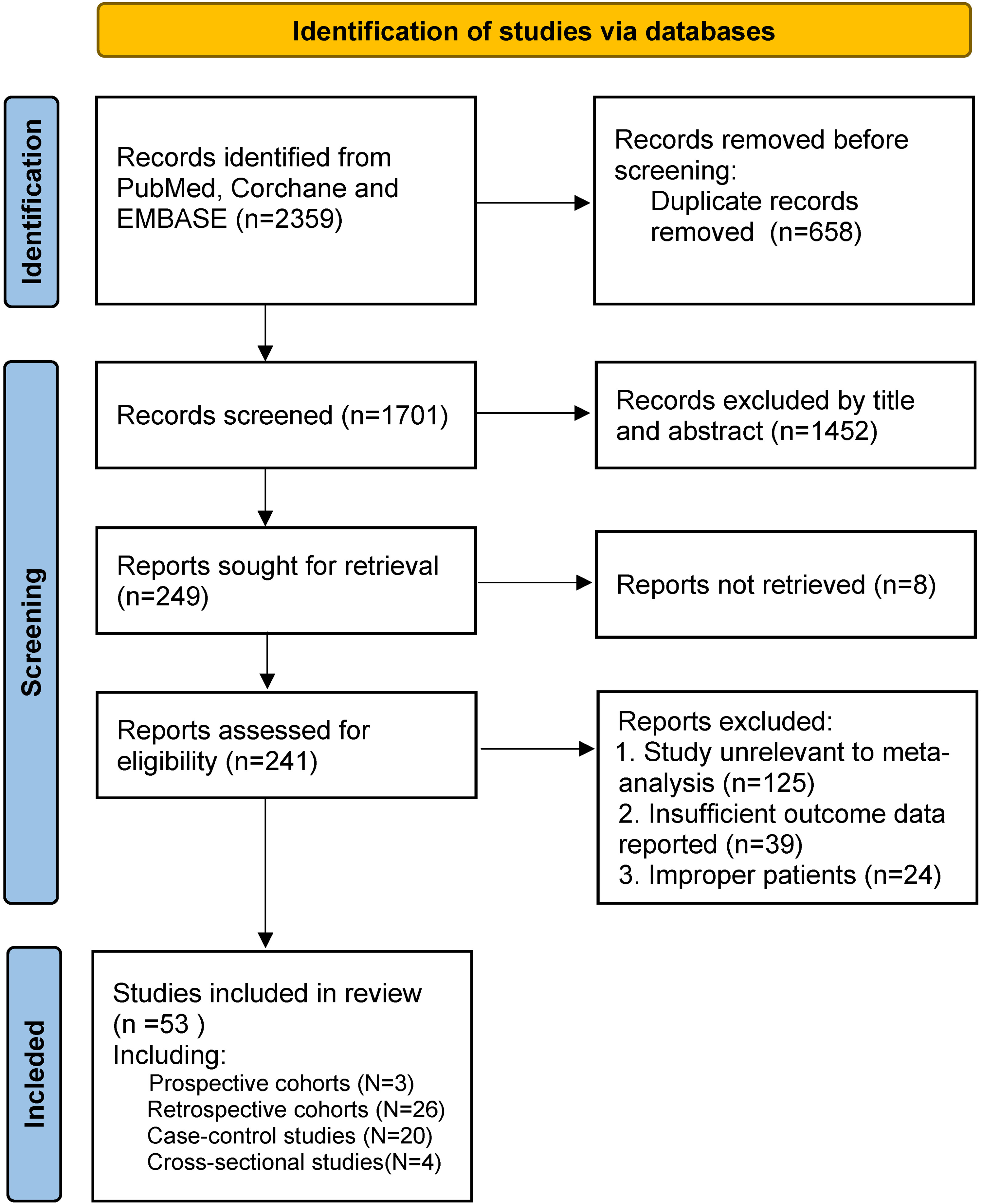
Stepwise literature review procedure.
