Abstract
Background and Aims:
Ketamine use as a recreational drug is becoming more popular nowadays. Ketamine-induced uropathy (KIU) is a late finding observed with long-term use of ketamine. A systematic review of Ketamine-Induced Uropathy was performed to emphasise its key clinical manifestations, mechanism of action and establish an effective treatment pathway.
Methods and Results:
A literature search was conducted in MEDLINE via Pubmed and Cochrane using the keywords ketamine and bladder, ketamine and uropathy, and ketamine and epidemiology. The search strategy was limited to articles published from 2000 to 2023. Both animal and human studies were included. A total of 101 papers were reviewed based on topic relevance from the title and abstracts available. While ketamine is a controlled drug in the United Kingdom (UK) and other countries, 283 ketamine-related deaths have been reported in the UK. There is no definite pathogenesis but multiple potential mechanisms that cause KIU and its related symptoms. KIU involves chronic inflammation of the bladder, ureteral wall thickening, hydronephrosis and finally, chronic renal failure. A multidisciplinary approach is paramount when managing these patients to break the vicious cycle. The mainstay of medical and surgical treatment pathways is continued abstinence to prevent symptom relapse. This review included the pathophysiology, novel medical treatments and surgical management of KIU.
Conclusion:
KIU is a rare but significantly disabling condition often seen among ketamine abusers. With the rising trend in drug addiction, KIU is expected to be more common. Unfortunately, it is a late complication in chronic ketamine abusers and is only partially reversible even with abstinence. This review discusses this rare entity’s newer medical treatments and surgical options.
Keywords
Introduction
Ketamine is a dissociative, hallucinogenic general anaesthetic agent. 1 Low cost, easy availability, friends, curiosity and psychomimetic effects have made Ketamine a desirable ‘rave’ drug.1–3 Cystitis was first reported in ketamine abusers in 2007. 4 Both upper and Lower urinary tract symptoms (LUTS) have been reported, such as increased urinary frequency, urge incontinence, painful haematuria, bilateral hydronephrosis and reduced renal function. 5 There is a perception of ketamine being a ‘safe’ drug with minimal risk of addiction. However, due to its short half-life (2–4 h), users become tolerant relatively quickly with the need to crave higher doses to reach the same experience as before.1,2 Nonetheless, Ketamine alone can kill, and the myth that ketamine-associated deaths rarely occur must be dismissed. 3 This article emphasises pathophysiology mechanisms and currently available treatment options.
Materials and methods
Literature search
A literature search was conducted in May 2023 in MEDLINE via PubMed and Cochrane databases using the keywords ‘Ketamine AND epidemiology’, ‘Ketamine AND bladder’ ‘Ketamine AND ureter’ and ‘Ketamine AND uropathy’. The search strategy was limited to articles published from 2000 until 2023.
Study selection
Two authors (TK and YEB) independently screened all articles retrieved from the literature. Discrepancies were solved by another author (DDC). Case reports, editorial letters and comments, non-English articles and abstracts were excluded. Human and animal studies were included in the review. Overall, a total of 101 papers were included based on topic relevance from the title and abstracts available. Figure 1 shows the PRISMA flow diagram for our evidence acquisition.
