Abstract
Background:
T1 high-grade (HG) non-muscle invasive bladder cancer (NMIBC) has a significant risk of recurrence and progression, and the European Association of Urology recommends a second transurethral resection of the bladder (ReTUR). Stage at ReTUR has been shown to be a reliable predictor of survival, therefore, we sought to assess clinical and pathological predictors associated with the persistence of T1 at ReTUR in our retrospective multicentric cohort.
Methods:
This is a retrospective multicentric study of T1 HG patients at transurethral resection of the bladder (TURB) who underwent subsequent ReTUR. All histological samples were sub-classified according to Rete Oncologica Lombarda (ROL) T1 sub-staging system.
Results:
One hundred and sixty-six patients were enrolled. Forty-four (26.5%) had T1 HG tumor at ReTUR while 93 (56%) had residual tumor of any stage. Lesion size was significantly greater in T1 HG patients at ReTUR, as well as the prevalence of multifocality. The multivariable logistic regression model showed lesion dimension and multifocality as predictors of T1 HG at ReTUR, after adjusting for significant covariables (CIS and detrusor muscle presence). ROL sub-staging system was not a significant predictor, but ROL2 prevalence was higher in the T1 HG at ReTUR group.
Conclusions:
Lesion size and multifocality were independent predictors of T1 HG persistence at ReTUR, and patients at risk should be promptly identified and treated accordingly. Our results could help physicians make patient-tailored decisions by identifying those most likely to benefit from a second resection.
Introduction
T1 high-grade (HG) non-muscle invasive bladder cancer (NMIBC) is a heterogeneous disease with a high risk of recurrence and progression.1 –3 According to the European Association of Urology (EAU) guidelines, a second transurethral resection of the bladder (ReTUR) within 2–6 weeks from the first one is recommended in patients with T1 disease before undergoing the induction course with Bacillus Calmette-Guerin (BCG) intravesical instillations. 4 This indication is based on the evidence of non-negligible rates of understaging and tumor persistence in patients diagnosed with T1 HG bladder cancer.
Nowadays, the persistence of T1 HG tumor at ReTUR is considered an essential predictor of survival in patients with T1 HG bladder cancer at first transurethral resection of bladder tumor (TURB).1,5 However, several studies raise doubts about the role of ReTUR in improving long-term oncological outcomes, especially in patients where the first resection is considered complete.1,6 –8 Considering the evidence suggesting the opportunity to safely avoid the ReTUR in selected cases where the tumor is macroscopically completely removed, we need more clinical and pathological reliable predictors of worse oncological outcomes to stratify T1 HG bladder cancer patients better and consequently differentiate those who might benefit the most from an early ReTUR. 6 Therefore, we sought to assess clinical and pathological predictors associated with the persistence of T1 disease at ReTUR in our retrospective multicentric cohort of T1 HG bladder cancer patients.
Materials and methods
This is a retrospective study enrolling patients who underwent TURB with a diagnosis of T1 HG bladder cancer and underwent ReTUR between 2015 and 2021 in two Italian referral centers (Department of Urology, IRCCS Humanitas Research Hospital, Rozzano; Urology Unit, Mater Domini Hospital, Castellanza). All patients with suspected bladder cancer at outpatient cystoscopy and/or with positive urine cytology underwent TURB followed by Narrow Band Imaging (NBI) resection of the margins and bed. Patients were positioned in the standard lithotomy position and procedures were performed with a 26 Ch resectoscope with bipolar loop electrode. After the resection, an 18–24 Ch three-way catheter was positioned with 24–48 h of continuous saline irrigation. All patients with a new diagnosis of T1 HG bladder cancer underwent computed tomography of the upper urinary tract to exclude the presence of upper tract urothelial carcinoma. ReTUR was performed after a median time of 6 weeks (IQR 5–7 weeks) from the first resection, including the resection of the primary tumor site and any areas suspicious of malignancy. Patients who did not undergo ReTUR, those with a concomitant urothelial carcinoma of the upper tract, and patients with incomplete clinical and pathological data were excluded from the analysis (Supplemental Figure 1). One hundred ten patients did not undergo ReTUR because of the following: indication of early radical cystectomy because of an unresectable tumor, uncontrollable hematuria, or very high-risk variants; in some cases, the ReTUR was avoided in consideration of the patient’s will after a thorough discussion on pro and cons of the procedure; some patients may have undergone ReTUR in another center. Pathologic samples from TURB were classified and reviewed by two uro-pathologists (P.C., M.V.) according to the grading of the WHO 2016 and WHO 1973 classifications. 9 We further sub-classified tumor samples in the first TURB using the ROL system (Rete Oncologica Lombarda) as previously described. 10 This pT1 sub-staging divided urothelial carcinoma depending on the type of lamina propria invasion: ROL 1 is characterized by a single focus or multiple foci summarized together ≤1 mm in dimension; ROL 2 is characterized by a single focus or multiple foci summarized together >1 mm in dimension. Specifically, ROL 1 refers to less than one power field of invasion (1PF, objective 20×, ocular 10×/field 22, diameter of 1 × 1 mm), corresponding to ≤ 1-mm invasion of the lamina propria (single or multiple foci); ROL 2 refers to more than one PF at 20× of invasion, corresponding to >1 mm invasion of the lamina propria (single or multiple foci). For this purpose, the major diameter of invasion, that is the extent of invasion in any direction, was measured.
