Abstract
Introduction:
We aimed to assess the sexual and the reproductive functions in males post COVID-19 infection in Beni-Suef Governorate.
Methods:
One hundred men were recruited in the current study. All participants were assessed by the Arabic validated version of the International Index of the Erectile Function 5 (ArIIEF-5), semen analysis, and Hospital Depression and Anxiety Scale (HADS). Finally, morning testosterone serum level was measured.
Results:
There were significant decreases in the sperm total motility, the ArIIEF-5 score, and serum total testosterone in the post COVID-19 patients after 3 months when compared to the controls. However, there were significant increases in the sperm total motility, the ArIIEF-5 score, and serum total testosterone in the post COVID-19 patients after 6 months group when compared to the post COVID-19 patients after 3 months group. Moreover, there was a significant increase in the HADS score in the post COVID-19 patients after 3 months when compared to the controls. In contrast, there was a significant decrease in the HADS score in the post COVID-19 patients after 6 months when compared to the post COVID-19 patients after 3 months.
Conclusion:
Our study had shown temporary impairment of sexual and reproductive functions in post COVID-19 convalescent males especially after 6 months from the onset of the infection.
Introduction
Coronavirus disease 2019 (COVID-19) pandemic globally devastated the lives of all people. 1 Mental health problems has occurred as a result of the imposed restrictions that led to marked changes in lifestyles. 1 COVID-19 features a state of hyper inflammation resulting in a “cytokine storm”, which results in micro-thrombosis and disseminated intravascular coagulation. 2 Despite the fact that patients in intensive care units (ICUs) suffer from erectile dysfunction (ED) as a minor problem, yet, the survivors are expected to suffer form it as a long term complication. 3 The etiology of ED due to COVID-19 is multifactorial including endothelial dysfunction, subclinical hypogonadism, psychological distress, and impaired pulmonary hemodynamics. 4 Additionally; exacerbation of cardiovascular conditions by COVID-19 further raises the risk of ED. 4 A variety of viruses including Zika, Ebola, and Marburg has been detected in male testes and semen. 5 Previous study on COVID-19 suggested that it could cause orchitis. 6 The illness in itself and the unprecedented lockdown measures are causes of stress as well as emotional distress following isolation, social distancing, and loss of relatives and friends. 7 We aimed to evaluate the effect of COVID-19 on male sexual and reproductive functions and depression and anxiety status of affected patients following recovery after 3 and 6 months, respectively.
Methods
A cross sectional study including 100 consecutive participants was conducted. The institutional review board revised our signed consents and approved the study work that conforms to Helsiniki declaration (2013). 8 They were divided into 40 recovered patients after 3 months, 40 recovered patients after 6 months as well as 20 controls.
All COVID-19 cases were diagnosed after using the reverse transcription polymerase chain reaction (RT-PCR) test of pharyngeal and nasal swabs. The severity of COVID-19 was determined following the guidelines stated by Xu et al. 9
Inclusion criteria
Married sexually active fertile recovered males (20–50) years old were recruited.
Exclusion criteria
Participants suffering from chronic medical illnesses (diabetes mellitus, hypertension, liver cirrhosis, and ischemic heart disease), receiving medications that affect the sexual function or suffering from ED or infertility were excluded. Also, patients suffering from psychiatric disorders that affect sexual function before the pandemic were also excluded. Finally, cases diagnosed as mild COVID-19 were also excluded.
All the participants were subjected to history taking and general and local examinations. Also, they were assessed by the Arabic validated version of the International Index of Erectile Function 5 (IIEF-5) 10 as well as the hospital depression and anxiety scale (HADS) 11 to determine the level of anxiety and depression among the participants. Furthermore, a semen analysis according to the WHO guidelines (2010) 12 was done for the participants. The participants abstained for 4–5 days before they brought their ejaculates into sterile containers. Another semen sample was provided 4 weeks apart. Finally, 5 cc blood was withdrawn the participants for total testosterone level measurement before 11 am.