Characteristics of the included studies.
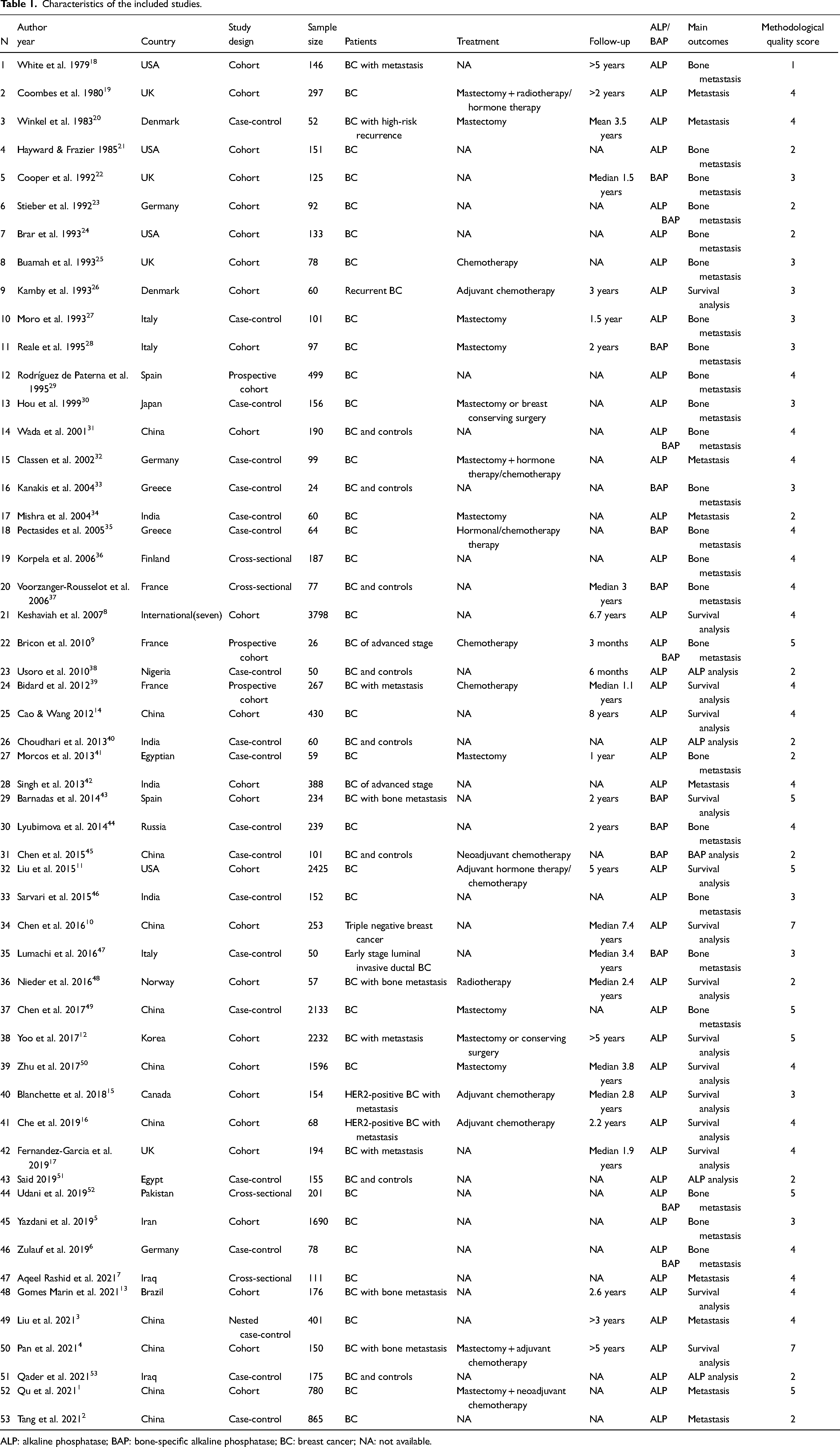
ALP: alkaline phosphatase; BAP: bone-specific alkaline phosphatase; BC: breast cancer; NA: not available.