Outcomes of the study
The primary outcome was to assess the preoperative clinical predictors of residual T1 HG tumor at ReTUR; the secondary outcome was to evaluate the role of the T1 ROL sub-staging system in identifying patients at risk of T1 HG tumor persistence at ReTUR.
Statistical analysis
Patients’ clinical, pathological, and demographic characteristics were reported as descriptive statistics. Continuous and categorical variables were reported as median with interquartile range (IQR) and frequencies, respectively. Mann-Whitney U test was used to compare continuous variables, while the chi-squared test was used to compare categorical variables. Univariable and multivariable logistic regression models were used to evaluate the predictors of adverse pathological findings at ReTUR. Even if not significant at univariable analysis, the presence of muscle layer and concomitant carcinoma in situ (CIS) at first TURB were computed in the multivariable analysis because they were considered clinically meaningful covariables at risk of confounding. To assess the potential center bias, main variables distributions among centers were tested and the final multivariable logistic regression was re-computed with the recruiting center as a covariable (Supplemental Table 1).
Two-sided p-value (p) < 0.05 was considered statistically significant. Analyses were performed with STATA 17.0 (StataCorp, College Station, Texas).
Results
From our prospective registry of NMIBC patients, we identified 166 T1 HG patients who underwent ReTUR, 91 and 75 from each recruiting center. There were no significant differences in the meaningful variables for the primary and secondary outcomes between the two recruiting centers, apart from a higher percentage of detrusor muscle in TURB specimens for center 2 (80.2 vs 93.3%, p = 0.015). Table 1 summarizes the overall cohort’s baseline characteristics and is divided between patients with and without the persistence of T1 HG tumor at ReTUR.
Main population characteristics.
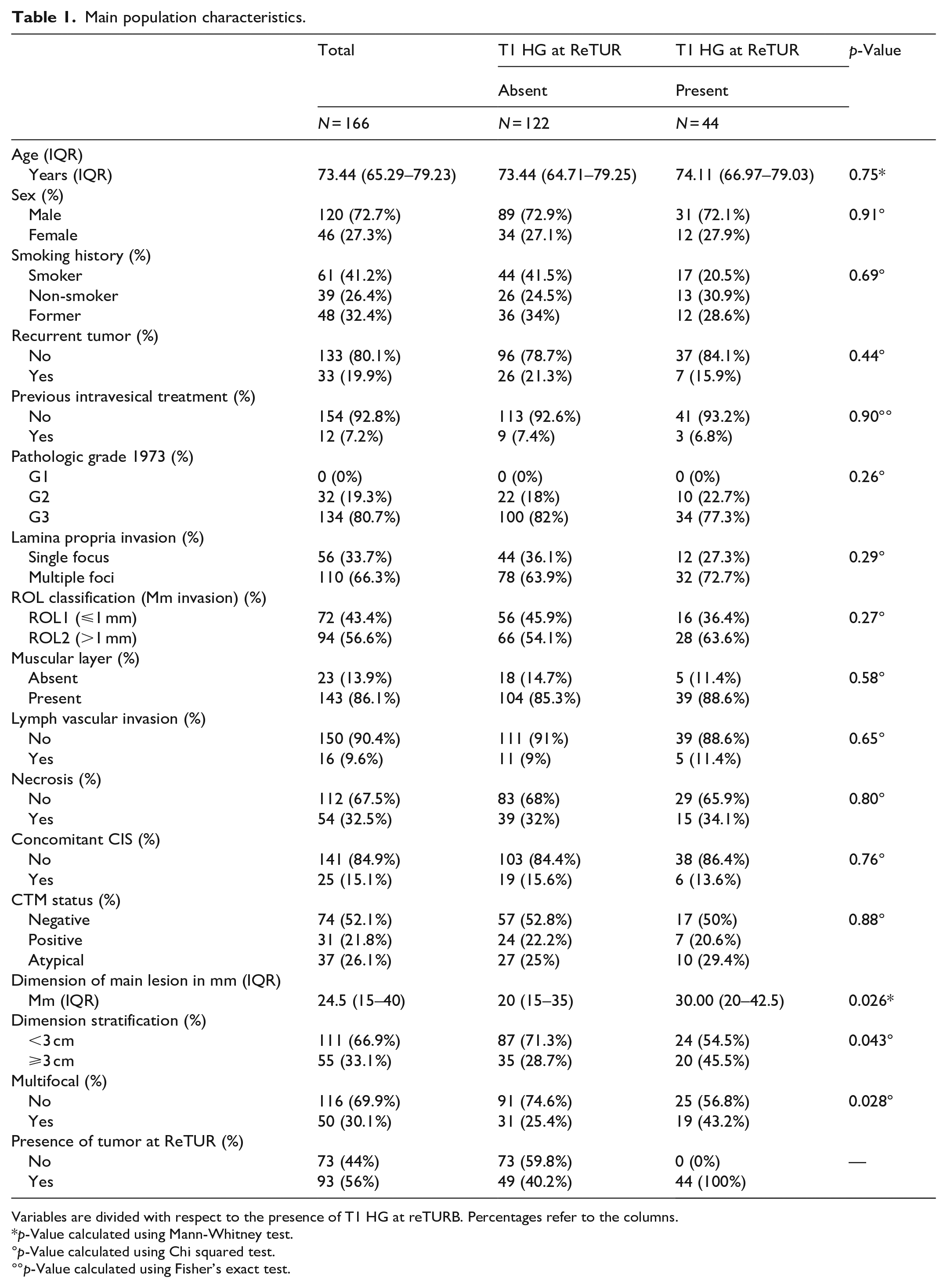
Variables are divided with respect to the presence of T1 HG at reTURB. Percentages refer to the columns.
p-Value calculated using Mann-Whitney test.
p-Value calculated using Chi squared test.
p-Value calculated using Fisher’s exact test.
Out of 166 patients, 120 (72.7%) were males, and 46 (27.3%) were females. The median age was 73 years old (IQR 65–79). All tumors were HG according to WHO pathologic grade 2016, while 134 (80.7%) were G3 and 32 (19.3%) were G2 according to WHO pathologic grade 1973. Thirty-three patients (19.9%) had a history of previous TURB, and 12 (7.2%) history of previous intravesical treatment. Overall, 44 (26.5%) patients had T1 HG at ReTUR while no patient was upstaged to T2, and 93 (56%) had residual tumor (any stage). The main differences between those with and without the persistence of T1 HG tumor at ReTUR were the dimension of the main lesion (p = 0.026) and multifocality (p = 0.028) (Figures 1 and 2). The muscular layer was absent in 23 (13.9%) patients, while CIS was present in 25 (15.1%), but the difference between those with and without the persistence of T1 HG at ReTUR was not significant (Figure 2).
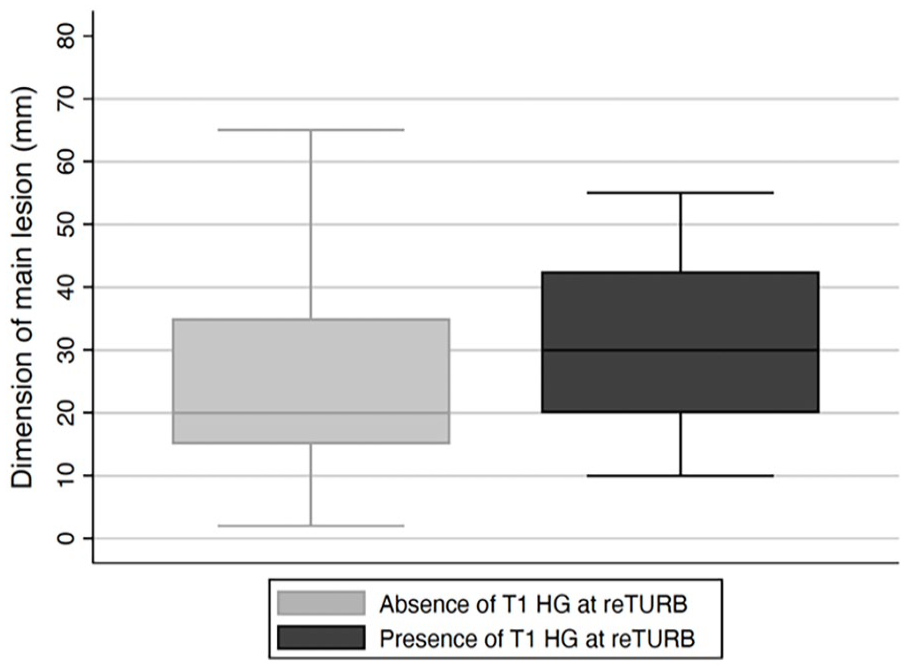
The figure shows the difference of the dimension of the main lesion at first TURB, see Table 1 for values.
