Abstract
Objectives:
We aimed to compare the efficacy and adverse events of Bacillus Calmette-Guérin (BCG) versus Mitomycin C (MMC) in high-risk Non-Muscle-Invasive Bladder Cancer (NMIBC) patients.
Methods:
This randomized controlled study was conducted over 24 months in four hospitals in Egypt. A sample of 90 patients was randomly assigned to either treatment group, with procedures including baseline examinations, a single postoperative instillation of chemotherapy, a 6-week induction cycle of the assigned drug, and regular follow-up cystoscopies and upper urinary tract imaging. Treatment results and side effects were monitored, with data analyzed via Statistical Package for Social Sciences (SPSS).
Results:
No significant differences were observed in mean age or tumor characteristics (p > 0.05). However, adverse reactions were significantly higher in the BCG group, including cystitis (40% vs. 17.78%, p = 0.020), hematuria (24.44% vs. 4.44%, p = 0.007), overall local reactions (75.56% vs. 26.67%, p < 0.001), fever (13.33% vs. 2.22%, p = 0.049), and fatigue (17.78% vs. 2.22%, p = 0.014). The MMC group had a slightly higher recurrence rate (28.89% vs. 17.78%, hazard ratio 1.89, 95% CI: 0.78–4.55, p = 0.15) with a shorter median time to recurrence (six vs. 12 months). Progression rates were similar (8.89% MMC vs. 4.44% BCG, p = 0.398).
Conclusion:
Although BCG and MMC have comparable efficacy in managing high-risk NMIBC, BCG demonstrated a higher rate of adverse reactions. Decision-making should consider this balance, patient preferences, and health status. Further research is needed for the validation and exploration of these findings.
Introduction
Non-Muscle-Invasive Bladder Cancer (NMIBC) is one of the most prevalent forms of urinary bladder malignancies.1,2 NMIBC accounts for approximately 75–85% of all bladder cancer cases globally,3–7 posing a significant burden on healthcare systems due to its high recurrence rate and lifelong surveillance. 8 Despite advancements in diagnostics and management strategies, the overall recurrence rates of NMIBC range from 30% to 70%,9–11 and progression rates to muscle-invasive bladder cancer stand around 10–30%. 12 These figures highlight the compelling need for efficacious treatment modalities that can mitigate disease progression and recurrence.
Intravesical therapy has emerged as a pivotal approach in managing NMIBC, particularly for intermediate- and high-risk patients. Intravesical instillation of therapeutic agents directly into the bladder allows for high local drug concentrations while mitigating systemic side effects. 13 Bacillus Calmette-Guérin (BCG), an attenuated strain of Mycobacterium bovis, has been the mainstay treatment for high-risk NMIBC for over four decades due to its demonstrated effectiveness in reducing recurrence and delaying progression.14,15
On the other hand, Mitomycin C (MMC), an antineoplastic antibiotic, has also been used as an intravesical therapy for NMIBC. Its cytotoxic effect results from DNA cross-linking, which inhibits DNA synthesis and cell proliferation. 16 While MMC is a well-established treatment for low-risk NMIBC, 17 its effectiveness in high-risk patients remains a point of contention, thereby necessitating further investigation.
Previous research has extensively compared BCG to other intravesical therapies in NMIBC. 18 However, there’s a noticeable gap in the literature when it comes to a direct comparative study between BCG and MMC, particularly in high-risk NMIBC patients. Such a comparison is essential to inform clinical decision-making and patient management. This study aimed to evaluate the risk of recurrence and progression after intravesical instillation of MMC and BCG in high-risk NMIBC patients. By doing so, we aim to contribute valuable insights that could optimize the selection of intravesical therapy, ultimately enhancing patient outcomes in NMIBC.
Methods
Study design and setting
This randomized controlled study was conducted over a period of 24 months, from March 2021 to March 2023, at four different locations: Helwan University Hospitals, Air Force Hospital, Al Maadi Military Hospital, and Kobri Al-Kobba Hospital. Patients were randomly assigned to BCG intravesical instillation or MMC intravesical instillation using the sealed envelope technique, with an allocation ratio of 1:1. Patient enrollment started in October 2020, and the last patient was enrolled on 14th February 2021. The study protocol was submitted to the Faculty of Medicine, Helwan University research ethics committee, and received approval before study initiation (
Participants
The study population comprised patients diagnosed with primary high-risk NMIBC who were under follow-up at the designated study locations. Inclusion criteria encompassed patients with high-risk Ta and T1 transitional cell carcinoma of the bladder per the 2020 European Guidelines classification. 8 On the other hand, immune-compromised patients, such as those with HIV, cancer, or transplant patients on immunosuppressive drugs and pregnant women, were excluded. Other exclusion criteria included patients with low and intermediate-risk NMIBC, recurrent TCC of the bladder, non-urothelial cancer of the bladder, and those with upper urinary tract affection.
