Abstract
Severe rhabdomyolysis, characterized by extensive muscle breakdown and release of myoglobin and creatine kinase (CK), is a life-threatening condition often complicated by acute kidney injury (AKI) and multi-organ failure (MOF). Even when conventional treatments such as fluid resuscitation and renal replacement therapy (RRT) are timely applied, severe cases remain challenging to manage. Among therapies available in this setting, hemadsorption with CytoSorb has the potential not only to treat rhabdomyolysis through removal of circulating molecules but also to limit or even prevent rhabdomyolysis-related renal failure and MOF. In this case series we present a preliminary experience with a novel use CytoSorb hemoadsorption therapy, which encompassed the use of two CytoSorb cartridges running in parallel, to enhance myoglobin and cytokine clearance. Clinical data from the three patients with severe rhabdomyolysis treated with dual concomitant CytoSorb treatments highlighted marked improvements in CK, renal, hepatic, and inflammatory markers, with possible remarkable impact in containing rhabdomyolysis-related organ failure and death. Further investigation is warranted to establish the opportunity of tailored protocols for critically ill patients.
Introduction
Rhabdomyolysis is a clinical syndrome characterized by the breakdown of striated muscle cells, leading to the release of intracellular components such as myoglobin, electrolytes, lactate dehydrogenase (LDH), and creatine phosphokinase (CK) into the bloodstream.1,2 Among its complications, acute kidney injury (AKI) is particularly severe, occurring in 15%–50% of cases.3,4 Key risk factors include extensive muscle injury, dehydration, and CK levels exceeding 15,000 U/L, which predispose patients to nephrotoxicity and systemic instability.5,6 The nephrotoxic effects of myoglobin and electrolyte imbalances are the primary mediators of organ damage. Myoglobin-induced AKI arises from oxidative tubular injury due to reactive oxygen species (ROS), tubular obstruction, and renal vasoconstriction. These mechanisms are often exacerbated by systemic inflammation, creating a vitious cycle of worsening organ dysfunction. 7
The initial management of rhabdomyolysis is centered on aggressive fluid resuscitation, administration of osmotic agents such as mannitol, and forced diuresis to prevent complications. 8 In cases of renal failure, continuous renal replacement therapy (CRRT) is commonly employed.9 –11 However, standard dialysis membranes are limited in their ability to clear myoglobin due to its molecular weight. High cut-off (HCO) membranes and hemadsorption devices, such as CytoSorb, provide superior myoglobin and cytokine clearance.12,13 CytoSorb in particular is a hemadsorption device capable of removing molecules up to 55 kDa that has shown valuable efficacy in removing myoglobin, although the risk of saturation should be taken into account. 14 Manufacturer recommendations include replacement of CytoSorb cartridge every 24 h, and with this regimen CytoSorb has proven valuable efficacy in different clinical contexts dominated by uncontrolled inflammatory response and hemodynamic instability. 15 However, it is arguable that in specific circumstances, such as in case of severe rhabdomyolyses with massive release of molecules that may precipitate renal function and MOF, patients may benefit from more intensive hemadsorption cycles compared to routine practice16,17 to contain the risk of kidney failure.
In the lack of any data testing specific hemadsorption protocol in this setting, we present a case series exploring the innovative use of two concomitant CytoSorb filters in parallel to treat severe rhabdomyolysis. 18
Methods
We retrospectively collected data from clinical record charts of all patients treated at our Institution who received two Cytosorb circuits in parallel in the context of severe rhabdomyolysis. All the patients signed an informed consent allowing for clinical data management for scientific purposes.
Results
Case 1
Medical history
A 26-year-old woman with no significant medical history presented with severe left knee pain since 3 days. CT Imaging revealed the presence of aorto-bisiliac thrombosis with embolization to both iliac arteries, pulmonary embolism (PE), and ischemic renal and splenic lesions, in the context of massive embolization from an atrial myxoma measuring 12 mm × 7 mm with a maximum thickness of 6 mm.Acute ischemia led to compartment syndrome of the left leg, necessitating fasciotomy. Cardiac evaluation revealed Takotsubo cardiomyopathy with an ejection fraction (EF) of 25% and apical akinesia. Imaging confirmed atrial myxoma and systemic ischemia affecting multiple organs. Surgical intervention included thrombectomy, fasciotomy, and surgical removal of the atrial myxoma was performed. At the referring hospital, the patient was initiated on CRRT in the emergency department due to persistent anuria and distributive shock, and norepinephrine support was started.
Cytosorb treatment
The dual standalone CytoSorb setup, configured in parallel, was applied immediately postoperatively to provide intensive removal of inflammatory mediators. The patient underwent four hemoadsorption cycles with parallel CytoSorb filters, with filter replacement every 8–12 h (total duration of dual CytoSorb therapy: 4 days). CK levels, initially >100,000 U/L, reduced to 8800 U/L by therapy’s end. Improvements were observed in renal, hepatic, and cardiac biomarkers, reflecting systemic recovery (Figure 1).
