Abstract
Background:
Periprocedural prophylaxis in medicine encompasses the set of measures (physical, chemical, and pharmacological) used to reduce the risk of infection. Antibiotic prophylaxis (AP) refers to the administration of a short-term regimen of antibiotics shortly before a medical procedure to reduce the risk of infectious complications that can result from diagnostic and therapeutic interventions. The outspreading growth of multidrug-resistant bacterial species and changes in the bacterial local ecosystem have impeded the development of a unique scheme of AP in urology.
Objectives:
To review the literature and current guidelines regarding AP for urological diagnostic and therapeutic procedures, and to define agents, timing, and occasions when administering pharmacological prophylaxis. Secondly, according to current literature, to open new scenarios where AP can be useful or useless.
Results:
Major gaps in evidence still exist in this field. AP appears useful in many invasive procedures and some sub-populations at risk of infectious complications. AP is not routinely recommended for urodynamic exams, diagnostic cystoscopy, and extracorporeal shock-wave lithotripsy. The available data regarding the use of AP during the transperineal prostate biopsy are still unclear; conversely, in the case of the transrectal approach AP is mandatory. AP is still considered the gold standard for the prevention of postoperative infective complications in the case of ureteroscopy, percutaneous nephrolithotomy, endoscopic resection of bladder tumor, endoscopic resection of the prostate, and prosthetic or major surgery.
Conclusion:
The review highlights the complexity of determining the appropriate candidates for AP, emphasizing the importance of considering patient-specific factors such as comorbidities, immunocompetence, and the nature of the urologic intervention. The evidence suggests that a one-size-fits-all approach may not be suitable, and a tailored strategy based on the specific procedure and patient characteristics is essential.
Keywords
Introduction
According to the Center for Disease Control and Prevention (CDC), healthcare-acquired infections (HAIs) are defined as any infection that arises in an inpatient or outpatient setting and appears 48 h after hospitalization or within 30 days after receiving healthcare assistance. 1
HAIs represent the sixth cause of death worldwide (100,000 deaths per year).2,3 Surgical site infections (SSIs) are defined as every type of infection that hits the patient at or near the surgical wound within 30 days of an operative procedure or within 1 year since device implantation. They can be divided into superficial incisional, deep incisional, and organ/space incisional. 2 Urinary tract infections (UTIs) and SSIs cause most HAIs. 4
Periprocedural prophylaxis in medicine, specifically in urology, represents the ensemble of measures (physical, chemical, and pharmacological) used to reduce the risk of infection. Non-antibiotic measures must be performed during the hospitalization and convalescence period to improve the effectiveness and reduce the complication rate of a urological treatment.5–7
Antibiotic prophylaxis (AP) consists of administering a short-term regime of antibiotics before or at the beginning of a medical procedure to reduce the risk of infectious complications resulting from diagnostic or therapeutic interventions. Even if AP does not eliminate microorganisms, it reduces the microbial burden at surgical sites, allowing the host’s immune system to manage any remaining bacteria. 8
The absence of well-conducted and statistically significant studies, especially in the cases of endoscopic procedures, has impeded the development of a proper scheme of AP in urology. This lack of strength led to divergences regarding the definition and evaluation of risk factors and substantial disparities in treatment protocols and therapy selections. Rigorous scientific evidence-based recommendations are strongly required in this domain. In the last 20 years, urologic surgery reduced surgical invasiveness (from open to endoscopic, laparoscopic, and robotic surgery) while the patients’ mean age and frailty increased. These changes may have an impact on AP policies. 9
AP is not designed to treat any post-procedural infections. The ideal approach is to administer specific therapy with appropriate dosages using antibiotics based on factors such as the type of infection, the patient’s clinical characteristics, and the epidemiological context, particularly in the case of nosocomial infection. 10
Although the rationale for using antibiotics in prophylaxis is widely acknowledged, inappropriate use can have detrimental effects on the patient (unexpected side effects, increased risk of infection), on the environment (increased resistance), and on the health system (waste of financial resources). 9 It is crucial to emphasize that AP represents a supplement to a good surgical technique in an aseptic setting and good hygiene, not their replacement. An effective strategy for controlling acquired infections must include prevention as one of its key elements.
This literature review aims to highlight the value of AP in urologic diagnostic and therapeutic procedures and to examine the following main elements of this topic:
Materials and methods
A literature review was conducted in October 2023, and no temporal limits were adopted. The search was performed on Pubmed and Web of Science databases, using the following keywords (title and abstract): “antibiotic prophylaxis” and “urologic surgery” or “urologic procedures” or “urologic interventions” or “urology” or “urodynamics” or “cystoscopy” or “stent removal” or “ureteral stent” or “urinary catheter” or “urethral catheterization” or “prostate biopsy” or “transrectal prostate biopsy” or “transperineal prostate biopsy” or “extracorporeal shock wave lithotripsy” or “ureteroscopy” or “percutaneous nephrolithotomy” or “TURB” or “TURBT” or “bladder resection” or “TURP” or “prostatectomy” or “nephrectomy” or “cystectomy” or “artificial urinary sphincter” or “penile prosthesis” or “sling” or “urological prosthetic device.” References of retrieved articles were also manually searched for other relevant citations. In addition, European Association of Urology (EAU) and American Urological Association (AUA) guidelines were consulted to identify specific recommendations for each urologic procedure. Evidence was pooled and compared with the results of articles, meta-analyses, and randomized trials. The evidence synthesis was conducted descriptively.