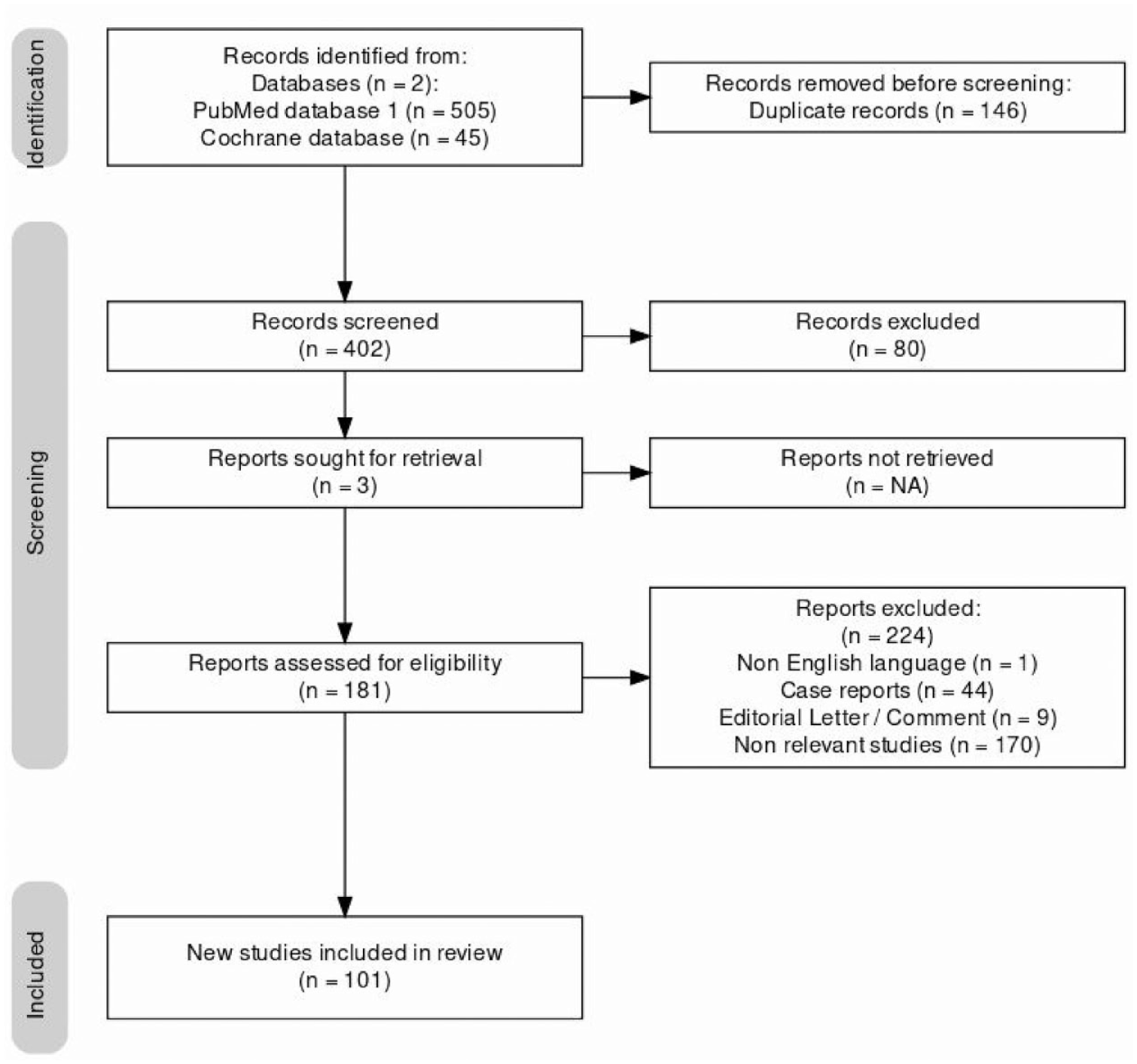
PRISMA flow diagram of the study search and selection process.
