Abstract
Introduction:
This study investigated the role of ambrisentan; the selective endothelin type-A receptor (ETAR) blocker on experimental diabetic erectile dysfunction in rats.
Materials and methods:
Eighty-four adult male Sprague Albino rats were divided randomly into 7 groups. Three control groups received 1 mL saline, 0.2 mg/kg/d ambrisentan and 1.5 mg/kg/d tadalafil, respectively orally for 4 weeks. The remaining four groups were fed high fat diet for 14 days. Diabetes was induced by a single intra-peritoneal injection of 40 mg/kg streptozotocin. After 72 h, diabetes was confirmed by plasma glucose level ⩾250 mg/dL. Diabetic rats were divided randomly into four groups, numbered from 4 to 7. The fourth group was the diabetic-control group, while the fifth and sixth groups received ambrisentan and tadalafil respectively. The seventh group received a combination of both drugs. Treatment continued for 4 weeks then, copulatory, intracavernous pressure measurement, and laboratory tests were conducted.
Results:
In diabetic rats, ambrisentan and tadalafil improved fasting glucose, insulin, insulin resistance, testosterone, nitric oxide, and rho kinase (ROCK) values compared to diabetic group with the maximum improvement achieved in ambrisentan/tadalafil group (p < 0.05). Ambrisentan also enhanced ICP and improved latency to erection and number of mounts with a tolerable SBP. Yet, ambrisentan/tadalafil combined therapy resulted in deterioration in SBP with consecutive worsening in ICP and mating indices.
Conclusion:
Ambrisentan showed significant therapeutic potential to prevent and improve diabetic ED in rats comparable to tadalafil.
Introduction
Although erectile dysfunction (ED) is not a life-threatening disease, yet it affects social wealth. Diabetes mellitus (DM) was found to result in ED in about 35%–62.9% of adult males. The pathophysiology of diabetic ED is complex, including abnormal neuro-endocrinal signaling and endothelial vascular dysfunction.1,2 Although phosphodiesterase type 5 inhibitors (PDE5Is) are among the first line therapies of ED, failure of PDE5I was reported in about 51.1% of treated patients. 2
Insulin resistance (IR) is one of the features of type 2, non-insulin dependent DM (T2DM). High insulin levels observed in T2DM patients with IR, were found to increase the circulating Endothelin (ET-1) plasma levels through stimulating the transcription of ET-1gene (edn1).3,4 ET-1 is known as the most potent vasoactive peptide in our bodies and abnormal ET-1 levels were encountered in many vascular disorders such as: pulmonary hypertension, heart failure, systemic hypertension, and some malignancies especially the ovarian cancer.
ET-1 acts upon two types of receptors: ET-type A receptor (ETAR) and ET-type B receptor (ETBR). ETBR are furtherly divided into two subtypes: endothelial ETB1 and ETB2 located on the vascular smooth muscle cells (VSMCs). ET-1 binding to ETAR and ETB2 receptors causes vasoconstriction (VC). 5 On the other hand, endothelial ETB1 activation results in a nitric oxide (NO)/cGMP dependent vasodilatation (VD). ETBR also contributes in the clearance of ET-1.6,7
In view of diabetic ED, higher ET-1 levels were observed in patients with ED than in healthy controls. 7 The upregulation and the long-lasting pressor action of ET-1 on the ETARs in the corpus cavernous (CC) tissue is hypothesized to maintain a flaccid penile state and mediate mitogenic dysfunction that predispose to diabetic ED. This action is at least partially mediated by an rho kinase (ROCK)-dependent Ca2+ sensitization mechanism.8,9 Contrarily, ETBR was found to possess a counter-protective effect and facilitates ET-1 degradation. 10 A previous study by Alkan et al. 7 demonstrated a beneficial effect of using bosentan (a non-selective endothelin receptor blocker) in treatment of diabetic ED. Alkan and his colleagues reported an abnormal high ET-1 level in DM rats that was decreased after bosentan treatment. They also mentioned that the CC is not only able to respond to ET-1, but also can synthesize it.
In the view of the above-mentioned information, this study aimed to demonstrate the effect of selectively blocking ETAR receptors using ambrisentan on diabetic ED. We hypothesized that ambrisentan can ameliorate the diabetic-dependent ET-1 hazards on the CC through blocking the ETARs. Moreover, by preserving the ETBRs actions, ambrisentan would improve the endothelial NO synthesis and restore the normal erection physiology. Ambrisentan is already approved as a treatment of pulmonary arterial hypertension due to its potent role as a vasodilator and an inhibitor of pulmonary vascular smooth muscle proliferation.11,12 Accordingly, the role of ambrisentan in diabetic ED was evaluated, either as a monotherapy or in combination with the tadalafil, and compared to tadalafil being a standard therapy for ED. We mainly relied on the copulatory tests to elucidate the real impact of ambrisentan on ED. Yet, for better understanding of the mechanism of action of ambrisentan on diabetic ED, we measured the change in the intracavernous pressure after ambrisentan therapy. We also evaluated the impact of ambrisentan on glucose homeostasis, insulin resistance, pancreatic β cells function, ROCK activity, NO level, and plasma testosterone level.