Aims and principles of antibiotic prophylaxis
The main purpose of AP is to reduce the risk of infections and to decrease antibiotic overuse, reducing postoperative complications and controlling the spread of antibiotic resistance. 11 AP can be useful, but it can also lead to adverse events, multidrug resistance, and more dangerous community-acquired infections.12,13
AP does not eliminate microorganisms from tissues but decreases microbial burden at the surgical site to a level that the host’s immune system can effectively manage. Bacterial contamination at the surgical site is inevitable due to the patient’s colonization and the surrounding environmental bacterial flora. 14
Different antibiotics should be administered when designing a prophylactic treatment depending on skin wound risk of infection, local pathogen prevalence, local resistance profiles and rates, patient drug-related allergies, and comorbidities. Ideally, pharmacological agents designed for AP should not be the same used to treat the possible after-treatment infection. 14
Risk factors
In some cases, due to factors linked to the patient, the microorganism, or environmental factors, the risk of contracting HAIs is high. Overall, risk factors can be classified into three different groups:
Patient’s condition according to the American Society of Anesthesiologists (ASA) score [from ASA-1 to ASA-6];
Patient’s general risk factors according to age, diabetes mellitus, immune-compromisation, malnutrition, altered weight (obesity or underweight);
Exogenous or endogenous risk factors: history of urological infections, use of a permanent urinary catheter, presence of bacteriuria and its urinary load, previous operations, genetic factors, urinary anatomic malformations.
The type of urological procedure can also be linked to an increased risk of infection. The National Nosocomial Infections Surveillance (NNIS) System risk score combines the ASA score, the duration of the surgical procedure, and the Altemeier classification (used to stratify the surgical procedures and wound infectious risk 15 ) to establish the postoperative infectious risk. 16
As the SSIs risk varies largely depending on multiple factors, it is extremely difficult to build a universal infection risk score and to build reliable recommendations for reducing the infection rate.
Recently, numerous scores have been proposed as indicators of the SSIs risk in every surgical area, to minimize the abuse of antibiotics, stop the spreading of antibiotic resistance, and identify the right patient to treat.17–19
Transurethral procedures are risk factors for postoperative infections themselves, moreover, urines are not considered sterile. As a result, the EAU guidelines classify every endoscopic procedure with a transurethral approach as “clean-contaminated.”20,21
Antibiotic selection and administration route
No clear indications are given in current EAU guidelines regarding agents used in AP. Ideally, the antibiotic agent used for AP should differ from those required for the treatment of the infections. 21
Target AP represents a topic of current debate. This process of care has been formally indicated in the case of transrectal ultrasound prostate biopsy22,23 while has not been assessed prior to other urological procedures. In high-risk patients, it would be useful to perform a preoperative urine culture to direct the AP.
When selecting an antibiotic for prophylaxis, attention should be given to adverse effects and allergy profiles. All antibiotics have the potential to cause an allergic reaction. β-Lactam allergies are some of the most frequent drug-related reactions. 24
Nowadays, the use of fluoroquinolones (FQs) in AP is becoming wider thanks to an undemanding oral and intravenous administration, a favorable oral bioavailability and safety profile, relatively low cost, and high penetration into the genitourinary (GU) system. On the other hand, their overuse has increased the resistance rate worldwide, especially among typical urologic pathogens like Escherichia Coli.25,26
The evaluation of local bacteria epidemiology and susceptibility profile is of utmost importance and should be performed in all healthcare facilities to identify the correct agent to administer.27,28
When the risk of negative outcomes in the case of SSIs is low or minimal, despite the presence of scant data on the topic, a good choice can be represented by an aminoglycoside or a second-generation cephalosporin, as an alternative.29,30
No pieces of advice are given in European guidelines about the type of drug to use in high-risk procedures. Regarding the administration route, if an antibiotic agent possesses adequate bioavailability, it can be administered either orally or intravenously; no differences in efficacy are shown. 11
Timing and duration
According to the current EAU guidelines, AP should be administered 1–2 h before the procedure when recurring to an orally administered drug, while it can be given at the time of induction of anesthesia if administered intravenously. 31
Duration should be reduced to the minimum, and continuing AP after the end of surgery should be avoided. Although a single dose of AP is preferred, duration should be administered depending on the procedure’s duration, the antibiotic’s half-life, and if significant blood loss occurs during surgery.14,32,33
Diagnostic procedures
Urodynamics