Quality of included studies
Our standardized quality assessment for observational studies is presented in Online Resource Table S1. With a total of 8 points, the mean quality score of the 53 included studies was 3.5 ± 1.2 points (range 1–7 points).
Effects of meta-analysis
Primary outcome of prognostic value of ALP and BAP for survival in BC patients
High levels of both ALP and BAP were associated with short survival time. As shown in Figure 2, the merged HR of high ALP levels in 10 studies of multivariate analysis for OS was 1.72 (95% CI 1.37, 2.16; P < 0.001; I2 = 61%, random-effects model). All the included studies were cohort studies; a subgroup analysis based on study design could not be performed. The subgroup univariate analysis of four studies showed a HR of 1.98 (95% CI 1.53, 2.56; P < 0.001; I2 = 56%, random-effects model). The result of BAP on OS was only reported by Barnadas 43 and the reported HR by multivariate analysis was 3.25 (95% CI 1.293, 8.189; P = 0.0120). As presented in Online Resource Figure S1(the mean survival time) the merged result showed that a high ALP level was associated with shorter survival time (mean difference 18.6 months, 95% CI 7.1, 30.1; P = 0.001, I2 = 94%, random-effects model).
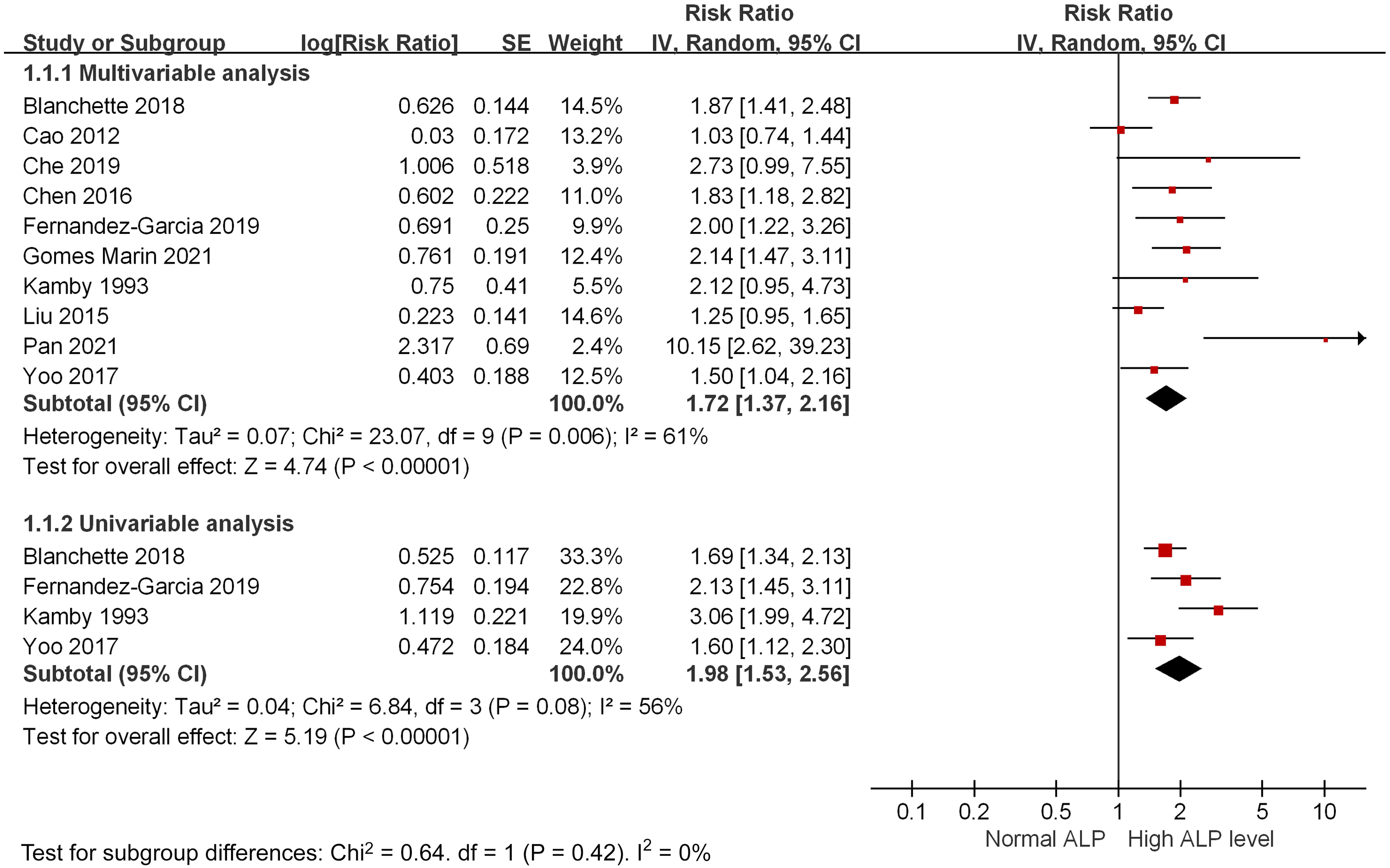
High ALP levels are associated with poor overall survival for breast cancers: forest plot merged by hazard ratio and sub-grouped by multivariate and univariate analysis.