Sample size and sampling
We employed a probability sampling method, specifically a simple random sample. Sample size calculation was derived from the progression-free survival (PFS) proportion among the group treated with BCG intravesical instillation and the other group treated with MMC intravesical instillation, as noted in the previous research. 19 Utilizing G*power version 3.0.10 for sample size computation based on a difference of 29.5%, a two-tailed test, α error of 0.05, and power of 80.0%, the total calculated sample size was 82 (41 in each group). An additional 10% was added to compensate for potential dropouts, rendering a total sample size of 90 (45 in each group) at the least.
Procedures
Study participants were recruited based on the pre-set inclusion and exclusion criteria. Subsequently, the study groups and baseline characteristics were matched by conducting a full history and clinical examination, pre-operative laboratory investigations, pelvi-abdominal ultrasonography, computerized tomography (CT) on the abdomen and pelvis with and without contrast, and transurethral resection of bladder tumor (TURBT) with a histopathological examination. All patients with T1 were subjected to a second TURB in addition to patients with incomplete initial TURB and those with no muscle in the specimen after initial resection. Participants then underwent an immediate single postoperative instillation of chemotherapy (MMC 40 mg/20 cc normal saline for 1–2 h) within 6 or 24 h after transurethral resection, irrespective of their group. This was succeeded by a 6-week induction cycle of either BCG (Tice strain; 1 vial [50 mg] in 50 ml normal saline) or MMC (40 mg/20 cc normal saline for 1–2 h), 3–4 weeks post-TURBT. MMC patients received maintenance therapy after the induction cycle, one instillation every 3 months for 1 year, while BCG patients received one intravesical installation each week for 3 weeks at 3 and 6 months after the BCG induction cycle, and then every 6 months thereafter for a total of 2 years. Finally, the outcome among the study groups was assessed by conducting follow-up cystoscopies every 3 months in the first year and every 6 months in the second year, coupled with upper urinary tract imaging every 6 months for the first 2 years. Any occurrences of recurrence and progression were duly documented.
Data analysis
Data were analyzed using the Statistical Package for Social Sciences (SPSS) version 24 and Jamovi software. Quantitative data were tested for normality by the Shapiro–Wilk test, described as the mean and standard deviation for normally distributed data and median and range for non-normally distributed. The Chi-square test was used to compare the categorical variables across both groups. A T-test was used to compare the continuous variables across the two groups. The recurrence and progression rates were determined using the Kaplan–Meier curve. The Cox regression analysis was performed to determine the predictors of recurrence and progression, and the data were presented as hazard ratio (HR) and 95% confidence interval. A p-value of less than 0.05 was considered significant.
Results
Demographic characteristics
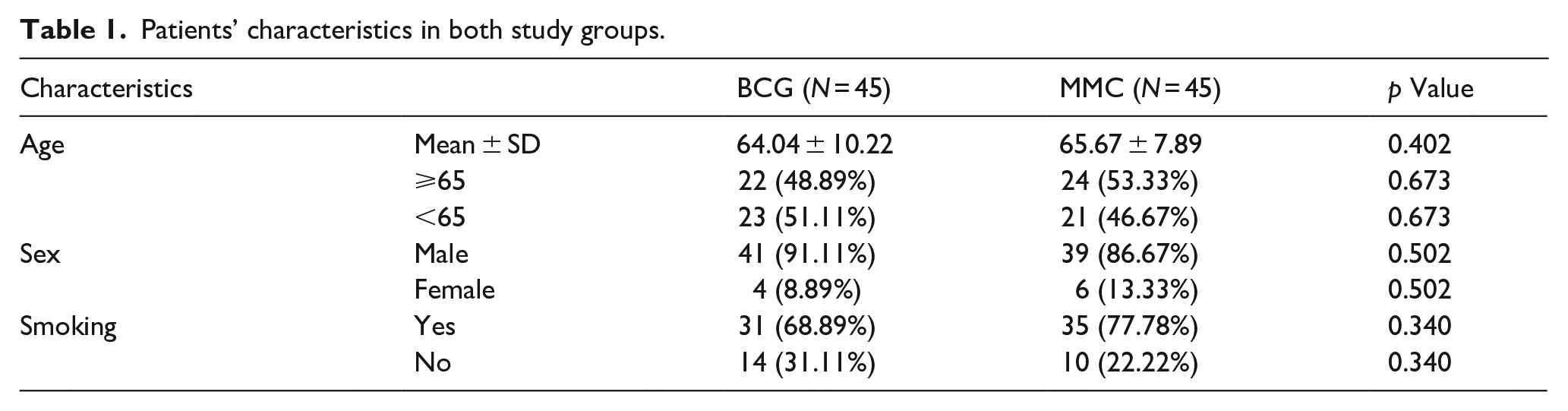
The baseline characteristics of the BCG and MMC groups were statistically comparable, with no significant differences observed. Both groups comprised 45 patients each, with a comparable age distribution and mean age of 64.04 ± 10.22 years in the BCG group and 65.67 ± 7.89 years in the MMC group (p = 0.402). The included patients were predominantly males, with 91.11% in the BCG group and 86.67% in the MMC group (p = 0.503). Furthermore, no significant difference was observed in the distribution of smoking status between the groups, with 68.89% and 77.78% smokers in the BCG and MMC groups, respectively (p = 0.340), as shown in Table 1.