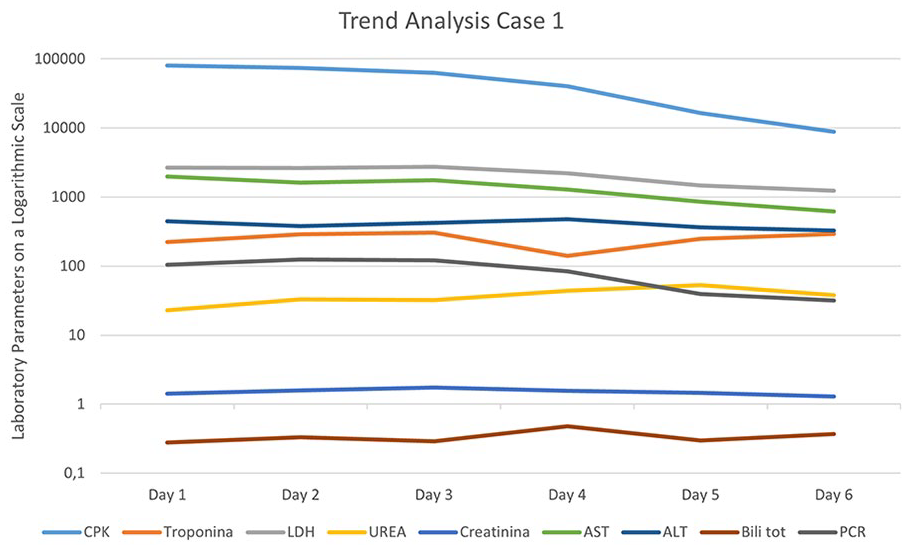
Temporal trend of laboratory parameters in Case 1. This figure illustrates the evolution of various laboratory biomarkers over 6 days of monitoring. The Y-axis represents laboratory values plotted on a logarithmic scale to better visualize differences in magnitude. The X-axis indicates the days of observation (Day 1 to Day 6). CPK: Creatine Phosphokinase; LDH: Lactate Dehydrogenase; AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; PCR: C-reactive protein; Urea; Creatinine; Troponin; Total Bilirubin. A decline in CPK and LDH levels is observed over time, while other biomarkers exhibit variable trends.
Clinical outcome
Norepinephrine was rapidly tapered off, and inotropic support with adrenaline was introduced, reaching a maximum dose of 0.03 µg/kg/min. The patient developed AKI with a peak plasma creatinine level of 1.73 mg/dL, necessitating the initiation of CRRT already prior to ICU admission. ICU stay was 6 days.
Case 2
Medical history
A 54-year-old woman with a history of folliculitis presented at emergency department with signs of cardio-septic shock. Initial symptoms included vomiting and malaise following a flu-like illness. Imaging identified pleural and pericardial effusions and possible cholecystitis. Despite interventions, including intubation, fluid resuscitation, and vasopressor, worsening shock led to the development of mixed acidosis and severe left ventricular dysfunction (EF 20%). Coronary angiography ruled out obstructive chronic coronary artery disease.
Clinical conditions further deteriorated with cardiac tamponade requiring pericardiocentesis and intra-aortic balloon pump (IABP) support for low cardiac output syndrome sustained by LV dysfunction. Diffuse muscle swelling triggered the use of dual CytoSorb therapy to manage inflammatory and myoglobin burden.
Cytosorb treatment
After the first 3 days of dual cartridges in parallel, CytoSorb treatment was continued with a single cartridge for a total of 12 days. The patient subsequently developed sepsis-associated multi-organ failure. Diagnosis of lymphocytic myocarditis associated with HHV-6 and West Nile virus was made. Dual CytoSorb therapy effectively reduced CK and inflammatory markers during the acute phase, possibly limiting renal failure extension and providing temporary stabilization. Renal function, as assessed through urea and creatinine levels, remained relatively stable throughout the observation period: despite this, the patient nevertheless required few cycles of continuous renal replacement therapy (CRRT) in the context of critical illness. Cardiac markers, including troponin, exhibited significant elevation, consistent with myocardial injury. Lactate dehydrogenase (LDH) levels mirrored this trend, further highlighting systemic tissue damage and hemolysis, which aligned with the diagnosis of lymphocytic myocarditis. Inflammatory markers such as C-reactive protein (PCR) displayed a progressive upward trend, underscoring the severity and persistence of the systemic inflammatory response driven by viral infections and sepsis. Together, these laboratory findings illustrate the profound multi-organ involvement during the patient’s clinical course (Figure 2).