Material and methods
Drugs and chemicals
Ambrisentan powder (AUG Pharma, Egypt).
Tadalafil powder (EVA Pharma, Egypt).
Streptozotocin powder (Sigma Aldrich, Egypt).
Estradiol benzoate ampoule (MARCH trademark).
Progesterone ampoule (IBSA)
Formalin: Formaldehyde sol. 38%–40% (AlNasr Pharmacy. Chemicals, Egypt).
Kits and instruments
Nitrite/nitrate colorimetric Assay Kits (Sigma-Aldrich, Egypt).
Rho-associated Kinase (ROCK) Activity Assay Kit. (Sigma-Aldrich, Egypt)
Handheld glucometer to measure blood glucose (Gluco Dr auto)
Rat/mouse insulin ELISA kit (Sigma-Aldrich, Egypt).
Testosterone test kit (Sigma-Aldrich, Egypt).
Invasive arterial blood pressure measurement (Powerlab AD Instruments ML866, 430-0820-Australia).
Rat tail sphygmomanometer (Panlab, USA).
Animals used
The study was conducted in accordance with the Institutional Animal Care and Use Committee (IACUC) Faculty of Medicine, Cairo University. Approval number: (CU, III, F, 83, 19)/April 2020, and in accordance with the Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the U.S. National Institutes of Health. Eighty-four adult Sprague Dawley male rats weighting between 300 and 350 g were included in this study. They were housed in standard laboratory conditions under a 12-h light/dark cycle and controlled temperature of 22°C ± 3°C, with free access to standard food and water. Before conducting experiments, animals were acclimatized to laboratory conditions for 7 days.
Induction of diabetes
Diabetes was induced in 48 rats after 14 days of feeding high fat diet (66.5%-commercial feed, 13.5%-lard, and 20%-sugar) to induce insulin resistance, 13 followed by intra-peritoneal injection of streptozotocin (STZ) at a single dose of 40 mg/kg body weight. After 72 h of STZ injection, fasting blood samples were collected via the tail veins to measure blood glucose levels by a handheld blood glucose monitor. Rats with plasma glucose level of 250 mg or higher were considered to be diabetic (as shown in Supplemental File Table 1). 14 Diabetic rats were then randomized on the four diabetic groups and treatment was started immediately. 8
Experimental design
As shown in Figure 1, the control rats (n = 36) were subjected to intra-peritoneal injection of 0.5 mL saline and divided randomly into 3groups (n = 12) as follows:
Group 1 (C):
Group 2 (C-Am):
Group 3 (C-Td):
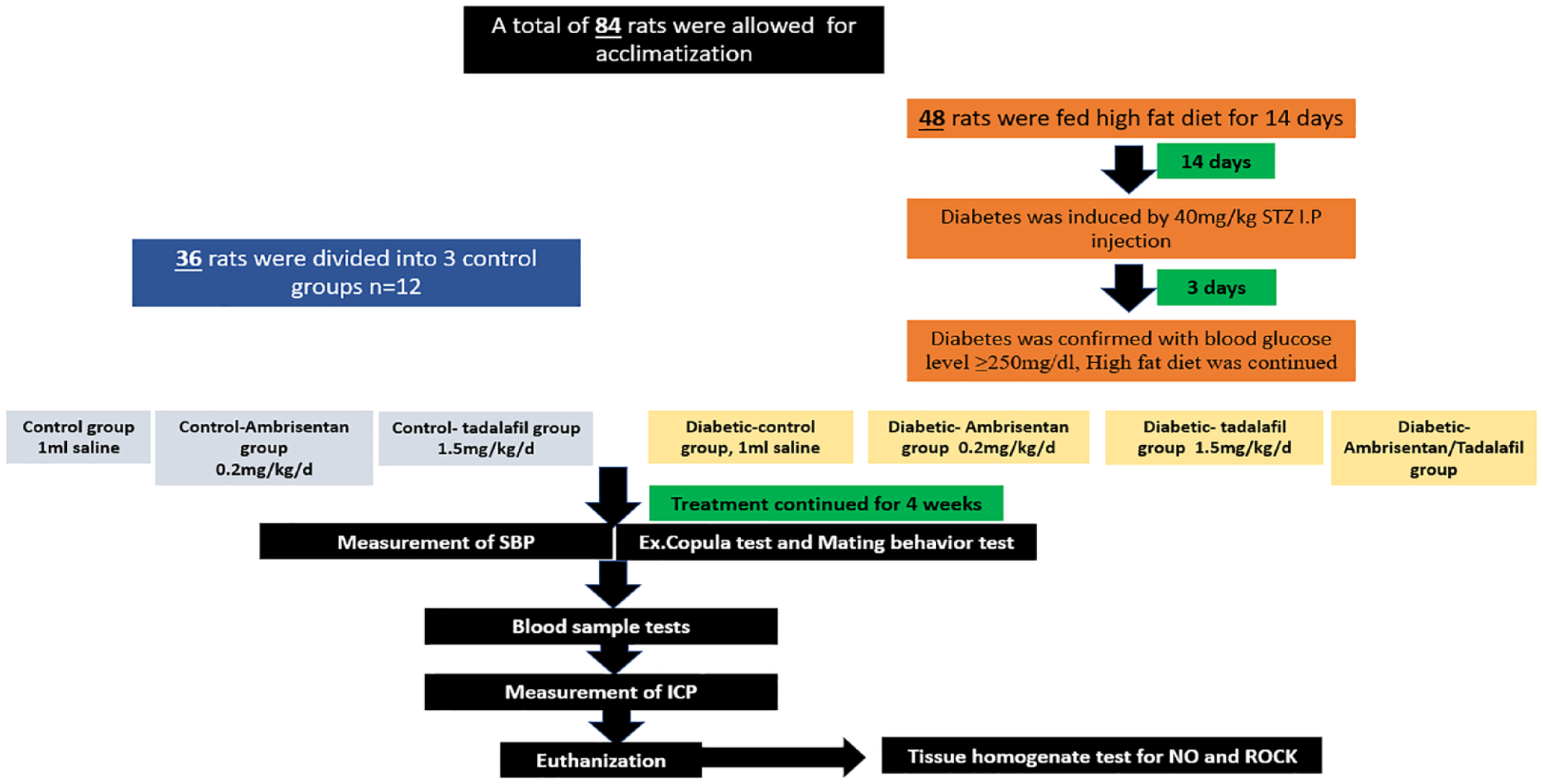
The experimental design of the current study.
As for the diabetic rats, they were randomly divided into four groups (n = 12) as follows:
Group 4 (D):
Group 5 (D-Am):
Group 6 (D-Td):
Group 7 (D-Am-Td):
Treatment lasted for 4 weeks in all groups. 8
Measurements
Pharmacological studies
Systolic blood pressure measurement