Results and discussion
Epidemiology
The use of ketamine as a recreational drug began in California in the early 1970s and was sold in the UK initially as ‘ecstasy’ during the 1990s.1,3,4 Pharmaceutical companies in the United States (US) developed a nasal form of S-Ketamine with promises in clinical trials for treatment-resistant major depressive disorder. 3 Still, the National Institute for Health and Care Excellence (NICE) in 2020 did not recommend the product for use in the UK, given uncertainties over its medical effectiveness and cost. 3
UK reclassified ketamine from class C to B in June 2014. 3 Nonetheless, from 1997 to 2020, 283 Ketamine-related deaths have been reported in England, most commonly attributed to accidental poisoning followed by hanging, accidental falls and road traffic accidents. 3
While Ketamine is a controlled drug in most countries, as per British Crime Survey (BCS) 2018/19, ketamine is the fourth most popular recreational drug in the UK after cannabis, powder cocaine and ecstasy. 6 Its use has dramatically risen to 3.1% between 2017/18 compared to 1.3% in 2016/17, especially among teenagers. 6 The precise incidence of ketamine-related urinary tract involvement is very challenging, firstly because many patients will not disclose ketamine use themselves and secondly because they will not seek medical attention until a late stage. 7 Ketamine usage rates are twice higher among males than females. 8
Clinical presentation
Sixty percent of patients diagnosed with KIU experienced increased urinary frequency, nocturia, urgency and intolerable suprapubic pain. 9 Dysuria, haematuria and urinary incontinence prevailed by 40%–50%. 9 KIU also involves chronic bladder inflammation, ureteral wall thickening, dysuria and upper urinary tract symptoms.10–12 Hydronephrosis affects 30% of ketamine patients, whereas nearly 20% presented with ureteric strictures and less than 10% with impaired kidney function. 9 Most recreational drug abusers are poly-substance users, and it is uncertain whether ketamine is directly related to urinary toxicity. 13 The timeframe required to cause irreversible damage to the urinary system remains unclear. 14 The onset of urinary tract symptoms varies from person to person to a month to years and progressively worsens at 1 year. 13
Pharmacokinetics and pharmacodynamics
Ketamine is sold in the street market as K, Special K, Kit Kat, Vitamin K, Super K, Keets, Super acid, green, Dorothy, cat valium, jet and super acid and is mainly snorted or smoked as a powder, often co-ingested with other illicit drugs.14–16 It is a structural analogue of phencyclidine extensively metabolised by the cytochrome P450 system, primarily CYP3A4 and CYP2B6, to its active metabolite norketamine. This is subsequently metabolised to hydroxynorketamines (HNKs) and dehydronorketamine.1,17 Ketamine poses analgesic, anti-inflammatory and anti-depressant effects depending on the dose, exposure time and administration route. 1
Mechanism of action
Ketamine is a non-competitive N-methyl-d-aspartate (NMDA) receptor antagonist as well as an α1 and β2 adrenergic receptor agonist. 3 The pharmacological effects of ketamine are not solely attributed to the NMDA receptors but also the hyperpolarisation-activated cyclic nucleotide (HCN) ion channels. 1
Pathophysiology of KIU
There is no definite pathogenesis but multiple potential mechanisms that cause ketamine-induced uropathy (KIU) and its related symptoms.16,18,19
Inflammatory cascade in ketamine induced uropathy (KIU)
Because ketamine and its metabolites, such as norketamine, are excreted by the urinary tract system, they interact directly with the bladder urothelium and induce immunological hypersensitivity and inflammatory cascade.12,19,20 Cells such as lymphocytes, plasma cells, mast cells and predominantly eosinophils within the denuded urothelium are commonly seen with ulceration and haemorrhage found throughout the layers of the urothelium.12,19,21
Juan et al. studied the inflammatory role of nuclear factor-kB (NF-kB) and COX-2 in response to ketamine. 22 NF-kB is a transcription factor activated by several inflammatory stimuli, and abnormal activation of NF-kB can cause the development of multiple autoimmune, inflammatory and malignant pathologies.23,24 The study found an increased expression of NF-kB and COX-2 and NF-kB translocation from cytoplasm to nucleus. 22
Jhang et al. evaluated serum IgE level, which was used as an indicator to measure immunological hypersensitivity and reported that the median serum IgE was significantly higher in patients with KC. 25 There was also a positive correlation between visual analogues scale score, bladder capacity and bladder eosinophil infiltration.19,25 Wang et al. used quantitative real-time PCR and found alterations in mRNA levels of cytokines and chemokines in the group treated with methoxetamine which is a ketamine analogue. 26 It showed considerably increased levels of pro-inflammatory cytokines such as IL-1β, IL-6, CCL-2, CXCL-1, CXCL-10, NGF and cyclooxygenase 2 (COX-2). 26