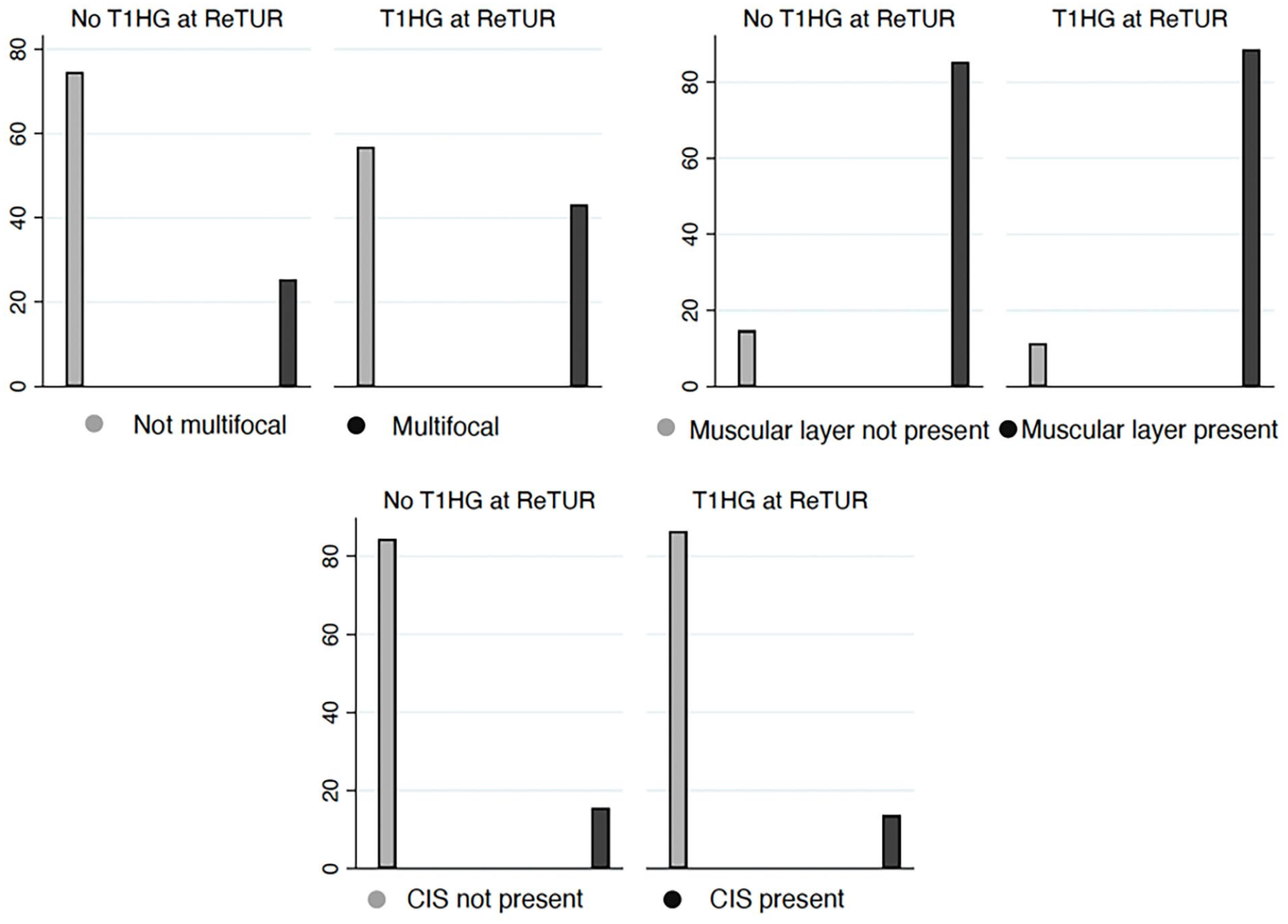
The figure shows the distributions of multifocality, presence of muscle layer and presence of CIS at first TURB stratified by T1 HG tumor at ReTUR. See Table 1 for values.