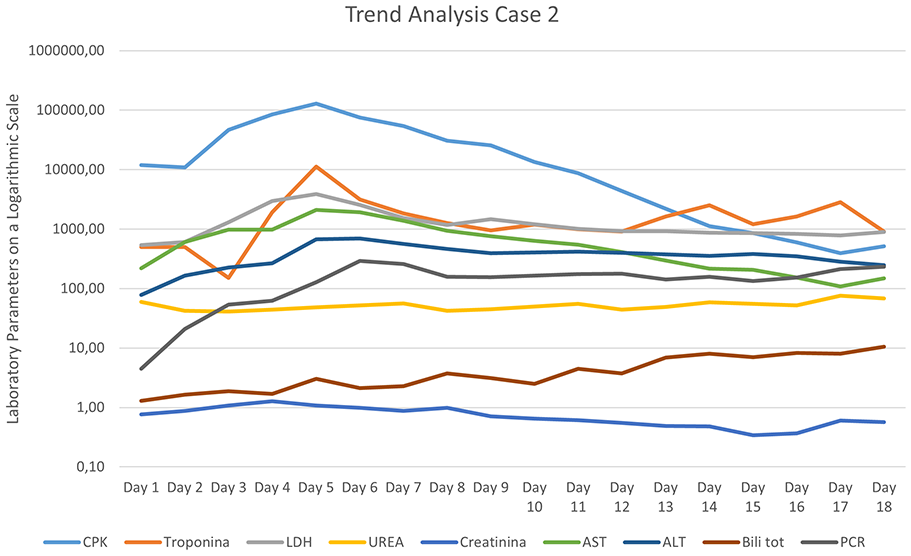
Temporal trend of laboratory parameters in Case 2. This figure illustrates the evolution of various laboratory biomarkers over 6 days of monitoring. The Y-axis represents laboratory values plotted on a logarithmic scale to better visualize differences in magnitude. The X-axis indicates the days of observation (Day 1 to Day 12). CPK: Creatine Phosphokinase; LDH: Lactate Dehydrogenase; AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; PCR: C-reactive protein; Urea; Creatinine; Troponin; Total Bilirubin. A decline in CPK and LDH levels is observed over time, while other biomarkers exhibit variable trends.
Clinical outcome
Despite escalation to full cardiorespiratory support with veno-arterial extracorporeal membrane oxygenation (VA-ECMO), the patient died due to MOF in the context of refractory cardiogenic after 17 days from ICU admission.
Case 3
Medical history
A 41-year-old man without comorbidities presented with sudden-onset left lower limb plegia due to acute type A aortic dissection involving also brachiocephalic trunk, left carotid artery, and left subclavian artery . Emergency surgery included ascending aorta replacement with debranching of the brachiocephalic trunk and limb revascularization. Severe rhabdomyolysis with CK levels exceeding 100,000 U/L accompanied by fulminant AKI dictated the need for CRRT combined with dual CytoSorb therapy from the first postoperative day.
Cytosorb treatment
Dual CytoSorb treatment was performed for two cycles followed by three single-filter cycles effectively and rapidly reduced CK to 55,069 U/L. Elevated AST, ALT, and troponin levels progressively declined with therapy, reflecting systemic recovery (Figure 3). Hemodynamic stabilization was progressively achieved with reduction of inotropic load in the days after surgery.
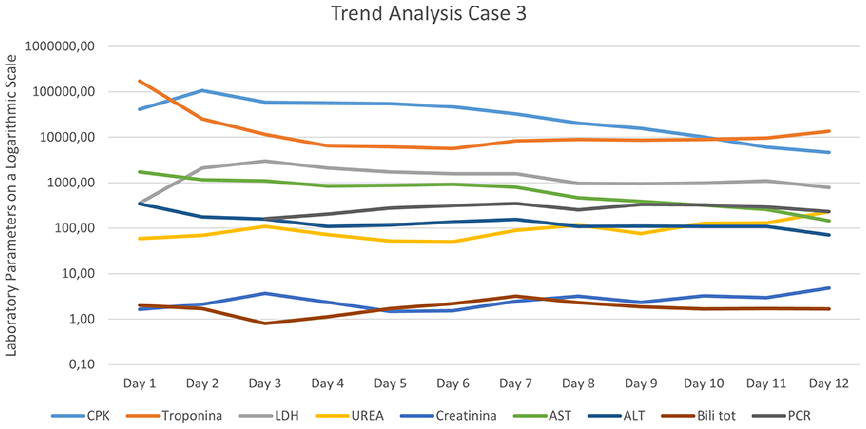
Temporal trend of laboratory parameters in Case 3. This figure illustrates the evolution of various laboratory biomarkers over 6 days of monitoring. The Y-axis represents laboratory values plotted on a logarithmic scale to better visualize differences in magnitude. The X-axis indicates the days of observation (Day 1 to Day 18). CPK: Creatine Phosphokinase; LDH: Lactate Dehydrogenase; AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; PCR: C-reactive protein; Urea; Creatinine; Troponin; Total Bilirubin. A decline in CPK and LDH levels is observed over time, while other biomarkers exhibit variable trends.