Statistical analysis
All data were collected and tabulated and were statistically analyzed using Statistical Package for the Social Sciences program version 23 (SPSS Inc., Chicago, IL, USA). Comparisons among groups were carried out with Student’s t-test. Correlations between different variables were tested by the Pearson test. Sample size calculation was done using F tests—ANOVA. 13
Results
A marked decrease in the total motility in post COVID-19 patients was observed after 3 months when compared to the controls (46.13 ± 17.85 and 55.00 ± 12.25, respectively; Table 1). However, a marked increase in the total motility in post COVID-19 patients was observed after 6 months when compared to post COVID-19 patients after 3 months group suggesting that this impairment in the semen parameters may occur for transient duration (53.38 ± 10.34, 46.13 ± 17.85, respectively). Remarkably, there were 29 patients in the recovered group after 3 months showing normal sperm motility compared to 37 patients in the recovered group after 6 months (Table 2). This change in the percentage of normal sperm motility between the two groups was statistically significant (p = 0.036; Table 2). Furthermore, a marked decrease in the ArIIEF-5 score in post COVID-19 patients was observed after 3 months when compared to the controls (19.08 ± 3.96, 21.25 ± 2.92, respectively; Table 3). In contrast, a marked increase in the ArIIEF-5 score in post COVID-19 patients was noted after 6 months when compared to post COVID-19 patients after 3 months suggesting also a temporary impairment in the sexual function (21.28 ± 3.13 and 19.08 ± 3.96, respectively; Table 3). Moreover, a marked increase in the HADS score in the post COVID-19 patients was noted after 3 months when compared to the controls (8.10 ± 2.25 and 7.20 ± 2.21, respectively; Table 3). In contrast, a marked decrease in the HADS score in post COVID-19 patients was detected after 6 months when compared to post COVID-19 patients after 3 months suggesting improvement in the levels of depression and anxiety in the recovered cases after 6 month from the onset of the infection (6.88 ± 1.95 and 8.10 ± 2.25, respectively; Table 3).
Compares between controls and recovered patients after 3 months and recovered patients after 6 months regarding semen parameters.
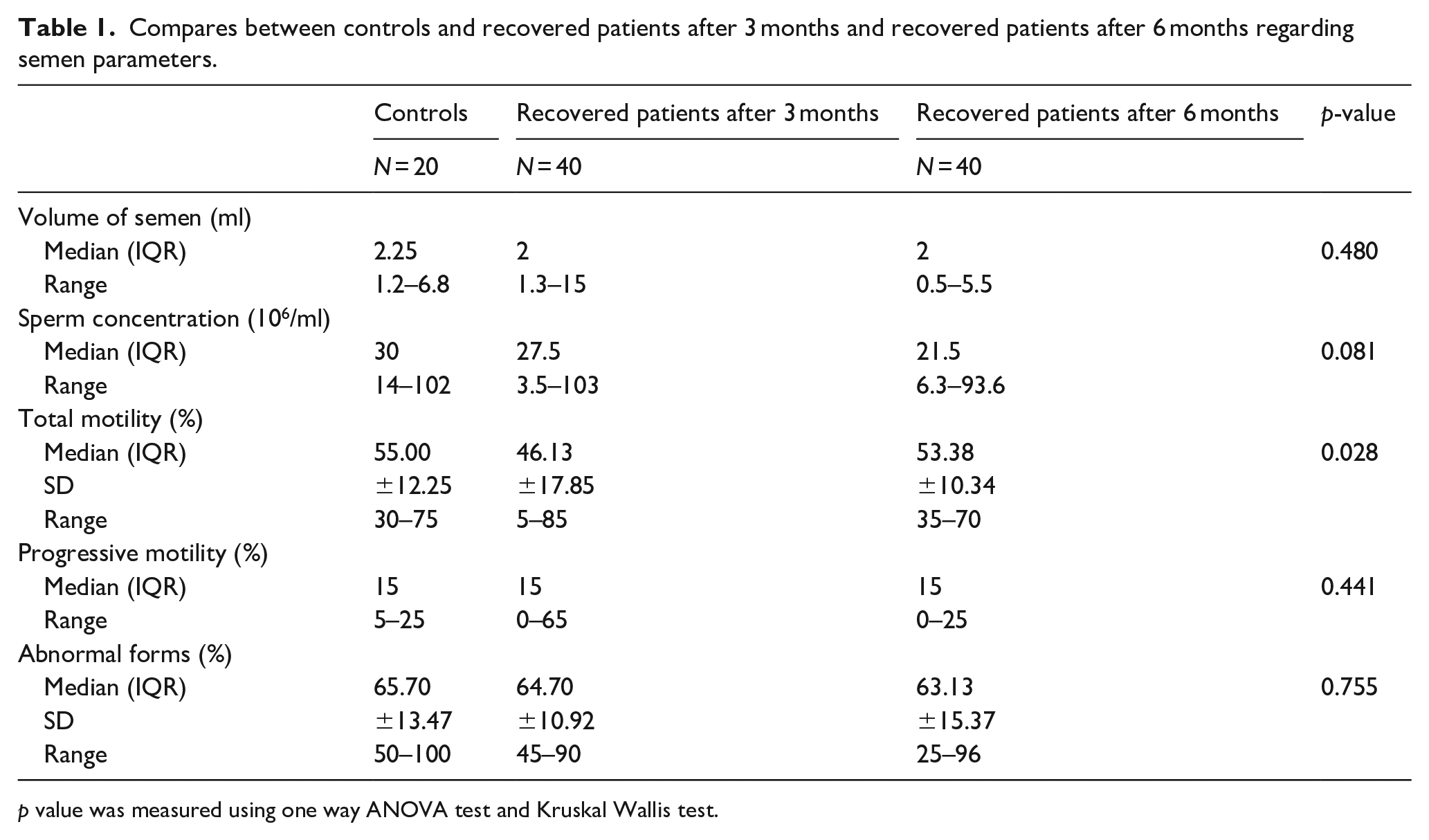
p value was measured using one way ANOVA test and Kruskal Wallis test.