A urodynamic study is a diagnostic, minimally invasive procedure used to investigate low urinary tract symptoms (LUTS) or pelvic floor dysfunction. 34 Using transurethral and rectal catheters, it measures the vesical volume and compliance, both abdominal and vesical pressures, and uroflow parameters. The occurrence of postoperative UTIs (symptomatic or asymptomatic) is variable, depending on several factors, ranging from 1.5% to 36%. 35
In a systematic review published in 2008, the administration of AP reduced the risk of significant bacteriuria when compared with no treatment [OR 0.39, 95% CI 0.24–0.61]. 36
According to a recent Cochrane review that compares the administration of AP with a placebo, no significant difference was observed regarding the risk of UTIs [RR 0.73, 95% CI 0.52–1.03], while the overall risk of asymptomatic bacteriuria (ASB) decreased in the treatment group [RR 0.35, 95% CI 0.22–0.56]. 37
Accordingly, Benseler et al. reported that AP could reduce the risk of bacteriuria but not clinical UTIs following urodynamic studies in women. 38
Conversely, a recent meta-analysis shows that AP usage, when compared with the placebo or no treatment, has a statistically significant impact in preventing not only ASB [RR 0.42, 95% CI 0.30–0.60] but also symptomatic UTIs [RR 0.65, 95% CI 0.48–0.88]. In addition, there was no statistically significant difference in the risk of adverse events between the AP group and no treatment group [RR 4.93, 95% CI 0.61–40.05]. 39
According to the most up-to-date EAU and AUA guidelines, AP should not be performed for simple outpatient urodynamics procedures.21,40
AUA guidelines mentioned some factors that appeared to increase the SSIs risk of urodynamic procedure (relevant GU anomalies, diabetics, prior GU surgery, a history of recurrent UTIs, post-menopausal women, recently hospitalized patients, patients with cardiac valvular disease, nutritional deficiencies, or obesity). The investigators, with low levels of evidence, suggested that there was an increased risk of infection for patients with neurogenic lower urinary tract dysfunction, outlet obstruction or an elevated post-void residual volume, frailty, indwelling catheters, or on clean intermittent catheterization. 40 AP in these higher-risk settings should consider trimethoprim-sulfamethoxazole. Alternatives include first- or second-generation cephalosporins, amoxicillin/clavulanate, or an aminoglycoside ± ampicillin. When indicated, a single oral dose given within an hour before the procedure is sufficient, although it depends on the agent’s oral pharmacokinetics. Oral dose was the route chosen in nearly all reviewed studies. 40
Cystoscopy
Cystoscopy is a common diagnostic procedure that involves the insertion of a thin (16–22 Ch), flexible, or rigid tube with a camera (cystoscope) into the bladder through the urethra. It is routinely performed to investigate hematuria, recurrent UTIs, LUTS, and for bladder tumor surveillance. 41 A working channel allows interventions such as bladder biopsies and diathermy. Additional applications include ureteral stent insertion and removal, as well as therapeutic interventions such as botulinum toxin injections. 42
Several past studies demonstrated the presence of ASB after cystoscopy in rates ranging from 2.7% to 7.5%.43,44 Some more recent meta-analyses measured the rate of symptomatic UTIs between the AP group compared with placebo or no treatment group. Results showed the reduction of symptomatic UTI episodes in the AP group (both in Carey et al. ’s [RR 0.34, 95% CI 0.27–0.47] 45 and Garcia-Perdomo et al. study [RR 0.53, 95% CI 0.31–0.90] 46 ).
In addition, Zeng et al. 47 evaluated the outcome of systemic UTI, showing no effect of AP compared with placebo or no treatment [RR 1.12, 95% CI 0.38–3.32]. The increased antibiotic resistance was observed as a secondary outcome [RR 1.73, 95% CI 1.04–2.87], with a very low level of evidence.
According to EAU and AUA guidelines, no use of AP is required in the case of routinary cystoscopy in the absence of infectious signs and symptoms.21,40
Stent removal and catheterization
After stone surgical treatments, indwelling ureteral stents are frequently used to ensure adequate drainage. Finally, they are extracted cystoscopically within a few weeks. 48 There is still considerable disagreement on using single-dose AP in cases involving indwelling catheters and stents beyond the immediate perioperative period. So far, limited high-quality literature is available.
The administration of AP during catheter removal has been a cornerstone in urological practice.49,50 A meta-analysis revealed that patients may benefit from AP at the time of catheter removal, as the incidence of symptomatic UTIs was much lower. The calculated number needed to treat was 17, indicating a favorable outcome in preventing UTIs. 51 In most of the analyzed studies the risk of selection and attrition bias was high, and the quality of evidence was inconsistent. However, as demonstrated by a study of renal transplant recipients, AP should be performed in high-risk sub-populations. 52
In contrast with the former statements, the AUA guidelines did not recommend AP for catheterization or catheter substitutions. 40
Given the high prevalence rate of carriers of multidrug-resistant bacteria (such as E. coli ESBL+), investigations based on local population and ecology should be used to determine the true cost-benefit profile. The uncontrolled use of AP in case of catheter or stent removal would have detrimental consequences on resistance selection and adverse effects.