Bladder dysfunction due to muscle dysfunction and fibrosis
P2X1 purinergic receptors found on the detrusor muscle are involved in bladder contraction. 27 Meng et al. compared the two groups of mice, the control and the group treated with ketamine (100 mg/kg). 28 It found a profoundly decreased inter-contraction interval and decreased bladder capacity in ketamine treated mice group. 28 Immunohistochemical investigation also found an increased adenosine triphosphate elicited detrusor contraction with increased P2X1 receptor expression, whereas M2 and M3 receptor expression remained the same, which could contribute to detrusor overactivity in KIU and cause bladder dysfunction.19,28
Yang et al. studied transient receptor potential cation channel subfamily V (TRPV) proteins in KIU. 29 The study found that TRPV1 and TRPV4 expression was higher in those with severe KIU bladder than those with mild KC bladder or in the control group, which may contribute to small bladder capacity and stronger contractility. 29 TRPV1 expression was higher in those with reduced maximal bladder capacity, whereas there was a positive correlation between the velocity of detrusor pressure rise to maximal flow rate and expression of TRPV4. 29
Another channel that may play a role in the detrusor muscle contractility causing bladder dysfunction, such as decreased voiding pressure and bladder compliance, is the L-type calcium channel (Ca1.2).19,30 Chen et al. presented that ketamine directly inhibited calcium influx and contraction of smooth muscles by antagonising the L-type Ca2+ channel (Cav1.2). 31 They found an accumulation of several genes in the extracellular matrix, which could correspond to connective tissue fibrosis progression and calcium signalling regulation responsible for urinary bladder smooth muscle contraction. 32
In ketamine-treated mice, fibrosis was often found in the submucosal layer. 28 Wan et al. looked at the effects of synthetic ketamine analogue methoxetamine. 26 It found higher collagen I (COL I), COL III, fibronectin and TGF-β expression and visible connective tissue proliferation. 26 Another study showed that genes such as FN1, fibulin2, fibrinogen-like 2, laminin ϒ (LAMC2) and collagen type 1α2 (COL1A2) were substantially upregulated, which could be associated with the increased connective tissue in the submucosa layer. 32
Deficiency of urothelial junction-associated protein
The protective layer called the glycosaminoglycan (GAG) layer, consists of glycoproteins and proteoglycans, which are based on the bladder urothelium. 19 Yeh et al. found that the GAG layer was impaired in rats treated with intravesical instillation of ketamine. 33 Lee et al. also found that rats treated with ketamine demonstrated a profound decrease in urothelial tight junction proteins (Claudin-4 and ZO-1), adhesion protein (E-cadherin) and UPIII, which could be responsible for the defect of the urothelial-lining layer.19,34
Role of autophagy in KIU
Autophagy has a vital role in cell growth and upholding cellular homoeostasis through the degradation and recycling of impaired cytoplasmic constituents, defective and toxic accumulated proteins, and damaged and malfunctioning organelles.19,35 Mammalian target of rapamycin (mTOR) inactivation and subsequent phosphorylation of class II phosphatidylinositol 3-kinase (PI3K-III) induces autophagy. 35 Multiple autophagy-related genes oversee the development and maturation of autophagosome, and notably, microtubule-associated protein light chain 3 (LC3) accumulation on autophagosomal membrane serves as signals for autophagy. 35 Lu et al. showed that ketamine and its metabolites increased the expression of mTOR phosphorylation and markedly increased the level of Beclin 1 and LC3. 35
Investigations for KIU
(i)
(ii)
(iii)
(iv)
Treatment
A multidisciplinary team approach is essential when managing these patients to determine optimal treatment strategies.
14
Up to date, there is no single definitive treatment for KIU.
39
The numerous and unclear pathophysiology mechanisms, small number of patients and loss of long-term follow-up are the main limitations in most studies.
40
Ketamine cessation should be the very first line treatment of KC, followed by symptom control and intravesical administration of urothelium protective agents.
40
Patients who continue using ketamine fail to respond to medical or intravesical treatments leading to poor quality of life secondary to severe urge incontinence, bladder pain, upper urinary tract involvement and eventually kidney failure.