On univariable analysis tumor dimension (OR = 1.028, 95% CI 1.004–1.052, p = 0.019), having a lesion size greater than 3 cm (OR = 2.071, 95% CI 1.017–4.219, p = 0.045), and multifocal lesions (OR = 2.231, 95% CI 1.083–4.595, p = 0.029) were predictors of persistence of T1 HG at ReTUR (Table 2).
The table shows the univariable logistic regression model of clinically relevant variables.
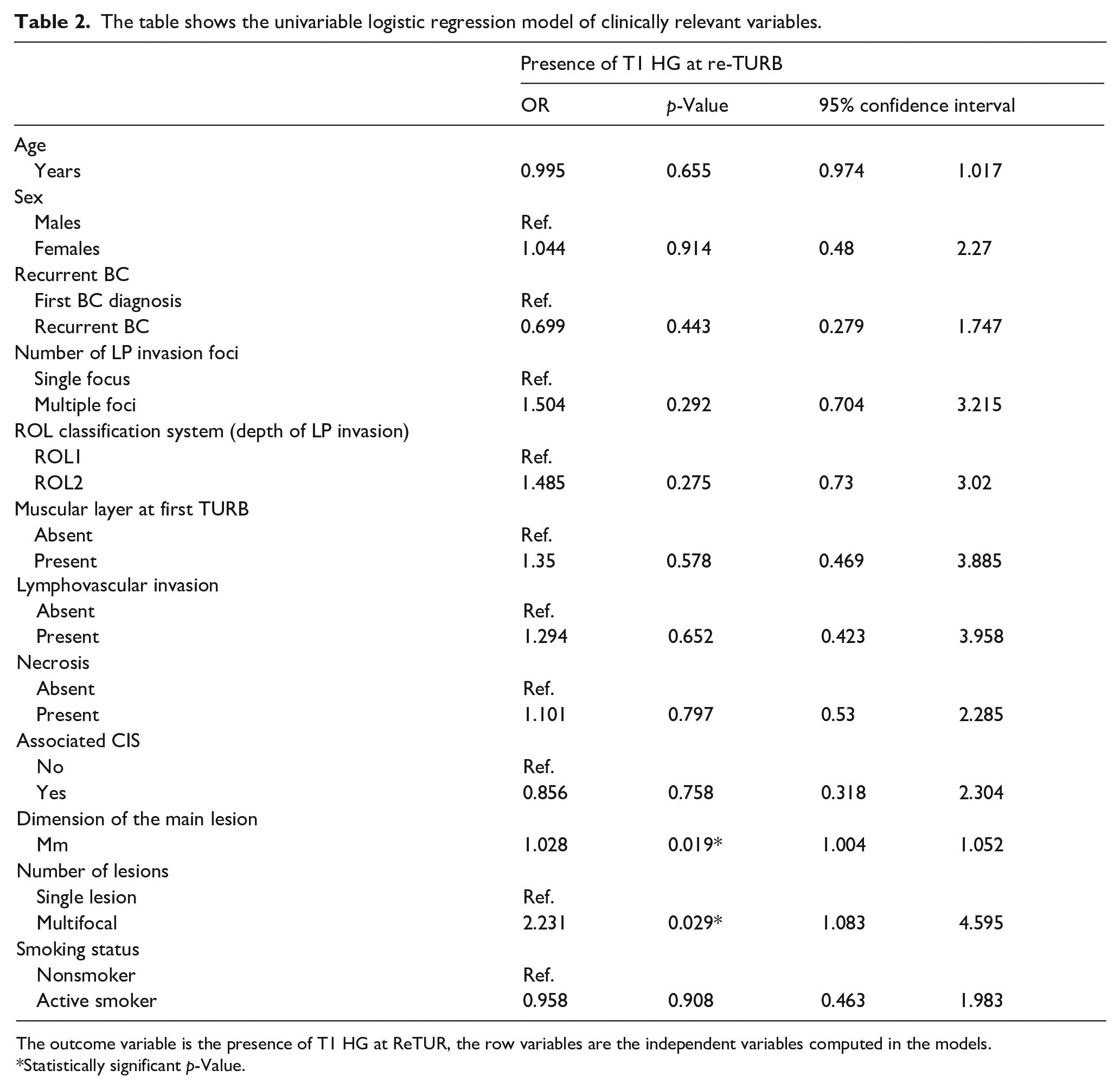
The outcome variable is the presence of T1 HG at ReTUR, the row variables are the independent variables computed in the models.