For the progression-free survival (PFS)/disease-free survival (DFS), the HR of high ALP levels were reported in six studies. As shown in Figure 3, the merged HR of multivariate analysis was 1.38 (95% CI 1.19, 1.61; P < 0.001; I2 = 1%, random-effects model). Subgroup univariate analysis of three studies showed that the HR was 1.55 (95% CI 1.07, 2.24; P = 0.02; I2 = 73%, random-effects model). The result of BAP on PFS was only reported by Barnadas, 43 and the reported HR by univariate analysis was 2.25 (95% CI 1.391, 3.626; P = 0.001).
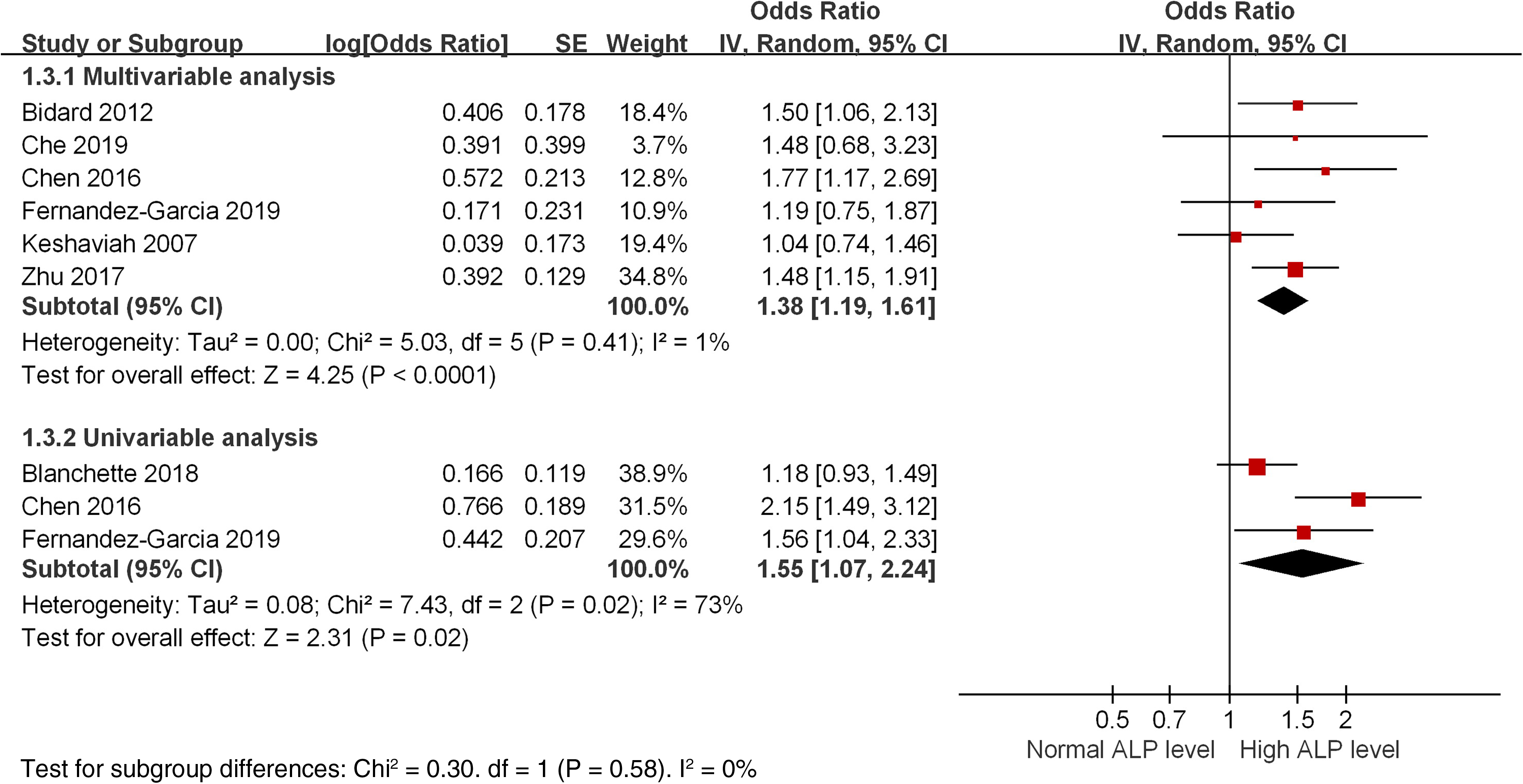
High ALP levels are associated with poor progression-free survival/disease free survival for breast cancers: forest plot merged by hazard ratio and sub-grouped by multivariate and univariate analysis.
Secondary outcomes of ALP/BAP levels in BC compared with healthy controls
High ALP levels were detected in BC compared with healthy controls. Because different ELISA kits were used to detect the ALP/BAP levels and the normal range varied greatly, SMD was utilized to compare the ALP/BAP level among the studies. Presented in Online Resource Figure S2, six studies compared the ALP level and four studies compared the BAP level in BC compared with healthy controls. The results showed high serum ALP levels in BC (P = 0.02; I2 = 96%, random-effects