Patients’ characteristics in both study groups.
Tumor characteristics
The comparison of characteristics between the BCG and MMC patient groups revealed no significant differences. Similar proportions were observed for tumor number and size (p = 1.00 and p = 0.788, respectively), presence of carcinoma in situ (p = 0.398), tumor stage (p = 0.172), and tumor grade (p = 0.091) between the two groups (Table 2).
Tumor characteristics.
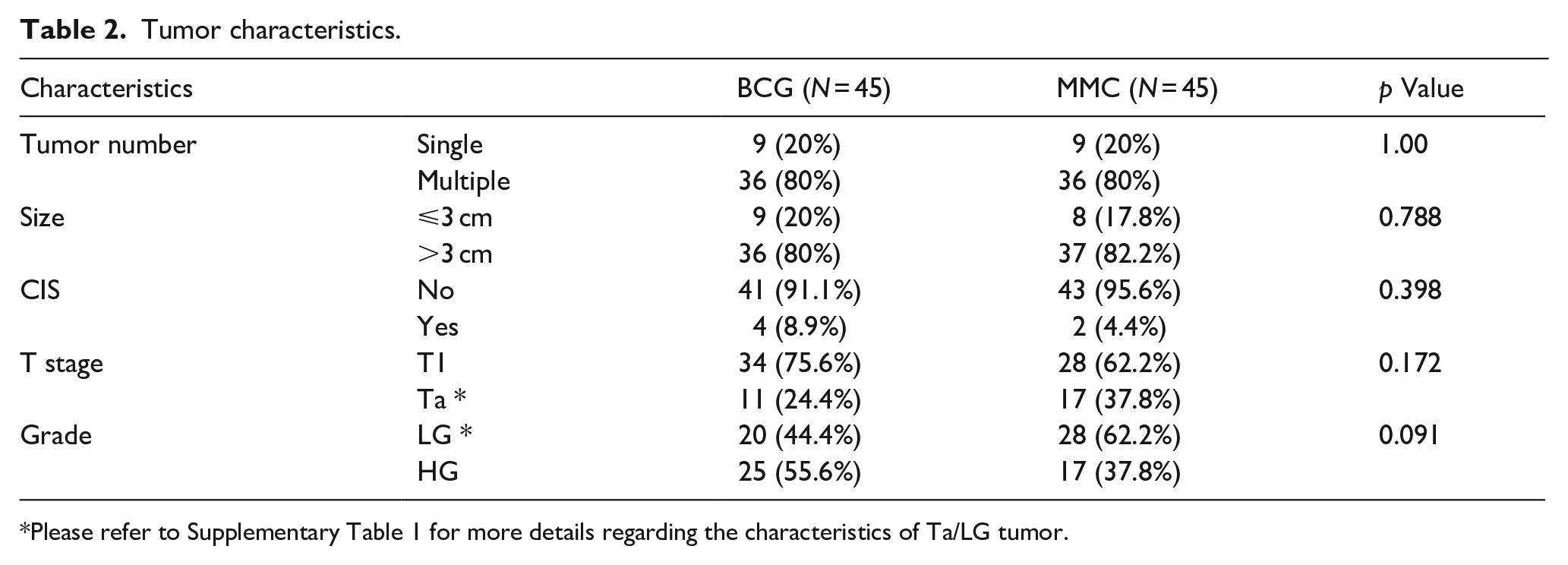
Please refer to Supplementary Table 1 for more details regarding the characteristics of Ta/LG tumor.
Local adverse reactions
Significantly more cases of cystitis (40% vs. 17.78%, p = 0.020), hematuria (24.44% vs. 4.44%, p = 0.007), and overall local adverse reactions (75.56% vs. 26.67%, p < 0.001) were observed in the BCG group compared to the MMC group. The BCG group also had significantly higher number of patients with adverse events (46.67% vs. 22.22%, p = 0.014). Bladder spasms were not significantly different between the groups (11.11% vs. 4.44%, p = 0.237), as shown in Table 3.
Local adverse reactions in both study groups.