The systolic blood pressure (SBP) of each rat was measured non-invasively using a rat tail sphygmomanometer and was calculated as the mean of three readings on three different days after being trained for 7 days for the measuring procedure before the actual measurement was recorded. 18
Extended Ex. copula erection test
The ex. copula test was performed by restraining the male rat on his back. The penile sheath is then retracted gently to touch the glans smoothly with a cotton pad. Latency for the first penile erection (the time elapsed from touching the glans to erection) and number of penile erections occurring during a 30 min-observation period were recoded. 19
Mating test
According to Mercier et al. 20 protocol, this test was carried on between 17:00 and 19:00 pm in a sound attenuated room. Each Male rat was placed individually in an observation glass chamber (30 × 30 × 50 cm) for 5 min for adaptation. The females were subcutaneously injected with 30 μg estradiol benzoate and 500 μg progesterone 24–48 h and 4 h before the test respectively, to induce estrous cycle. 21 The following were recorded in a period of 30 min: (1) latency to first mount (defined as when a male rat mounts the female from the rear and grasps her flanks with his front feet as shown in the Supplemental Photo S1), (2) number of mounts, (3) number of intromissions, (4) number of ejaculations, and (5) duration of copulation activity. The test was stopped if no intromissions occurred within 15 min. Ejaculations were confirmed by the presence of a semen plug in the female’s vagina. 22
Intra-cavernous penile pressure (ICP) measurement
Under isoflurane 2.5%, male rats were anesthetized and the skin overlying the penis was incised, and the prepuce was degloved to expose the corpora cavernosa. A 26 -gauge needle filled with heparinized saline was carefully inserted into the corpus cavernosum (CC) on one side to measure the ICP. The needle was sealed to a polyethylene 50 tubing of 20 cm length filled with heparinized saline 23 as shown in Supplemental Photo S2, connected to a pressure transducer (Powerlab AD Instruments ML866, 430-0820-Australia).
Biochemical study
At the end of the ICP measurement study and with ensuring 6 h fasting, a blood sample was collected from the lateral vein of the rat tail to test for measuring:
Fasting plasma glucose (FPG) using handheld blood glucose monitor in mmol/L.
Fasting plasma insulin (FPI) using a rat/mouse ELISA kit in mU/L according to the manufacturer instructions. 24
Insulin resistance (IR) and pancreatic β (B) cell function were evaluated using the Homeostatic model assessment (HOMA) for insulin resistance (HOMA-IR) and for β cell function (HOMA-%B) as described by Wallace et al.