model) while there was no difference in BAP levels (P = 0.18; I2 = 76%, random-effects model). All the included studies were non-cohort studies; a subgroup analysis based on study design could not be performed.
Secondary outcomes of ALP/BAP as risk factors for bone metastasis in BC
High levels of both ALP and BAP were risk factors of bone metastasis. Presented in Online Resource Figure S3, four studies in the ALP analysis and two studies in the BAP analysis showed that high ALP and BAP were associated with bone metastasis (high ALP OR 4.62; 95% CI 2.34, 9.15; I2 = 56%; P < 0.001, random-effects model; high BAP OR 26.96; 95% CI 10.06, 72.28; I2 = 0%; P < 0.001, random-effects model). As shown in Figure 4, the ALP level was compared in 14 studies and BAP in 10 studies between the BC bone-metastatic group and the BC non-bone-metastatic group. Merged results showed that high levels of both ALP and BAP could be detected in the bone-metastatic group (ALP: P < 0.001; I2 = 93%, random-effects model; BAP: P < 0.001; I2 = 89%, random-effects model). The mean difference of ALP was 67.7 U/L (95% CI 38.9, 96.6). As shown in the Online Resource Figure S4 and Figure S5 of the subgroup analysis based on study design of the cohort studies versus non-cohort studies found that the study design had little influence on the risk effect of ALP/BAP levels for bone metastasis in BC.