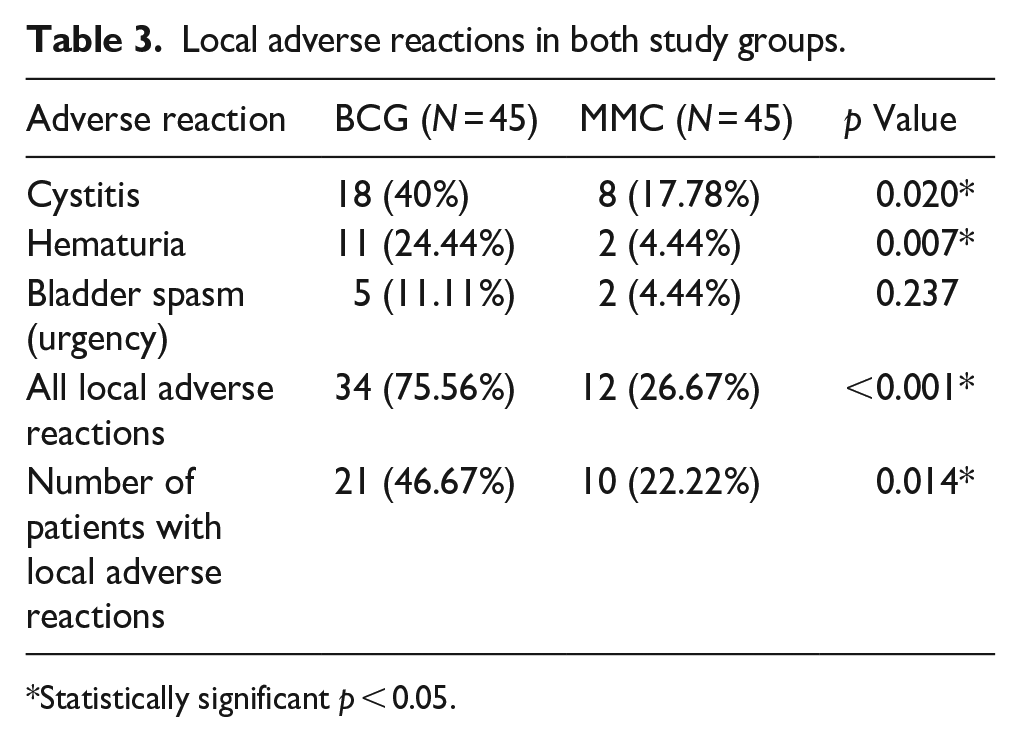
Statistically significant p < 0.05.
Systemic adverse reactions
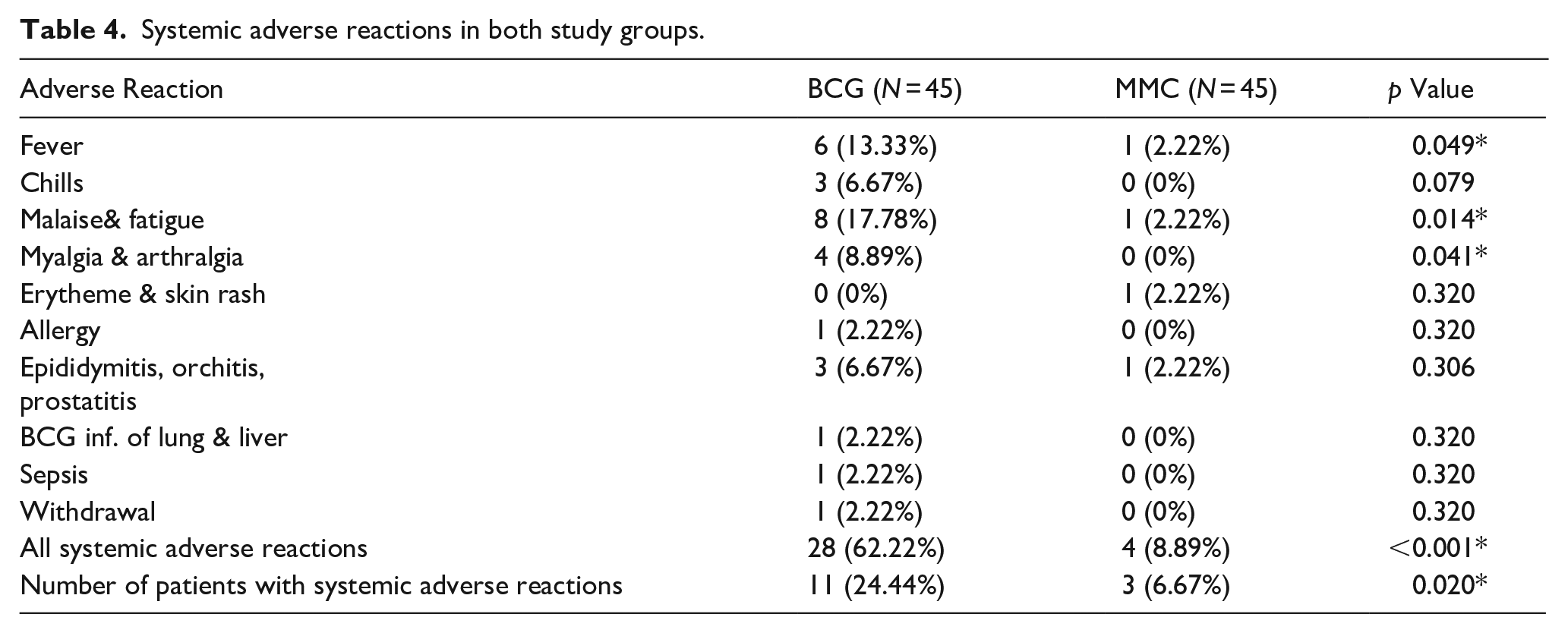
Significant differences were noted in the occurrence of fever (13.33%vs 2.22%, p = 0.049), malaise and fatigue (17.78% vs 2.22%, p = 0.014), myalgia and arthralgia (8.89% vs 0%, p = 0.041), and total systemic adverse reactions (62.22% vs 8.89%, p < 0.001) between the BCG and MMC groups, respectively. Furthermore, more affected cases were reported in the BCG group compared to the MMC group (24.44% vs 6.67%, p = 0.020), as shown in Table 4.
Systemic adverse reactions in both study groups.
Recurrence and progression