Statistically significant p-Value.
Dimension of the main lesion (OR = 1.029, 95% CI 1.005–1.055, p = 0.015) and multifocality (OR = 2.352, 95% CI 1.119–4.942, p = 0.024) were significant predictors of persistence of T1 HG bladder cancer at ReTUR adjusted multivariable logistic model (Table 3). By including the fixed effect of the recruiting center in the final multivariable model we tested the possible center bias, which proved unremarkable (Supplemental Table 1).
The table shows the multivariable logistic regression model of the significant variables found at ULRM.
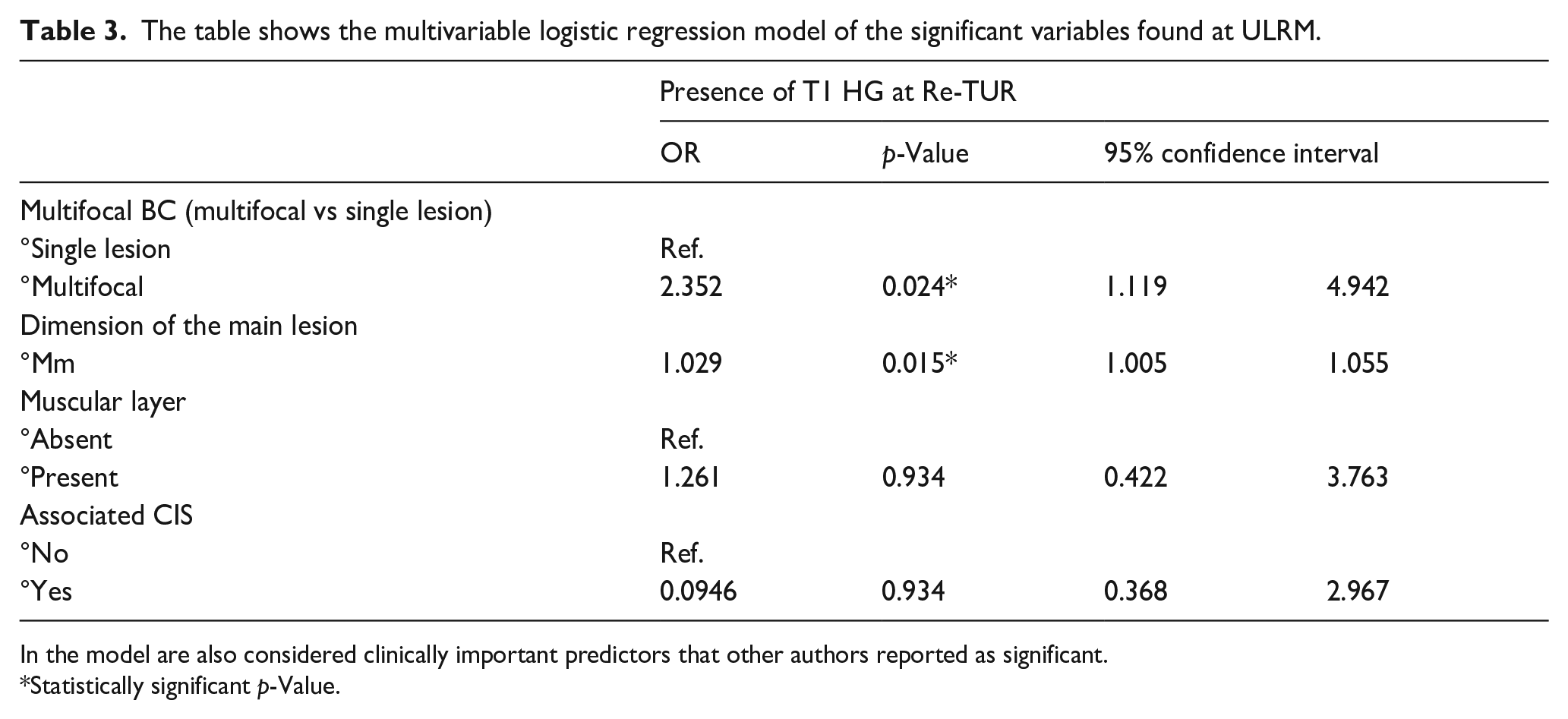
In the model are also considered clinically important predictors that other authors reported as significant.