25
using the following equations: HOMA − IR = (FPI × FPG) / 22.5 HOMA − %B = (20 × FPI)/ (FPG − 3.5)
FPI: is the fasting plasma insulin concentration in mU/L and FPG: is the fasting plasma glucose in mmol/l.
Testosterone level using a rat/mouse ELISA kit in ng/dL according to the manufacturer instructions 26
By an esthetic overdose, rats were killed at the end of the experiment. Cavernous tissues were dissected gently to the level of crus penis. The penis was resected from the proximal of the cruses and then the cavernous tissue was frozen at −80°C to be prepared as a tissue homogenate to test the following:
Nitric oxide (NO) in µmol/L using colorimetric laboratory method. 27
Rho-Kinase (ROCK) in mU activity according to the manufacturer instructions. 28
Statistical analysis
Statistical Package for the Social Sciences (SPSS) version 24 was used for data coding and entering. Data was summarized using mean and standard error of mean (SEM) in quantitative data and using frequency (count) and relative frequency (percentage) for categorical data. Comparisons between the different groups were done using ANOVA with post hoc Tukey test in normally distributed quantitative variables. For non-parametric tests, independent Kruskal Wallis test was used with adjusted Mann Whitney test as the post hoc test. p-Value less than 0.05 was considered as of statistical significance. 29
Results
In vivo results
Systolic blood pressure (SBP)
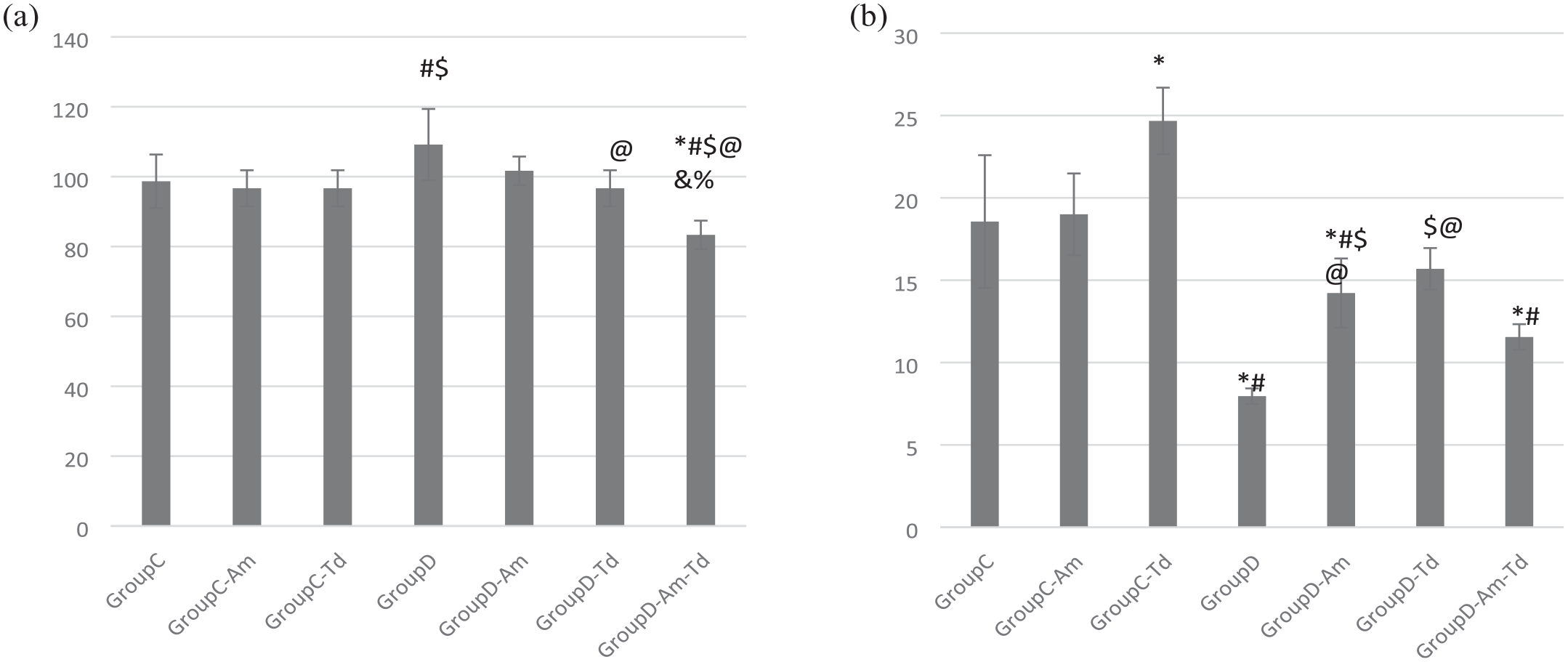
(a) Systolic blood pressure (SBP) and (b) intracavernous pressure (ICP) values in the different studied groups.