Frequency of abnormal semen parameters among controls and recovered patients after 3 months and recovered patients after 6 months.
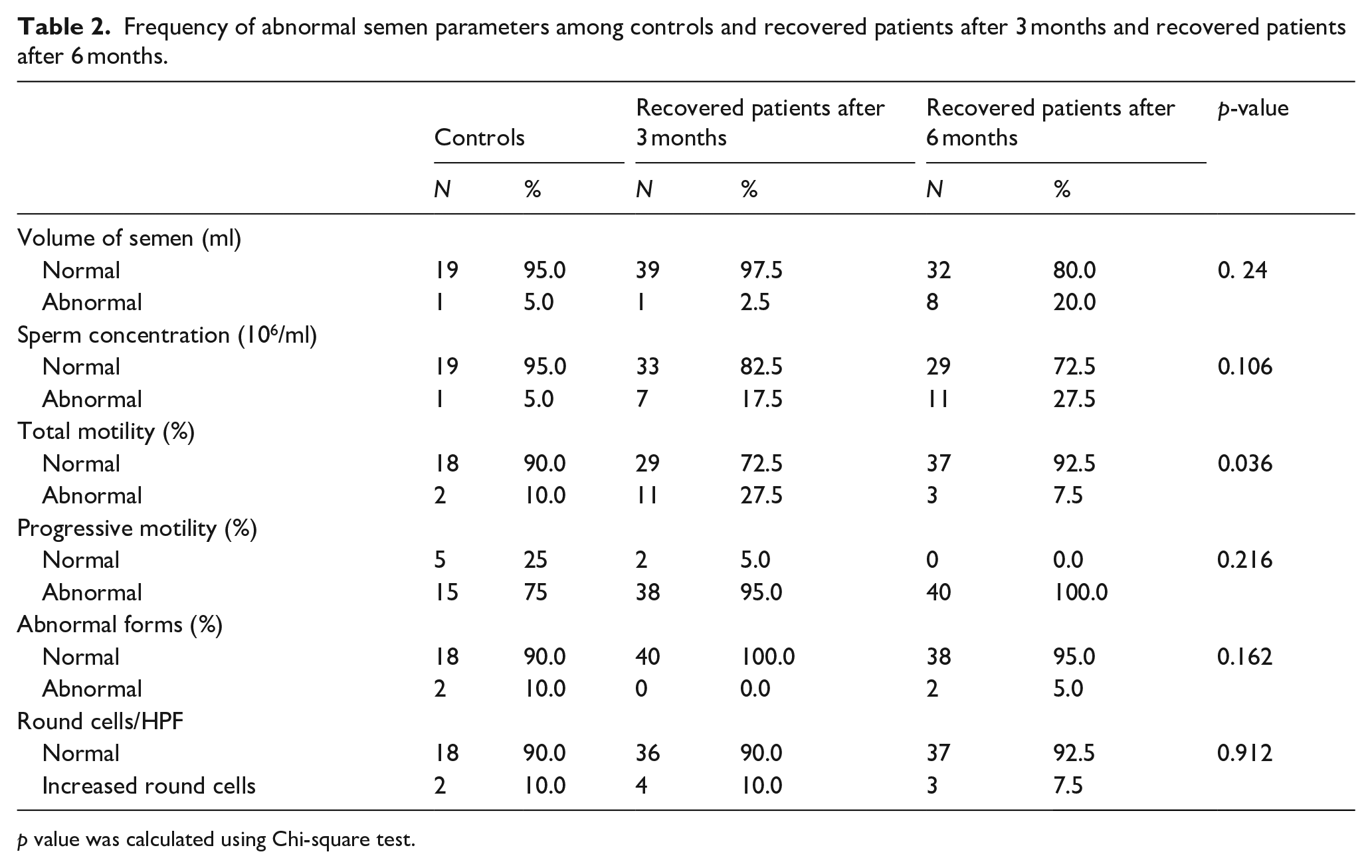
p value was calculated using Chi-square test.
Compares between controls and recovered patients after 3 months and recovered patients after 6 months regarding IIEF-5, HADS, and total testosterone.
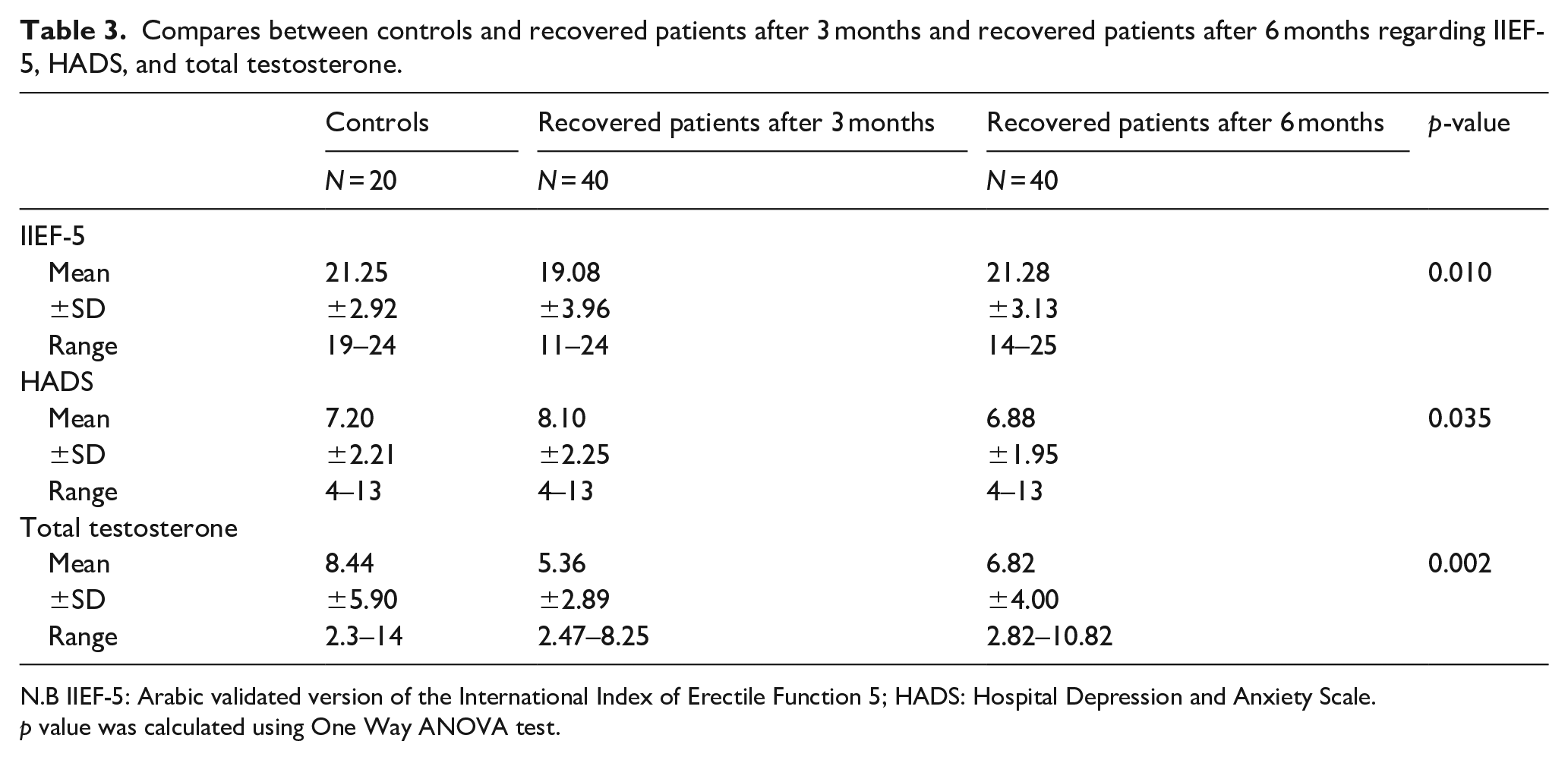
N.B IIEF-5: Arabic validated version of the International Index of Erectile Function 5; HADS: Hospital Depression and Anxiety Scale.
p value was calculated using One Way ANOVA test.
Finally, a marked decrease in the serum total testosterone in post COVID-19 patients was detected after 3 months when compared to the controls (5.36 ± 2.89 and 8.44 ± 5.90, respectively; Table 3). On the contrary, serum total testosterone showed an increase the post covid-19 patients after 6 months when compared to the post COVID-19 patients after 3 months (6.82 ± 4.00 and 5.36 ± 2.89, respectively; Table 3). Post Hoc analysis by LSD demonstrated a statistically significant decrease in the IIEF-5 score in post COVID-19 patients after 3 months when compared to the controls (p = 0.023). A marked increase in the IIEF-5 score in post COVID-19 patients was noted after 6 months when compared to post COVID-19 patients after 3 months (p = 0.005). Regarding the HADS score, a post hoc analysis demonstrated a statistically significant increase in the post COVID-19 patients after 3 months when compared to the controls (p = 0.026). A marked decrease in the HADS score in post COVID-19 patients was observed after 6 months when compared to post COVID-19 patients after 3 months (p = 0.012). Finally, a post hoc analysis demonstrated a statistically highly significant decrease in total testosterone in post COVID-19 patients after 3 months group when compared to the controls (p = 0.001). A marked increase in total testosterone in the post COVID-19 patients was detected after 6 months when compared to post COVID-19 patients after 3 months (p = 0.001).
Discussion
The current study had revealed a significant decrease in total sperm motility in the recovered patients after 3 months. Interestingly, total sperm motility significantly increased in recovered patients after 6 months. Furthermore, sperm concentration insignificantly declined after diagnosing COVID-19.