Antimicrobials are not indicated for the reduction of SSIs in case of catheterization during the postoperative period as they do not reduce the risk of catheter-associated urinary tract infection. 40
AP may be considered at the time of clinical procedures such as ureteral stent or nephrostomy tube placement, especially when risk factors are present.
When intraoperative circumstances change and a wound is considered contaminated, a shift up in AP coverage should occur. For example, if purulence is discovered at the time of a routinary stent exchange, then cultures should be obtained, and the antimicrobial agent(s) should continue until the culture results are known. 40
A Randomized Controlled Trial (RCT) of 58 patients by Bradshaw et al. found a low risk of infectious complications during cystoscopic stent removal, regardless of the use of AP. Urine culture positivity rate before stone surgery (16.7% vs 11.8%, p = 0.819) and at the time of stent removal (16.0% vs 11.1%, p = 0.648) was not significantly different in treatment versus control groups, respectively. The postprocedural infection risk profiles would seem to change intuitively from more invasive cystourethroscopic procedures to the minimally disruptive nature of stent removal. 48 However, this study had a very small sample size and therefore represents a low level of evidence. EAU guidelines do not provide recommendations regarding catheterization, ureteral stent, and nephrostomy tube management. 21
Further studies are needed to clarify the role of AP in these urological settings.
Prostate biopsy
Biopsy of the prostate is a urological procedure used to definitively diagnose the presence of a prostatic tumor. It can be performed using a transrectal or transperineal approach. 53
According to EAU guidelines, 21 the transrectal approach should be abandoned in favor of the transperineal approach, as in the case of transrectal biopsy the risk of infection [RR 2.48, 95% CI 1.47–4.20] and the sepsis rates (transrectal 0.9% vs transperineal 0.1%) increase significantly.54,55
Beyond these guidelines, global consensus is missing in this topic. During the annual AUA meeting in April 2023, results from the first RCT showed no differences in terms of composite infections rate over 30 days and noninfectious complications between transrectal and transperineal arms [RR 1.06, 95% CI 0.43–2.65]. 56
However, there is still a hot debate on this topic and for a stronger recommendation it is necessary to wait for the results of ongoing RCTs (NCT04815876; NCT05179694).57,58
Transrectal prostate biopsy
During the transrectal prostate biopsy, the needle is inserted in the rectum, approaching the prostate from behind. This procedure allows bacteria to migrate from the lower intestine to the urinary tract, resulting in an increased risk of infection and sepsis. Even if its use is not recommended, it is still performed.
Regarding the non-pharmacologic prophylaxis, rectal povidone-iodine preparation before the biopsy is mandatory to reduce by half the rate of infectious complications [RR 0.47, 95% CI 0.36–0.61].59,60
No differences were found in terms of risk of infection regarding needle guide type, needle type, needle size, or number of anesthetic injections for nerve-blockage. 54
Rectal culture should be performed to avoid infective complications following the procedure and a targeted prophylactic treatment should be started. In the study of Tops et al. 61 patients were divided in a control group (who took ciprofloxacin empirically) and in a treatment group (antibiotic choice was based on the preoperative rectal culture and antibiogram). Patients who underwent a rectal culture sample collection showed a statistically significant reduction in infectious complication rates when compared to empirically treated patients [OR 0.52, 95% CI 0.27–0.99].
According to the literature, AP should be performed in case of transrectal prostate biopsy, allowing the patient to reduce the risk of serious infections and systemic sepsis. 62 Whereas FQs are considered the prophylactic gold standard in the case of prostate biopsy, a recent South Korean study showed that fosfomycin has a similar impact on preventing an infectious onset. 63
EAU guidelines recommend the use of target prophylaxis based on rectal swabs or stool culture for transrectal biopsy. An alternative is augmented prophylaxis (two or more different classes of antibiotics) without FQs or alternative antibiotics (e.g. fosfomycin trometamol, cephalosporin, aminoglycoside). The use of FQs should be avoided in favor of other antibiotic classes as a result of the European Commission decision on EMEA/H/A-31/1452. 21
According to the AUA guidelines, the risk of SSIs and sepsis in healthy individuals is considerable with transrectal prostate biopsy; as such, AP is mandatory. No further information is provided by the American panel. 40
Transperineal prostate biopsy
A recent study by Castellani et al. 64 showed no association between AP and reduction of infectious complications [RR 2.09, 95% CI 0.54–8.10].