Ex. copula test
As shown in Table 1,
Comparison between the different study groups as regards: Ex. copula test and mating behavior parameters.
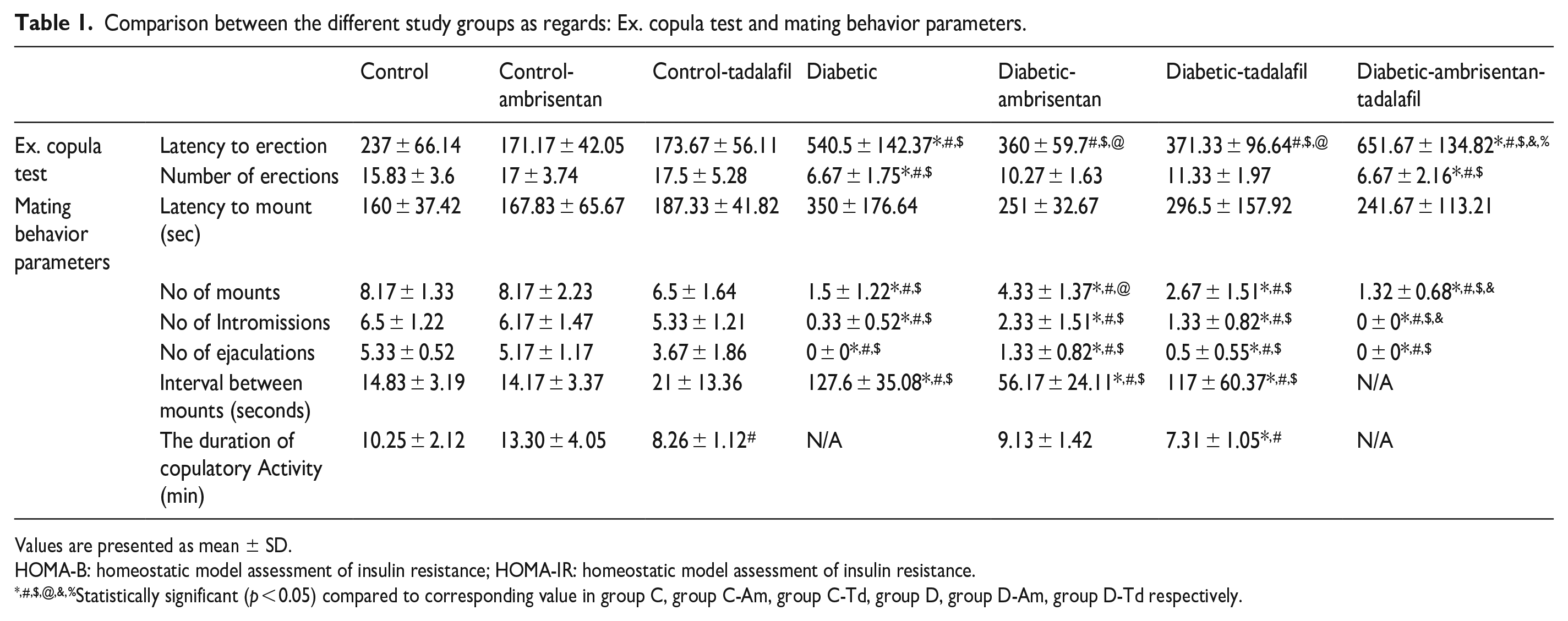
Values are presented as mean ± SD.
HOMA-B: homeostatic model assessment of insulin resistance; HOMA-IR: homeostatic model assessment of insulin resistance.
Statistically significant (p < 0.05) compared to corresponding value in group C, group C-Am, group C-Td, group D, group D-Am, group D-Td respectively.
Mating behavior
Intracavernous pressure (ICP)
Biochemical results
Blood samples results
Fasting plasma glucose (FPG)
Comparison between the different study groups as regards glucose and insulin profiles.