The overall recurrence rate was 17.78% in the BCG group and 28.89% in the MMC group (p = 0.107). The recurrence-free survival (RFS) was comparable in both groups (HR = 1.89, 95% CI: 0.78–4.55, p = 0.15; Figure 1(a)). On the other hand, the time to first recurrence was significantly (p = 0.003) shorter in the MMC group compared to the BCG group (8.54 ± 5.59 months vs 11.63 ± 3.74, respectively). The 1-year RFS was (86.2%, 95% CI: 76.6% to 97.1%) in the BCG group and (75.0%, 95% CI: 63.2% to 89.0%) in the MMC group. While the 2-year RFS was and the 2-year RFS was 81.6%, 95% CI: 70.8% to 94.0%) in the BCG group and (70.0%, 95% CI: 57.5% to 85.1%) in the MMC group. Supplementary Table 2 shows the RFS of both groups at all follow-up intervals.
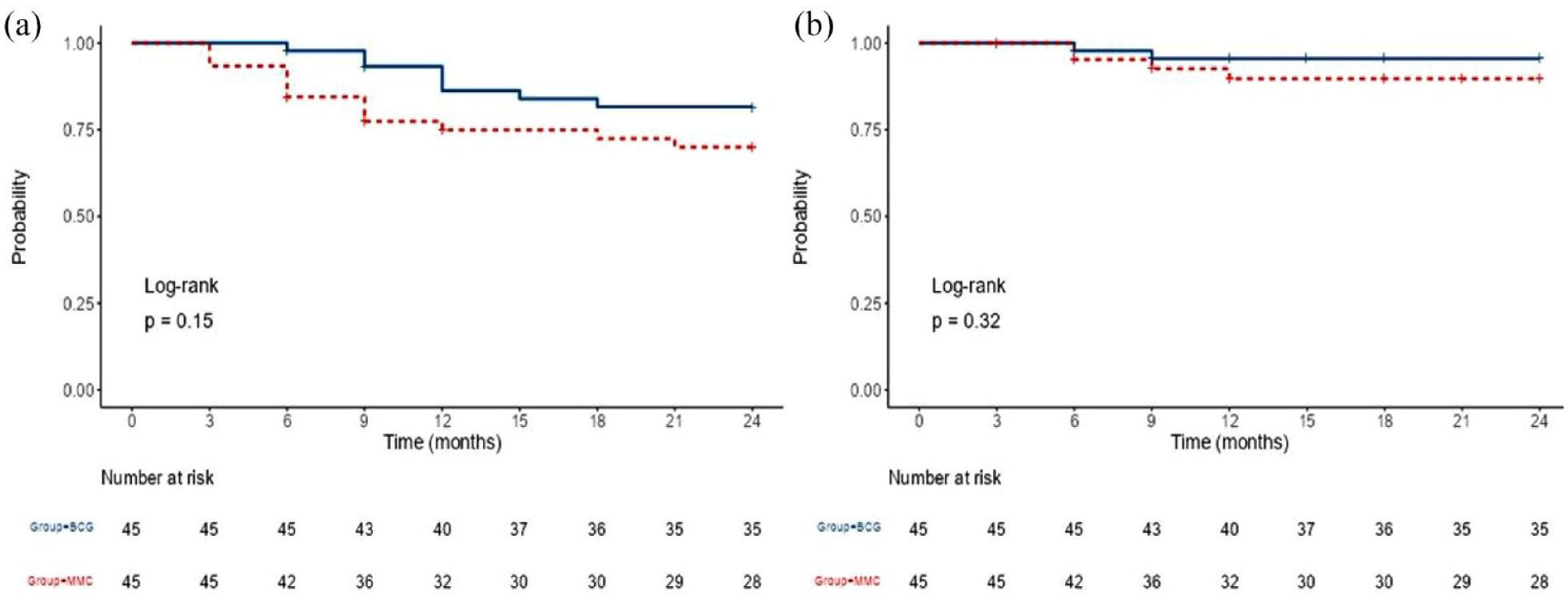
Kaplan–Meier curve of (a) recurrence and (b) progression.