Accordingly, a randomized, open-label, non-inferiority trial directed by Jacewicz et al. demonstrated that AP could be omitted in patients undergoing transperineal prostate biopsy; however, high-risk patients were not included in the study. 65
A smaller RCT in 85 patients showed no differences in terms of UTI or sepsis rates when performing or not performing AP before transperineal prostate biopsy. 66
These results are in line with another meta-analysis that showed no differences in terms of post-procedural infections between patients who received AP and patients who did not. 67
Differently, Benli et al. demonstrated that AP, especially when targeted and based on a rectal swab performed some days before the biopsy, could effectively prevent infective complications following transperineal prostate biopsy. 68
Interestingly, research on single-dose antibiotic prophylaxis performed before transperineal biopsies showed that it could lead to changes in gut microbiota composition. 69
According to EAU guidelines, prostatic biopsy should be performed using a transperineal approach due to the lower risk of infective complications. Routine surgical disinfection of perineal skin should be performed. Although several studies reveal that AP may not be required when performing the transperineal prostate biopsy, the EAU Panel chose to wait for other ongoing RCTs on the topic before elaborating a statement. No clear evidence is reported about the use of target prophylaxis based on rectal swabs or stool culture. 21
The AUA guidelines provide no recommendations regarding AP in transperineal biopsy. 40
Therapeutic procedures
Extracorporeal Shock-Wave Lithotripsy
The extracorporeal Shock-Wave Lithotripsy (ESWL) is a non-invasive option for treating renal lithiasis. Several studies investigated the effect of AP in patients undergoing this procedure, either with or without preoperative bacteriuria. 70
In the study of Lu et al., no association was found between AP and the rate of fever [RR 0.36, 95% CI 0.07–2.36], rate of positive urine culture [RR 0.77, 95% CI 0.54–1.11], the incidence of UTIs [RR 0.54, 95% CI 0.29–1.01] in patients with pre-procedural sterile urines. 71 Mrkobrada et al. showed no evidence of an association between AP and the risk of post-procedural UTIs [RR 0.76, 95% CI 0.39–1.48] and indicated that, according to the Canadian Urological Association (CUA) Guidelines Committee, AP could be considered in patients at high risk of infectious complications. 72 Hsieh et al. evidenced that no benefits in terms of postoperative pyuria, bacteriuria, and symptomatic UTIs were shown in patients who took levofloxacin orally as a prophylactic agent. 73
In conclusion, no study showed a benefit in performing AP in patients undergoing ESWL. No AP is needed before or during this procedure according to EAU guidelines. 21 According to the AUA guidelines, ESWL does not require AP if the pre-procedure urine culture is negative.40,74,75
Ureteroscopy and endoscopic lithotripsy
Ureteroscopy is an endoscopic procedure that allows the surgeon to investigate and treat different conditions at the level of the ureter and renal pelvis. It can be used as a diagnostic or a treatment procedure, either for lithiasis and urothelial tumors of the upper urinary tract (UTUC).
A meta-analysis by Deng et al. 76 evidenced that AP could have an impact on pyuria [OR 0.42, 95% CI 0.25–0.69], and ASB [OR 0.25, 95% CI 0.11–0.58], but it does not affect UTIs [OR 1.00 95% CI 0.26–3.88].
When comparing FQs and fosfomycin as prophylactic agents, two RCTs showed similar effects on the risk of systemic inflammatory response syndrome (SIRS).77,78 In both articles, a benefit regarding the risk of infection was observed in the group that recurred to AP.
Due to the lack of high-quality papers regarding the use of AP in ureteroscopy, the recommendation about AP in EAU’s latest guidelines is weak. 21 More data should be collected.
In the AUA guidelines ureteroscopy is considered a clean contaminated procedure and no benefit of AP has been demonstrated. Selective use of AP for higher-risk individuals is encouraged. 40
Urine culture is not mentioned in European Guidelines. Its role in the prevention of UTIs has been analyzed by El-Agamy et al. 79 A randomized comparison was performed between standard AP and enhanced prophylactic measures using urine cultures and targeted pharmacological therapy. Results demonstrated that an antibiotic-culture-based therapy could significantly reduce the risk of UTIs in patients undergoing ureteroscopy when compared with a traditional empirical antibiotic.
Percutaneous nephrolithotomy
Percutaneous nephrolithotomy (PCNL) is currently the gold standard treatment for kidney stones larger than 2 cm. 80 Despite its efficacy, PCNL is associated with complications including urinary leakage, blood transfusion, postoperative fever, and sepsis, 81 and shares the same risk factors for infectious complications with therapeutic ureteroscopy.
Charton et al. 82 observed that: bacteriuria developed in 35% of cases and fever without bacteriuria developed in 10% of patients post-PCNL who did not receive AP. They also showed that 32% of patients required postoperative antibiotic therapy due to increased transitory fever, and 3.5% developed sepsis with the need for antibiotic therapy.
In their two RCTs, Fourcade 83 and Doğan et al. 84 compared patients who underwent AP with those untreated. Findings revealed a substantial reduction in infectious complications among patients who were offered AP, regardless of the choice of antibiotic.