Clinical outcome
The postoperative recovery was complicated by sternal wound infection with need for surgical revision and VAC therapy. The patient has recovered from end-organ dysfunction and sepsis, and was discharged from ICU after 73 days.
Discussion
This case series highlights the clinical relevance of rhabdomyolysis in critical care. Though not frequent, severe rhabdomyolysis can arise in various clinical scenarios, and if massive, it may lead to hyperacute kidney failure, jeopardizing patient outcomes. 19 Our case series underscores that rhabdomyolysis typically arises in critically ill patients who are often young and without comorbidities. While renal replacement therapy (RRT) is frequently employed to manage the complications of acute kidney injury (AKI),20,21 an ideal strategy in the setting of rhabdomyolysis should also addressing the need to contain rhabdomyolysis related renal insult in a second time. In particular, addressing the underlying drivers of rhabdomyolysis—such as massive myoglobin release and systemic inflammation—early may theoretically help to mitigate kidney injury and multi-organ failure (MOF) more effectively. Therapies like CytoSorb hemoadsorption offer a targeted approach to rapidly clearing myoglobin and inflammatory mediators, which could help to preserve renal function and improve overall outcomes. 22 Therefore, a shift toward preventive strategies in managing severe rhabdomyolysis, rather than solely relying on RRT, is essential to optimize patient care and reduce mortality.
Although hemoadsorption has significant potential in the management of severe rhabdomyolysis, however, in cases of acute severe rhabdomyolysis with massive toxin release, the rate of clearance achieved by conventional hemoadsorption approaches may not be sufficient to prevent rapid organ damage. Of note, it has been shown that Cytosorb® adsorption capacity of hemoglobin decreases rapidly after few hours. 23 It is therefore arguable, that early change of the adsorber in patients with severe rhabdomyolysis might increase its efficacy. To address this limitation, we introduced an innovative approach utilizing dual CytoSorb filters in a parallel configuration. This strategy effectively maximized adsorption capacity, accelerating the removal of myoglobin and inflammatory mediators in emergency settings. By reducing the time required for toxin clearance, this approach creates a critical temporal window to intervene and prevent or limit the entity of renal and multi-organ damage. Furthermore, it can be postulated that the saturation of the cartridge is delayed when using two circuits running in parallel, 23 thus providing further protection toward rhabdomyolysis related organ failure. The dual CytoSorb setup proved particularly effective in rapidly mitigating systemic inflammation, providing essential support during the acute phase of severe rhabdomyolysis. Furthermore, our strategy proved safe and low invasive, as it is Cytosorb therapy deployed routinely with a single cartridge. This approach represents a significant advancement over conventional CytoSorb use and was shown to be both safe and feasible in our extremely critically ill cases. Therefore, our findings highlight the potential of dual CytoSorb therapy as an advanced solution for the management of severe rhabdomyolysis, warranting further investigation in broader clinical applications. Furthermore, the preliminary findings of our work may be transferred for application also to other clinical settings, where massive release of inflammatory or lysis molecules in the bloodstream occurs, such as in tumor lysis, cytokine storm, or polytrauma settings. We also acknowledge that Cytosorb treatment affects platelet count and potentially influence the efficacy of concomitant therapies. However, in a recent study 17 we showed that anticoagulation and antibiotic agents pharmacokinetics and dynamics during Cytosorb treatment remained reliable and reproducible, suggesting that, adopting appropriate dosing monitoring of drugs as recommended in such critically ill patients with organ failure, the clearance of administered drug by hemadsorption circuit does not represent a major concern.
We also acknowledge some limitations of this work. Being a case series, experience is limited and we are aware that we were able to provide only preliminary data. Given the retrospective collection of data, some parameters that could interesting to study, cannot be provided since they were not collected as part of our routine clinical practice (including cytokine or interleukin levels or dosages of other molecules removed by CytoSorb). Similarly, we did not collect data on inlet and outlet pressures from Cytosorb cartridge, as it was not part of our routine clinical practice. We also claimed that our approach with dual CytoSorb treatment may support renal function and possibly reduce the degree of renal failure. Given the extreme critically illness of our patients, however, renal replacement therapy was needed in all three patients; therefore, further data on larger numbers of patients are needed to better study the impact of dual CytoSorb treatment in preventing (or reducing) severe renal failure.
In conclusion, dual CytoSorb therapy offers a transformative approach to severe rhabdomyolysis, demonstrating potential for organ preservation and recovery in critically ill patients. Future studies are warranted to further evaluate its efficacy and refine its application in acute multi-organ failure contexts.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