Statistically significant p-Value.
Regarding the ROL classification system at the first TURB, it was feasible in 100% of the cases and 94 (56.6%) patients were ROL2, while 72 (43.4%) were ROL1. We found that patients with T1 HG at ReTUR had a higher prevalence of ROL2 at first TURB (63.6 vs 54.1% of patients), but the association was not statistically significant (p = 0.27). Fifty-six (33.7%) patients had a single focus of invasion of the lamina propria, while 110 (66.3%) had multiple foci of LP invasion (p=0.29).
Discussion
This retrospective multicenter study shows that about 25% of the patients presented a residual T1 HG tumor at ReTUR. Furthermore, multifocality and lesion dimensions were demonstrated as independent predictors of residual high-risk disease at ReTUR.
It is commonly recognized that the ReTUR stage holds an important role as a prognostic indicator of disease recurrence, progression, and cancer-specific survival. Herr et al. found that T1 HG BC tumor at ReTUR is associated with an 82% risk of disease progression to MIBC at 5 years. 5 Recently, Palou et al. 1 found that 25% of T1 cases at ReTUR experienced progression to muscle-invasive disease at 5 years. Literature reported that residual tumor could range between 33 and 58%, while the rate of upstaging to muscle-invasive disease ranged from 1–4 to 10 25%.2,7,11 –15 In our series, 56% of patients had a residual tumor, while there was no upstaging to MIBC. Many factors were demonstrated to affect the correct staging at first TURB, such as surgeon and pathologist experience. A recent systematic review of the literature found a negligible risk of upstaging in many series (1−4%), underscoring a possible effect of surgeons’ experience on primary TUR outcomes. 12 Another possible explanation may be that most urologists in our center usually sample the bed of the resection to avoid any understaging. Arguably, we still reported that 13.9% of the pathological samples did not have the muscular layer. However, that number in our series may be too small to encounter an upstaging, which is a varying and uncommon event among centers. Also, we refer patients harboring extended T1 HG tumors with uncontrollable hematuria to radical cystectomy. These unusual cases did not enter the study cohort as they did not undergo ReTUR and may have been the most likely to experience an upstaging. Overall, the apparent contradiction of finding 56% of the patients harboring residual tumor while 86% of the samples had the muscular layer may be related to the intrinsic biological aggressiveness of T1HG tumors, which are a high-risk entity of NMIBC under every oncological aspect.
Previous studies investigated the predictors of persistence of T1 HG at ReTUR. Ferro et al. 16 found a statistically significant association between the presence of T1 HG BC at ReTUR and multifocal tumors, tumors greater than 3 cm, and the presence of CIS at first TURB. Kamiya et al. 17 showed that multifocality at first TURB is an independent predictor of T1 HG at ReTUR. Furthermore, Palou et al. 1 found that T1 HG at ReTUR is more likely if the detrusor muscle is absent at first TURB. Interestingly, BMI and being overweight were proven to be significantly associated with residual HG tumor at ReTUR, alongside tumor size and concomitant CIS. 18 The evidence is heterogeneous, and large studies do not consistently demonstrate the same predictors to be significant. However, most articles found that lesion size and multifocality were common independent predictors of T1 HG tumor at ReTUR.
Whether performing ReTUR has a positive impact on oncological outcomes remains controversial. A large study, including data from 2451 patients, showed no benefit from ReTUR if detrusor muscle was present at first TURB.7,11,19 –22 Furthermore, a recent metanalysis by Krajewski et al. 8 including 3257 patients failed to show any oncological outcome improvement after ReTUR, except for studies that included both HG and Low grade (LG) T1.