Several RCTs comparing different antibiotic regimes are available: no differences in SIRS or sepsis rates were observed between patients receiving sulbactam/ampicillin versus cefuroxime 85 ; or ceftriaxone versus ceftriaxone plus an oral third-generation cephalosporin 86 ; or ceftriaxone versus cefazolin 87 ; or ciprofloxacin versus cefotaxime. 88
A large meta-analysis of 13 comparative studies (1549 patients) 89 revealed that a single dose of AP administrated before surgery significantly decreased postoperative sepsis and fever rates [OR 0.31, 95% CI 0.20–0.50 and OR 0.26, 95% CI 0.14–0.48, respectively]. Additionally, preoperative AP reduced the rate of positive pelvic urine cultures and positive stone cultures. No difference in sepsis rate between patients receiving or not receiving postoperative prophylaxis was observed. 89
Jung et al. in a meta-analysis of 10 articles, found no significant differences between single-dose and extended-dose of antibiotics regarding the rate of fever [OR = 0.96, 95% CI 0.44–2.13]. On the other hand, the incidence of SIRS was lower in the extended-dose group compared to the single-dose group, especially in high-risk patients [OR = 1.81, 95% CI 1.30–2.53]. The impact of therapy duration was not assessed and in the included studies the duration varied too much. 90
Danilovic et al. 91 showed in a recent meta-analysis that a one-week preoperative cycle of oral antibiotics was a protective factor from developing SIRS/sepsis [OR = 0.37, 95% CI 0.23–0.53]. Patients who received preoperative antibiotic therapy had lower positive intraoperative urine culture and stone culture than the control group.
These studies moderately evidenced that a single dose of antibiotics can be effective and sufficient, however, in patients at high risk for post-PCNL infections, the use of extended doses may be required.
According to the EAU and AUA guidelines, only a single-dose AP is strongly recommended to reduce the rate of clinical UTIs.21,40
Transurethral resection of the prostate
Transurethral resection of the prostate (TURP) is widely recognized as the gold-standard surgical intervention for the management of benign prostatic hyperplasia. Infectious complications are particularly increased in males with preoperative bacteriuria. 92 Postoperative UTIs and urosepsis represent common complications, with incidence rates ranging from 6% to 13% and 4% to 12%, respectively.93,94
Berry and Barratt 95 systematically reviewed 32 RCTs (4260 patients), demonstrating that AP use in patients with preoperative sterile urine undergoing TURP significantly decreased the bacteriuria incidence from 26% to 9% [RR reduction 65%, 95% CI 56–73] and the clinical septicemia incidence from 4.4% to 0.7% [RR reduction 77%, 95% CI 55–88].
Qiang et al., 96 in a systematic review including 28 RCTs (4694 patients) showed similar results. Specifically, AP was significantly more effective than placebo in reducing postoperative TURP complications such as bacteriuria, high-degree fever, and bacteremia. Both reviews showed that short-term prophylaxis lasting more than 72 h is more effective than a single dose but less effective than therapy administered for a week.
Dahm and Dmochowski 97 confirmed the abovementioned results in a systematic review of 39 RCTs. The use of AP compared to placebo showed a RR reduction for sepsis [RR 0.51, 95% CI 0.27–0.96], for fever [RR 0.64, 95% CI 0.55–0.75], and for bacteriuria [0.37, 95% CI 0.32–0.41].
Postoperative bacteriuria may be prevented by careful hemostasis, prevention of postoperative catheter disconnections, and limitation of the duration of postoperative catheterization. 98 In summary, there is significant evidence acquired from high-quality research indicating that AP for TURP effectively decreases the rate of postoperative bacteriuria, fever, and sepsis.
EAU guidelines recommended the use of AP to reduce the rate of infectious complications in males undergoing TURP. 21
A single dose of AP is also indicated in the AUA guidelines, and, in addition, the antibiotic agent choice requires knowledge of the urine culture results, the local antibiogram, and the patient’s associated risks. 40
Transurethral resection of bladder tumor
Transurethral resection of bladder (TURB) tumor is one of the most frequently performed urological procedures. Despite its widespread use, there is a lack of high-quality evidence evaluating AP in TURB.
Bausch et al., 99 in a meta-analysis including seven studies (1725 patients), showed no significant effect of AP on postoperative UTIs incidence [OR 1.55, 95% CI 0.73–3.31] and ASB [OR 0.43, 95% CI 0.18–1.04]. Age, preoperative pelvic radiation, preoperative ASB, and pyuria have been identified as risk factors for UTIs following TURB.
In an RCT conducted by Yang et al., 100 the efficacy of a single oral dose of fosfomycin 3 g administered the night before surgery was compared to the intravenous administration of 2 g of cefoxitin, given 30 min pre- and 24 h post-surgery. The oral single dose of fosfomycin was found to be non-inferior to intravenous administration of cefoxitin in the prevention of post-TURB UTIs, even in patients considered high-risk.
Baten et al., 101 in a multi-centered RCT, investigated the safety of omitting AP in TURB in patients without a preoperative indwelling catheter/double-J/nephrostomy and with a negative preoperative urinary culture. They found no significant differences between groups for developing postoperative fever (p = 0.44).