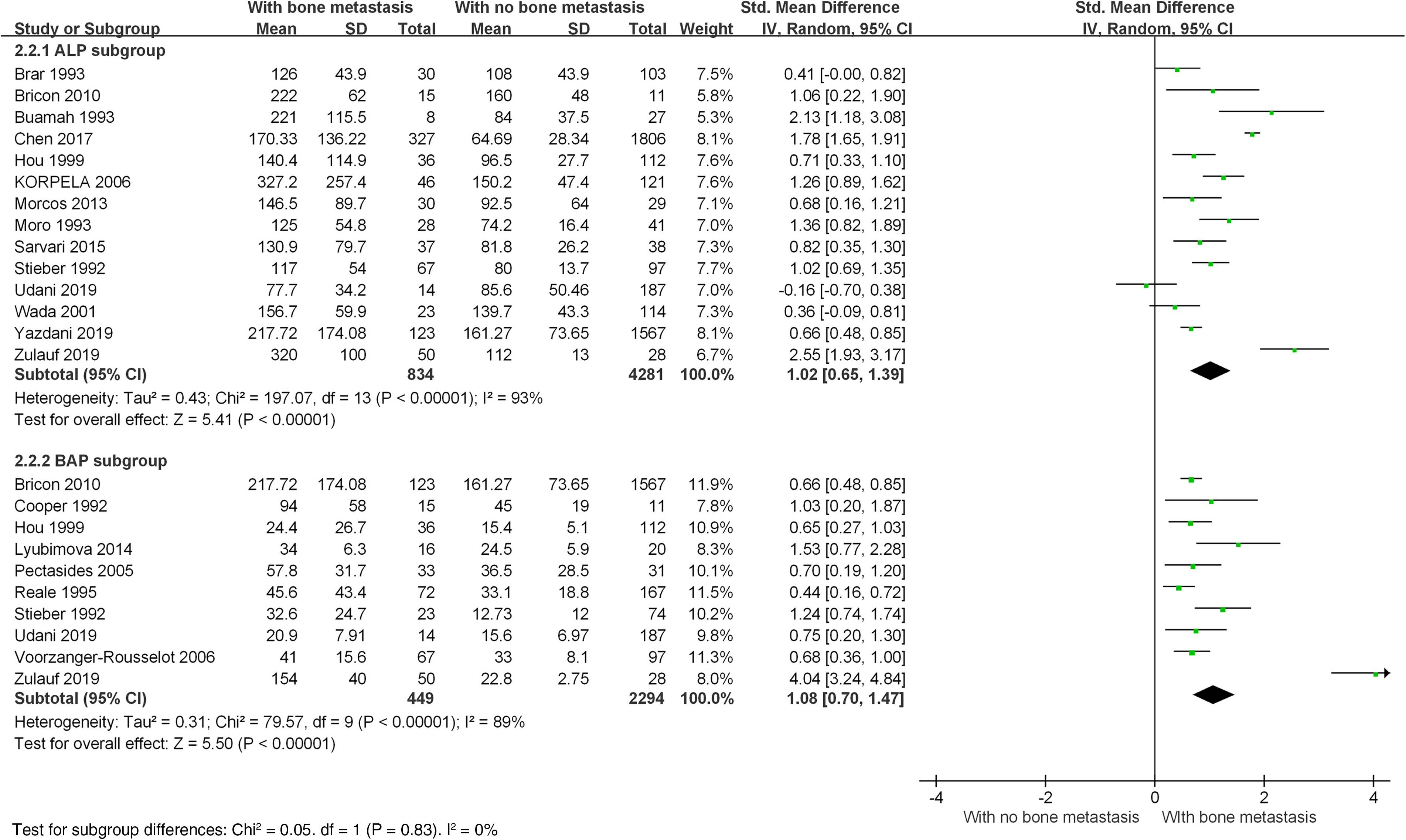
High ALP and BAP levels are associated with bone metastasis in breast cancers: forest plot merged by standardized mean difference.