The overall progression rate was 4.44% in the BCG group and 8.89% in the MMC group (p = 0.398). The mean time to progression in the BCG group (7.5 ± 2.12 months) was slightly shorter than the MMC group (8.25 ± 2.87 months), with no statistically significant difference (p = 0.16). The progression-free rate was comparable in both groups (HR = 2.29, 95% CI: 0.42–12.52, p = 0.32), as shown in Figure 1(b). The 2-year PFS was (95.5%, 95% CI: 89.6% to 100%) in the BCG group and (89.7%, 95% CI: 80.6% to 99.9%) in the MMC group.
Follow-up data
At the 3-month follow-up, recurrence was observed in 0% of patients in the BCG group compared to 6.67% in the MMC group (p = 0.241). At 6 months, recurrence was found in 2.22% and 9.52% (p = 0.167) and progression in 2.22% and 4.76% (p = 0.557) of BCG and MMC patients, respectively. The 9-month follow-up saw recurrence in 4.65% of BCG patients and 8.33% of MMC patients (p = 0.645) and progression in 2.33% and 2.78% (p = 1.00), respectively. At 12 months, recurrence was at 7.5% in the BCG group and 3.13% in the MMC group (p = 0.306), with progression at 0% and 3.13% (p = 0.320), respectively. Recurrence at 15 months and 18 months was observed in 2.7% and 2.78% of BCG patients and 0% and 3.33% of MMC patients, while progression remained at 0% in both groups. At 21 and 24 months, both recurrence and progression were at 0% in both groups, as shown in Table 5.
Follow-up data in both study groups.
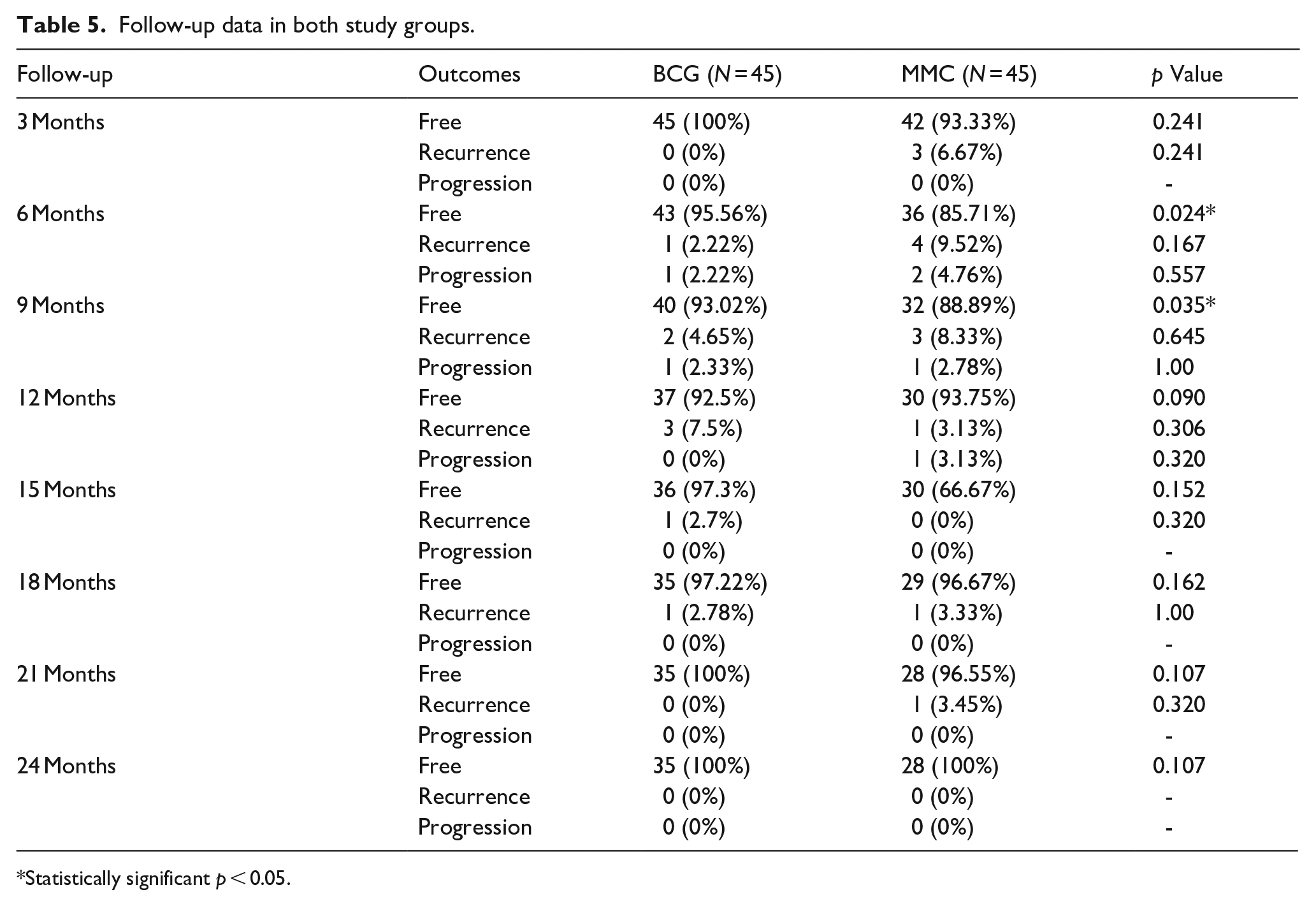
Statistically significant p < 0.05.
Predictors of recurrence and progression
The Cox regression analysis showed that patients with Ta tumor with multiple risk factors, including tumor size >3 cm, multiple papillary tumors, and age >70 years, were associated with a significantly higher risk of recurrence compared to those with T1 tumor (HR = 2.67, 95% CI: 1.04–6.83; p = 0.041). On the other hand, there was no significant association between other patients’ and tumors’ characteristics and risk of recurrence or progression ( Supplementary Figures 1 and 2 ).
Discussion
The management of NMIBC presents a challenge due to high recurrence rates despite the current standard treatments. In recent years, the use of intravesical therapies has grown considerably due to the promising results they have demonstrated in disease control. 20 This randomized controlled trial, carried out at four medical institutions over a period of 24 months, compared the efficacy and safety of BCG intravesical instillation versus MMC in patients with high-risk NMIBC. Our findings demonstrated similar efficacy between BCG and MMC treatments in preventing recurrence and progression, although the BCG group reported a significantly higher rate of local and systemic adverse reactions.
In terms of recurrence and progression, no significant difference was found between BCG and MMC over 24 months. However, our study noted a shorter median time to recurrence in the MMC group (6 months) compared to the BCG group (12 months), suggesting an earlier onset of tumor recurrence in patients treated with MMC. Despite this, the survival analysis found no significant difference in the RFS and PFS between both groups. This could potentially be attributed to the more intensive maintenance therapy schedule applied to MMC patients, compensating for the earlier onset of recurrence. Our findings are consistent with the results of previous studies.21,22