Kohada et al., 102 in a retrospective study, demonstrated that AP was effective for patients who underwent TURB; history of pelvic radiotherapy, high age, preoperative hospital stay, and large tumor size were risk factors as well as pyuria and postoperative bacteriuria.
In high-risk patients undergoing TURB (large or necrotic tumors and prolonged operation time), the EAU guidelines recommendations reflect the low level of evidence and provide a weak recommendation in favor of AP. 21 The AUA guidelines state that AP may not be necessary for simple bladder biopsies performed with periprocedural uninfected urine. No more indications are given. 40
Slings and prosthetic device implantation
The use of AP in incontinence surgery remains a topic with limited available research.
One systematic review and meta-analysis 103 was performed on UTIs prevention after synthetic midurethral slings implantation in women. Only one of the seven analyzed studies assessed AP for midurethral sling surgery and was halted early due to a low rate of infectious events. No statistically significant differences between Cefazolin and placebo group regarding wound infections [1 (3.3%) and 0 (0%)] or bacteriuria [3 (10%) and 1 (3.5%)], were found.
A registry-based cohort study 104 of 28,687 women undergoing midurethral sling implantation showed that AP led to a reduction in the occurrence of postoperative infections and extended pain after surgery but did not offer protection against tape exposure. However, the variations in the success rate were minimal and likely not significant from a clinical perspective. The authors concluded that if a slight increase in surgical site infections is considered acceptable, it may be possible to avoid the use of AP.
AUA guidelines 40 recommend AP targeting common skin flora, such as coagulase-negative staphylococci, and other gram-negative bacteria, such as Pseudomonas species, for implantation of prosthetic device surgery.
It is worth noting that recent research105,106 has indicated a decline in the overall incidence of prosthetic infections. Nevertheless, when infections do occur, relatively higher rates of anaerobic infections, methicillin-resistant Staphylococcus aureus (MRSA), and fungal infections are potentially identified.
The effectiveness of periprocedural non-pharmacological prophylactic measures has an impact on reducing colonization and the presence of positive postoperative cultures after the placement of artificial urinary sphincters. However, establishing a clear correlation with periprocedural infectious complications is challenging due to the low prevalence of SSIs. 107
Currently, there is a substantial lack of standardized practice in terms of AP for prosthetic devices and the existing evidence is relatively limited. 40 While the majority of the authors use an extended AP in this setting, other authors reported similar results with a single-dose administration for both sling and artificial urinary sphincter in males.108,109
Urgent studies are required to fill these gaps, as the risk associated with extended antibiotic courses and the use of vancomycin is notably higher compared to short-course first-generation cephalosporins. Similarly, the efficacy of wound irrigation in the absence of prosthetic infection or erosion is being actively investigated, along with methods to reduce biofilm formation. 106
Major surgery
No RCTs evidenced the role of AP in major urological surgery. 110 So far, the knowledge about AP in urology comes from other surgical specialties. The pieces of evidence about this topic should be deepened. The infectious risk is linked to the procedure:
In clean procedures involving non-infected tissues, AP is not recommended 111 ;
In clean-contaminated procedures involving non-infected opened urological structures (e.g.: radical prostatectomy, pieloureteroplasty), preoperative AP is recommended when risk factors are present112,113;
In contaminated procedures, for example, in case of intestinal manipulation during radical cystectomy, AP is mandatory. 114
Consensus exists about the minor incidence of infective complications in the case of laparoscopic or robotic procedures when compared to traditional open procedures. 115
No specific recommendations are available in the EAU and AUA guidelines regarding major surgery procedures (in which AP represents common practice).
Conclusions
AP plays a critical role in reducing the risk of HAIs in urological diagnostic and therapeutic procedures; however, scientific evidence remain scant and antibiotic resistance is a major issue.
The European CDC 14 identified five modalities that should be used when creating local policies for antibiotic prophylaxis use. All these five key points, shown in Table 1, support the harmonization of the administration of AP and increase awareness in hospitals, which would eventually lead to a decrease in antibiotic use and antibiotic resistance.