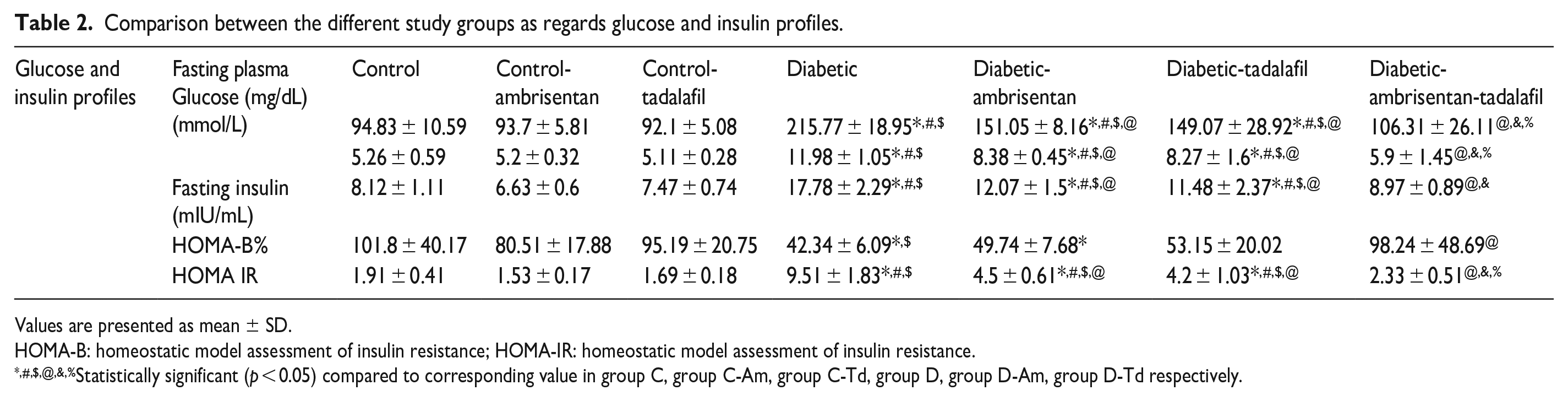
Values are presented as mean ± SD.
HOMA-B: homeostatic model assessment of insulin resistance; HOMA-IR: homeostatic model assessment of insulin resistance.
Statistically significant (p < 0.05) compared to corresponding value in group C, group C-Am, group C-Td, group D, group D-Am, group D-Td respectively.
Fasting plasma insulin (FPI)
HOMA-IR
HOMA-B
Testosterone
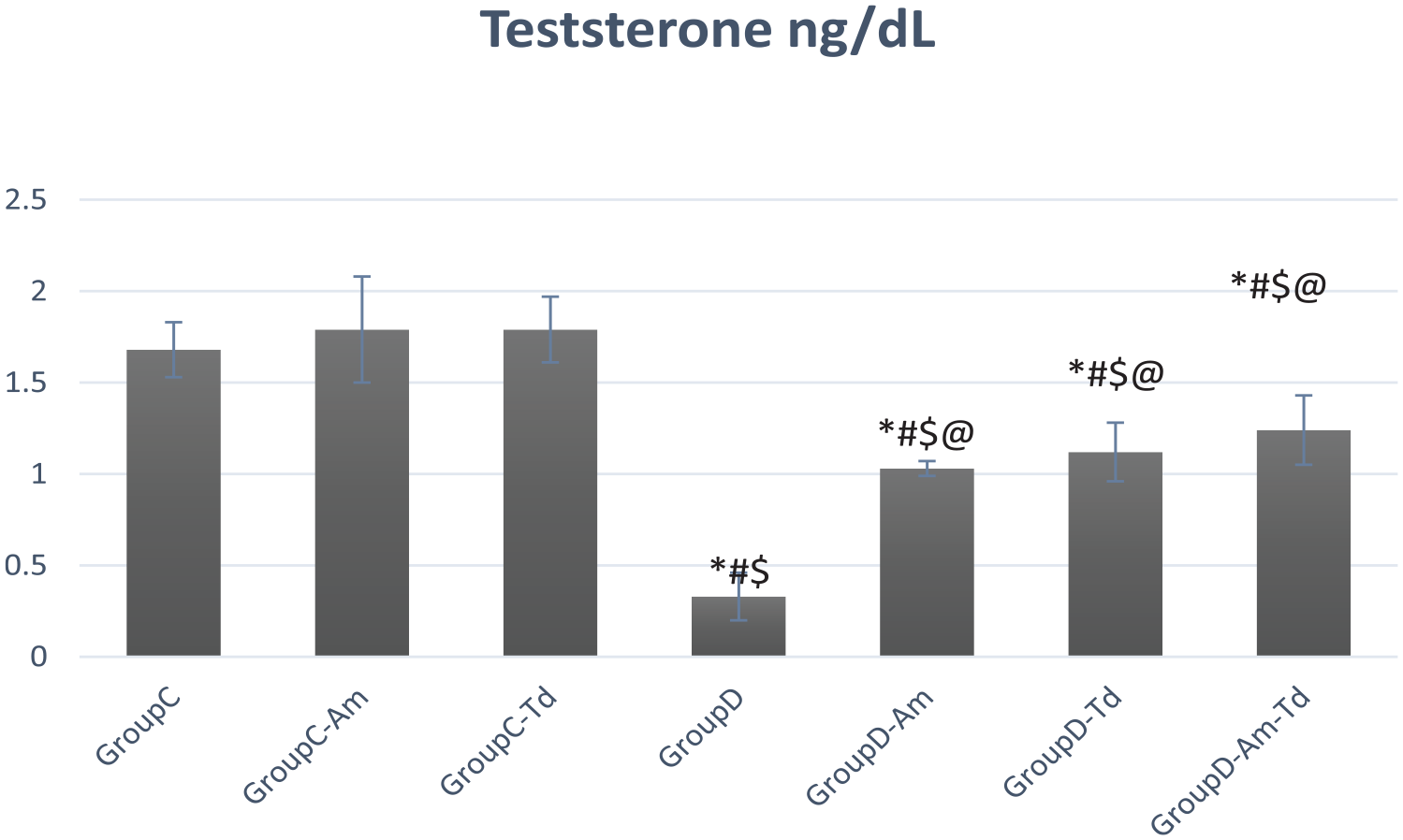
Testosterone values in the different studied groups.