Schmidt et al. ’s analysis observed a potential decrease in time to recurrence with BCG as compared to MMC (HR 0.88, 95% CI 0.71–1.09), although their evidence was of low certainty. This reduction could translate into 41 fewer recurrences per 1000 participants treated with BCG at 5 years. Nevertheless, they also noted that BCG might not influence the time to progression (HR 0.96, 95% CI 0.73–1.26). 21 Similarly, Malmström et al.′s meta-analysis demonstrated that there was no statistically significant differences were discerned between BCG and MMC regarding progression, overall survival, and cancer-specific survival. Additionally, they found no significant difference in time to first recurrence (p = 0.09) between BCG and MMC, as 43% recurred after a median follow-up period of 4.4 years. 22 In Shelley et al.′s meta-analysis, they arrived at a similar conclusion, noting that while BCG might outperform MMC in reducing tumor recurrence, particularly in high-risk patients, there was no observable difference in disease progression or survival rates. They also highlighted that the choice between the two therapies could be influenced by the frequency of adverse events and cost considerations. 23 Overall, these studies emphasize that disease progression, overall survival, and cancer-specific survival remain comparable between the two treatments. On the other hand, a meta-analysis of six studies showed that BCG was more effective than MMC in terms of PFS (OR = 0.53; 95% CI, 0.38–0.75; p < 0.001). However, the included studies had patients with superficial NMIBC, which may explain the contradictory findings. 24 Additionally, these meta-analyses showed that BCG after TURB was significantly associated with a reduced recurrence rate of NMIBC compared to TURB alone or TURB + chemotherapy.22,23,25,26 Moreover, another meta-analysis demonstrated a 32% reduction in the risk of recurrence for BCG compared to MMC in trials with BCG maintenance but a 28% increase for patients treated without BCG maintenance. These findings were also supported by those of Järvinen et al., who found that maintenance BCG was a sustained and significant long-term reduction in recurrence compared to maintenance MMC instillation therapy. 27
As for adverse reactions, we found significantly more local (cystitis and hematuria) and systemic (fever, malaise & fatigue, myalgia & arthralgia) side effects in the BCG group. This finding is consistent with established literature indicating a higher incidence of side effects associated with BCG therapy. 23 While the increased rate of adverse reactions may negatively impact patient quality of life, it is important to note that the side effects observed were largely manageable and did not result in treatment withdrawal in a majority of cases.