Perioperative antibiotic prophylaxis modality. Reproduced and adapted from European Centre for Disease Prevention and Control Technical Report on perioperative antibiotic prophylaxis. 14

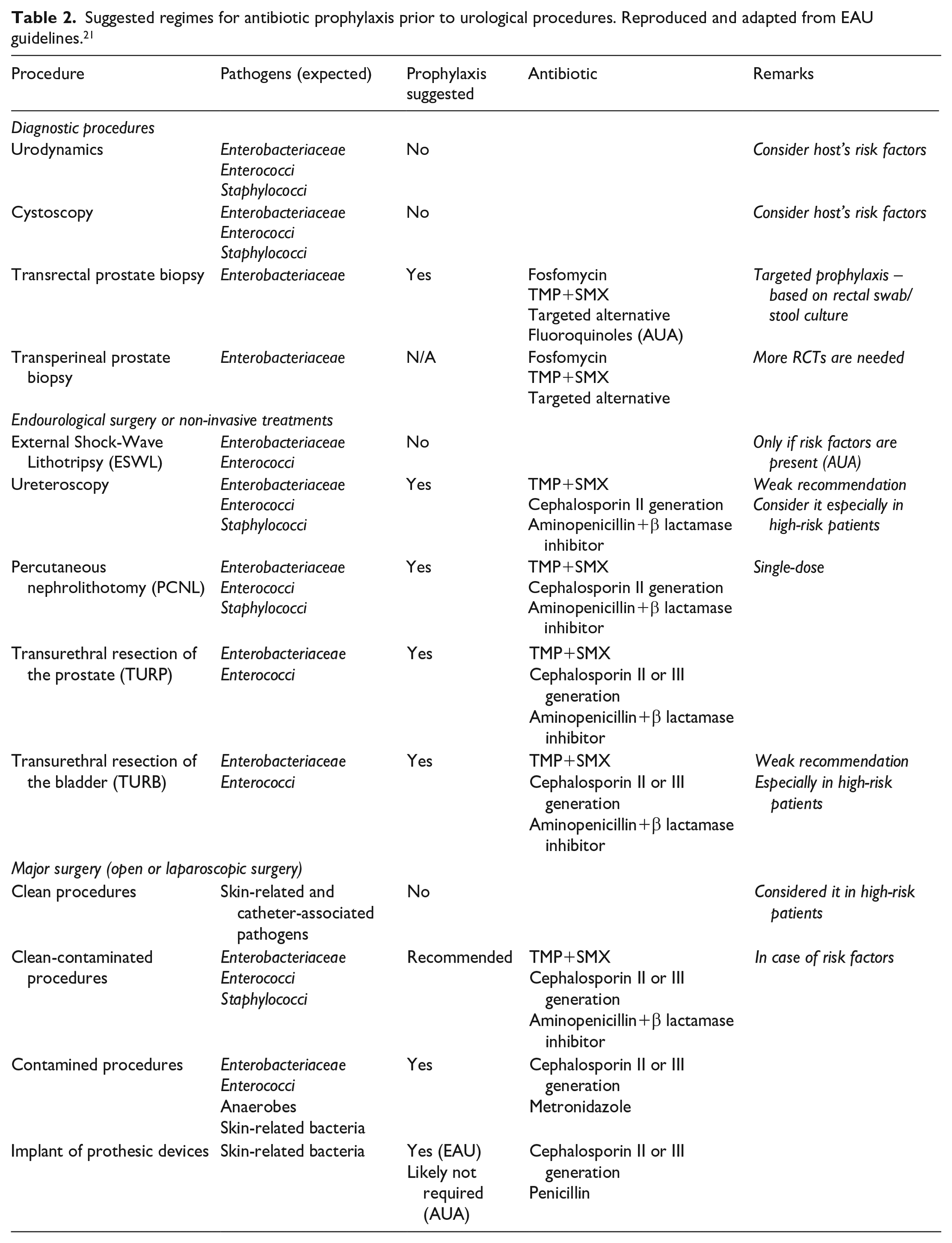
Single-dose AP is suggested before undergoing any procedure for treating benign prostatic hyperplasia, TURB, open and laparoscopic/robotic stone surgery, PCNL, endoscopic stone removal, and prostate transrectal biopsy. Currently, no strong scientific evidence supports the use of multiple doses of antibiotics when there is no symptomatic infection before surgery. Furthermore, several RCTs with moderate-quality evidence do not demonstrate any advantages in extending AP beyond the time of the surgery’s completion. 116 Table 2 summarizes evidence and recommendations for peri-procedural prophylaxis and the suggested regimes for each kind of urological procedure according to EAU guidelines. Table 3 summarizes the principal studies that were examined during the drafting of this review.
Suggested regimes for antibiotic prophylaxis prior to urological procedures. Reproduced and adapted from EAU guidelines. 21
Resuming principal articles on AP in urological procedures.

Future research should focus on refining AP guidelines and individualizing prophylactic approaches to ensure effective infection prevention while minimizing the risks of antibiotic overuse and resistance. In this regard, the DEEP-URO (DEEscalation of antibiotic Prophylaxis in UROlogical procedures) study was set in motion during the EAU23 Congress in Milan with the aim to generate high-level evidence for the appropriate use of AP in urology. 127 It aims to increase our understanding of rates and risk factors for infectious complications in urology and to determine the actual need for AP in urological procedures. The primary objective is to identify the limits of AP de-escalation by comparing 30-day infection rates (deep tissue, skin, urinary tract, and sepsis) relative to contemporary intensive AP protocols.
In summary, this review encourages a nuanced and patient-centered perspective on AP, underscoring the evidence gaps in the field and advocating for a judicious use of antibiotics to minimize the risk of infections while simultaneously addressing concerns related to antibiotic resistance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