Secondary outcomes of ALP as a risk factor for non-bone metastasis in BC
A high level of ALP (not BAP) was a risk factor for non-bone metastasis (mainly liver metastasis). As presented in Online Resource Figure S6, non-bone metastasis was compared in five studies. Combined results showed that a high ALP level was associated with non-bone metastasis (OR 8.32; 95% CI 1.57, 44.21; I2 = 91%; P < 0.001, random-effects model). As shown in Online Resource Figure S7, eight studies compared ALP levels in the BC non-bone metastatic group with the BC without metastasis group. The combined result showed high ALP levels in the metastatic BC group (P < 0.001; I2 = 98%, random-effects model). Only one study reported the BAP result and no difference was found. As presented in the Online Resource Figure S8 of the subgroup analysis based on the design of cohort studies versus non-cohort studies found that the study design had little influence on the risk effect of ALP levels for non-bone metastasis in BC.
Sensitivity analysis
As shown in Online Resource Figure S9, a sensitivity analysis was performed by omitting one study at a time and calculating the pooled HRs for the primary outcome of ALP prognostic value for OS. Results revealed that no individual study significantly changed the pooled HRs, which confirms the credibility of the meta-analysis. The sensitivity analysis for the risk effect of ALP/BAP for bone metastasis is shown in Online Resource Figure S10 and Figure S11.
Publication bias
Funnel plots of the primary outcome of ALP prognostic value for OS is presented in Online Resource Figure S12. The shape of the funnel plots did not have obvious asymmetry. Additionally, the results of the Begg's test (P = 0.721, presented in Online Resource Figure S13) and Egger's test (P = 0.499) provided statistical evidence of funnel plot symmetry, suggesting the non-existence of significant publication bias. The funnel plot of ALP/BAP for bone metastasis is presented in Online Resource Figure S14.
Discussion
The meta-analysis was performed to summarize the prognostic value of ALP and BAP in BC. There were 53 studies with 22,436 patients included. For the primary outcome of survival, high levels of both ALP and BAP were associated with short survival time (OS and PFS/DFS). For the secondary outcomes, a high ALP level (not BAP) was detected in BC compared with healthy controls, and high levels of both ALP and BAP were risk factors for bone metastasis, while ALP (not BAP) was a risk factor for non-bone metastasis. Thus, the biomarkers could clinically be used as early diagnostic assessments and monitoring biomarkers in the follow-up of BC patients.
BC is the most common female malignancy worldwide. Metastases, especially bone metastases—which usually occur at an early stage—play an important role in the prognosis of the malignancy.4–6 It is reported that approximately 60%–75% of metastasis in BC could be diagnosed as bone metastasis which significantly reduces 5-year survival and causes serious complications. 5 Therefore, early diagnostic assessment and treatment of metastases—especially bone metastases—is required, 6 and biomarkers are crucial. In the present meta-analysis, the statistical results showed that the serum markers of ALP and BAP can help to monitor the disease and metastases.
ALP is a group of phosphatidylinositol-anchored membrane proteins with wide substrate specificity. It comprises a group of enzymes that catalyze the hydrolysis of phosphate esters in an alkaline environment, generating an organic radical and inorganic phosphate. 42 Like other enzymes, ALP has many isozymes. In healthy adults, this enzyme is mainly derived from the liver and bones, and in lesser amounts from the intestines, placenta, kidneys, and leukocytes. Elevated serum ALP is predominantly seen with more specific disorders, including bile duct obstruction, bone disease, hepatitis, malignancy (liver, bone, breast, lymphoma), etc.9,42,54 BAP is one of the ALP isozymes that is produced by osteoblasts during the bone formation phase by clipping off the membrane and releasing it into circulation. It is produced in extremely high amounts and is, therefore, an excellent indicator of bone formation activity. 33 In the present meta-analysis, statistical results showed that in BC patients high serum levels of both ALP and BAP are risk factors for bone metastasis and associated with short survival times. Furthermore, ALP is a risk factor for non-bone metastasis, especially in liver metastasis.