We believe that a personalized approach should be defined in the future years, allowing for fewer operations and consequently lowering patients’ stress and health system burden. In this scenario, ReTUR might be performed only in patients with a significant risk of T1 HG persistence, while well-selected patients could promptly start intravesical therapy. In this regard, Contieri et al. evaluated the chance of avoiding ReTUR in patients where the tumor was macroscopically completely removed at first TURB and had both negative urine cytology and cystoscopy at 4–6 weeks after the resection. They found that among those not undergoing the early second resection, only 17.5% experienced recurrence of the disease during the follow-up without evidence of upstaging. 6
These findings support that ReTUR indication should be tailored to the patient risk factors for T1 HG persistence because it is the only solid prognostic indicator. In this scenario, ReTUR might be avoided not only in Ta HG patients that underwent complete first resection with the presence of detrusor muscle in the pathological specimen as prescribed by EAU guidelines, but also in selected small solitary T1 tumors. Further prospective trials, including prognostic factors, molecular markers, and new diagnostic tools, are needed to correctly identify those patients who could benefit from a more aggressive treatment.23 –26 Moreover, some interesting findings depicted De Ritis Ratio and Neutrophil-to-Lymphocytes Ratio as possible indicators of locally aggressive tumors and may be considered in the classification of patients, as in the article by Uleri et al. 27
We have previously documented an interesting predictive role of ROL system for progression in T1 HG tumors on a retrospective series that has been also introduced by WHO 2022 classification as one of the feasible methods to stratify pT1 tumors in TURB.10,28 This is the first study investigating the role of ROL T1 sub-staging on ReTUR outcomes. Although we did not find a significant difference between ROL 1 and ROL 2 in patients with persistence of T1 HG at ReTUR, we demonstrated a possible effect that may be relevant in larger sample sizes. Notably, the validity of the ROL sub-staging system for oncological outcomes was recently evaluated and the system reliably stratified patients, with ROL 2 having lower PFS and RFS. 29 Further studies will help consolidate the oncological impact of this innovative staging system. A critical issue with T1 sub-staging systems is their feasibility, which is not the same among different systems. In our population, the ROL system was feasible in 100% of the cases. A new, intriguing topic regarding T1 sub-staging emerging from recent literature is how they relate to the en-bloc resection of bladder tumors. A recent randomized non-inferiority trial demonstrated that en-bloc resection, compared to conventional TURB, has a significantly higher T1 a/b/c sub-staging feasibility (100 vs 80%, p = 0.02). 30 Considering the possible influence of the T1 sub-staging system on different outcomes, its feasibility must be considered and reported in future literature.
The present study has some limitations. First, due to its retrospective design, it has some intrinsic drawbacks. For this reason, we were forced to eliminate some records due to missing or inaccurate data. Second, there was significant heterogeneity in surgeons’ experience, considering that patients were enrolled from two hospitals with residents in training. Third, the study does not report oncological outcomes as RFS, PFS, and OS. Fourth, we do not investigate the surgical technique and in particular the en-bloc resection, which seems to provide accurate staging and oncological outcomes.31,32 Fifth, the study sample is relatively small, which may impact on the reliability and reproducibility of the results.
Conclusions
There is no conclusive evidence on whether ReTUR improves oncological outcomes and which patients would benefit from it the most. We found that lesion size and multifocality were independent predictors of T1 HG persistence at ReTUR. Neither CIS nor detrusor muscle presence, as other studies had shown, were associated with T1 HG persistence however, there is discordance among current literature and further data are needed. Our results could help physicians make patient-tailored decisions by identifying those most likely to benefit from a second resection.
Supplemental Material
sj-docx-1-urj-10.1177_03915603231181619 – Supplemental material for Clinical and pathological predictors of persistent T1 HG at second resection
Supplemental material, sj-docx-1-urj-10.1177_03915603231181619 for Clinical and pathological predictors of persistent T1 HG at second resection by Andrea Gobbo, Vittorio Fasulo, Roberto Contieri, Alessandro Uleri, Pier Paolo Avolio, Nicola Frego, Giovanni Lughezzani, Cesare Saitta, Gianluigi Taverna, Matteo Zanoni, Stefano Mancon, Piergiuseppe Colombo, Marina Valeri, Alberto Saita, Massimo Lazzeri, Nicolò M Buffi, Rodolfo Hurle and Paolo Casale in Urologia Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was was funded by BIBLIOSAN.
Institutional review board statement
Ethical review and approval were not inquired for this study in consideration of its retrospective design. All patients referring to the two centers that contributed to this study signed informed consent for data gathering, elaboration, and publication in anonymous form for retrospective studies. The study was conducted according to the Declaration of Helsinki and Good Clinical Practices Guidelines.
Informed consent statement
Informed consent was obtained from all subjects involved in the study for anonymous retrospective data collection and publication.
Data availability statement
Data are available under request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