In this study, the duration of treatment of the BCG group was 2 years and for the MMC group, it was 1 year due to the adverse events associated with MMC. According to the recent EAU guidelines, BCG can be administered from 1 to 3 years; however, expanding the duration of treatment beyond 1 year should be considered in the view of costs, adverse events, and availability of BCG. 28 However, further evidence is needed to confirm the superiority of 3 years of BCG over one or 2 years.
The idea of combining BCG and MMC as adjuvant intravesical therapy was explored in the study by Hayne et al. Their results pointed out that despite optimal BCG treatment, around 30% of patients still experienced disease recurrence or progression. 29 On the other hand, BCG therapy is not a universal solution for NMIBC. As Zhu et al. have pointed out, the utility and efficacy of BCG therapy depend on factors such as the dosing schedule and possible combined therapies. 30 They conducted a comprehensive meta-analysis on this subject and found that patients who received maintenance BCG therapy demonstrated significantly better recurrence-free survival rates than those who received induction alone. Their results also suggested that the use of standard-dose BCG, compared to low-dose, yielded better oncologic outcomes. Moreover, they found that combining BCG with epirubicin resulted in improved patient outcomes, a finding that adds a new dimension to our understanding of BCG therapy’s potential.
Interestingly, an experimental study by Arends et al. evaluated the potential benefits of chemohyperthermia (CHT) using MMC as an adjuvant treatment for intermediate- and high-risk NMIBC. In a randomized controlled trial that compared CHT with MMC to BCG, they discovered that the group receiving CHT showed a promising recurrence-free survival rate. Despite some limitations of their study, such as potential bias and premature closure, their findings suggest that CHT is a viable alternative treatment for NMIBC, especially for patients with intermediate and high-risk diseases. 31
Finally, Oosterlinck et al. focused on the treatment of patients with CIS of the urinary bladder. They explored whether sequential MMC plus BCG after transurethral resection is worthy of further study in NMIBC patients with CIS. Despite the acceptable toxicity levels and comparable complete response (CR) and disease-free rates to BCG alone, the study concluded that more extensive trials are required to fully assess the potential benefits of this combination. 32
Demographic characteristics and tumor characteristics showed no significant difference between the two groups, aligning with our randomization process, which effectively minimizes bias and ensures the comparability of the two groups. Except for Ta with other risk factors, Cox regression analysis showed no significant associations between patient and tumor characteristics and the risk of recurrence or progression. This suggests that factors may not significantly impact the efficacy of BCG or MMC treatment, although larger and more diverse studies may be needed to fully explore these relationships.
This study has several strengths, including its randomized design, the rigorous protocol for data collection, and a reasonable sample size. However, the results must be interpreted in light of certain limitations. First, our sample size was calculated based on the difference between BCG and MMC in terms of PFS alone, without considering the RFS, which may result in an underpowered study to detect minor differences between the two groups. Also, the exclusion of immune-compromised patients may affect the generalizability of the results, given the potential influence of immune status on treatment response.
In conclusion, our findings indicate that while BCG and MMC demonstrate comparable efficacy in preventing the recurrence and progression of high-risk NMIBC, BCG was associated with a higher rate of adverse reactions. These findings suggest that decision-making for the management of such cases should weigh the balance between efficacy and side-effect profile, taking into consideration patient preferences and overall health status. Further large-scale and long-term studies are warranted to verify and expand on these findings.
Supplemental Material
sj-docx-1-urj-10.1177_03915603231206603 – Supplemental material for Comparative study between mitomycin C versus Bacillus Calmette-Guérin (BCG) in high-risk non-muscle-invasive bladder cancer
Supplemental material, sj-docx-1-urj-10.1177_03915603231206603 for Comparative study between mitomycin C versus Bacillus Calmette-Guérin (BCG) in high-risk non-muscle-invasive bladder cancer by Mohamed Mahmoud Abdelfatah Zaza, Tarek Abd El-Mageed Salem, Ahmed Mohamed El-Sadat and Mohammed Hassan Ali in Urologia Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