41
In most patients with severe KC, hydronephrosis and Urinary Tract Infections (UTI) may remain with reduced severity even if ketamine use has been stopped.42,43 The quality of life of female patients who abused ketamine for 2 years and more was significantly impaired (
Medical management
Several therapeutic agents have been reported for symptomatic control, including simple analgesics (e.g. paracetamol), anticholinergics, pregabalin, antibiotics, opioids and non-opioids. 4 Urinary frequency and urgency might result from non-cholinergic neurotransmitters; hence the efficacy of anticholinergics is limited. 44 Instead, active inflammation might contribute to an overactive bladder; theoretically, an anticholinergic drug is not useful. 44 Tsai et al. also reported that most patients responded poorly to these. 13 Whilst there is a lack of evidence, analgesics and anticholinergics are reasonable first-line treatments for patients with early and mild KC symptoms. 44 LUTS can be resolved by ketamine cessation before irreversible damage occurs in the urinary bladder. 13 Urinary symptoms improved in 51% of patients who stopped ketamine use. 44
Minimally invasive treatments
Intravesical administration of Hyaluronic Acid (HA) has been used in KC patients who failed oral management since its use has been supported by many studies in treating Bladder Pain Syndrome/Interstitial Cystitis (BPS/IC). 45 Only those who successfully abstained from ketamine abuse demonstrated noticeable symptom improvement. 46 Meng et al. studied the efficacy of intravesical HA instillation after hydrodistention, once weekly for 6 weeks, followed monthly for 3 months. 45 Compared to hydrodistention alone, HA and hydrodistention can provide longer but temporary relief in voiding symptoms and pain. 45 However, storage symptoms were not significantly improved, suggesting a more severe urothelial damage in KC compared to BPS/IC. 45
Similarly, 200 IU of intra-detrusor administration of Botulinum toxin A plus hydrodistention significantly improved LUTS and bladder capacity after a month in 36 patients. 19
Endoscopic treatments
Bladder Auto augmentation by Transurethral Vesicomyotomy (BATV) and Bladder Hydrodistention (BH) can be performed in patients with a curative intention and is an option between conservative and surgical management. 47 Tan et al. studied the safety and efficacy of both in 53 patients with KC, of whom 41 underwent BH and 12 BATV. 47 Indications for surgery included patients with worsening Upper Urinary system, Maximum Cystometric Capacity (MCC) <150 ml, bladder compliance <30 ml/cmH2O, refractory and/or relapse KC. 47 BATV involves longitudinal incision of the urinary bladder mucosa and detrusor with the usage of cystoresectoscope. 47 During BH, the pressure was continuously maintained at 60 cmH2O for 10 min. After 1 year of a follow-up, BATV proved superior to BH in relation to Pelvic Pain and Urgency / Frequency (PUF) symptom score and MCC. 47 The Postvoidal Urinary Volume (PVR) was significantly raised in patients who underwent BATV at 12-month follow-up compared to BH patients with no significant change at any follow-up interval. 47
Surgery for KIU
Augmentation enterocystoplasty
The efficacy of Augmentation Enterocystoplasty (AE) was studied in 14 patients with chronic ketamine cystitis (KC) and ketamine-related severe bladder pain by Chung et al. 41 The criteria for AE included a cystometric bladder capacity of <100 ml of low bladder compliance with or without VUR. 41 Ureteral re-implantation was additionally performed only in those patients with grade III-IV vesicoureteral reflux (VUR) or hydronephrosis secondary to extreme ureteral obstruction. 41 During the AE, the base of the urinary bladder and trigone were maintained. 41 No major perioperative complications were reported. 41 Hydronephrosis and VUR persisted in patients with simple AE without ureteral reimplantation. 41 Three months post-operatively, all patients’ cytometric bladder capacity increased from 50.9 ml to 309.2 ml and increased maximum flow rate (Qmax). 41 Hydronephrosis disappeared only in those who underwent AE with ureteral reimplantation. 41 Frequency urgency and small Functional Bladder Capacity (FBC) persisted until 1 month after the surgery. 41 Increased FBC was reported in 92.8% of patients, and everyone had less urinary bladder pain after 17.4 months except for a few patients who experienced persistent bladder pain and UTIs given Ketamine reuse. 41 However, their FBC remained improved compared to their baseline. 41