In contrast to our findings, Holtman et al. 14 demonstrated that sperm concentration and motility significantly decreased in post COVID-19 patients when compared to the controls. Additionally, Ma et al. 15 revealed low sperm motility in four patients after analyzing the semen samples of 12 patients after 56–109 days from the diagnosis of COVID-19. However; the previous study did not include control group as well as very low number of recruited patients in order to demonstrate change in semen parameters. 15 The changes in the semen parameters in our study can be attributed to the following facts. Firstly, SARS-CoV-2 may cause orchitis or spermatogonial stem cell infection leading to male infertility. 16 The orchitis caused by COVID-19 is facilitated by the angiotensin enzyme 2 as it acts as a cell entry receptor on the testis. 17 Furthermore, Yang et al. 18 had demonstrated testes from COVID-19 patients that exhibited significant seminiferous tubular injury, reduced Leydig cells, and mild lymphocytic inflammation. Our study revealed marked decline in total testosterone level in 3 months group when compared to the controls. However; there was a marked rise in total testosterone level in recovered patients after 6 months when compared to the recovered patients after 3 months. On the other hand, Ma et al. 15 did not report any significant difference in total testosterone and FSH levels between COVID-19 cases and controls. Meanwhile, LH level was significantly higher and total testosterone/LH ratio markedly declined in COVID-19 patients. 15 The initial decrease in the total testosterone reported in our study can be explained by the following facts. Firstly, hypergonadotropic hypogonadism state can independently develop due to testicular damage.4,19 –22 Furthermore, stimulation of the hypothalamohypophyseal axis as a result of emotional, physical, or psychological stresses and pain associated with infections leading to an abnormal hypothalamic pituitary axis’s function as well as irregular LH secretion rhythm may also be a reason. 15
Moreover, the active free testosterone decreases by binding to sex hormone binding globin. 15 In our study, there was a marked decline in the IIEF-5 score in the post COVID-19 patients after 3 months when compared to the controls. Interestingly, a marked increase in the IIEF-5 score in the post COVID-19 patients after 6 months when compared to the post COVID-19 patients after 3 months which suggest that this impairment in sexual function may occur for a transient duration. Quite the reverse, Saad et al. 23 stated that ED occurs in healthy individuals after recovery from severe COVID-19 as a consequence. In our study, there was a markedly increase in the HADS score of Post COVID-19 patients after 3 months group when compared with controls. However; decrease in the HADS score in post COVID-19 patients after 6 months when compared to the recovered patients after 3 months. In the same context, men and women suffered from temporary sexual dysfunctions due to an overall improvement that happened concomitantly with the end of lockdown. 24 Furthermore, this was observed especially in younger individuals with higher psycho-physical well-being. 24 On the contrary, Omar et al. 25 stated that COVID-19 pandemic was associated with lower sexual satisfaction in both genders. Additionally, females are at greater risk of sexual dysfunctions and sexual dissatisfaction as result of more anxiety and depression. 25 Consistently, COVID-19 lockdown negatively affected psychological, relational, and sexual health of the population in a dramatic way. 26 Interestingly, our study is one of the few studies that evaluated the effect of COVID-19 on the sexual and reproductive functions after 3 and 6 months, respectively. it delivered out transient negative impact that can be explained by elevation of total testosterone level approaching normal levels as well as improvement in the depression and anxiety levels. Admittedly, there are several limitations of our study that should be recognized. Firstly, the small sample size of our study is the major limitation.
Additionally, the nature of our study was cross sectional and not prospective as we evaluated two different groups of patients recovered from moderate to severe COVID-19 after 3 and 6 months can be also seen as a weakness point. Finally, we evaluated the patients by the HADS which is not validated in Arabic can be added as another limitation.
Conclusion
Our study had shown temporary impairment of sexual and reproductive functions in post COVID-19 Convalescent males especially after 6 months from the onset of the infection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