Biochemical results in tissue homogenate
Nitric oxide (NO) level
Ambrisentan and tadalafil caused a slight and a non-significant elevation in NO level compared to
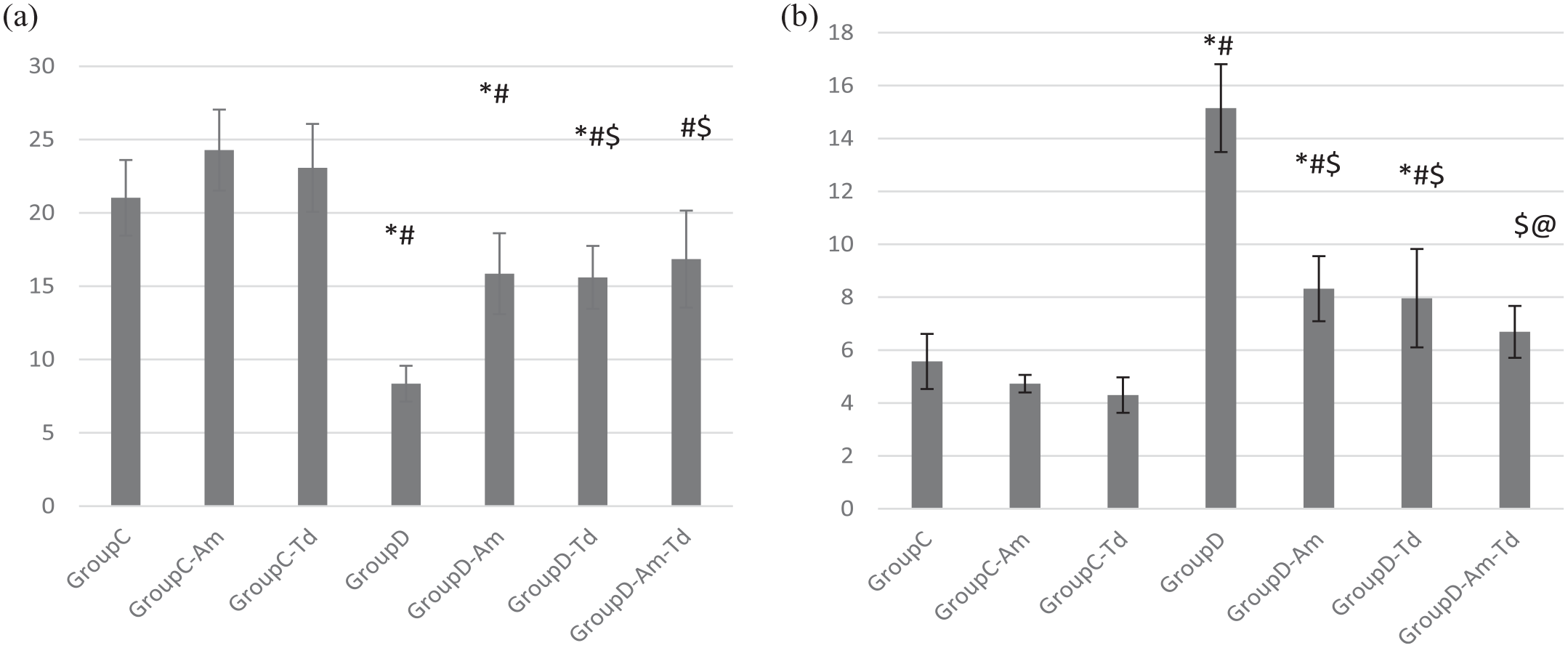
(a) Nitric oxide and (b) Rho kinase values in the different studied groups.