To our knowledge, our study is the first systematic analysis to summarize the prognostic value of ALP and BAP in BC. However, previous meta-analyses exploring the value of ALP and BAP in various malignant carcinomas showed some consistency with our results. Du et al. 55 in 2014 reported that BAP was a biomarker for bone metastasis in various malignant carcinomas. Ren et al. 56 in 2015, Hao et al. 57 in 2017 and Gu and Sun 58 in 2018 have reported that high ALP is associated with poor survival and metastasis in osteosarcomas. In 2015, Zhao et al. 59 reported that high BAP was associated with bone metastases in lung carcinomas. Li et al. 60 in 2018 and Mori et al. 61 in 2020 reported that high ALP was associated with poor survival in prostate cancer. In 2020, Sun et al. 62 in 2020 reported that high ALP was associated with poor survival in hepatocellular carcinoma. All the above meta-analyses were partly consistent with our results.
Our meta-analysis has several limitations. First, most of the included studies were retrospective observational studies that were sensitive to selection bias, performance bias, and detection bias—especially publication bias and reporting bias—and the quality of the included studies was relatively low. Second, because different ELISA kits were used to detect the ALP/BAP levels and the normal range varied greatly among the included studies, SMD was utilized to compare the effects among the studies. The exact predictive effects of ALP/BAP levels according to different ELISA kits still need further research. Third, heterogeneity was relatively high among the included studies caused by the variability in population, ALP/BAP measurements, BC treatments, and follow-up periods. Although efforts were made through subgroup analysis, the heterogeneity could not be eliminated, and the random-effects model was chosen over the fixed-effects model. Lastly, because the present study was focused on the prognostic value of bone metastasis and survival, further meta-analysis is still needed to evaluate the diagnostic value (sensitivity and specificity) of ALP/BAP levels for bone metastases and non-bone metastases. All results of this review should be interpreted within the above limitations.
Conclusion
This meta-analysis has provided an overview of current knowledge concerning the predictive effects of ALP and BAP in BC. By the statistical evidence of subgroup, sensitivity, and publication bias analyses, the credibility of this meta-analysis could be confirmed. There were 53 studies with 22,436 patients included. For the primary outcome of survival, high levels of both ALP and BAP were associated with short survival time. For the secondary outcomes, high ALP levels (not BAP) were detected in BC compared with healthy controls, and high levels of both ALP and BAP were risk factors for bone metastasis while ALP (not BAP) was a risk factor for non-bone metastasis. Thus, clinically, the biomarkers could be used as early diagnostic assessments and monitoring biomarkers in the follow-up of BC patients.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231154662 - Supplemental material for Prognostic value of alkaline phosphatase and bone-specific alkaline phosphatase in breast cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-1-jbm-10.1177_03936155231154662 for Prognostic value of alkaline phosphatase and bone-specific alkaline phosphatase in breast cancer: A systematic review and meta-analysis by Chengying Jiang, Fangke Hu, Xiaoqing Xia and Xiaojing Guo in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155231154662 - Supplemental material for Prognostic value of alkaline phosphatase and bone-specific alkaline phosphatase in breast cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-2-jbm-10.1177_03936155231154662 for Prognostic value of alkaline phosphatase and bone-specific alkaline phosphatase in breast cancer: A systematic review and meta-analysis by Chengying Jiang, Fangke Hu, Xiaoqing Xia and Xiaojing Guo in The International Journal of Biological Markers
Footnotes
Acknowledgements
None.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Chengying Jiang, Fangke Hu, and Xiaoqing Xia. The draft of the manuscript was written by Chengying Jiang and Fangke Hu. Chengying Jiang and Fangke Hu contributed equally to this work. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Tianjin Key Medical Discipline (Specialty) Construction Project, (grant number 81772840, TJYXZDXK-012A).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