On the other hand, the largest by far retrospective study series performed by Sihra et al. focussed on the rate of postoperative complications after major reconstructive surgeries in ketamine patients. 14 Fourteen patients in total, underwent a major reconstruction, including cystectomies with neobladder formation, ileal conduit and augmentation cystoplasty. 14 Ten out of fourteen (71.4%) required further interventions such as nephrostomies, laparotomy with adhesiolysis and bowel resection for small bowel obstruction, and ureteral re-implantation for avulsed ureters. 14
Six out of fourteen patients underwent augmentation cystoplasty, four underwent Mitrofanoff, and two underwent ureteric re-implantation. 14 Complications such as worsening renal function, which later required dialysis, three bilateral ureteric strictures at the anastomosis site, a post-operative intra-abdominal collection with conservative management, urosepsis and two uretero-ileal leaks have been reported. 14
Urinary diversion with or without cystectomy
Urinary diversion without cystectomy may be a temporary solution, but preserving a defunctioned bladder has higher morbidity and increases the risk of chronic bladder pain, haematuria, pyocystitis and occasionally malignancy by 60%. Total cystectomy for benign diseases carries as high as a 73% complication rate, suggesting that this technique must be very carefully performed in selected patients, such as those with small fibrotic bladders and chronic bladder pain. 14
Ketamine users who underwent reconstructive surgery had a much higher rate of postoperative complications than patients suffering from other diseases. 14 Severe pan-urothelial damage with ischaemia and ureteral involvement secondary to prolonged and/or higher ketamine dose might be the reasons behind the discrepancy in peri and postoperative complications between these two studies.
Potential novel treatments
Rapamycin is an mTOR signalling pathway inhibitor that reduces eosinophil-mediated inflammation in ketamine rats. 35 Intraperitoneal administration of Rapamycin (autophagy inducer) and wortmannin (autophagy inhibitor) was performed in 40 KIU female rats for 3 months. 35 Autophagy-associated proteins were significantly raised in the ketamine-rapamycin group compared to the ketamine group. 35 Ketamine metabolites and toxins were reduced in the bladder tissue. 35 The ketamine-rapamycin and ketamine-wortmannin groups showed a significant reduction in micturition frequency and pressure and increased bladder capacity compared to the ketamine group. 35 Additionally, wortmannin which is a potent PI3Ks inhibitor, exerts angiogenesis in ketamine rats by reducing basophil-induced inflammation, increasing Vascular Endothelial Growth Factor (VEGF) expression and capillary density which subsequently improves bladder repair.19,35 The precise role and mechanism of autophagy remain unclear.
Bay K8644, a Cav1.2 agonist, was intravesically or intraperitoneally administered in ketamine mice. It completely reversed ketamine-associated voiding dysfunction and improved bladder smooth muscle function in KC. 19
Increased levels of pro-inflammatory cytokines such as IL-1β, IL-6, CCL-2, CXCL-1, CXCL-10, NGF and cyclooxygenase-2 have been noted in KIU and COX-2 inhibitors have been tried in several studies with success. 26
Multipotent Mesenchymal Stem Cells (M-MSCs) might be a promising option in treating KC. The intravesical injection of a low dose (1 × 105) M-MSCs on KC-induced rats restored bladder function and prevented fibrosis, mast cell infiltration and apoptosis. However, further studies are required to assess their long-term safety. 48
Conclusion
This review focussed on KIU pathology, investigations and available treatment options. A holistic approach is vital to manage these patients in order to break the vicious cycle between physical and/or psychological pain and ketamine abstinence. Discrepancies between reports on the efficacy of various treatments is due to each patients’ response which in turn depends on the extent of the disease and combination with ketamine cessation. Unfortunately, ketamine is not part of the standard urine toxicology screen and might have to be considered whenever abuse is suspected. Despite the persistence of urinary symptoms in ex-ketamine users, the mainstay of treatment is continued abstinence from ketamine.
Footnotes
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics of approval statement
Not applicable.
Patient consent statement
Not applicable.
Permission to reproduce material from other sources
Not applicable.
Clinical trial registration
Not applicable.
Data availability statement
Data will be provided on request.