Rho associated protein kinase (ROCK)
Discussion
Although the great success in the management of ED using PDE5Is, failure of PDE5Is therapy was reported to range between 30% and 35% in diabetic ED. 2 Chen et al. 30 found that 52% out of 238 men complaining of ED, suffers from IR which was the most prevalent independent factor associated with ED. IR upregulates ET-1, where the latter acts on ETAR receptors causing vasoconstriction and penile flaccidity. High ET-1 can also decrease testosterone production, which would affect the sexual desire and the normal physiology of erection. It can increase the production of pro-inflammatory cytokines that play an important role in diabetic insults as well.7–9,30 It was noted that prolonged high ET-1 levels result in downregulation of endothelin receptors. Loss of ETBR actions will result in a non-opposed penile vasoconstriction. All together, abnormal ET-1 reported in DM, is speculated to contribute to diabetic-ED.
According to Alkan et al., 7 the non-selective ET-1blocker bosentan showed a promising therapeutic potential in the management of diabetic ED through antagonizing the ET-1 medicated VC and decreasing inflammatory cytokines production. However, one of the main contributors of diabetic-ED is the impaired endothelial NO, responsible for penile engorgement and erection. Accordingly, in this study we speculated an additive profit from preserving the vasodilatory actions of ETBR using the selective ETAR blockade, ambrisentan as ETBR is also responsible for almost 80% of ET-1 clearance in the lungs. 6
In this study, ambrisentan was compared to and combined with tadalafil where the latter role in ED is well established through increasing NO/cGMP levels and augmenting ICP,31,32 and that was documented also in this study. In addition, tadalafil can decrease ROCK activity possibly through enhancing protein kinase G (PKG) which phosphorylates the ROCK-guanosine triphosphatase unit
33
which explain the significant decrease in ROCK levels in the
As regards ambrisentan and concerning the actions of ambrisentan on glucose homeostasis, the improved FPG and HOMA-IR results are in concordance of Lteif et al. 38 who found that BQ123; a selective ETAR blockade, increased glucose uptake in hyper-insulinemic euglycemic obese patients due to the enhanced distribution of blood and glucose to skeletal muscles by an insulin-dependent VD. Although Ambrisentan didn’t improve HOMA-B, Eibl et al. 39 reported beneficial role of selective ETAR blockade in acute severe pancreatitis by decreasing the capillary permeability possibly by decreasing post-capillary resistance. However, more clinical research is needed to study the role of ambrisentan on pancreatic β cells function.
A realistic evaluation of a medication tested as a potential treatment of ED requires performing a copulatory test to mimic the real physiological situation where there is a harmony between the neuro-endocrinal-vascular and musculoskeletal systems. In this perspective and as regards the ex. Copula test, there was a significant improvement in latency to erection in the
Conclusion
A possible potential of ambrisentan on copulation through: improving sexual desire, muscular capacity and increasing ICP can be suggested. This favorable potential can be a direct effect (through improving the penile perfusion and counteracting the ET-1-mediated VC and vascular dysfunction by restoring ROCK/NO balance), or an indirect effect (resulting from its beneficial action on glucose homeostasis, insulin resistance, and testosterone levels). However, long-term clinical trials are recommended to validate these pre-clinical findings on the real scale of diabetic-ED patients
Yet, it would be of interest to test lower doses of ambrisentan (whether alone or with tadalafil) and after ensuring proper glycemic control to elicit the maximum potential of ambrisentan in the management of diabetic ED. Also, it would be of interest to perform a comparative study between bosentan and ambrisentan to elucidate the different mechanisms by which both drugs may improve ED, with special attention to their possible combination with other medications approved for ED management and their interaction with PDE5Is.
Supplemental Material
sj-docx-1-urj-10.1177_03915603231192737 – Supplemental material for The potential effect of ambrisentan as monotherapy and combined with tadalafil on diabetic erectile dysfunction in rats
Supplemental material, sj-docx-1-urj-10.1177_03915603231192737 for The potential effect of ambrisentan as monotherapy and combined with tadalafil on diabetic erectile dysfunction in rats by Omnia Azmy Nabeh, Fatma Ahmed El-Batrawy, Omayma Anwar Khorshid and Ghada Farouk Soliman in Urologia Journal
Footnotes
Acknowledgements
The authors would like to thank professor Dr Laila Rashed, professor of biochemistry Kasr alainy faculty of medicine for the biochemical results in this study.
Authors’ contributions
All authors contributed to the study conception and design. O.A.N was responsible for data collection and analysis. All authors contributed in the scientific writing, read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Kasr Alainy Faculty of medicine, Cairo university, Egypt.
Ethics approval statement
Institutional Animal Care and Use Committee (IACUC) Faculty of Medicine, Cairo University. Approval number: (CU, III, F, 83, 19)/April 2020.
Patient consent statement
Not applicable.
Permission to reproduce material from other sources
Not applicable.
Clinical trial registration
Not applicable.
Data availability statement
All data generated or analysed during this study are included in this published article and the supplementary file.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
